Abstract
Background:
Cochlear implantation (CI) has revolutionized auditory rehabilitation for patients with severe to profound hearing loss secondary to bacterial meningitis. This systematic review synthesizes current evidence on CI outcomes in this patient population. Demographic characteristics, surgical techniques, and audiometric outcomes across studies published from 1993 to 2024 are highlighted.
Methodology:
In a systematic search of electronic databases, 19 eligible studies from Europe, North America, China, and Turkey were identified. These studies included retrospective cohort designs, prospective cohorts, and case series that assessed various aspects of CI outcomes. Data extraction was focused on patient demographics, preimplantation characteristics, surgical details, and postoperative outcomes.
Results:
The studies reported heterogeneous but positive audiological outcomes following CI. Significant improvements in speech perception and quality-of-life metrics were observed across different patient subgroups. This review identified variability in surgical approaches (eg, cochleostomy and circumodiolar drill-out), implant devices (eg, nucleus and advanced bionics), and speech processing strategies (eg, SPEAK and ACE). Audiological gains were influenced by factors such as age at implantation, duration of deafness, and presence of cochlear ossification.
Conclusion:
CI offers significant benefits for patients with postmeningitis hearing loss, although the outcomes vary based on patient-specific factors and surgical techniques. The findings from this review underscore the importance of personalized treatment approaches and highlight areas for future research, including standardized outcome measures and long-term follow-up, to optimize patient outcomes and refine clinical protocols.
Introduction
Meningitis remains a significant global health concern that affects millions of individuals annually and often results in serious consequences, including sensorineural hearing loss (HL).1,2 HL is a common and devastating complication of bacterial meningitis that affects a significant proportion of survivors, particularly in pediatric populations.1,3 The prevalence of postmeningitis HL varies, with estimates suggesting that approximately 5% to 35% of patients develop some degree of hearing impairment following bacterial meningitis.4,5 Severe-to-profound HL is observed in approximately 4% to 10% of these patients, which necessitates interventions such as cochlear implantation (CI) for auditory rehabilitation.6,7
While advancements in medical treatments have improved survival rates, the residual hearing impairment poses considerable challenges, particularly in children and young adults.3,8,9 Sensorineural HL due to meningitis is often severe to profound and affects both ears asymmetrically, rendering conventional hearing aids ineffective. 1
CI has been recognized as an effective intervention for restoring auditory function in patients with severe-to-profound HL, including those whose HL results from meningitis.10,11 However, the specific efficacy and outcomes of this procedure in patients with postmeningitis HL have been a subject of ongoing research and debate. 11 Understanding the benefits, limitations, and considerations for CI in this population is crucial for clinicians, researchers, and affected individuals seeking optimal auditory rehabilitation strategies.
This systematic review aimed to provide an updated synthesis of the outcomes of CI in patients with postmeningitis HL, building on previous reviews and incorporating the latest data from studies published between 1993 and 2024. The last comprehensive systematic reviews on this topic was published in 2020 and 2024, and both studies were without meta-analysis highlighting the benefits of CI in this patient population and identifying gaps in the literature and areas for future research.12,13 By updating the evidence base, this review intends to augment our comprehension of the factors influencing CI outcomes and inform clinical practice and policy. This systematic review and meta-analysis aimed to evaluate the outcomes of CI in postmeningitis HL. In addition, the impact of CI on speech perception and recognition in these patients is discussed. The timing of the intervention in relation to hearing outcomes and the correlation between hearing improvement and radiological grade of cochlear ossification are explored.
Methodology
Methods
This study was performed in accordance with the guidelines of Preferred Reporting Items for Systematic review and Meta-Analysis (PRISMA) 11 and was registered in the PROSPERO website [CRD42024543650]. Moreover, this study adhered to the PICO protocol, as follows:
Population: Patients with postmeningitis severe-to-profound HL
Intervention: Cochlear implantation
Comparison: Preoperative hearing status, congenital HL
Outcomes: Postimplantation hearing outcomes, speech perception, speech recognition, and quality-of-life improvement after CI in patients with postmeningitis HL. Association of the time of intervention with the grade of hearing improvement. Association of the grade of hearing improvement with the grade of preimplantation cochlear ossification.
Study Design
This systematic review and meta-analysis was conducted via a comprehensive search of electronic databases, including PubMed, Scopus, Google Scholar, MEDLINE, EMBASE, and the Cochrane Library, to identify all relevant studies published up to April 2024. The search strategy involved using keywords related to “cochlear implant” OR “cochlear implantation” AND “postmeningitis hearing loss,” combined with terms such as “cochlear ossification,” “severe to profound hearing loss,” “hearing improvement,” “outcomes,” and “quality of life.” The search included studies from 1993 to 2024, encompassing retrospective cohort designs, prospective cohorts, and case series. Data extraction focused on patient demographics, preimplantation characteristics, surgical details, and postoperative outcomes.
Selection Criteria and Screening Process
Studies identified through the database searches were collected using the Rayyan software 14 for screening and selection, with no restrictions on timeframe. Duplicate articles were eliminated prior to the title and abstract screening. Two reviewers independently evaluated the titles and abstract of the articles. In cases of disagreement, the article underwent full-text screening. Subsequently, 2 independent reviewers conducted full-text screening of the selected papers based on the inclusion and exclusion criteria, with a third senior reviewer addressing any discrepancies.
Eligibility
The inclusion criteria were as follows:
Studies published without timeframe limitations.
Studies that reported the number of patients who underwent CI surgery because of postmeningitis deafness.
Studies published in the English language.
Prospective cohorts, randomized controlled trials, retrospective cohorts, case series, case-control studies.
Studies involving pediatric or adult patients.
Studies reporting at least 3 months of postimplantation audiological assessment.
Studies reporting outcomes of interest relevant to the clinical questions.
The exclusion criteria were as follows:
Those with incomplete data on postimplantation hearing outcomes and speech perception.
Studies not focused on postmeningitis HL.
Data Collection
Data collection was conducted using a standardized data extraction form completed by 2 independent pairs of authors. This form captured a variety of information including study characteristics (author and year of publication), participant demographics (age and sex), and preoperative details such as hearing level, speech perception, and speech recognition. Intervention specifics were meticulously recorded, detailing the type and timing of the surgery, the side of the surgery (left, right, or both), whether the surgery was bilateral or unilateral, the duration between the incidence of meningitis and CI, the radiological grade of postmeningitis cochlear ossification, and electrode insertion depth. Postoperative outcomes were also documented, focusing on hearing levels, speech perception and recognition, and improvements in quality of life.
Data Analysis
Meta-Analysis
The data were statistically pooled using appropriate meta-analytical techniques (eg, random-effects models) to estimate the overall effect size of CI on hearing outcomes.
Assessment of Heterogeneity
Heterogeneity among the studies was assessed using Cochran’s Q test and the I2 statistic. Substantial heterogeneity was predefined as I2 >50%. Sensitivity analyses and subgroup comparisons were performed to identify potential sources of heterogeneity (eg, study quality and geographical location).
Publication Bias
Publication bias was evaluated using statistical tests (eg, Egger’s test) to assess the likelihood of small-study effects influencing the overall results.
Quality Assessment of the Studies
Newcastle–Ottawa Scale for Cohort Studies
The methodological quality of the included cohort studies was assessed using Newcastle–Ottawa Scale (NOS). This scale evaluates studies based on 3 broad categories: selection of study groups, comparability of groups, and determination of outcomes. Each study is rated on a star system, with a maximum of 9 stars indicating the highest quality.
Each included study was independently assessed by 2 reviewers, and any discrepancies were resolved via discussion to ensure consensus. The studies were categorized as high quality (7-9 stars), moderate quality (4-6 stars), or low quality (0-3 stars) based on their total score on NOS.
This systematic approach to data analysis and quality assessment ensured the reliability and validity of the findings regarding the outcomes of CI in postmeningitis HL and the associated outcomes.
Results
Primary Search Results
A comprehensive review was conducted on 438 articles sourced from 4 databases (PubMed, Science Direct, Web of Science, and EMBASE). After duplicate removal, 223 articles proceeded to title and abstract screening. Of these, 157 studies advanced to full-text review. Ultimately, 19 studies published between 1993 and 2024 meeting the inclusion criteria were selected (Figure 1).
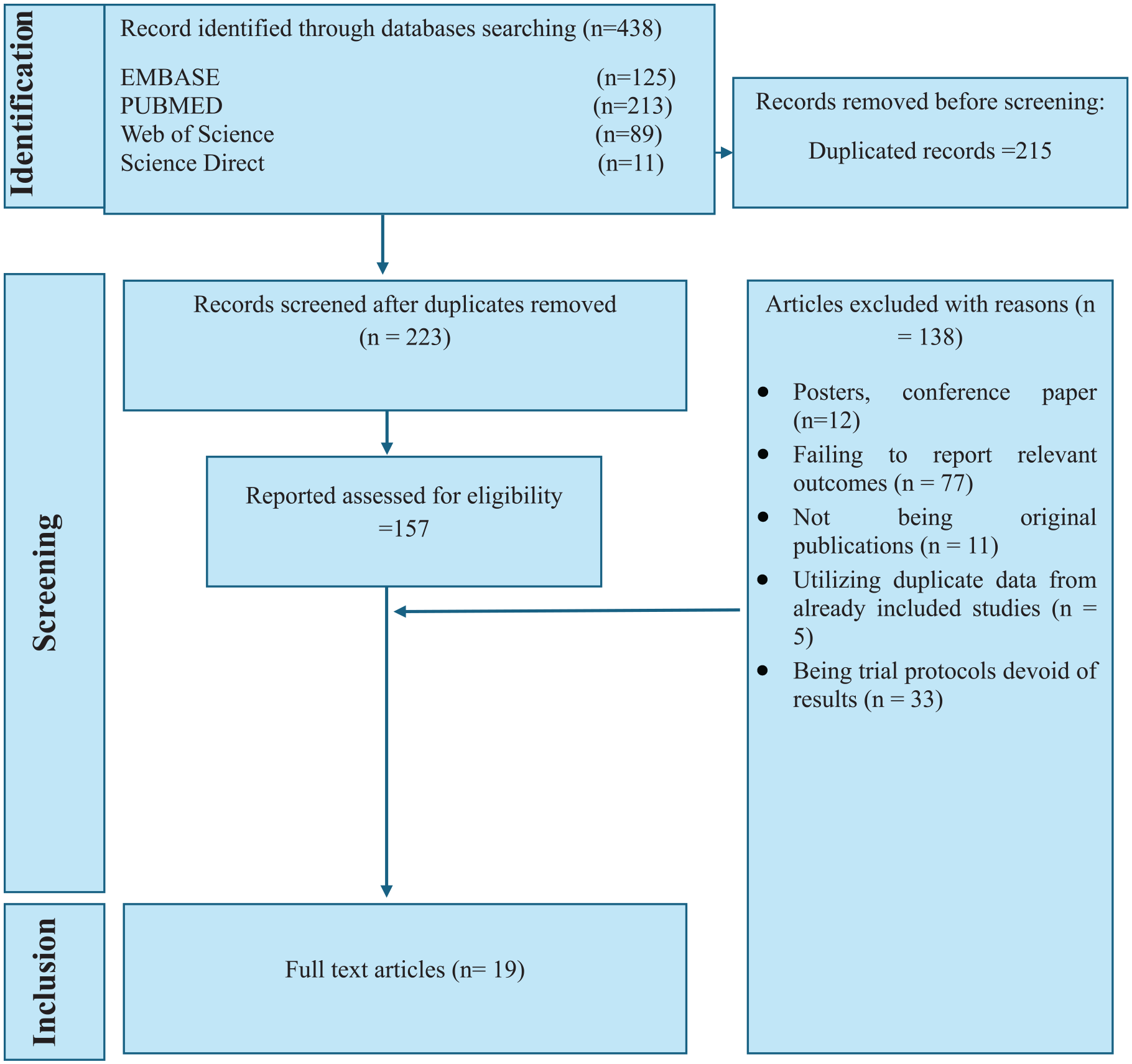
PRISMA figure showing the steps to choose the studies for a systematic review.
Study Characteristics
This systematic review identified 19 studies published between 1993 and 2024 that met the inclusion criteria.15-33 These studies varied in terms of design and geographic location, with the majority conducted in Europe and North America. The study designs included retrospective cohort studies, prospective cohorts, and retrospective case series. The earliest study by Parisier et al 15 in 1993 and the most recent one by Chen et al 33 in 2024 provided insights into the outcomes of CI in patients with postmeningitis HL. Each study offered unique perspectives on factors such as patient demographics, preimplantation characteristics, and outcomes related to speech perception, speech recognition, and quality of life improvement (Table 1).
Study Characteristics.

Abbreviation: CI, cochlear implantation.
Quality Assessment of the Studies
The methodological quality of the included studies was evaluated using the NOS. Overall, most studies demonstrated high methodological rigor, with a focus on representative participant selection, group comparability, and reliable outcome measures. However, studies such as those by Parisier et al, 15 Steenerson et al, 18 Mitchell et al, 19 Cordero et al, 22 Beadle et al, 23 and Liu et al 30 were rated as moderate quality and scored 4 to 6 stars. These studies exhibited adequate representation of study groups but showed some limitations in comparability or outcome assessment criteria (Table 2).
Quality Assessment of Studies.
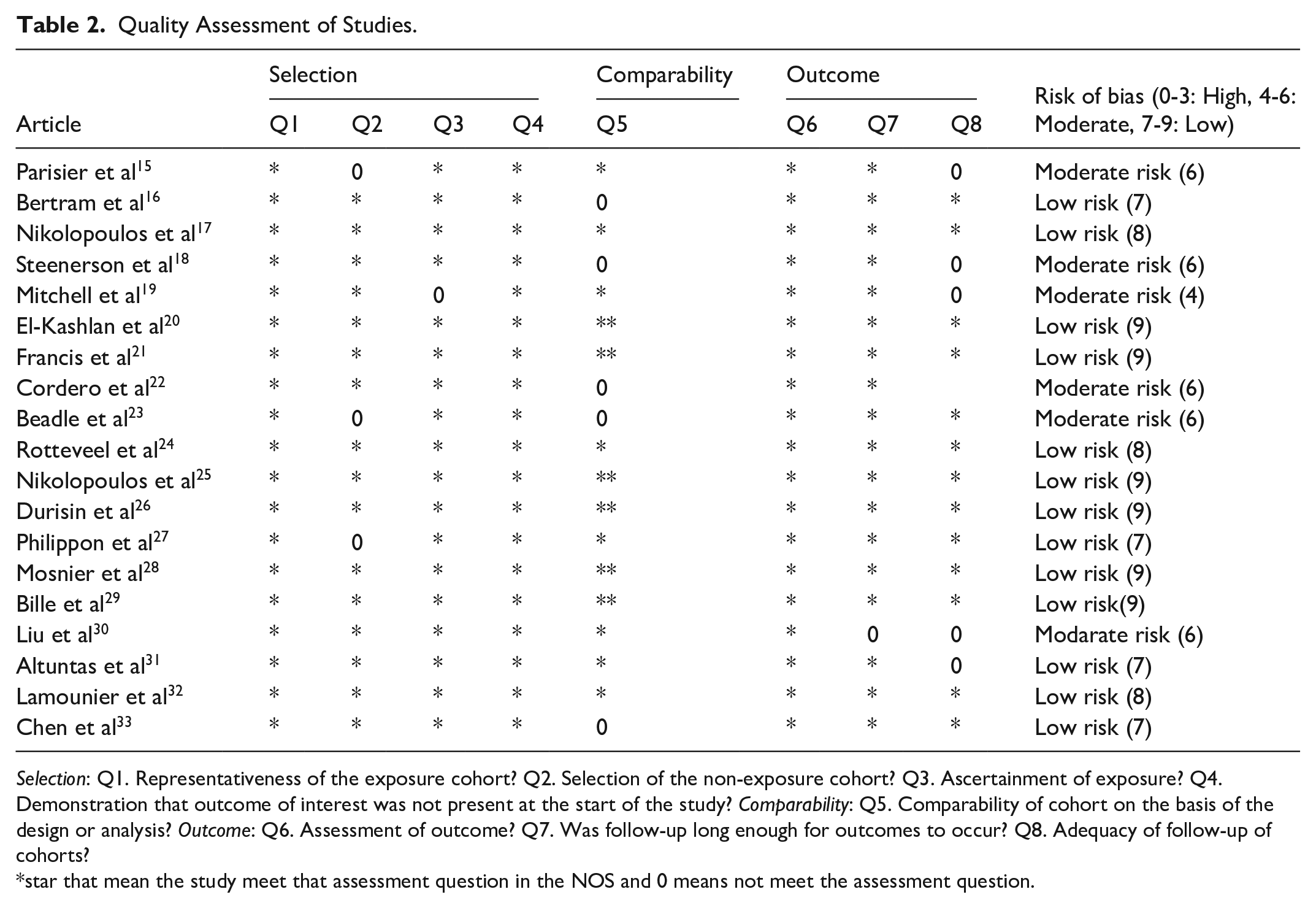
Selection: Q1. Representativeness of the exposure cohort? Q2. Selection of the non-exposure cohort? Q3. Ascertainment of exposure? Q4. Demonstration that outcome of interest was not present at the start of the study? Comparability: Q5. Comparability of cohort on the basis of the design or analysis? Outcome: Q6. Assessment of outcome? Q7. Was follow-up long enough for outcomes to occur? Q8. Adequacy of follow-up of cohorts?
star that mean the study meet that assessment question in the NOS and 0 means not meet the assessment question.
Patient Characteristics
Table 3 summarizes the demographic and clinical characteristics of patients across the included studies. The eligibility criteria for patient selection varied slightly among the studies, with common criteria including severe-to-profound deafness resulting from meningitis and candidacy for CI. The number of patients with meningitis included in each study ranged from 8 to 60; the age distribution varied but was focused on pediatric populations. Causative organisms for meningitis included Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis, Streptococcus suis meningitis, and unspecified pathogens. The onset of ossification following meningitis typically occurs between 4 to 8 weeks and studies often categorized patients into subgroups based on cochlear ossification patterns observed radiologically. These subgroups included minimal, partial, and total ossification, which influenced the surgical techniques and outcomes. Furthermore, several studies have demonstrated a correlation between the time of CI postmeningitis and both the incidence and severity of cochlear ossification. These studies indicate that early CI (within 6 months of meningitis) is associated with improved auditory outcomes, minimized ossification progression, and higher likelihood of successful full electrode insertion. Conversely, prolonged delays (>6 months postmeningitis) correlate with poorer auditory performance, advanced ossification patterns, and an increased likelihood of partial electrode insertion due to obstructed cochlear lumina.15,20,22,24,26,29 Chen et al 33 demonstrate that S. suis meningitis triggers earlier ossification onset, occurring as early as 2 to 10 days postinfection, with rapid progression to fibrosis within 3.5 months. Moreover, early CI performed within 4 to 6 weeks of diagnosis reduces the incidence of cochlear ossification. In contrast, delayed CI beyond 6 weeks associated with elevated neural response telemetry thresholds, indicative of greater neural damage. In addition, certain studies reported the presence of neurological sequelae or learning disabilities in the patients, although the specific prevalence varied (Table 3).
Patient Characteristics.
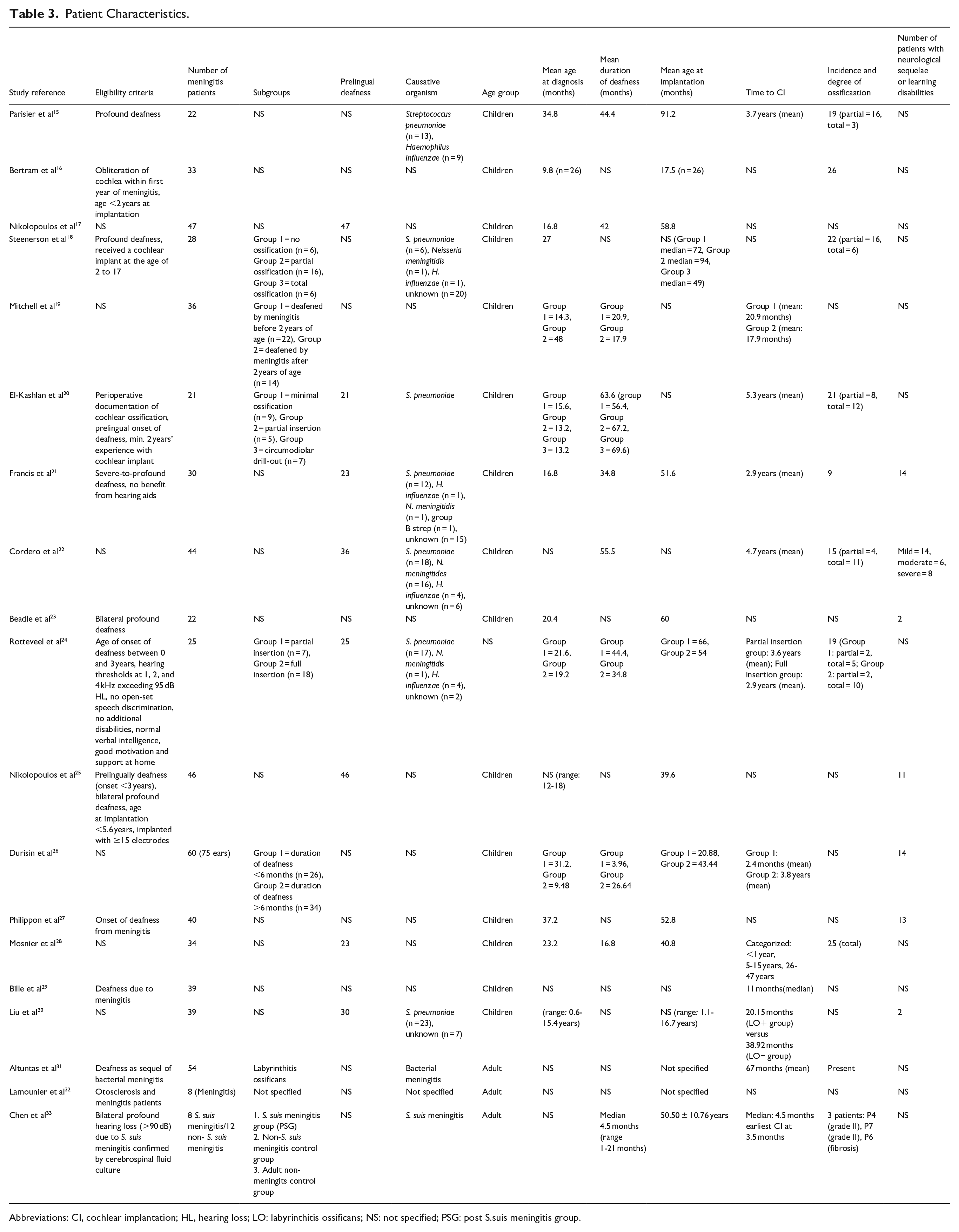
Abbreviations: CI, cochlear implantation; HL, hearing loss; LO: labyrinthitis ossificans; NS: not specified; PSG: post S.suis meningitis group.
Surgical and Implantation Details
This systematic review included 19 studies that described various surgical techniques, implant devices, and speech processing strategies employed in CI for patients with postmeningitis HL. The surgical techniques varied across the studies, with cochleostomy and circumodiolar drill-out being the common approaches. The implant devices predominantly included models from Nucleus (Nucleus 22 and Nucleus 24) and other manufacturers such as Advanced Bionics and MED-EL. The processing strategies varied widely and encompassed SPEAK, MPEAK, ACE, and other proprietary algorithms tailored to individual patient needs.
The mean length of follow-up ranged from 12.0 to 89.8 months across the studies. The feasibility and outcomes of full insertions were reported, with varying success rates that were influenced by ossification levels and surgical approaches. Preoperative imaging using computed tomography (CT) or magnetic resonance imaging (MRI) was commonly used to assess cochlear anatomy and guide surgical planning. The insertion methods targeted the scala tympani, although variations such as scala vestibuli and circumodiolar drill-out were noted (Table 4).
Surgical and Implantation Details.
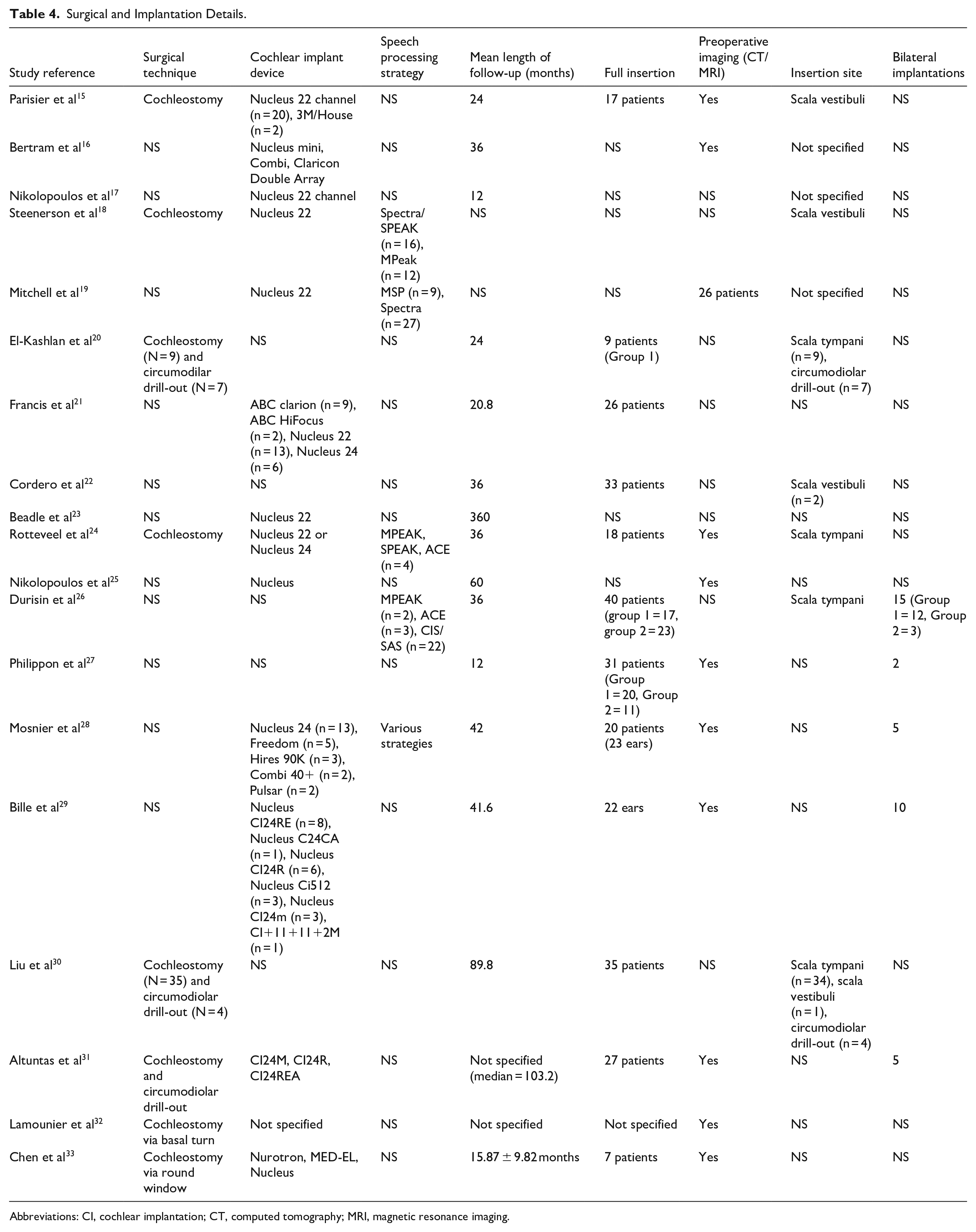
Abbreviations: CI, cochlear implantation; CT, computed tomography; MRI, magnetic resonance imaging.
Audiological Outcomes
All studies reported a consistent improvement in the audiological outcomes following CI. Of the 11 studies that described the preimplantation hearing status, all patients exhibited severe-to-profound bilateral HL. Of these, 6 studies employed statistical analysis to demonstrate significant enhancements in audiological outcomes after CI.20,21,24-26,30 The commonly-used primary outcomes were speech perception scores and categorical auditory performance (CAP).15,17,25,27,29
A meta-analysis of closed-set speech perception tests across 3 studies16,18,21 (n = 110 participant) yielded a pooled mean score of 58.4% (95% CI: 54.3-62.6) with moderate heterogeneity (P = .135; I² = 43.1%) (Figure 2).
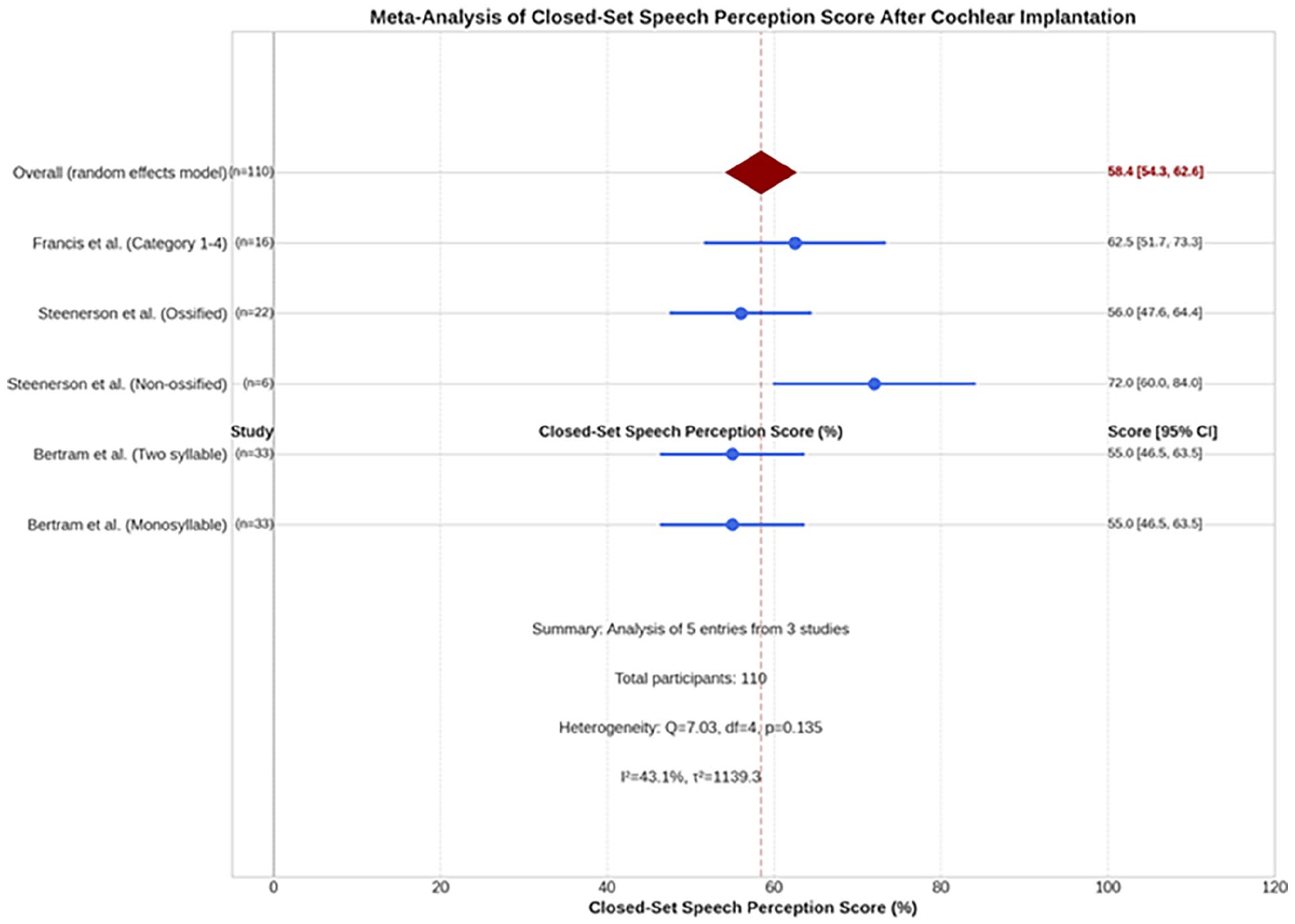
Forest plots of 3 studies exhibiting the mean score of close-set speech perception after cochlear implantation in postmeningitis hearing loss. The overall pooled estimate was 58.4% (95% CI: 54.3-62.6) with moderate heterogeneity (I² = 43.1%, P = .135). n, number; CI, confidence interval.
Moreover, a meta-analysis examined open-set speech perception outcomes included 7 studies18,19,21,22,24,30,32 (n = 194 participants). The pooled mean estimate of the open-set speech perception score was 46.3% (95% CI: 26.3-66.2) with substantial heterogeneity across studies (Q statistic, I² >50%) (Figure 3). Subgroup analysis was conducted based on cochlear ossification status and electrode insertion depth. Four studies18,22,24,30 (n = 58) involving patients with non-ossified cochleae and full electrode insertion reported significantly-better speech perception outcomes, with a pooled mean estimate of 76.2% (95% CI: 65.8-86.7) and moderate heterogeneity (I² = 42.3%). Conversely, Four studies18,22,24,30 (n = 62) involving patients with ossified cochleae and partial electrode insertion demonstrated significantly-poorer outcomes, with a pooled mean estimate for this cohort was 31.4% (95% CI: 0.2-62.7) and substantial heterogeneity (I² = 93.7%) (Figure 4).
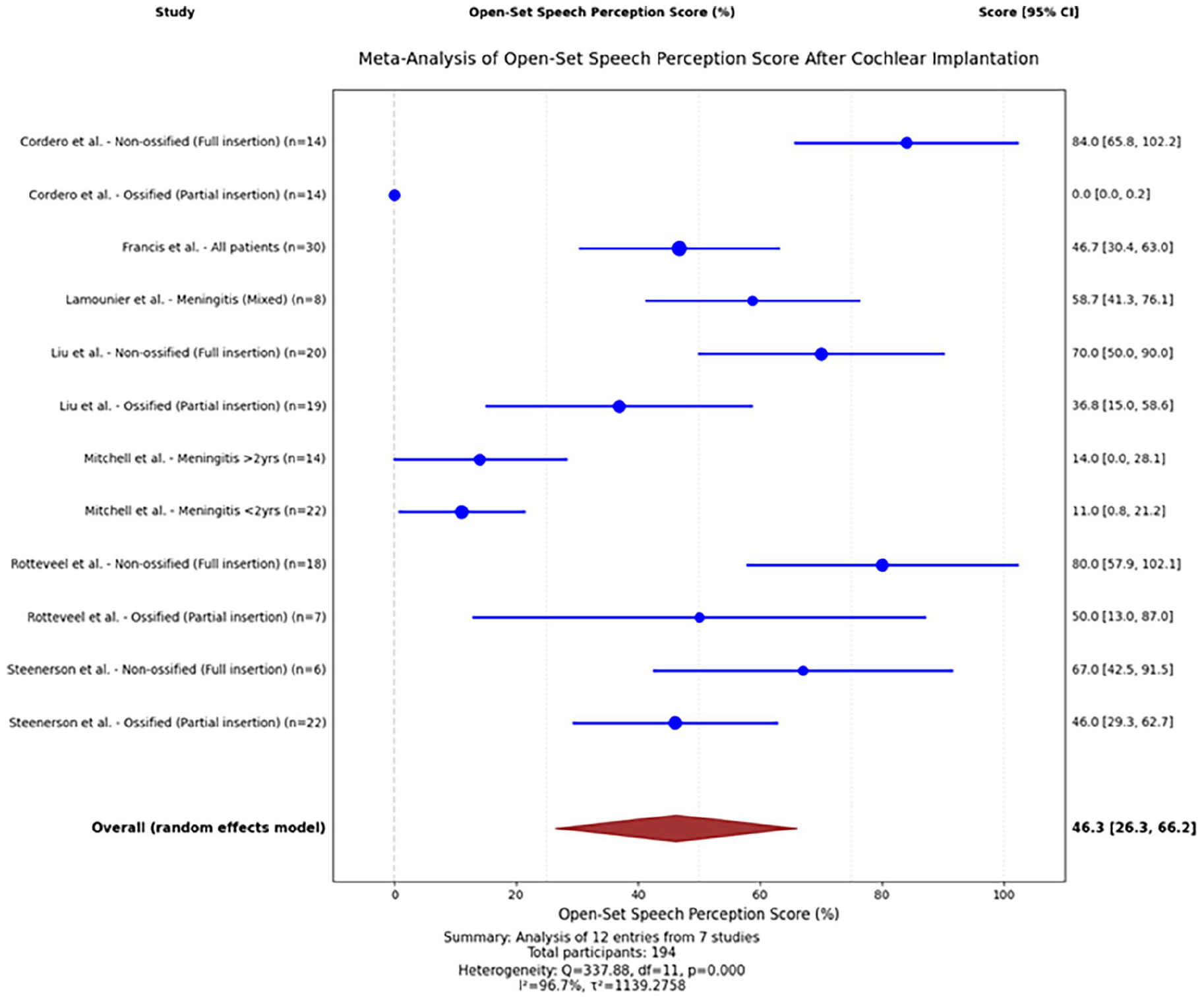
Forest plot of 7 studies assessing the open-set speech perception score after cochlear implantation in patients with postmeningitis hearing loss. Scores varied widely across studies (range: 0.0%-84.0%). Pooled estimate: 46.3% (95% CI: 26.3-66.2; high heterogeneity: I² = 98.7%, P < .001). n, number; CI, confidence interval.
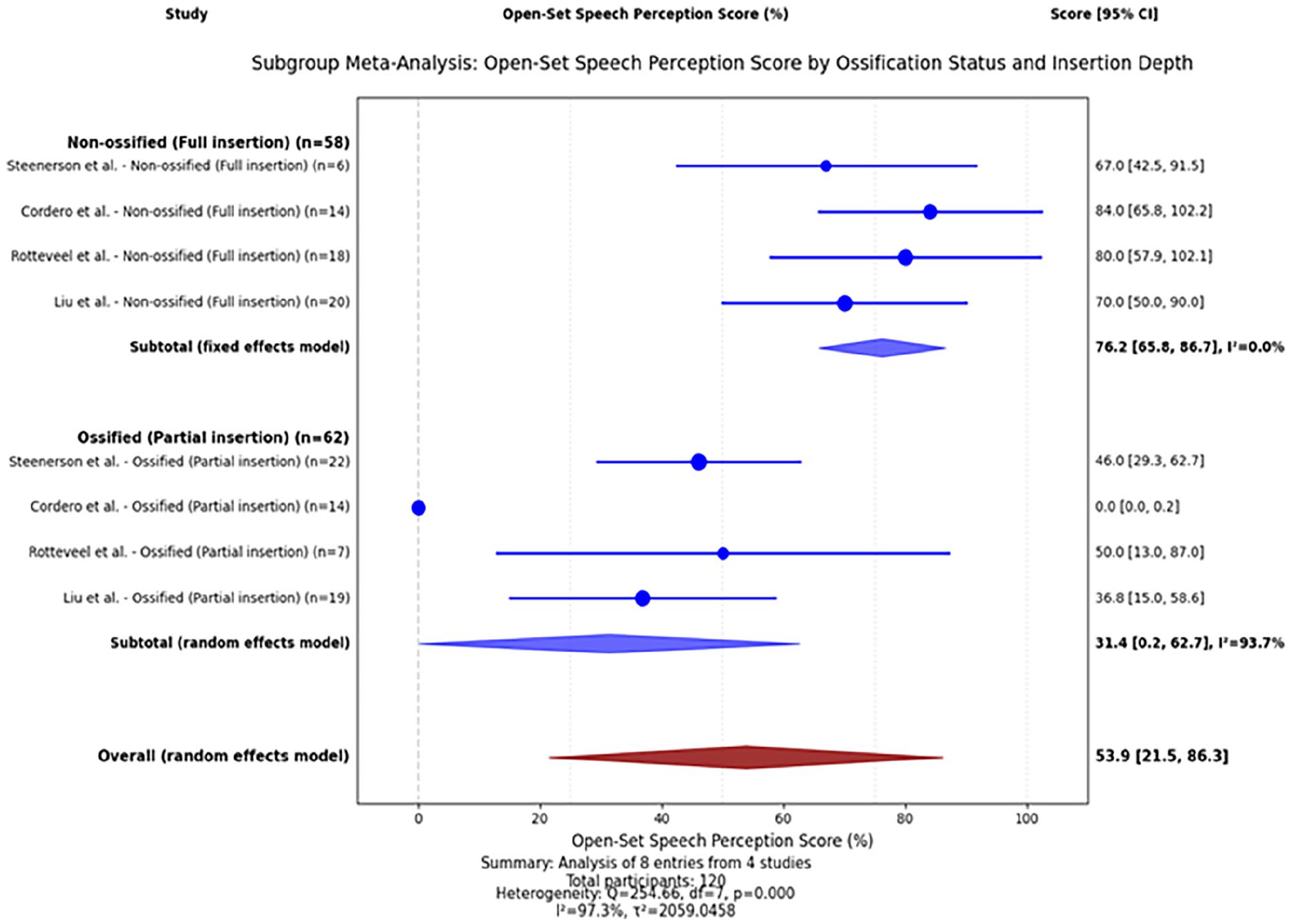
Forest plots showing the subgroup analysis of open-set speech perception score according to cochlear ossification status and electrode insertion depth after cochlear implantation in patients with postmeningitis hearing loss. Non-ossified cochleae with complete electrode insertion had better speech perception scores (76.2%) versus ossified cochleae with partial insertion scores (31.4%). Heterogeneity was low (I² = 0%) and high (I² = 93.7%). Overall heterogeneity (I² = 97.3%, P < .001). n, number; CI, confidence interval.
A meta-analysis of 6 studies15,23,25,29,31,33 (n = 170 participants) revealed a pooled mean CAP score of 5.5 (95% CI: 5.3-5.7) with a statistically-significant heterogeneity (P < .0001, I² = 96.4%) (Figure 5). Subgroup analyses showed that studies of non-ossified cochlea with complete electrode insertion reported a pooled mean CAP score of 5.3 (95% CI: 4.9-5.7). In contrast, studies of ossified cochleas with incomplete electrode insertion yielded a pooled mean CAP score of 3.8 (95% CI: 3.3-4.3) (Figure 6). Additionally, in 3 studies involving a total of 88 patients, the mean postimplantation CAP exceeded 5.17,25,27
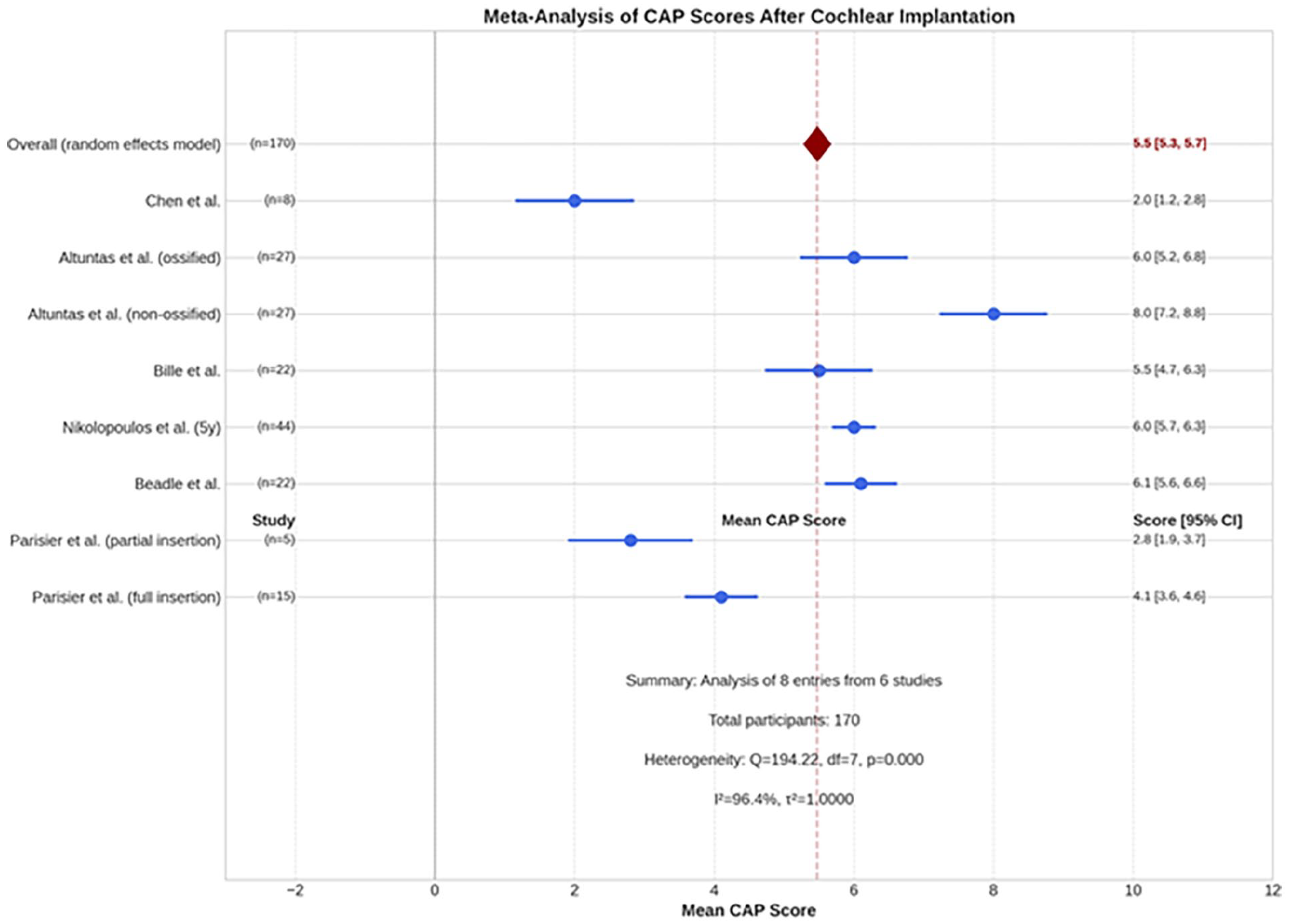
Forest plot of 6 studies assessing the categories of auditory performance (CAP) score after cochlear implantation in patients with postmeningitis hearing loss. Pooled mean score: 5.5 (95% CI: 5.3-5.7). Individual scores ranged from 2.0 (Chen et al) to 8.0 (Altuntas et al, non-ossified). Significant heterogeneity (I² = 96.4%, P < .001). n, number; CI, confidence interval.
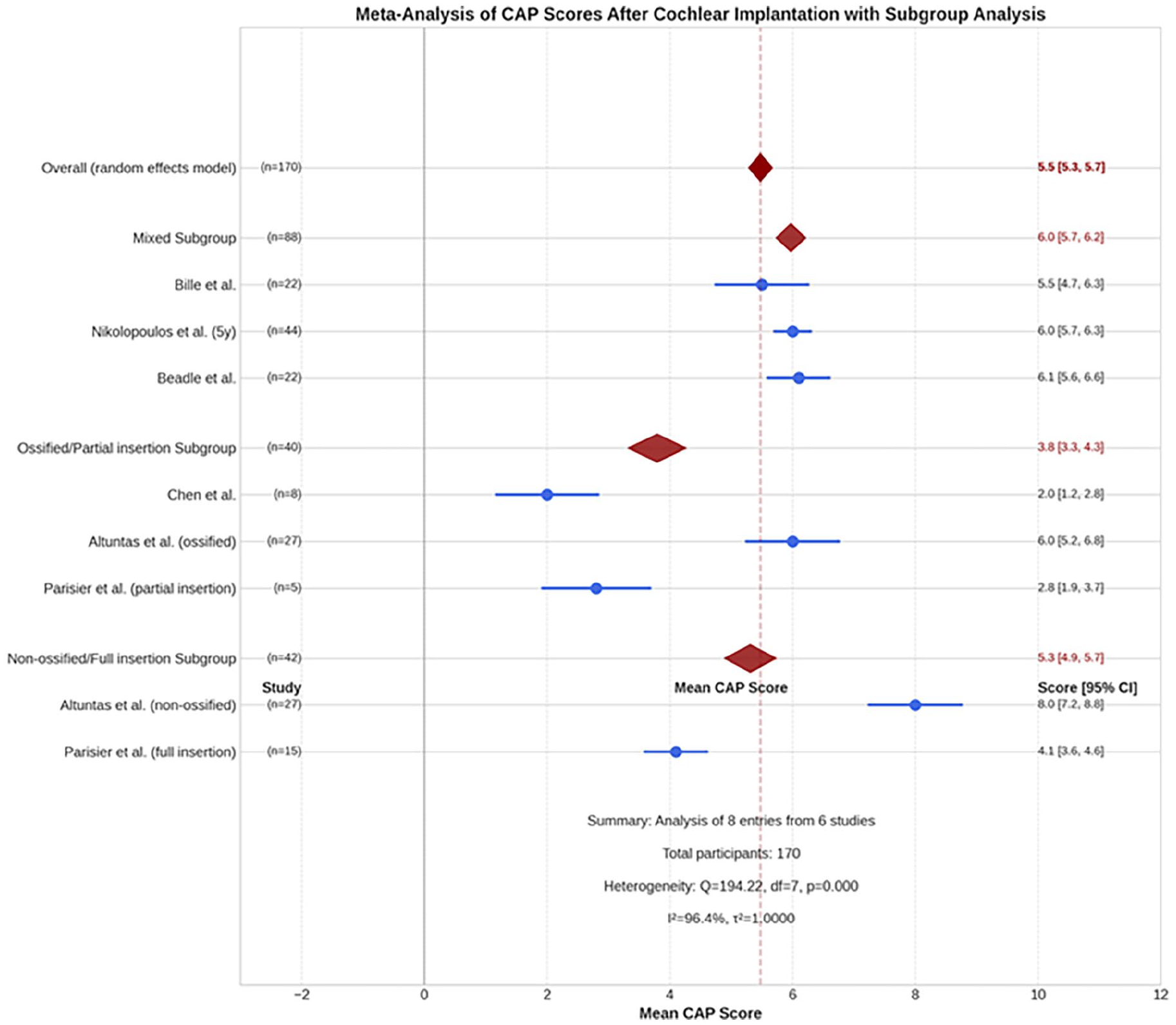
Forest plots showing the subgroup analysis of the categories of auditory performance (CAP) score according to cochlear ossification status and electrode insertion depth after cochlear implantation in patients with postmeningitis hearing loss. Subgroups (mixed, ossified/partial insertion, non-ossified/full insertion) demonstrated varying means with significant heterogeneity (I² = 96.4%, P < .001). n, number; CI, confidence interval.
On the other hand, Chen et al 33 report that patients with post-S. suis meningitis exhibited significantly-lower CAP scores than the non-S. suis meningitis control group, despite having equivalent preoperative hearing status and follow-up duration. Notably, even with a shorter duration of deafness, the CAP score in the post-S. suis meningitis group remained lower than that in the non-S. suis meningitis control group.
Bilateral implantations were addressed in several studies, which highlighted the benefits and considerations associated with simultaneous implantation. These benefits included improved speech perception in the presence of noise, better sound localization, enhanced overall auditory experience compared with unilateral implantation and mitigates the risk of complications associated with advanced ossification, such as partial electrode insertion or the necessity for extensive surgical interventions, including circummodiolar drill-out procedures. Durisin et al 26 reported that simultaneous bilateral implantation resulted in significantly-better open-set test outcomes, which showed the benefits of receiving auditory input from both ears and avoid the complications associated with advanced ossification. This observation is in line with the findings of Nikolopoulos et al 25 who noted that bilateral implants provided a more balanced and complete auditory experience, which considerably enhanced the patients’ quality of life. Furthermore, this approach decreased the overall anesthesia exposure and recovery time.
Patients who undergo CI within a short duration of deafness achieve better audiological outcomes. For instance, Durisin et al found that patients implanted within 6 months of their deafness onset exhibited superior results in open-set tests compared with those implanted later than 6 months after the onset. 26 Similarly, Philipon et al reported that 37% of pediatric patients (mean duration of deafness = 25 months) achieved open-set speech discrimination, whereas only 23% of adult patients (mean duration of deafness = 336 months) reached this milestone. 27
In addition, the outcome of postoperative hearing rehabilitation can be influenced by factors such as the age at which deafness began, the duration of normal auditory function before meningitis, and the age at which CI was performed. Investigations have noted that older age at the onset of deafness, a period of normal auditory experience before illness, and younger age at implantation were linked to favorable outcomes in postoperative hearing rehabilitation. In their study, Mosnier et al 13 examined the influence of advancements in CI technology by comparing the audiological outcomes between patients implanted before and after 2001. The researchers observed considerably-greater improvement in patients implanted after 2001, and speech scores comparable to those of control subjects were achieved. 28 The detailed audiological outcomes are presented in Table 5.
Primary Outcomes (Audiometry and/or Speech Perception and/or Speech Production).
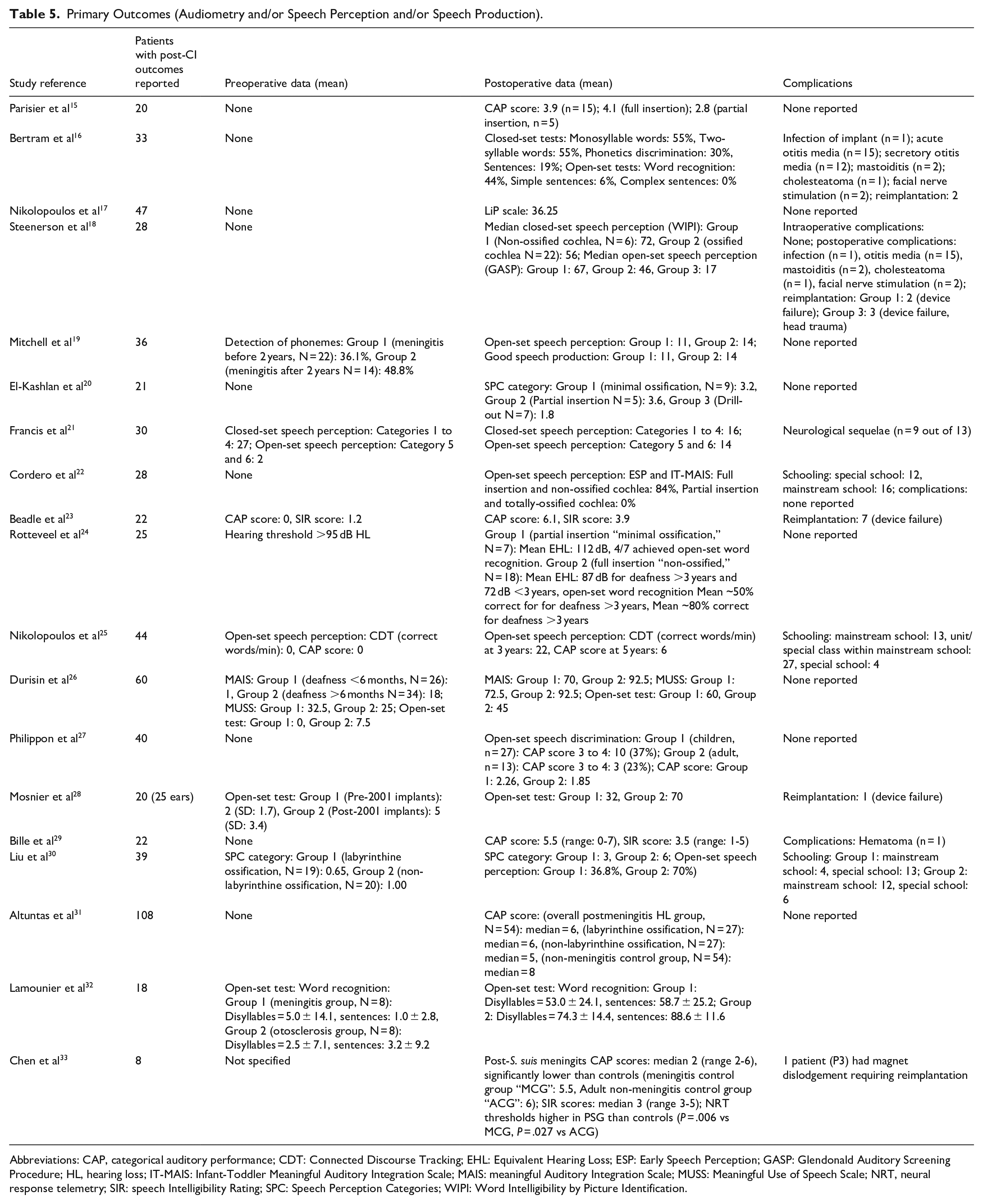
Abbreviations: CAP, categorical auditory performance; CDT: Connected Discourse Tracking; EHL: Equivalent Hearing Loss; ESP: Early Speech Perception; GASP: Glendonald Auditory Screening Procedure; HL, hearing loss; IT-MAIS: Infant-Toddler Meaningful Auditory Integration Scale; MAIS: meaningful Auditory Integration Scale; MUSS: Meaningful Use of Speech Scale; NRT, neural response telemetry; SIR: speech Intelligibility Rating; SPC: Speech Perception Categories; WIPI: Word Intelligibility by Picture Identification.
Surgical Complications and Reimplantation
Postoperative complications were reported in 5 studies16,23,27,28,33 that observed different complications such as infection of the implant, stimulation of the facial nerve, secretory otitis media, acute otitis media, wound infection, and hematoma above the insertion site. In addition, 13 patients experienced device failure, and 16 patients required reimplantation because of infection of the implant, head injury, or magnet dislodgement. The specific results are presented in Table 5.
Discussion
This systematic review of CI outcomes in patients with postmeningitis HL highlights the significant advancements and challenges in managing this complex condition. Across the reviewed studies, which were performed from 1993 to 2024 and were focused on European and North American populations, several critical themes emerged regarding patient demographics, surgical techniques, audiological outcomes, and broader implications for clinical practice and patient care.
A consistent finding across the studies was the heterogeneous nature of patient demographics and clinical presentations among individuals with postmeningitis HL. Patients with severe-to-profound deafness following meningitis caused by various pathogens, such as Streptococcus pneumoniae, Haemophilus influenzae, S. suis meningitis, and Neisseria meningitidis, were included in this study. The mechanism of HL after acute meningitis can involve either direct damage to the organ of Corti or spiral ganglion owing to the toxic effect of bacteria or the formation of cochlear ossification. Impairment of the central auditory pathway is another possible etiology. This etiological diversity emphasizes the complex pathophysiological mechanisms underlying postmeningitis hearing impairment, which can influence the severity and progression of auditory deficits.3,17,34,35 The mean age at diagnosis ranged from early childhood to adolescence, which reflects the age range at which meningitis-induced deafness manifests. Mean ages at implantation varied across studies and were influenced by factors such as clinical protocols and individual patient circumstances. The mean length of follow-up ranged from 12 to 89.8 months across the studies, which indicates the longitudinal nature of CI outcome assessment.
Moreover, the categorization of patients based on cochlear ossification patterns provided valuable insights into the anatomical changes that affect surgical planning and implantation outcomes. Previous studies have often classified patients into groups with minimal, partial, or total cochlear ossification, which significantly influenced the feasibility of electrode insertion and subsequent auditory nerve stimulation.12,36,37 These classifications are aligned with findings in the broader literature, which suggests that cochlear ossification complicates surgical outcomes and may necessitate modified approaches to optimize electrode placement and functional outcomes.38,39
The variability in surgical techniques, including cochleostomy and circumodiolar drill-out, shows that strategies to address the anatomical challenges in postmeningitis cochleae are constantly evolving. More extensive ossification is associated with greater challenges in electrode insertion and may necessitate advanced procedures such as the circummodiolar drill-out technique. El-Kashlan et al 20 and Parisier et al 15 documented the increased complexity of surgeries in patients with severe ossification and stated that these cases often require more intricate surgical approaches to ensure successful electrode placement. Each approach is tailored to mitigate the risks associated with inner ear pathology and optimize the electrode positioning within the scala tympani or scala vestibule.20,26
Advances in imaging modalities, such as CT and MRI, have facilitated preoperative planning by providing detailed anatomical assessments, guiding surgeons in selecting the most appropriate insertion method based on individual cochlear morphology. 28
Furthermore, the selection of implant devices from various manufacturers, such as Nucleus, Advanced Bionics, and MED-EL, signifies the ongoing innovations in electrode design and speech processing algorithms. These developments are aimed at enhancing the auditory perception and speech discrimination capabilities tailored to the specific needs of patients with postmeningitis HL, thereby improving the overall communication outcomes.28,29 Importantly, the depth of electrode insertion emerged as a critical factor that influences the audiological outcomes. Subgroup meta-analyses of open-set speech perception and CAP scores, stratified by cochlear ossification status and electrode insertion depth, demonstrating superior performance in patients with full electrode insertions than those with partial insertions (Figures 4-6). This finding align with broader trends, suggesting that cochlear ossification and partial electrode insertion may adversely affect auditory performance outcomes post-CI.
Shorter electrodes are often preferred for patients with partial cochlear ossification to facilitate insertion and avoid complications. Rotteveel et al 24 and Durisin et al 26 reported the use of various electrode designs tailored to the degree of cochlear ossification, noting that shorter electrodes can help achieve full insertion and improve the hearing outcomes. This customization of the electrode choice indicates the need for individualized surgical planning to maximize the benefits of CI. Surgical precision and intraoperative monitoring are thus vital for optimizing the electrode placement and maximizing the neural stimulation, as validated by studies evaluating the electrode position and the associated auditory nerve response.39,40,41
The findings from this systematic review, supported by existing literature, strongly advocate for the consideration of simultaneous bilateral CI in patients with postmeningitis HL.32,42 Notably, a key prognostic factor in meningitis-related hearing impairment is cochlear ossification, which can rapidly progress and significantly impact surgical outcomes. Since advanced ossification can hinder electrode insertion and reduce CI effectiveness, simultaneous bilateral CI is strongly recommended for patients with bilateral severe-to-profound HL to maximize the window of surgical opportunity before ossification occurs in both cochleae. When evaluating the advantages of bilateral implantation, the primary focus should be on the risk of progressive cochlear ossification and fibrosis processes that can advance within weeks or even days rather than on concerns about auditory deprivation in the second ear, which unfolds over a much longer timespan, typically measured in years.13,33,43
The duration of deafness before the implantation and the timing of implantation relative to the onset of ossification are a crucial factors influencing the CI outcomes. Studies have observed that cochlear ossification can begin as early as 2 to 4 weeks postmeningitis. Additionally, our findings support the notion that a shorter duration of deafness before implantation is associated with better audiological outcomes. Durisin et al 26 demonstrated that patients implanted within 6 months of the onset of deafness achieved superior open-set speech perception compared with those with a longer duration of deafness. This finding has been corroborated by Philippon et al, 27 who found that a shorter duration of deafness resulted in a higher speech discrimination score, underscoring the importance of timely intervention to avoid the complications associated with advanced ossification. These findings reinforce the importance of early diagnosis and intervention to achieve optimal benefits from CI in patients with postmeningitis HL.
The audiological outcomes following CI consistently demonstrated significant improvements in speech perception and auditory performance across the reviewed studies.
CAP scores, along with close- and open-set speech perception, are crucial measures of CI success. This review identified that a meta-analysis of CAP scores showed significant improvements, indicating substantial improvements in auditory performance (Figure 5). Moreover, Durisin et al 26 reported CAP scores of >5 in a high proportion of patients, which denotes the ability to understand speech without visual cues. Similarly, a meta-analysis of close- and open-set speech perception exhibited significant gains (Figures 2 and 3). In addition, studies by Francis et al 21 and Nikolopoulos et al 25 have established the improvement in audiological outcomes following CI, which confirms the effectiveness of the device in restoring hearing in patients with postmeningitis deafness. These findings agree with the broader literature documenting the efficacy of CI in restoring the auditory function for patients with severe HL of varied etiologies.44-48
The presence of neurological disabilities significantly affects the CI outcomes. Francis et al 21 observed that patients with additional neurological impairments, such as hydrocephalus, exhibited poorer speech perception outcomes than those without such disabilities. This difference could be attributed to impaired central auditory pathway or impaired intellectual ability of the patient, which may affect the audiological rehabilitation after CI. This finding suggests that while CI can provide significant benefits, the extent of improvement may be limited by underlying neurological conditions. In addition, the age at the onset of deafness and the presence of a period of normal auditory input are critical factors. Cordero et al 22 noted that children with an early onset of deafness and no prior auditory input had more challenging outcomes than those with some period of normal hearing, which stresses the role of early-auditory experience in optimizing the CI benefits.
Finally, despite advancements in surgical techniques and implant technologies, key challenges related to postoperative complications and reimplantation were identified in this review. Infection rates, device failures, and middle ear complications were documented in several studies. Therefore, vigilance is needed in managing the perioperative risks and optimizing the long-term implant durability. 26 Comprehensive preoperative assessment and postoperative care protocols are required to mitigate the adverse events and ensure optimal patient outcomes.
Limitations of the Study
Although this systematic review on CI outcomes in patients with postmeningitis HL is comprehensive, several limitations warrant consideration. First, the heterogeneity in study designs, patient demographics, and outcome measures across the included studies might introduce inherent variabilities and potential biases in synthesizing the findings. Variations in surgical techniques, implant devices, and follow-up durations among the studies might limit the generalizability of the findings and complicate direct comparisons of intervention outcomes. Moreover, the predominantly-retrospective nature of many of the included studies imposes constraints in establishing causal relationships between CI and long-term audiological, educational, and societal outcomes. In addition, the exclusion of non-English language publications and unpublished studies may introduce publication bias and potentially skew the overall findings toward studies with positive outcomes. Moreover, the limited number of studies focusing on certain aspects, such as the effect of specific pathogens or the influence of preimplantation hearing rehabilitation, indicates gaps in current literature that may affect the comprehensiveness of our conclusions. Finally, while the review provides a broad overview of the outcomes, detailed patient-level data and standardized reporting of complications were not consistently available across all studies, which limits the depth of analysis in certain areas.
Conclusion
CI is a transformative intervention for patients with severe HL resulting from meningitis, offering substantial improvements in auditory function and communication abilities. The integration of multidisciplinary approaches, including surgical expertise, technological innovation, and comprehensive rehabilitation strategies, is essential to optimize the outcomes and meet the diverse clinical and functional needs of postmeningitis CI recipients.
Footnotes
ORCID iDs
Ethical Consideration
No ethics approval or informed consent is required for this systematic review and meta-analysis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
