Abstract
Objective:
To compare the outcomes of endoscopic sinus surgery (ESS) and dupilumab using nasal nitric oxide (nNO) as a biomarker of mucosal health, Lund-Kennedy endoscopy score (LKES), and Sino-Nasal Outcome Test-22 (SNOT-22).
Methods:
A prospective observational cohort of chronic rhinosinusitis with nasal polyposis (CRSwNP) patients who underwent ESS followed by standard postoperative therapy was compared with another prospective cohort of CRSwNP patients who received dupilumab. In addition to baseline characteristics, nNO production, LKES, and SNOT-22 levels were compared between the 2 cohorts at the 1st month and 6th month posttreatment.
Results:
A total of 28 and 37 patients were included in the ESS cohort and the dupilumab group, respectively. At the 1stmonth and 6th month posttreatment, nNO levels were comparable between 2 groups. Compared to baseline, the ESS cohort showed increased nNO levels at 1st month posttreatment, while the dupilumab group did not. At the 6th month posttreatment, both groups exhibited a significant increase in nNO levels. Similar trend was observed in LKES. SNOT-22 decreased at the 1st month and 6th month posttreatment compared with that in baseline.
Conclusion:
ESS and dupilumab confer comparable benefits in terms of nNO changes, symptoms, and endoscopy findings at the 6th month posttreatment. ESS may result in more rapid improvement in the outcomes than in dupilumab as evidenced by the 1 month posttreatment values.
Introduction
Chronic rhinosinusitis (CRS) is a heterogeneous inflammatory disease of paranasal sinuses and affects 10% to 15% of the population.1,2 Chronic rhinosinusitis with nasal polyposis (CRSwNP) is a subtype of CRS characterized by type 2 inflammation and can be therapeutically challenging especially in patients with comorbid asthma, aspirin-exacerbated respiratory disease (AERD), and allergic fungal sinusitis.3-5 The conventional treatment pathway of CRSwNP includes standard medical therapy (topical corticosteroids and oral corticosteroids) and endoscopic sinus surgery (ESS) to further facilitate the delivery of medical therapy.4,5 Since the U.S. Food and Drug Administration approval in 2019, dupilumab has emerged as a promising novel biologic therapy for CRSwNP, as it targets interleukin (IL)-4-mediated and IL-13-mediated pathways to subdue type 2 inflammation.6-8 Phase-3 clinical trials have showed favorable sino-nasal outcomes with improved patient-reported quality of life, nasal polyp score, and smell.6,8
Nasal nitric oxide (nNO) is a useful tool to monitor sinus mucosal health and response to therapy as shown in previous studies, as it is produced constitutively by the mucosa of the paranasal sinuses via the actions of inducible nitric oxide (NO) synthase.9-16 Physiologically, the role of nNO has been hypothesized to maintain health and sterility of paranasal sinuses via mucociliary regulation, antimicrobial activity, airbone messaging, and modifications to the inflammatory pathway.12,15-17 It has been shown to correlate well with endoscopic and radiological markers of disease severity.9-14 In addition, nNO has been shown to be decreased in eosinophilic CRS compared with that in non-eosinophilic CRS. 14 In other words, a Th-2 polarized inflammatory environment such as CRSwNP would lead to lower nNO levels.10,14 Although a recent study compared the clinical outcomes between ESS and dupilumab using extracted data from a prospective cohort study and clinical trials, 18 there has not been a comparative study using type 2 inflammatory biomarkers such as nNO. Therefore, the objective of our study was to extract and compare data from 2 real-world studies of patients with CRSwNP undergoing ESS and dupilumab treatment with nNO as a primary outcome measure with secondary clinical outcomes of the Sino-Nasal Outcome Test-22 (SNOT-22) and the clinician-derived Lund-Kennedy endoscopy score (LKES).
Methods
Study Design and Patient Population
Approval of this study was granted by the Research Ethics Board at St. Michael’s Hospital, Toronto, Canada (#12-202), and at Humanitas Research Hospital, Milan, Italy (#59/20).
Adult study participants were prospectively enrolled into an observational cohort study evaluating treatment outcomes following ESS (Toronto, Canada) between January 2013 and January 2015 and dupilumab (Milan, Italy) between March 2021 and January 2022. Results from the individual cohorts have been previously published.10,19 Patients with CRSwNP were enrolled. Exclusion criteria included patients with unilateral disease, cystic fibrosis, primary ciliary dyskinesia, or immune deficiency. The diagnosis of CRSwNP was made according to the American Academy of Otolaryngology – Head and Neck Surgery Guidelines.20,21
The patients in the ESS cohort underwent complete ESS by a senior author (J.M.L.) with bilateral maxillary antrostomy, total ethmoidectomy, sphenoidotomy, and frontal sinusotomy (at least Draf2a) after having failed appropriate medical therapy. Postoperatively, all patients were started on 2 week course of oral antibiotics with a short course of oral steroids and were maintained on saline irrigations and intranasal steroids.
The patients in the dupilumab cohort were treated with dupilumab after having failed appropriate medical therapy and/or ESS. For patients who failed surgery, they were started on dupilumab at least 12 months after the last surgery. All patients received dupilumab 300 mg subcutaneously every 15 days, apart from those with concomitant severe asthma that received a loading dose of 600 mg followed by 300 mg every 15 days.
Outcome Measures
The primary outcome measure was nNO levels measured pretreatment, at the 1stmonth and 6th month posttreatment. In the ESS cohort, nNO measurements were performed with a nasal NO analyzer (Eco Medics CLD 88sp NO analyzer; Eco Physics Inc, Ann Arbor, MI, USA). Each nostril was measured independently at a fixed flow rate of 300 mL/minute. This was in accordance with the American Thoracic Society.22,23 In the dupilumab cohort, nNO measurements were performed with an electrochemical analyzer (FEN0+; MediSoft, Sorinnes, Belgium) in accordance with European Respiratory Society technical standard.24,25 Each nostril was measured independently at a fixed flow rate of 350 mL/minute. In both cohorts, nNO values were recorded after a steady-state plateau was achieved and the mean value was recorded. Due to differences in the sample gas flow rate, nNO concentration values were standardized as nNO production (nL/minute), which is the product of nNO concentration in parts per billion and flow rate in liters per minute. 26
The secondary outcome measures included SNOT-22 and LKES at each aforementioned time point. The clinician performing endoscopic examination was blinded to nNO measurements at each visit. Baseline CT scans were reviewed and scored with Lund-Mackay score (LMS).
Statistical Analysis
Demographic and clinical characteristics were summarized descriptively by reporting the median and range or interquartile range (IQR) for continuous variables and the frequency and proportion for categorical variables. Differences in characteristics between 2 comparison groups were compared using the Mann-Whitney test for continuous variables and the chi-squared test for categorical variables. The Friedman test and Dunn’s multiple comparisons test were used for comparison of means at each time point. Spearman correlations were used to examine potential relationships between nNO and the clinical parameters of LMS, SNOT-22, and LKES. All analyses were carried out using SPSS Statistics (IBM SPSS Statistics Version 28; IBM Corp, Armonk, NY, USA), and statistical significance was set at an alpha level of .05.
Results
Study Population and Baseline Characteristics
There were 28 patients in the ESS cohort and 37 patients in the dupilumab cohort. Baseline characteristics are summarized in Table 1. The mean age and proportion of each sex were comparable between the 2 groups. In the dupilumab group, there were significantly-more patients with AERD (48.6% vs 17.9%, P = .010) and previous ESS (91.9% vs 60.7%, P = .002). The patients had comparable LMS at baseline between the ESS and the dupilumab group (18.5 [IQR 16.0-22.0] vs 21.0 [IQR 18.5-22.3], P = .19).
Baseline Demographics.
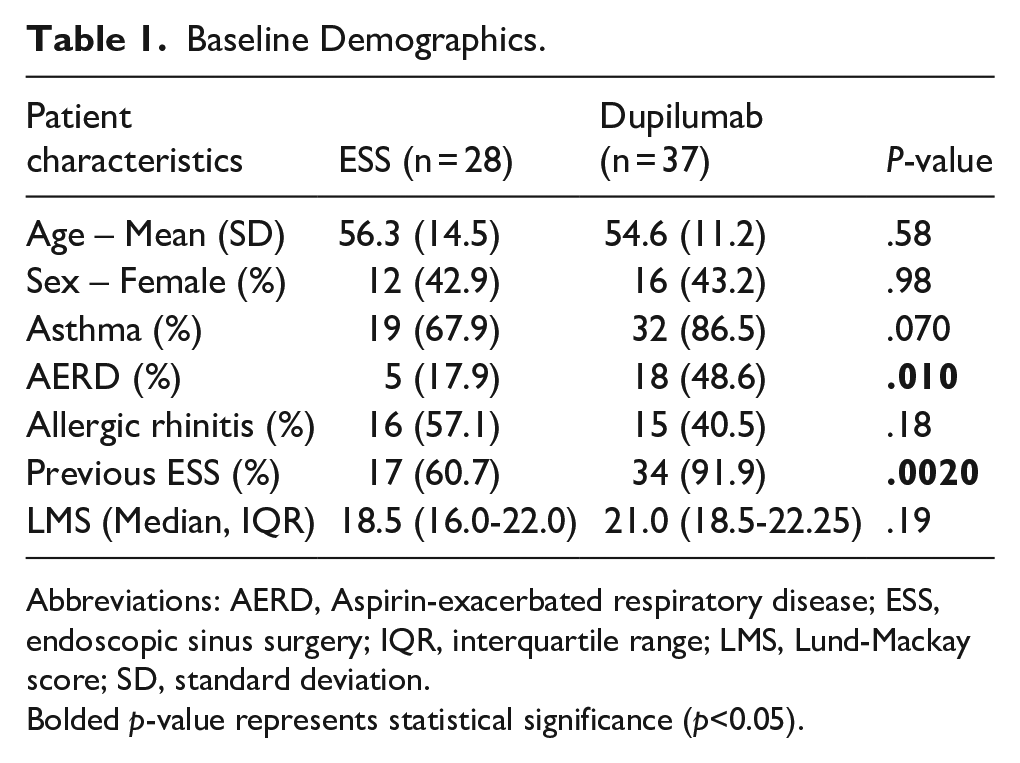
Abbreviations: AERD, Aspirin-exacerbated respiratory disease; ESS, endoscopic sinus surgery; IQR, interquartile range; LMS, Lund-Mackay score; SD, standard deviation.
Bolded p-value represents statistical significance (p<0.05).
nNO Measurements
At baseline, the dupilumab group exhibited higher nNO values (in nL/minute) than the ESS group (139.3 [IQR 56.2-242.2] vs 80.8 [IQR 17.3-134.6], P = .032). At the 1st month and 6th month posttreatment, nNO values were comparable (Table 2, Figure 1). At the 1st month posttreatment, the ESS cohort demonstrated a significant increase in nNO levels from baseline (174.3 [IQR 90.2-264.7] vs 80.8 [IQR 17.3-134.6), P < .001) while the dupilumab did not (197.9 [IQR 130.6-289.4] vs 139.3 [IQR 56.2-242.2], P = .42). At the 6th month posttreatment, both cohorts demonstrated a significant increase in nNO levels from baseline (P = .002 for surgery, P = .003 for dupilumab). There was no statistical difference in nNO levels between the 1st month and 6th month posttreatment times for both cohorts.
Outcome measures for the ESS and dupilumab cohorts.
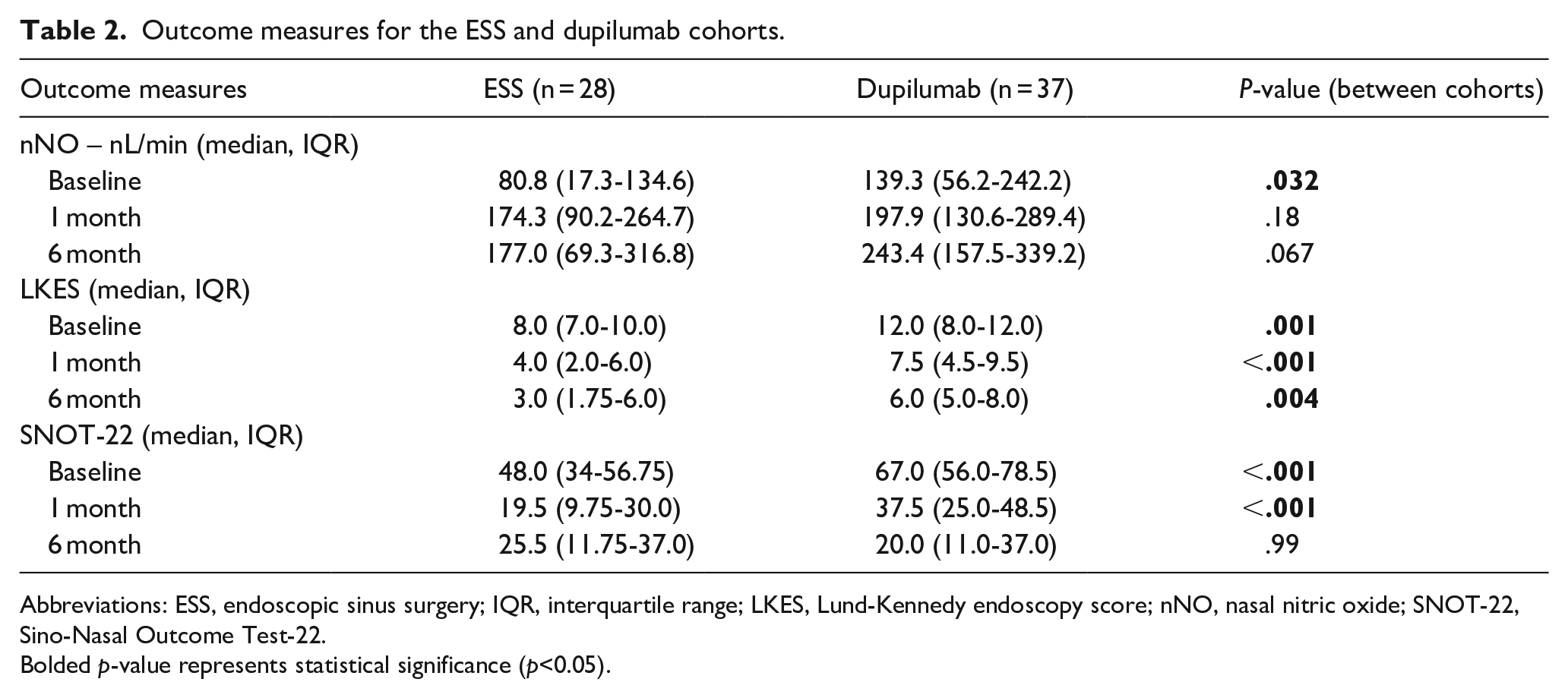
Abbreviations: ESS, endoscopic sinus surgery; IQR, interquartile range; LKES, Lund-Kennedy endoscopy score; nNO, nasal nitric oxide; SNOT-22, Sino-Nasal Outcome Test-22.
Bolded p-value represents statistical significance (p<0.05).
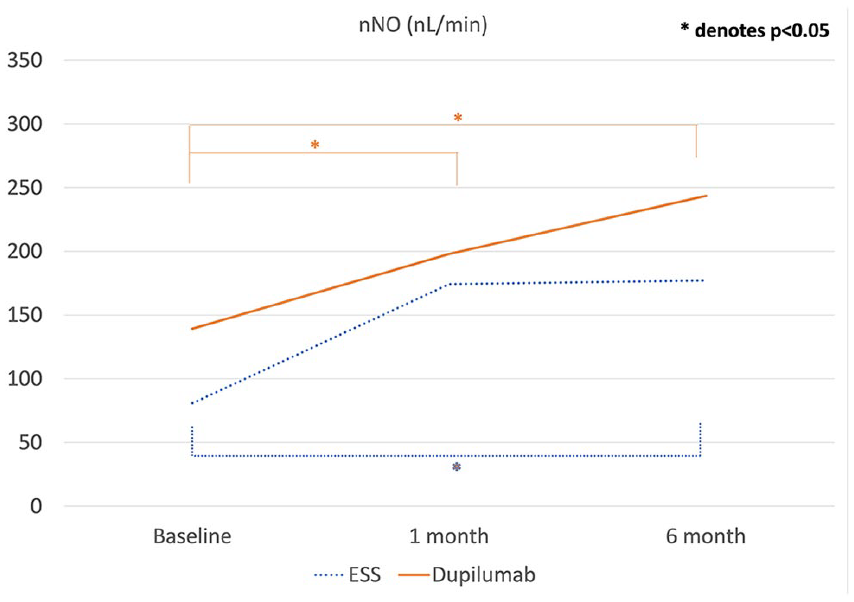
nNO trends in the ESS and dupilumab cohorts. nNO, nasal nitric oxide; ESS, endoscopic sinus surgery.
Clinical Outcomes (LKES and SNOT-22) and Relationships Among Measurements
LKES and SNOT-22 values are summarized in Table 2 and Figure 2. Similar to nNO, at the 1st month posttreatment, LKES demonstrated improvement in the surgical cohort (P < .001) but did not show improvement in the dupilumab cohort (P = .16). At the 6th month posttreatment, both cohorts demonstrated improvement in LKES compared with baseline (P < .001 for surgery, P = .002 for dupilumab). There was no difference in LKES between 1 month and 6 month intervals for both cohorts. While SNOT-22 did not change for ESS between 1 month and 6 month posttreatment, it did significantly improve for dupilumab (P = .043).
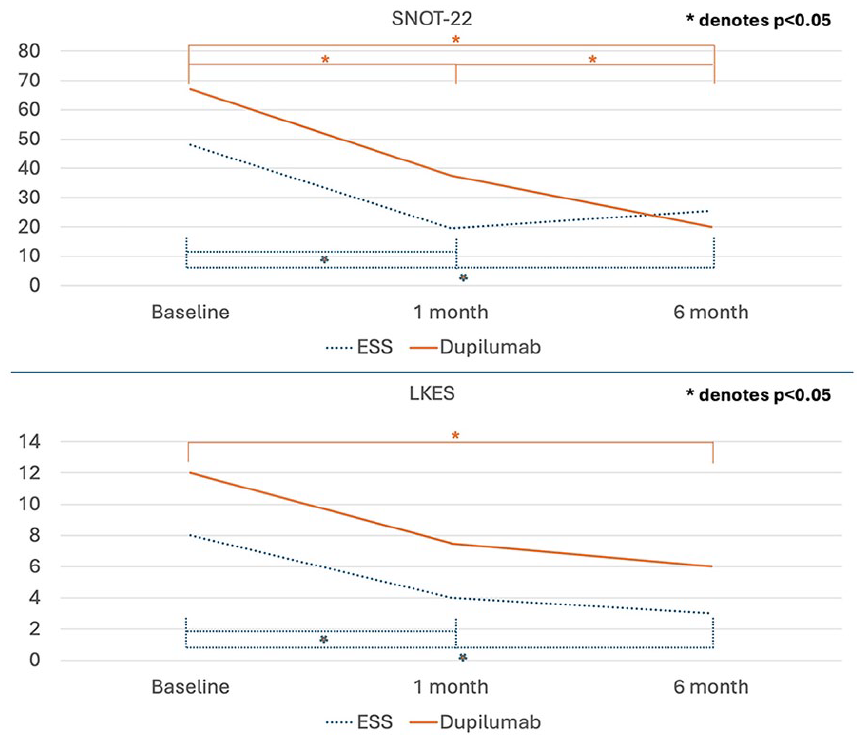
LKES and SNOT-22 trends in the ESS and dupilumab cohorts. LKES, Lund-Kennedy endoscopic score; SNOT-22, Sino-Nasal Outcome Test-22; ESS, endoscopic sinus surgery.
We explored a correlation between nNO and LMS as well as LKES. At baseline, the nNO levels were negatively correlated to LMS (r = −.376, P = .004) and LKES (r = −.286, P = .042). Following the treatment, there was a negative correlation between nNO and LKES at the 1st month (r = −.306, P = .028) and 6th month posttreatment (r = −.435, P = .001). There was no correlation between nNO and SNOT-22 at any time points.
Discussion
To our knowledge, this is the first exploratory study to use nNO as an outcome measure when comparing ESS or dupilumab therapy for CRSwNP. Prior investigations demonstrated the role of nNO as a biomarker of type 2 inflammation, which correlates well with objective markers of disease severity, such as LKES.9-14 In the current study, ESS and dupilumab resulted in comparable outcomes in terms of nNO levels at the 1st month and 6th month posttreatment among patients with CRSwNP.
While absolute values do demonstrate comparable outcomes between 2 treatment modalities, it is important to evaluate the overall trend in nNO flow rates. While both ESS and dupilumab resulted in increase in nNO levels in 6 months, only the ESS cohort experienced a significant increase in nNO levels at the 1st month posttreatment. This was corroborated in endoscopy scores as shown in LKES. This is not surprising given that patients would have had complete removal of nasal polyps after ESS. Biologics, on the other hand, reduce nasal polyp burden to over longer periods of time, as demonstrated in clinical trials.7,8 Interestingly, in the surgical cohort, SNOT-22 was sustained between 1 month and 6 month posttreatment after the initial decrease in SNOT-22 from baseline while SNOT-22 continued to decrease in the dupilumab cohort between 1 month and 6 month posttreatment. Overall, the results of our study indicate that both ESS and dupilumab confer comparable benefits in terms of quality of life, nasal polyp burden, and type 2 inflammation, as demonstrated with SNOT-22, LKES, and nNO flow rate.
In literature, there are 2 other studies that directly compare the outcomes of ESS and biologics.18,27 Dharmarajan et al used a retrospectively-matched cohort design, which showed comparable SNOT-22 outcomes between ESS and dupilumab and superior nasal polyp score for ESS at variable follow-up timepoints. 27 Miglani et al utilized outcomes from a prospective cohort study for ESS and results from clinical trials for biologics. 18 In this study, ESS and dupilumab had comparable SNOT-22 scores at 24 weeks whereas ESS resulted in lower nasal polyp score than dupilumab. These findings are in line with the results from our paper which employed a similar method of extracting data from 2 separate cohorts. Distinctive features in our methodology however include the use of real-world prospectively-collected cohort data for both ESS and dupilumab (as opposed to trial data) and the use of nNO as a biomarker of type 2 inflammation in addition to SNOT-22 and LKES.
In the lower airways, exhaled NO has garnered attention as a biomarker of type 2 inflammation.28,29 The distinction, however, is that in the lower airway, there is a positive correlation between the level of exhaled NO and the level of type 2 inflammation.28,29 In other words, higher levels of exhaled NO are indicative of type 2 inflammation.28,29 In fact, the American Thoracic Society clinical practice guideline recommends the incorporation of exhaled NO to guide, monitor, and calibrate the management of asthma.28,29 Similarly, while nNO demonstrates an inverse relationship with type 2 inflammation, the results of our study demonstrates that nNO has a potential to be used to monitor the treatment responses of those with CRSwNP.
There are limitations in the current study. First, the dupilumab cohort had a higher proportion of AERD patients and those undergoing revision ESS with resultant discrepancies in LKES and SNOT-22. However, it is reassuring that the proportion of patients with comorbid asthma and the baseline LMS are similar between the 2 cohorts. In addition, there is discrepancy in baseline nNO levels between the 2 groups; given the exploratory nature of this study, we focused on the trend of nNO changes. Furthermore, our study lacks results beyond 6 months mark as the initial objectives of data collection did not expand beyond this. Future studies should have comparison with longer terms. Another limitation is the discrepancy between LKES and SNOT-22 scores at baseline. We provide within-cohort analysis to investigate the trend along with between-cohort analysis to provide comprehensive statistics and potentially mitigate this issue. Lastly, there was discrepancy in the method of nNO measurement. We standardized the nNO measurement as nNO production (nL/minute), as recommended by the American Thoracic Society. 26
Conclusion
ESS and dupilumab result in comparable benefits in terms of changes in nNO, symptoms, and endoscopy findings at the 6th month posttreatment. ESS may result in more rapid improvement in the outcomes than dupilumab as evidenced by the 1 month posttreatment values. Further work is needed to establish a long-term comparison between ESS and dupilumab with the use of nNO and other biomarkers in conjunction with other clinical outcomes.
Footnotes
Author Contributions
Study concept and design: D.L., G.P., E.H., J.L. Acquisition of data: D.L., G.P., G.G., R.P. Statistical analysis of data: D.L. Interpretation of data: D.L., G.P., G.G., E.G., R.P., E.H., J.L. Drafting of the manuscript: D.L. Critical revision of the manuscript for important intellectual content: D.L., G.P., G.G., E.G., R.P., E.H., J.L. Administrative, technical or material support: D.L., G.P., E.H., J.L. Study supervision: E.H., J.L. All authors have given their agreement for this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Harry Barberian Scholarship (Toronto, Canada), The Dunin Foundation (Toronto, Canada), Chiesi Italia (Milan, Italy).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Daniel Lee is on the advisory board for Sanofi Regeneron and received fees for speaker activities/honorarium from Sanofi Regeneron and GSK. Giovanni Paoletti received fees for speaker activities and/or advisory board participation from Novartis, Lusofarma, outside of the submitted work. Enrico Heffler received fees for speaker activities and/or advisory board participation from Sanofi, Regeneron, GSK, Astrazeneca, Novartis, Circassia, Stallergenes-Greer, Nestlè Purina, Chiesi, outside of the submitted work. John Lee is on the advisory board for Sanofi Regeneron. All other authors declare no conflict of interest.
Data Availability Statement
Data generated or analyzed during this study are available from the corresponding author upon reasonable request.
