Abstract
Objectives:
This study reviews current postoperative care recommendations following endoscopic sinus surgery (ESS) for chronic rhinosinusitis (CRS), with the goal of optimizing recovery, minimizing complications, and reducing the risk of disease recurrence.
Methods:
A comprehensive literature review was conducted to identify studies on postoperative care following ESS for CRS. Findings were synthesized to develop evidence-based recommendations for optimal postoperative management.
Results:
Postoperative care following ESS for CRS requires a multifaceted approach. Pain management should prioritize acetaminophen and nonsteroidal anti-inflammatory drugs, with opioids reserved for severe pain. Nasal irrigation, topical corticosteroids, and debridement are recommended for all patients. Nasal packing and drug-eluting devices may offer additional benefits, but are not universally required. The use of antibiotics and oral corticosteroids should be selective and guided by individual patient factors and risk profiles.
Discussion:
While general recommendations are made, patient-specific factors such as comorbidities, financial constraints, and adherence to care must be considered. Further evaluation of these factors is essential to establish a more comprehensive and individualized standard of care for postoperative management.
Conclusions:
Effective postoperative care following ESS for CRS should be individualized, multifaceted, and focused on improving recovery, minimizing complications, and ensuring the best possible patient outcomes.
Keywords
Introduction
Chronic rhinosinusitis (CRS) describes prolonged inflammation of the paranasal sinuses lasting more than 12 weeks.1,2 CRS is associated with a variety of symptoms, including nasal obstruction, nasal drainage, facial pain or pressure, and decreased or lost sense of smell. 2 These symptoms often significantly impact quality of life and lead to substantial healthcare expenditures each year.2,3
Historically, CRS has been classified phenotypically, contingent on the presence of nasal polyps, CRS with nasal polyps (CRSwNP), or their absence, CRS without nasal polyps (CRSsNP). CRSsNP is more common than CRSwNP, representing ~70% of total cases. 4 However, recent evidence has better described CRS as a heterogeneous disease representing several unique inflammatory processes with distinguishing cytokine profiles.5-7 CRSwNP is often associated with a predominantly Th2-driven inflammatory process, whereas CRSsNP has been linked to non-Th2-mediated processes. Further, other CRS endotypes like allergic fungal rhinosinusitis and aspirin-exacerbated respiratory disease (AERD) have been linked to their own unique inflammatory pathways.7,8 In reality, recent findings have shown there to be a mix of inflammatory processes present in the numerous CRS endotypes, which may be influenced by genetics, environmental, and geographic factors. 7
CRS can respond to medical therapy to varying degrees. For most patients, medical treatment includes some combination of nasal rinses, corticosteroids, and antibiotics. 3 However, CRS patients fail to respond to medical treatment up to half of the time. 9 For patients with CRS who are unresponsive to appropriate medical therapy, endoscopic sinus surgery (ESS) is an effective option that aims to improve sinus ventilation and drainage and enhance the delivery of topical medications.2,10,11 ESS has been associated with improvements in both global and disease-specific quality-of-life scores and objective improvements in endoscopic scoring scales.12,13
The postoperative period following ESS is critical to ensure satisfactory outcomes for patients. Effective postoperative care following ESS has been shown to reduce the incidence of disease recurrence and ensure adequate sinonasal healing. 14 Poor healing following ESS can result from inflammation, refractory polyposis, adhesion formation, postoperative crusting, scarring, and stenosis of sinus ostia15,16(pp308-315)—all of which can be minimized with appropriate postoperative management. This review discusses current recommendations for the postoperative care of patients who have undergone ESS for CRS.
Materials and Methods
A comprehensive literature review was conducted using PubMed, MEDLINE, and Google Scholar databases to identify studies focused on postoperative care following ESS for CRS. The search included articles published between January 2000 and December 2024, with a focus on studies that assessed pharmacologic, behavioral, and procedural management strategies for post-ESS care. The following search terms were used in various combinations:: “functional endoscopic sinus surgery,” “postoperative care,” “postoperative management,” “analgesics,” “nasal rinses,” “intranasal steroids,” “systemic steroids,” “antibiotics,” “nasal packing,” “debridement,” and “complications.” The resulting publications were reviewed to provide evidence-based recommendations for optimal postoperative management.
Results and Discussion
Analgesics
Although postoperative pain following ESS is generally low, proper analgesia is essential for effective care. Despite being one of the most commonly performed otolaryngologic surgeries, ESS lacks a fully standardized postoperative pain management regimen. 17 Prescription patterns vary among providers and institutions.
Historically, nonsteroidal anti-inflammatory drugs (NSAIDs) have been avoided following ESS due to the fear of postoperative bleeding. However, recent studies have failed to substantiate these concerns, showing that the benefits of NSAIDs generally outweigh the risks for most patients.18-21 An evidence-based review by Svider et al. gave NSAIDs grade A evidence, finding they reduce opioid consumption and improve pain metrics following ESS, noting that some patients experience potential for NSAID intolerance. 20 In a 2019 study, 166 patients undergoing endoscopic nasal surgeries received either acetaminophen 325 mg with hydrocodone–acetaminophen 5/325 mg as needed or ibuprofen 200 mg and acetaminophen 325 mg, with the opioid reserved for breakthrough pain. 19 The group receiving NSAIDs had significantly lower visual analog scores of pain (P = 0.006) and took significantly fewer opioid pills (P = 0.018), with no bleeding complications in either group. 19 A recent 2023 systematic review and meta-analysis found that while patients receiving NSAIDs reported similar pain scores to those who did not, they demonstrated a reduced need for rescue opioids and had a lower nausea risk. 21 Further, the same group found NSAID use did not increase the risk of epistaxis (relative risk (RR) = 0.72). 21 NSAIDs should be used with caution in patients with AERD postoperatively, as they have the potential to trigger hypersensitivity reactions. However, selective COX-2 inhibitors can be considered. Bavbek et al. found that 7.5 mg meloxicam was a safe option for patients with asthma and/or nasal polyps with proven aspirin hypersensitivity. 22 Further, a 2014 meta-analysis found that selective COX-2 inhibitors were generally safe in patients with stable mild-to-moderate asthma with AERD, though they did cause respiratory symptoms in 1 in 13 patients with AERD. 23
In addition, evidence exists to suggest that many patients can achieve adequate postoperative pain control with scheduled acetaminophen. 24 A 2021 multicenter randomized trial evaluated acetaminophen as a first-line postoperative analgesic for ESS. 25 All postoperative patients received scheduled acetaminophen 650 mg, after which they were randomized to receive either oxycodone 5 mg as second-line therapy or ibuprofen 600 mg as second-line therapy, with oxycodone 5 mg reserved for breakthrough pain. 25 The study found that 63% of patients in the second-line oxycodone group had adequate pain control with acetaminophen alone, while 73% of patients in the second-line ibuprofen group achieved pain control with either acetaminophen alone or acetaminophen plus ibuprofen. 25 Ibuprofen use did not significantly affect epistaxis levels in this study. 25 This suggests that for many patients, postoperative pain can be effectively managed with acetaminophen and NSAIDs without the need for opioids. A 2023 study found that gabapentin could also reduce opioid consumption in the postoperative period following ESS or septoplasty. 26 Patients receiving gabapentin on postoperative days 1, 2, and 4 in addition to hydrocodone–acetaminophen and/or acetaminophen as needed, reported a significant reduction in opioid use during the first week following surgery compared to those who did not receive gabapentin (gabapentin group: −9.54 mg hydrocodone on postoperative day 7, P = 0.025). 26 Both groups reported similar levels of pain and adverse events. 26
In some cases, opioids may be required during the first few days following ESS. Despite many studies suggesting patients utilize opioids minimally following ESS and a 2020 study reporting that opioids were prescribed in excess of 85% of the time for sinonasal surgery, there is little consensus on the amount of opioids that should be prescribed.27-29 A 2021 study found that of patients receiving opioids, 68% endorsed usage on day 2, with usage decreasing to 43%, 23%, 9%, and 3% on days 4, 6, 8, and 10, respectively. 30 Zheng et al. found that the majority of patients may only require 10 to 15 doses of opioids for adequate pain control following ESS. 31
In conclusion, postoperative pain management following ESS can be effectively achieved with a multimodal approach, minimizing opioid use. Scheduled acetaminophen, often in combination with NSAIDs, can provide sufficient analgesia for many patients, reducing the need for opioids. However, NSAIDs should be used with caution in individuals with AERD or bleeding risks. Gabapentin may also offer additional benefits in reducing opioid consumption without compromising pain control. Although opioids may be necessary in the early postoperative period, most patients require only minimal doses. At our institution, we typically encourage routine acetaminophen 650 mg every 4 to 6 hours and prescribe ten 5 mg oxycodone tablets for breakthrough pain. Given the variability in prescribing patterns, further standardization of postoperative pain management protocols is warranted to optimize patient outcomes and reduce opioid dependence following ESS.
Saline Irrigations
There is substantial evidence supporting the use of saline irrigations as part of postoperative care, beginning 24 to 48 hours after ESS. 2 Nasal irrigation helps clear secretions from the nasal cavity, maintains moisture in the area, reduces mucosal edema, and improves mucociliary function, ultimately aiding in the healing of the sinuses and nasal passages.32-35
There remains some debate regarding the choice of solution for irrigations in the postoperative period. A 2014 study randomly assigned 74 patients to use either normal saline, lactated Ringer’s solution, or hypertonic saline for postoperative high-volume rinses, three times a day for 6 weeks. The study reported that lactated Ringer’s solution resulted in better subjective outcomes compared to normal saline or hypertonic saline, while hypertonic saline led to less polypoidal mucosa on endoscopic examination at the 6-week time point. 36 However, in a 2018 meta-analysis of five studies with a total of 331 participants, Chen et al. reviewed lactated Ringer’s solution, hypertonic saline, electrolyzed acid water, and Amphotericin B solutions as choices for irrigation mediums, concluding that none was superior to normal saline in terms of endoscopic and subjective assessments. 37
In contrast, a randomized, double-blind, controlled study in which participants received either hydrogen-rich saline (hydrogen concentration >0.6 mmol/L) or normal saline twice a day for 12 weeks demonstrated that hydrogen-rich saline resulted in significantly improved subjective symptoms and endoscopic outcomes when compared to isotonic saline alone, with similar adverse events. 38 Later, a systematic review and meta-analysis by Lima et al., which included 479 patients from seven studies, reported that irrigation with hypertonic saline was superior to isotonic saline for nasal crusting at 30 to 45 days post-surgery (RR = 0.65; 95% confidence interval (CI): 0.49-0.87; P = 0.004), severe crusting at 14 to 21 days (RR = 0.59; 95% CI: 0.38-0.91; P = .02), and the persistence of polypoid mucosa at 14 to 21 days (RR = 0.53; 95% CI: 0.43-0.65; P < 0.00001). 39 Further, patients receiving hypertonic saline had significant improvements in subjective symptom scores as compared to isotonic saline at 30 to 45 days after surgery. 39 This meta-analysis reported similar adverse events between the hypertonic and isotonic groups, but prior studies have associated hypertonic saline with higher pain scores.39,40
Although the choice of solution may depend on factors such as cost, availability, potential side effects, and institutional preferences, evidence supports the use of high-volume irrigation for all patients following ESS for CRS. Current recommendations suggest beginning high-volume saline irrigations (>60 mL) within 24 to 48 hours after ESS and continuing rinses for at least 8 weeks. 3 For most patients, we recommend beginning low-pressure, high-volume saline irrigation with isotonic saline using a device such as the NeilMed Sinus Rinse (NeilMed Pharmaceuticals Inc., USA), at least twice per day starting on postoperative day 1.
Corticosteroids
Both topical and systemic corticosteroid use are established treatment strategies for CRS due to their anti-inflammatory properties. 41 However, most studies have not demonstrated significant benefits from systemic corticosteroids in the postoperative setting that outweigh the associated risks, which include mood changes, insomnia, hyperglycemia, increased intraocular pressure, and immune dysfunction.2,41-44 A 2009 randomized, double-blind, placebo-controlled trial on patients with CRSwNP who underwent ESS administered 30 mg of prednisone 5 days prior to surgery and continued it until postoperative day 9 in the intervention group. 45 The study found that compared to placebo, patients on systemic corticosteroids had improved Lund–Kennedy endoscopy scores at time points up to 6 months (P < 0.001), but showed no improvements in postoperative symptom scores. 45 However, a prospective double-blinded, placebo-controlled, randomized clinical trial on patients with CRSsNP found no difference in Sinonasal Outcome Test-22 (SNOT-22) scores or Lund–Kennedy endoscopy scores at any postoperative time point up to 6 months in patients who received either a 12-day oral prednisone taper or a placebo following ESS. 43 Notably, the group that received prednisone had worse SNOT-22 psychologic subdomain scores than the placebo group. 43 Another randomized, double-blind, placebo-controlled trial showed no postoperative difference in endoscopic, quality of life, or symptom burden scores between patients with CRSwNP receiving prednisone 30 mg for 7 days versus placebo after having already received a steroid-eluting middle meatus spacer. 44 While systemic steroids may be appropriate for some patients with CRSwNP, they are not recommended as the standard of care for all patients following ESS.
Topical intranasal corticosteroids offer the benefits of local anti-inflammatory effects while largely avoiding systemic side effects and are generally favored in the postoperative setting.3,41 A 2013 systematic review, which included twelve studies, found that topical intranasal steroids led to significant improvements in symptom scores (P = 0.0002), polyp scores (P = 0.007), and polyp recurrence rates (P = 0.002) in patients with CRSwNP. 46 However, a 2015 meta-analysis evaluating eighteen randomized controlled trials found no significant difference in postoperative symptom scores between steroid and placebo groups. 47 It did, however, find significant improvements in endoscopic scores in the steroid group at 6 and 12 months in both patients with CRSwNP and CRSsNP, as well as a reduction in CRS recurrence in patients with CRSwNP. 47 Of note, this analysis included studies utilizing both topical and systemic steroids postoperatively. In a later meta-analysis, Yoon et al. specifically evaluated corticosteroid nasal irrigations and found they were associated with significant improvements in both endoscopic scores and quality-of-life metrics compared to pre-treatment values, with no significant adverse effects. 48 However, these results were not significant when compared to normal saline irrigations alone. 48 In evaluating the choice of agent, a 2019 double-blind randomized controlled trial determined that both topical nasal dexamethasone 0.032% and fluticasone propionate demonstrated significant symptomatic and endoscopic improvement in patients with CRSwNP at 4-, 8-, and 12-week intervals with twice daily use. 49 Patients in both groups of this study also performed nasal saline irrigations postoperatively during the study interval. 49
While the benefits of topical corticosteroids in the postoperative setting may not be as profound as those of saline irrigation, there is sufficient evidence to recommend their use following ESS for most patients, given their low side-effect profile compared to systemic corticosteroids.2,3,41 Patients with CRSwNP may experience the most benefits from systemic corticosteroids. At our institution, we typically offer a 9-day taper of prednisone, starting at 30 mg and decreasing by 10 mg every 3 days for patients with CRSwNP.
Antibiotics
In the past, a short course of antibiotics was considered to be the standard of care following ESS to prevent toxic shock syndrome associated with nasal packing and to mitigate infections that could delay healing.41,50 However, the lack of robust evidence supporting their efficacy in the postoperative setting has led to a decline in their use in recent years. 50 Several studies have evaluated the effectiveness of antibiotics following ESS, though their methodologies are heterogeneous.45-51
Some studies have shown no benefit in terms of subjective or endoscopic measures following antibiotic use,51,52 while others have demonstrated early postoperative improvements in endoscopic appearance and sinonasal crust formation. 53 A 2012 meta-analysis by Saleh et al., which pooled data from three trials, found that postoperative antibiotic prophylaxis did not result in statistically significant reductions in the incidence of infection, endoscopic scores, or symptoms. 54 A 2014 study, which considered intraoperative culture data, found that endoscopic outcomes improved when culture-appropriate postoperative antibiotics were used. 55 Zhang et al. demonstrated that patients treated with trimethoprim-sulfamethoxazole and clindamycin for 2 weeks postoperatively had worse quality-of-life metrics at 1 and 3 months when this antibiotic regimen was retrospectively found to be culture-inappropriate, although this difference was not observed at the 6 months timepoint. 56
A meta-analysis by Shu et al. assessed the long-term effects of postoperative macrolide antibiotics and found that while macrolides did not significantly improve quality of life or disease severity, they did result in improved nasal endoscopy scores (standardized mean difference = −0.32; 95% CI: −0.62 to −0.03). 57 In contrast, a systematic review and meta-analysis by Swords et al., which included seven publications, concluded that there was no level 1 evidence suggesting antibiotics improve patient outcomes following sinus surgery. 58 However, they noted the heterogeneity in outcome measures and recommended further investigation. 58
Ultimately, the decision to prescribe antibiotics following ESS for CRS should be made on a case-by-case basis, considering factors such as surgical findings (e.g., purulence), culture data, and the side-effect profiles of antibiotics. However, routine antibiotic therapy for all patients undergoing ESS for CRS is not recommended in the postoperative setting. At our institution, for patients with nonabsorbable packing, we typically provide an antibiotic course lasting between 10 and 21 days.
Unmedicated Packing
Nasal packing is commonly used in the perioperative setting to maintain postoperative hemostasis following ESS. 59 In addition to hemostasis, packing materials serve as scaffolds that can facilitate wound healing and prevent complications such as adhesions, synechiae, and stenosis.2,60 However, concerns have been raised regarding increased patient discomfort associated with packing. 3 There are two primary types of packing: nonabsorbable, which requires removal, and absorbable, which biodegrades over time.
Historically, nonabsorbable packing has been the most commonly used following ESS, with Merocel® (Medtronic Xomed, USA), being the most frequently studied option. 59 A 2009 randomized, controlled trial demonstrated that Merocel® packing in the middle meatus reduced postoperative adhesion rates compared to no packing. 61 Further studies, such as one by Kim et al., showed that the use of a gloved finger over the Merocel® pack significantly reduced pain levels, bleeding at the time of pack removal, and adhesion rates, while improving endoscopic scores at 4 weeks compared to an ungloved Merocel® group. 62 Similarly, Akbari et al. found that gloved Merocel® improved comfort during pack removal, although they did not show it bettered sinonasal mucosal inflammation or discomfort prior to removal. 63
Several studies have investigated how absorbable packing materials compare to nonabsorbable materials in terms of bleeding rates and wound healing. Randomized controlled trials have demonstrated that Nasopore® (Polyganics, The Netherlands), HemoStase® (CryoLife Inc., USA), Sepragel® (Genzyme Biosurgery, USA), and Floseal® (Baxter International Inc., USA) are each not associated with increased pain scores compared to unpacked cavities.64-67 Nasopore®, Floseal®, Surgicel® (Ethicon Inc., USA) Cutanplast® (Mascia Brunelli S.p.A., Italy), and oxidized cellulose have also been found in separate randomized controlled trials to be associated with less bleeding than Merocel® at the time of pack removal; however, none evaluated gloved Merocel®.68-72 A 2012 randomized trial comparing Nasopore® with gloved Merocel® found that the absorbable packing did not reduce bleeding or discomfort and was associated with slower mucosal healing over the first 3 months. 73 Chitogel® (Chitogel Limited, New Zealand) was shown to improve sinus ostial patency compared to no packing, though the results on adhesion formation were mixed. 74
Neither Floseal®, oxidized cellulose, nor Nasopore® were shown to confer a benefit in wound healing compared to Merocel®,68,71-73 and some studies even showed that Floseal® was associated with higher rates of symptomatic adhesion formation when compared to thrombin-soaked gelatin foam.75,76 Carboxymethylcellulose was demonstrated to result in lower synechia formation rates and improved pain scores compared to gloved Merocel® at time points up to 8 weeks, 77 but was not shown to improve wound healing compared to no packing in another randomized controlled trial. 78 A 2021 systematic review and pooled analysis by Huang and Zhou evaluated four studies with 148 participants and demonstrated absorbable packing may offer a reduction in adhesion formation at 6 to 8 weeks (odds ratio (OR) = 0.3864) and 12 weeks (OR = 0.2396) compared to no packing but did not show any difference in crusting, edema, or granulation formation. 79
A systematic review and meta-analysis by Yan et al. suggested that biodegradable packing materials reduced bleeding at time of removal, pain in situ, and nasal blockage compared to “conventional packing.” 80 Similarly, a 2015 systematic review of five studies and 241 nasal cavities evaluating outcomes with absorbable and nonabsorbable packing found postoperative bleeding to be lower with absorbable packing, but found no significant differences in postoperative edema or bleeding and pain upon pack removal. 81 A subsequent meta-analysis of two studies found no difference in synechia formation, though heterogeneity between studies was noted. 81
Despite the potential benefits of packing, some studies argue that nasal packing may not be necessary for hemostasis in most patients undergoing ESS.82-85 A 2016 retrospective study by Kim et al., which involved 490 patients who underwent ESS for CRS, compared nasal packing with electrocauterization and found no significant differences in primary (P = 0.058) or secondary bleeding (P = 0.142) between the two methods, though a trend favoring packing was present. 83 Similarly, a 2017 decision analysis by Stern-Shavit et al. concluded that while absorbable packing may be less detrimental to quality of life compared to nonabsorbable materials, middle meatal packing was generally not advantageous. 84 A more recent 2024 systematic review, evaluating 139 patients randomized to receive middle meatal packing in either nostril, found no difference in synechia formation between packed and unpacked sides at 8 weeks (RR at 8 weeks = 0.77; 95% CI: 0.48-1.25) and at 12 weeks (RR = 0.68; 95% CI: 0.42-1.20). 85
The literature on postoperative nasal packing following ESS remains inconclusive. Given the wide range of available packing materials and individual patient factors, it is difficult to make definitive recommendations. The decision to use packing, as well as the choice of material, should consider factors such as cost, availability, patient comorbidities, and the likelihood of complications such as bleeding and synechia formation. For most patients, packing following ESS for CRS is not necessary. However, when packing is indicated, gloved Merocel® or certain absorbable materials provide an optimal balance of hemostatic control, wound healing, and patient comfort and are regularly utilized for our patients.
Drug-Eluting Packing, Stents, and Spacers
Drug-eluting packing, stents, and spacers have been developed to enhance medication delivery, improve compliance with postoperative regimens, and maintain nasal airway patency. When combined with corticosteroids or antibiotics, these devices provide localized medication delivery to the sinus and nasal mucosa, promoting healing and reducing the likelihood of disease recurrence. 2
In 2008, Huvenne et al. demonstrated that a doxycycline-eluting frontal sinus stent significantly improved sinus symptoms and increased osteogenesis markers compared to a placebo stent. 86 Two randomized controlled trials evaluating triamcinolone-soaked nasal packing against saline packing found reduced polyp recurrence and improved endoscopic scores in the corticosteroid group at 1 week, 2 weeks, 3 months, and 6 months.87,88 A 2012 meta-analysis of two randomized trials involving the Propel™ implant (Medtronic, Ireland)—containing 370 µg of mometasone furoate embedded in a polymer matrix—showed that the device significantly reduced postoperative interventions (P = 0.0008), including the need for lysis of adhesions (P = 0.0016) and oral corticosteroid use (P = 0.0023). In addition, endoscopic evaluations revealed a significant reduction in polyposis (P < 0.0001) in comparison to the control group. 89
Gyawali et al. conducted a randomized controlled trial comparing triamcinolone-impregnated polyvinyl alcohol packs to saline-impregnated packs, using a within-patient design. 90 They found that the steroid-impregnated side showed significant reductions in Lund–Kennedy scores (P = 0.0001), Perioperative Sinus Endoscopy scores (P = 0.002), and edema (P = 0.027) at 3 weeks postoperatively. 90
A 2019 meta-analysis of seven studies further supported the efficacy of steroid-eluting stents. 91 The analysis showed significant improvements at ~30 days postoperatively with stent use, including a reduced need for postoperative interventions (OR = 0.45; 95% CI: 0.33-0.62; P < 0.001), revision surgery (OR = 0.30; CI: 0.18-0.52; P < 0.001), and oral steroid use (OR = 0.58; CI: 0.40-0.84; P = 0.004). 91 The stent group also demonstrated improved frontal sinus ostia patency (OR = 2.53; CI: 1.61-3.97; P < 0.001), fewer cases of moderate-to-severe adhesions and scarring (OR = 0.28; CI: 0.13-0.59; P < 0.001), and fewer instances of increased polyp scores (OR = 0.42; CI: 0.25-0.74; P = 0.002) compared to controls. 91 However, a more recent randomized clinical trial found that, compared to patients receiving Merocel® packs wrapped in a non-latex glove, those receiving the Propel™ implant had worse lower middle turbinate lateralization scores at the 10-day postoperative visit, worse SNOT-22 scores at the 20-day visit, and comparable Lund–Kennedy scores at up to 3 months postoperatively. 92
Overall, the use of drug-eluting stents, spacers, and packing in ESS has yielded mixed results in terms of postoperative outcomes. Several studies and meta-analyzes have shown that steroid-eluting devices significantly reduce the need for postoperative interventions and medications, and improve endoscopic scores and wound healing. Similarly, antibiotic-eluting devices have demonstrated positive effects on symptom relief and wound healing. However, these devices may cause local irritation, and the removal of nonabsorbable devices can be painful for some patients.
Although drug-eluting stents show promise in certain postoperative contexts, further research is needed to confirm their long-term efficacy and identify the patient populations most likely to benefit from their use. Propel™ implants are often used at our institution, especially for patients with CRSwNP who may benefit most from corticosteroids.
Debridement
Sinonasal debridement has often been done in the postoperative setting to remove tissue and material that may lead to inflammation and poor healing.2,3,41 Evidence supports its efficacy in reducing complications. Bugten et al. demonstrated that debridements reduced crusting and middle meatal adhesion rates at both 3 months and 1 year in two randomized controlled trials.93,94 A 2011 randomized controlled trial comparing debrided and non-debrided sides within the same patient found that, while debridement reduced adhesions, it did not improve endoscopic scores at 3 months. 95 However, a systematic review by Green et al. reported that, although debridement did not result in significant improvements in endoscopic scores, it led to reductions in both short- and long-term symptom scores. 96 Similarly, a 2018 Cochrane review, which included four studies, found no significant differences in subjective symptom scores or endoscopic scores at 6 months between patients who underwent debridements and those who did not. 97 However, the same review found that debridements significantly reduced adhesion rates. 97
In a 2016 randomized controlled trial, Varsak et al. compared patients who received a single debridement at postoperative week 1 with those who received debridements at weeks 1, 2, and 4. 98 The single debridement group had lower discomfort scores (P = 0.004) and less negative impact on work-related activities (P = 0.013), with no significant differences in Lund–Kennedy or SNOT-22 scores between the two groups.
While research supports the notion that debridements help minimize certain complications, their use has not been shown to consistently improve endoscopic or symptom scores. Nevertheless, routine debridement is recommended for patients undergoing ESS to prevent adhesions and promote healing. At our institution, endoscopic debridement is traditionally performed between 4 and 10 days postoperatively. If steroid-eluting stents are placed, they are typically removed at this time to reduce crusting. Further studies are needed to better define the role of debridement and establish an optimal routine to improve long-term surgical outcomes.
Conclusion
Postoperative care following ESS for CRS focuses on several key strategies to optimize recovery and minimize complications. Pain management should prioritize acetaminophen and NSAIDs, with opioids reserved for severe pain, balancing effective pain relief with responsible prescribing habits. Nasal irrigation is recommended for all patients undergoing ESS to support mucosal healing and improve sinus function, although the ideal solution remains debated. Corticosteroids should be administered to most patients, with topical agents preferred over systemic steroids due to their localized effects and lower risk of systemic side effects. Antibiotics should be prescribed selectively based on individual patient factors, surgical findings, and culture data, as routine use has not shown clear benefits. Nasal packing can aid in hemostasis and wound healing, though its use is not universally required. Drug-eluting devices, such as stents and spacers, may enhance outcomes by delivering medications directly to the nasal mucosa, but further research is needed to identify the patient populations that would benefit most from their use. Sinonasal debridement, performed at least 1 week postoperatively, is recommended to prevent complications and promote healing, although the evidence regarding its impact on endoscopic and symptom scores is mixed (Table 1).
Postoperative Recommendations Following ESS for CRS.
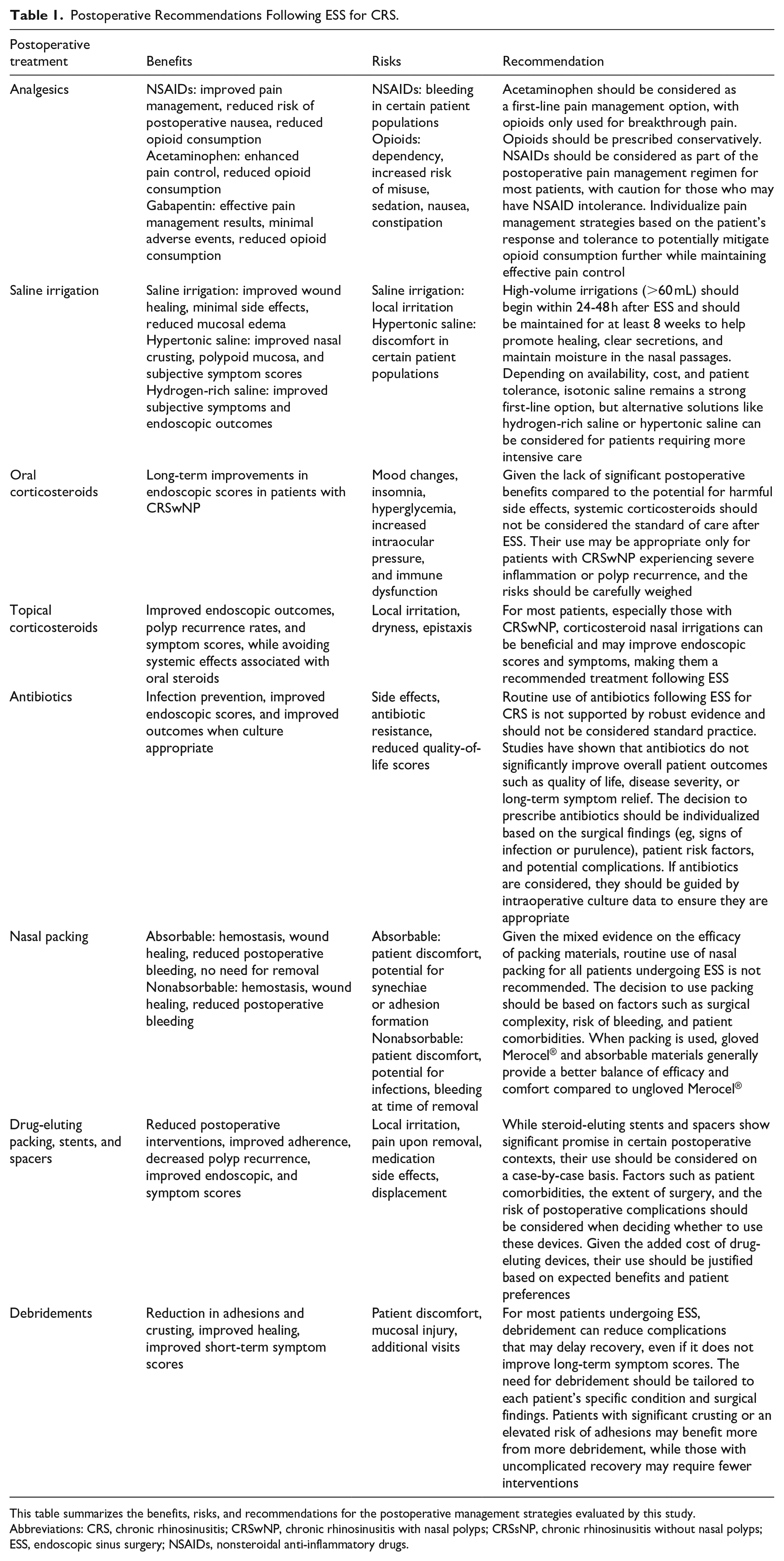
This table summarizes the benefits, risks, and recommendations for the postoperative management strategies evaluated by this study.
Abbreviations: CRS, chronic rhinosinusitis; CRSwNP, chronic rhinosinusitis with nasal polyps; CRSsNP, chronic rhinosinusitis without nasal polyps; ESS, endoscopic sinus surgery; NSAIDs, nonsteroidal anti-inflammatory drugs.
Footnotes
Ethical Considerations
All reported studies/experiments reported by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki Declaration and its amendments, institutional/national research committee standards, and international/national/institutional guidelines).
Consent to Participate
This review article did not involve the recruitment or direct participation of human subjects by the authors. All data presented and discussed were obtained from previously published studies in which informed consent was obtained by the original investigators in accordance with applicable ethical standards and institutional guidelines.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data availability is not applicable to this review article as it utilizes data from previously published studies. All relevant data from the included studies are available in the respective original publications.
