Abstract
Introduction:
Nasal septal deviation (NSD) is a leading cause of nasal airway obstruction, a condition that adversely affects quality of life. Septoplasty, typically performed with inferior turbinate reduction (ITR), is a common treatment to correct NSD.
Methods:
We conducted a prospective feasibility study of a novel, minimally-invasive implant (SeptAlignTM) to correct cartilaginous NSD as an alternative to traditional open septoplasty. The implant was inserted either in the operating room or at the medical office. ITR was performed in conjunction with the implant procedure.
Results:
We present 12 month results for 14 participants with mobile cartilaginous NSD. The procedure and implant were well-tolerated. At the 12th month post-procedure, the median Nasal Obstruction Symptom Evaluation (NOSE) score improved 71.8% from baseline (58-15; P = .001) and the median Sino-Nasal Outcome Test-22 (SNOT-22) score improved 60.3% from baseline score (37-8; P = .004). Most of the improvement in symptoms occurred within the first month following the procedure at which point 92.9% and 78.6% of participants were considered NOSE and SNOT-22 responders, respectively.
Conclusion:
These initial findings support the safety, tolerability, and early efficacy of the SeptAlignTM implant with concomitant ITR and suggest that the implant procedure can be safely performed in a medical office setting. Based on these promising initial data, larger, randomized, controlled studies should be conducted.
ClinicalTrials.gov:
NCT05967169; https://clinicaltrials.gov/study/NCT05967169?term=Spirair&rank=2
Keywords
Introduction
Nasal septal deviation (NSD) is a common pathology encountered in otolaryngology practice. While many cases are asymptomatic, NSD can result in abnormal nasal airflow and increased nasal airway resistance ultimately leading to nasal airway obstruction (NAO) symptoms. NAO symptoms, in turn, can be associated with a significant impact on patient quality of life and resultant patient morbidity.
In the event that NSD symptoms remain refractory to medical management, surgical intervention is commonly recommended 1 with more than 260,000 septoplasties performed in the United States annually.2,3 Inferior turbinate reduction (ITR) is commonly performed as a concomitant procedure given the underlying inferior turbinate hypertrophy that emerges as a compensatory response.4,5
A variety of septoplasty techniques have been described in the literature,6-8 and most involve resection of the involved quadrangular cartilage and bony septum; however, there is increasing interest among nasal surgeons to preserve septal cartilage when possible. L-strut preservation, in particular, is imperative to maintain structural integrity and to avoid complications such as saddle nose deformity.8,9 At the same time, studies have shown that anterior caudal deviations in the region of the nasal valve result in a disproportionate increase in nasal airway resistance. 10 The surgical challenge is to maintain balance between the correction of the problem and the maintenance of structural integrity. As a result, these cases are commonly referred to a subspecialist with training in facial plastic and reconstructive surgery. 11
The purpose of this first-in-human feasibility study was to gain initial clinical experience with a novel, minimally-invasive device that enables mechanical correction of cartilaginous NSD (including L-strut deviations) without cartilage resection using a bioabsorbable implant in either the office or operating room (OR) setting. The SeptAlignTM device (Spirair, South San Francisco, CA) is cleared by the US Food and Drug Administration for mechanical correction of minor cartilaginous NSD. The use of the implant does not require removal of cartilage or bone.
We report 12 month results from this preliminary study of 14 patients who received a SeptAlignTM implant and concomitant ITR. The potential benefits of this minimally-invasive approach include the correction of cartilaginous NSD (including anterior caudal deviations) without cartilage resection, potentially reducing surgical complications, shortening recovery time, and facilitating office-based treatment with local anesthesia. This study evaluated the tolerability, safety, and feasibility of this approach and the extent to which correction of cartilaginous NSD with ITR results in clinically-significant improvement in NAO symptoms.
Patients and Methods
Patients and Eligibility
This clinical trial was conducted at 2 centers in the United States. The study began in July 2022 and enrollment closed in November 2023. Eligible participants included adults ≥21 years of age, with noncalcified, mobile cartilaginous type I, II, or III (Mladina scheme) 12 NSD and a Nasal Obstruction Symptom Evaluation (NOSE) 13 score ≥30. Participants were excluded if they had prior septoplasty or rhinoplasty, other nasal surgeries within the past 6 months, or pathology, other than turbinate hypertrophy, that may be the primary contributor to airway obstruction.
Complete a priori inclusion and exclusion criteria are included in the Appendix. A post hoc inclusion criterion was undergoing concomitant ITR to ensure that all study participants received the same interventions.
Study Design
This was a prospective, feasibility, pilot study to evaluate the safety, tolerability, and efficacy of the SeptAlignTM implant in adults with symptomatic mobile cartilaginous NSD. Following informed consent, participants were assigned to 1 of the 2 cohorts. Participants in Cohort 1 had the implant placed in an OR and received general anesthesia and intravenous antibiotics per standard OR procedures. Once safety was demonstrated in Cohort 1, enrollment was opened for Cohort 2 and participants in this cohort had the implant placed in the medical office setting under local anesthesia. In both cohorts, implantation was performed by qualified otolaryngologists on study day 0, along with ITR.
All participants were followed up for safety and efficacy after implantation (days 1, 3; week 2; and months 1, 3, 6, 12) with follow-up ongoing.
The study was approved by an Institutional Review Board (Advarra, Inc (IRB#00000971/FWA00023875)) and is registered on clinicaltrials.gov (NCT05967169). Informed consent was obtained from all participants. The study was conducted according to the guidelines of Good Clinical Practice as established by the International Conference on Harmonization. 14
Outcome Measures
Safety was assessed by the investigator by way of nasal examination, and participant reported adverse events (AEs) associated with the procedure or the implant.
Procedure tolerability was assessed by participants’ reported pain/discomfort immediately prior to discharge using a numeric rating scale (NRS) from 0 (“no pain”) to 10 (“worst possible pain”). Additionally, office-based Cohort 2 participants were asked to rate the tolerability of the procedure on a scale from 1 (“mild”) to 5 (“not tolerable”).
Ongoing tolerability was assessed by participants’ responses to the question, “Is there any residual (ongoing) discomfort from the procedure?,” where 0 = “none” and 5 = “not tolerable.”
Efficacy was assessed by participants’ responses to the NOSE scale and the Sino-Nasal Outcome Test-22 (SNOT-22), both validated patient reported outcomes tools. The NOSE scale describes participant perception of NAO on a scale from 0 to 100 categorized into 4 categories: mild (5-25 points), moderate (30-50 points), severe (55-75 points), and extreme (80-100 points). A NOSE responder is defined as participants who had either ≥1 category reduction or >20% score reduction at follow-up. 15 The SNOT-22 includes 22 items, and ranges from 0 to 110, with higher scores indicating worse symptoms. Responders to SNOT-22 were defined as participants who experienced a decrease of at least 8.9 points, the minimal clinically-important difference. 16
Implant Device
The SeptAlignTM system consists of a bioabsorbable implant made from poly(dioxanone) (Figure 1), which is preloaded into a single-use delivery device (Figure 2).
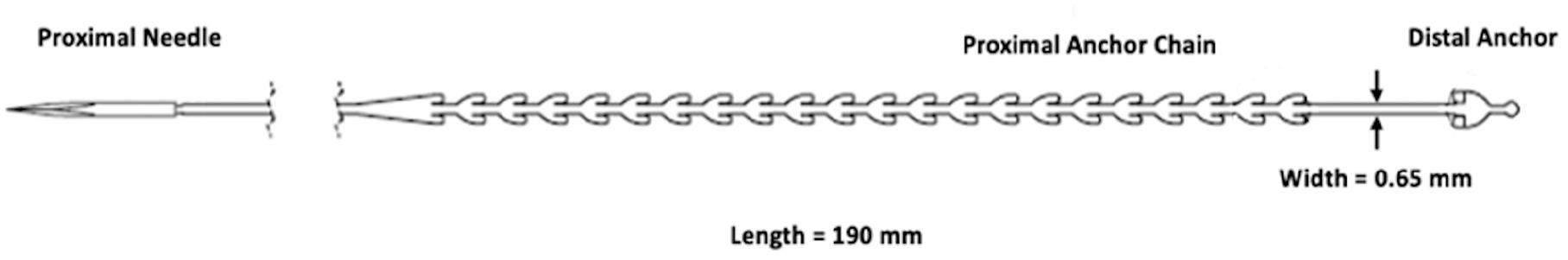
Bioabsorbable implant. Polydioxanone implant with bidirectional anchors (distal and proximal) designed to hold septal cartilage under tension during remodeling. Excess implant length, including pre-attached proximal needle used for puncturing cartilage and unused proximal anchors, is trimmed off after placement.
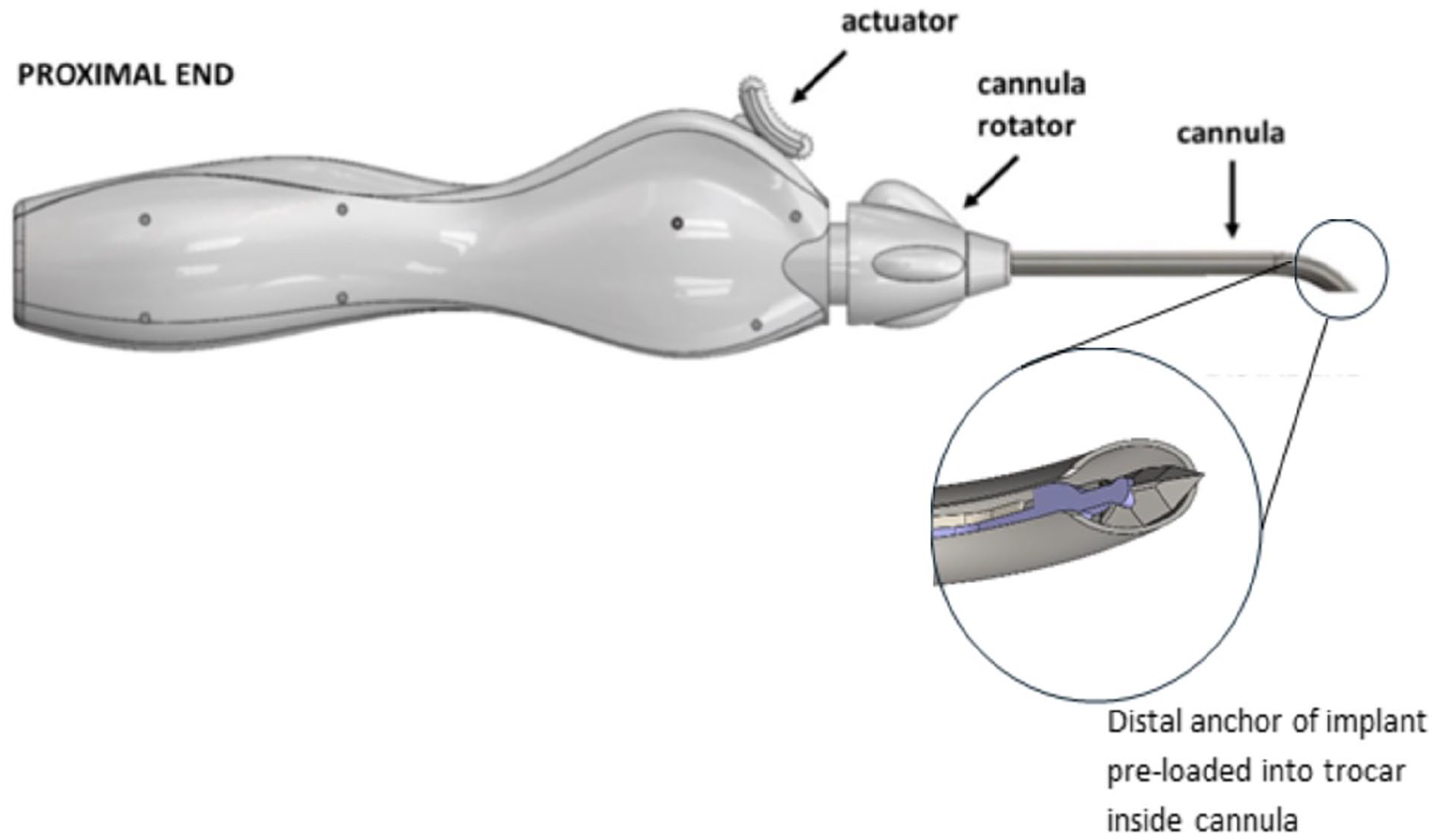
Delivery device. Single-use delivery device includes a rotating cannula, which houses the trocar and preloaded implant. Actuator slides proximally to release trocar and deliver the distal anchor of the implant.
Surgical Technique
To place the implant, the #15 blade is used to make a hemi-transfixion or Killian incision on the convex side of the deviation. A Cottle elevator is used to separate the mucoperichondrium from the septal cartilage sufficiently to insert the delivery device cannula submucosally along the convex side of the deviation. The surgeon then positions the cannula tip at the distal end of the deviation and activates the device. A sharp trocar punctures the cartilage, pushing the implant’s distal anchor through the septum, atop the mucosa on the contralateral side of the septum. The delivery device is then removed, unsheathing the implant in the process. The implant has a pre-attached proximal needle, which the surgeon drives through the cartilage proximal to the septal deviation and enables the implant to be pulled through the cartilage to the contralateral side. As the implant is pulled through the cartilage, small proximal anchors secure it in place. A Freer or blunt surgical tool is used to deflect the cartilage medially to the desired degree of correction as the implant is pulled flat against the cartilage. Once in place, the excess portion of implant, including the proximal needle, is trimmed leaving a proximal anchor on the contralateral side. The mucosa is then reapproximated (Figure 3). Once in place, the implant holds the straightened portion of the septal cartilage in tension while the cartilage remodels (usually over 5-10 weeks). Resorption of the implant begins around 10 weeks and is completed within 6 months.17-19 Of note, bone resection was not performed.
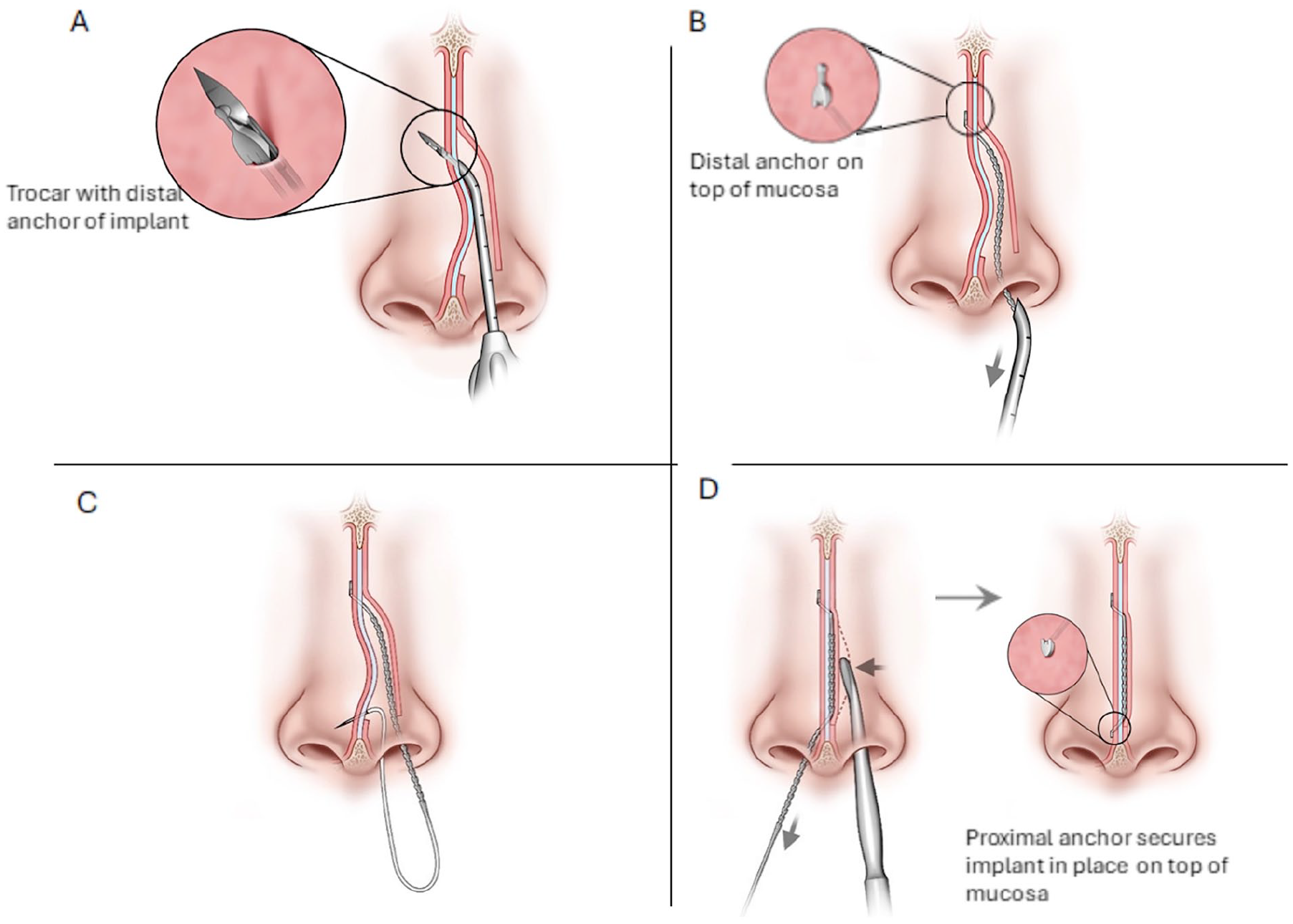
Implant placement. (A) After hemi-transfixion or Killian incision and elevation of the mucoperichondrium, the delivery device cannula is inserted submucosally along convex side of the deviation. A sharp trocar pushes distal anchor of implant through the septum. (B) Trocar is retracted into cannula and delivery device is removed from nose unsheathing the implant body. (C) The implant needle punctures the septum proximal to deviation, and the implant body is pulled through the septum. (D) The cartilage is deflected medially to the desired degree of correction via Freer as the implant is pulled flat against the cartilage engaging the proximal anchors. Once in place, implant is trimmed. The mucosa is then reapproximated.
Turbinate Reduction
In the OR, turbinate reduction was performed by submucosal resection. In the office setting, turbinate reduction was performed by radiofrequency reduction.
Statistical Analysis
No power calculations were performed for this first-in-human feasibility study. Two-sided Wilcoxon signed-rank tests were used to compare median NOSE and SNOT-22 scores over time. There were no missing data.
Results and Conclusions
Participants
Seventeen participants were enrolled in the trial. For this analysis, we report on 14 participants who (a) had the implant concomitantly with ITR and (b) completed the study through the 12 month time point. Three participants were excluded from this subgroup: 1 participant did not undergo turbinate reduction and was excluded from analyses based on the post hoc inclusion criteria, 1 participant was discontinued due to lack of successful implantation after 3 attempts (per protocol), and a third participant unintentionally removed the implant 12 days post-procedure. Among the 14 participants in this subgroup, 10 were enrolled into Cohort 1 (performed in the OR), and 4 were enrolled into Cohort 2 (performed in the medical office).
Table 1 shows the demographics and baseline characteristics of the participants. Twelve out of the 14 participants were determined to have caudal septal deviation by investigator evaluation of baseline coronal Computed Tomography (CT) scans.
Demographics and Baseline Characteristics.
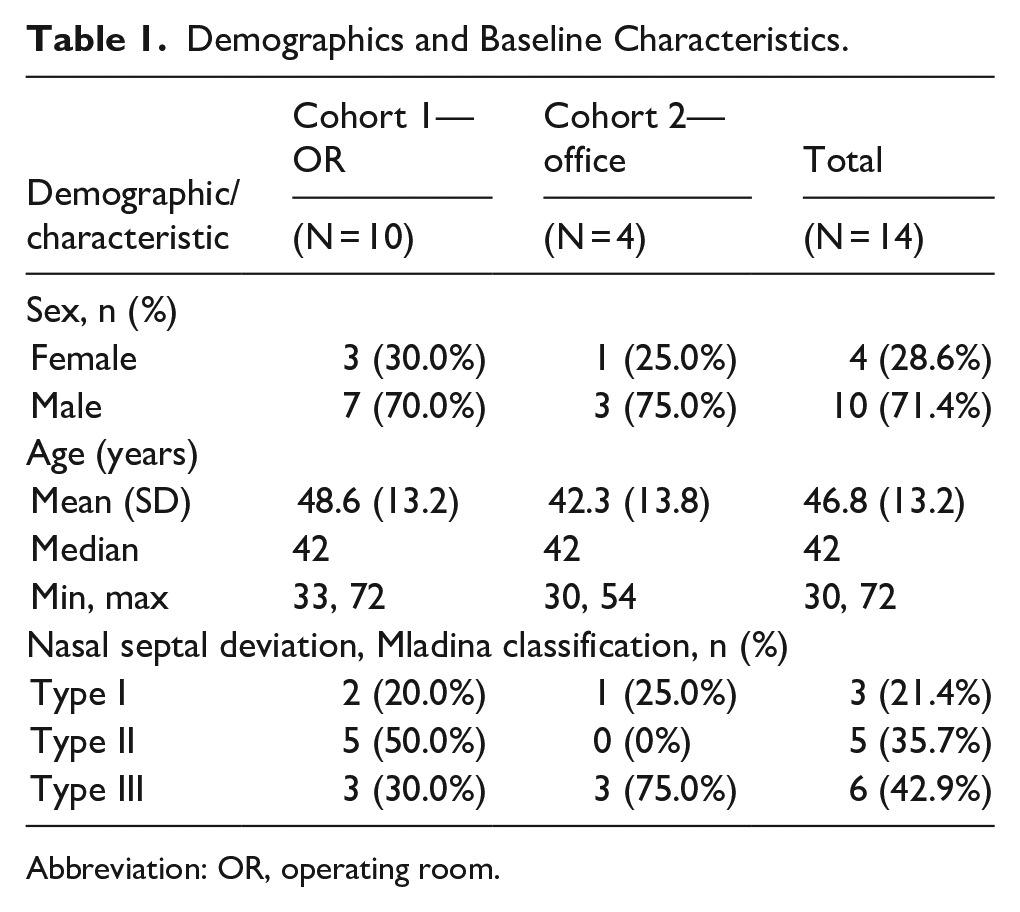
Abbreviation: OR, operating room.
No AEs occurred during the implant procedure and no serious AEs (SAEs) or unanticipated AEs occurred during the study. There were 3 AEs reported by 3 participants; all were mild. Two participants reported headaches (days 48 and 57 post-procedure) deemed unlikely related to the procedure or the implant. A third participant experienced 2 small septal perforations at the locations of the distal and proximal anchors with onset 84 days after the implant procedure. The implant placement occurred in the setting of a major protocol deviation as the investigator placed 2 implants with 1 implant on top of the mucosa rather than submucosally. The AE was mild and deemed related to the procedure.
The procedure was well-tolerated. The mean (standard deviation) NRS pain score (0-10) post-procedure and predischarge for the 14 participants reported herein was 3.64 (SD 3.15). Only the office-based cohort (n = 4) was asked to rate the tolerability of the procedure immediately following the procedure; 1 (25%) participant reported mild or minimal discomfort and 3 (75%) moderate discomfort. All 4 participants (100%) indicated that the injection of local anesthesia was the least tolerable aspect of the procedure.
The majority of participants (12/14) reported none to mild residual discomfort at day 3 following the implant procedure. At the 6th and 12th month following the implant procedure, 13 of 14 participants reported no discomfort. One participant, who was a nonresponder, reported persistent discomfort associated with residual NAO symptoms.
NOSE and SNOT-22
Figures 4 and 5, respectively, show a sustained reduction in NOSE and SNOT-22 median scores over time. The NOSE score decreased by a median of 35.0 points from baseline (57.5) at month 1, corresponding to a 60.0% reduction. This improvement in NOSE score was maintained at the 3rd, 6th, and 12th month with a reduction of 35.0 points (72.5%), 40.0 points (75.3%), and 35.0 points (71.8%), respectively. Reduction in NOSE score was statistically significant across all time points (P ≤ .001). Similarly, the month 1 SNOT-22 score decreased by a median 26.5 points from baseline (37.0), corresponding to a 67.9% reduction. This statistically-significant improvement in SNOT-22 score was also sustained at the 3rd, 6th, and 12th month with a reduction from baseline of 29.8 points (76.9%), 24.5 points (62.8%), and 23.5 points (60.3%), respectively (P ≤ .004).
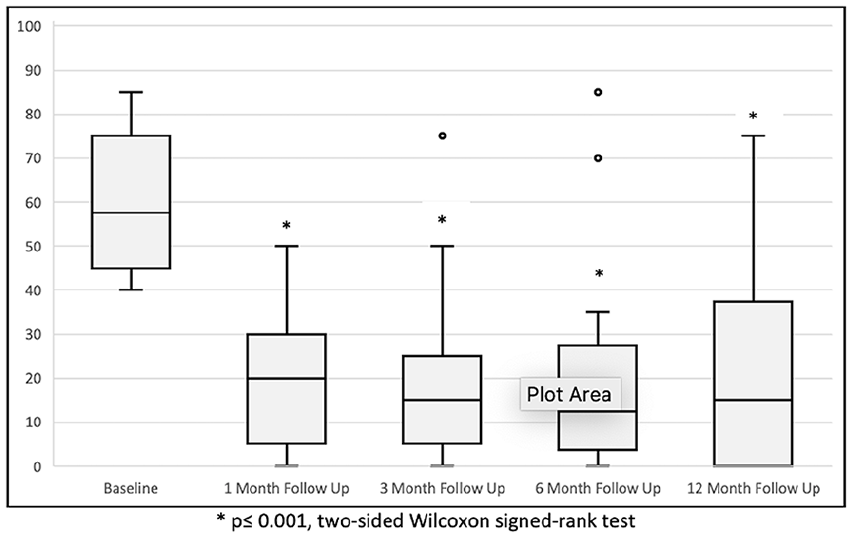
NOSE scores over time. The horizontal line through the middle of each box represents the median. The box contains the 25th to 75th percentile of the dataset. The vertical lines from the lower and upper bounds of the box mark the 5th and 95th percentile. Dots indicate outliers.
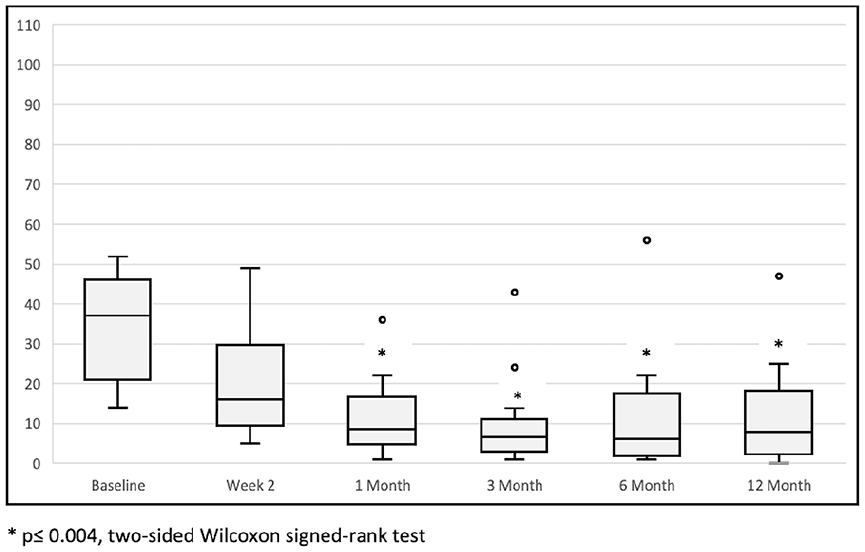
SNOT-22 scores over time. The horizontal line through the middle of each box represents the median. The box contains the 25th to 75th percentile of the dataset. The vertical lines from the lower and upper bounds of the box mark the 5th and 95th percentile. Dots indicate outliers.
At the 1st month visit, 13 of 14 (92.9%) participants were NOSE responders (95% CI: 66.1%-99.8%). Similarly, 11 of 14 (78.6%) participants were SNOT-22 responders (95% CI: 49.2%-95.3%). This was sustained through 12 months.
Discussion
This first-in-human feasibility study demonstrates the potential of the SeptAlignTM device to provide a minimally-invasive, cartilage-preserving solution for correcting cartilaginous NSD, including deviations involving the caudal septum. Additionally, this study shows that the SeptAlignTM device can be used in the office setting under local anesthesia for surgeons who offer office-based procedures. These results show early promise regarding the device’s safety, tolerability, efficacy, and durability profiles when used in conjunction with ITR.
The study’s primary outcomes, measured by reductions in NOSE and SNOT-22 scores, underscore the potential benefit of the SeptAlignTM device when combined with ITR. The results of this study also demonstrate the durability of the treatment at the 12th month as well as the comparability of treatment response relative to traditional septoplasty techniques. Median NOSE scores showed a sustained reduction of 35.0 (71.8%) through 12 months. Given the implant is known to be resorbed by 6 months, stable 12 month NOSE scores are indicative of a durable response. Moreover, the observed improvement in NOSE scores is comparable to that reported in studies looking at traditional septoplasty combined with concomitant ITR. For example, Samarei and Mabarian 4 noted a 73% improvement in the NOSE score at the 12th month.
Importantly, these results were achieved with the use of a device that allows for NSD correction without cartilage or bone resection. Cartilage-sparing techniques typically result in improvements in patient recovery, complications, and long-term structural integrity. 20 In addition, cartilage preservation can help improve the outcome of future revision surgery, should it be necessary.
The ability to address caudal septal deviations is also a significant advantage of the SeptAlignTM device. Deviations in this region are known to be associated with increased nasal airway resistance, 10 and are more likely to be symptomatic. Moreover, literature has demonstrated that up to 72% of revision septoplasties involve the caudal region. 21 Given such a high prevalence of caudal deviations among revision cases, it stands to reason that aggressive upfront caudal correction could contribute to reduced rates of revision. To this point, other authors have noted that careful attention to the caudal region at the time of primary surgery may be critical to reducing the rate of treatment failure and the need for revision surgery. 22
The safety profile of the SeptAlignTM device was favorable, with no SAEs reported. AEs were mild and occurred in the context of a protocol deviation or were not deemed related to the device or procedure. Importantly, no AE resulted in long-term morbidity or required surgical intervention. This suggests that with appropriate training and adherence to protocol, the device can be safely utilized.
This study is not without limitations. As is expected of early-feasibility studies, the number of participants enrolled is small and there is no control group. Also, all subjects analyzed had concomitant ITR because the goal of the study was to reflect the general standard of care; however, this may confound overall interpretation of the data. Additionally, the study was completed at only 2 sites, which may limit the generalizability of the observed results. Finally, while the anecdotal experience of the investigators noted faster patient recovery, metrics such as return-to-work were not included to formally assess the impact of this minimally-invasive treatment. A larger prospective, multicenter, open-label study is currently underway (NCT06163404), and this study will address many of the limitations of the current study.
The findings of this study suggest that the SeptAlignTM implant could be an appropriate tool used in the treatment paradigm of patient’s with NSD, particularly those who may not desire traditional septoplasty. Additionally, the implant may offer several potential advantages over traditional septoplasty. First, it eliminates the need for cartilage resection or grafting, preserving the structural integrity of the septal cartilage while enabling the correction of what can otherwise be challenging deviations in the L-strut region. Second, the minimally-invasive nature of the procedure may translate to a reduction in postoperative complications and shorter recovery times, which could lead to fewer disruptions to daily life. Third, the ability to perform the procedure in an office setting reduces the need for general anesthesia and its associated risks and may increase accessibility for patients.
Footnotes
Appendix
Inclusion and Exclusion Criteria.
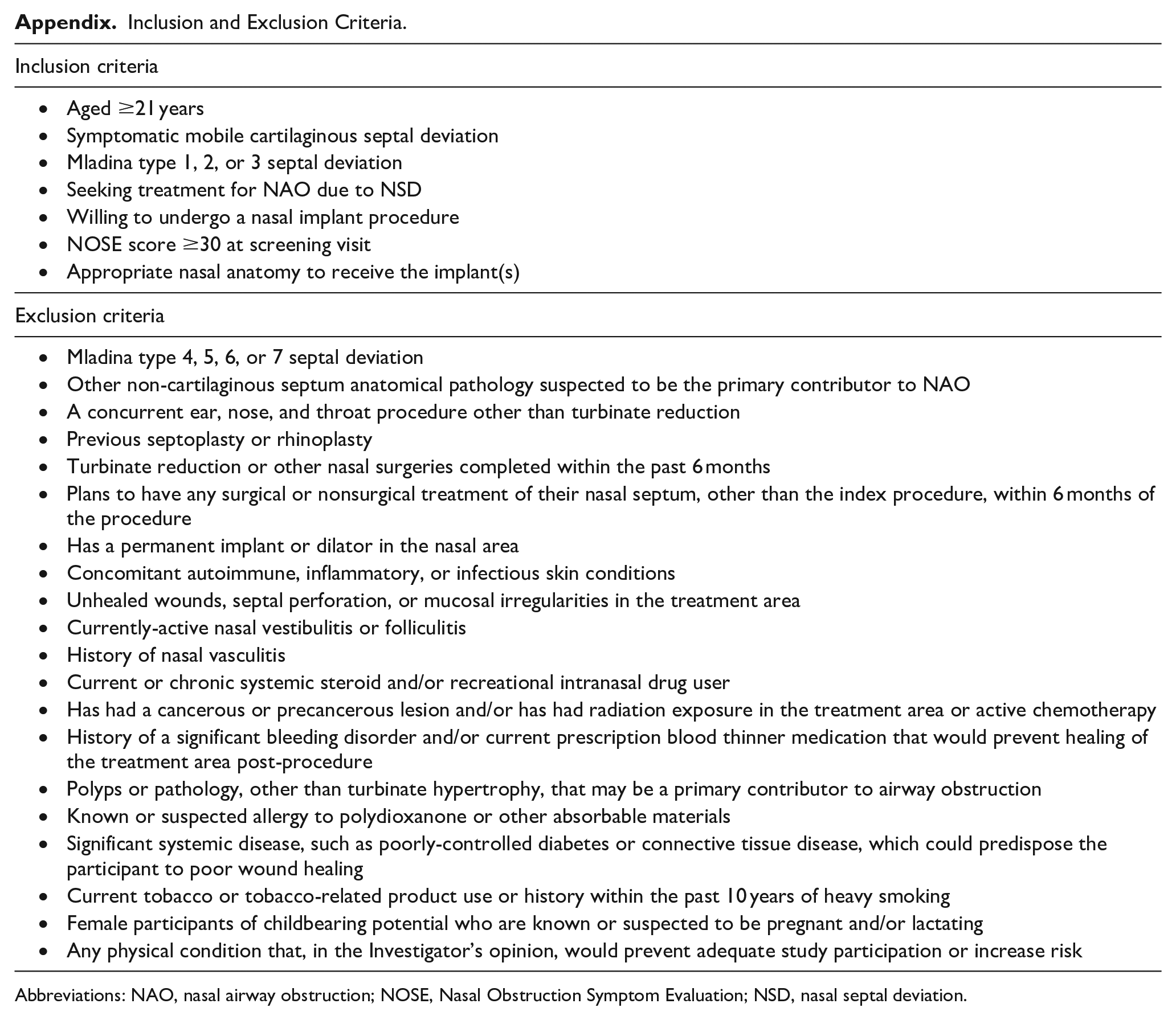
| Inclusion criteria |
|---|
| • Aged ≥21 years |
| • Symptomatic mobile cartilaginous septal deviation |
| • Mladina type 1, 2, or 3 septal deviation |
| • Seeking treatment for NAO due to NSD |
| • Willing to undergo a nasal implant procedure |
| • NOSE score ≥30 at screening visit |
| • Appropriate nasal anatomy to receive the implant(s) |
| Exclusion criteria |
| • Mladina type 4, 5, 6, or 7 septal deviation |
| • Other non-cartilaginous septum anatomical pathology suspected to be the primary contributor to NAO |
| • A concurrent ear, nose, and throat procedure other than turbinate reduction |
| • Previous septoplasty or rhinoplasty |
| • Turbinate reduction or other nasal surgeries completed within the past 6 months |
| • Plans to have any surgical or nonsurgical treatment of their nasal septum, other than the index procedure, within 6 months of the procedure |
| • Has a permanent implant or dilator in the nasal area |
| • Concomitant autoimmune, inflammatory, or infectious skin conditions |
| • Unhealed wounds, septal perforation, or mucosal irregularities in the treatment area |
| • Currently-active nasal vestibulitis or folliculitis |
| • History of nasal vasculitis |
| • Current or chronic systemic steroid and/or recreational intranasal drug user |
| • Has had a cancerous or precancerous lesion and/or has had radiation exposure in the treatment area or active chemotherapy |
| • History of a significant bleeding disorder and/or current prescription blood thinner medication that would prevent healing of the treatment area post-procedure |
| • Polyps or pathology, other than turbinate hypertrophy, that may be a primary contributor to airway obstruction |
| • Known or suspected allergy to polydioxanone or other absorbable materials |
| • Significant systemic disease, such as poorly-controlled diabetes or connective tissue disease, which could predispose the participant to poor wound healing |
| • Current tobacco or tobacco-related product use or history within the past 10 years of heavy smoking |
| • Female participants of childbearing potential who are known or suspected to be pregnant and/or lactating |
| • Any physical condition that, in the Investigator’s opinion, would prevent adequate study participation or increase risk |
Abbreviations: NAO, nasal airway obstruction; NOSE, Nasal Obstruction Symptom Evaluation; NSD, nasal septal deviation.
Acknowledgements
We thank Amy Morris and Amy Bronstone for assistance with manuscript writing.
Ethical Considerations
This study was approved by an Institutional Review Board (Advarra, Inc) on April 27, 2022.
Consent to Participate
Written informed consent was obtained from all participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study and for medical writing services was provided by Spirair, Inc.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.E.D. is a paid consultant for Spirair, Inc, Aerin Medical, Medtronic, Neurent Medical, and Regeneron. G.E.D. is on the Advisory Boards of Medtronic, Neurent Medical, Optinose, and ChitoGel. G.E.D. has received research funds from Neurent Medical. S.E.D. is a paid consultant for Spirair, Inc, Medtronic, and Lyra Therapeutics.
Submission Statement
Submission was performed by a third-party (eg, Tracey Henry, an employee of Spirair, Inc, performed data entry into electronic portal) with authorization and consent of all contributing authors; listed authors have approved all statements and declarations.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
