Abstract
Adenoid cystic carcinoma (ACC) is a malignancy most commonly found in the major salivary glands. It can, however, arise from minor salivary glands throughout the upper aerodigestive tract. It is typically slow-growing and asymptomatic, though presentation varies with tumor location, pattern of infiltration, and degree of perineural invasion. ACC is characterized as an aggressive malignancy due to high rates of local recurrence and distant metastasis, with the lungs, bone, and liver commonly implicated. ACC spreading to the internal auditory canal (IAC) is highly unusual and has not been reported. We present the case of a 40 year-old male with no significant past medical history, found to have nasopharyngeal ACC that spread to the IAC resulting in multiple cranial neuropathies. The challenging location and complex path of spread along the skull base presented a significant barrier to surgery, though after completion of definitive radiation therapy, the patient experienced substantial reductions in mass effect and symptomatic relief. This report offers additional insight into the patterns of spread for ACC, emphasizing the importance of recognizing the IAC as a potential site of involvement. Early identification may significantly expedite management and prevent serious morbidities.
Introduction
Adenoid cystic carcinoma (ACC) is a rare malignancy of secretory glands with a propensity for perineural invasion and hematogenous spread. ACC accounts for 1% of all head and neck malignancies and 10% of all salivary gland malignancies. 1 It occurs more frequently in the minor salivary glands but may also occur in the parotid, submandibular, and sublingual glands. Other reported locations in the head and neck include the tongue, palate, paranasal sinuses, nasopharynx, and external auditory canal (EAC). ACC typically presents as an asymptomatic, slow-growing mass in the head and neck region. Pain and paresthesia are rarely encountered and are likely a consequence of the tumor’s tendency for perineural invasion.2,3 Presentations may vary substantially based on tumor location. For example, nasopharyngeal tumors may result in epistaxis, nasal obstruction, and eustachian tube dysfunction, while tumors located in the skull base may cause ocular dysfunction or cranial nerve palsies. 4 Epidemiologically, ACC has a slight female predominance (60%-40%) and occurs more frequently in the fifth and sixth decades of life. 4
ACC is associated with high rates of both local recurrence and metastasis. The most common sites of metastasis are the lungs followed by bone, liver, skin, breast, and sometimes the brain. Although ACC can disseminate to various locations via perineural invasion, hematogenous spread, and the lymphatic system, it has not been documented to spread to the internal auditory canal (IAC). Involvement of the auditory canal—specifically the IAC—is exceedingly rare. This article details a case of ACC of the nasopharynx with involvement of the IAC, highlighting an unusual presentation.
Case Presentation
A 40 year-old male with no known past medical history presented to the emergency department with 2 days of right-sided facial droop and right eye sensitivity to light. He was initially diagnosed with Bell’s Palsy and prescribed acyclovir. After 1 month, the patient returned to the emergency department with intermittent epistaxis and persistent right-sided facial droop, at which point he was discharged with supportive care instructions. Three months later the patient again returned to the emergency department, this time with a new headache, right jaw pain, and vertigo. The patient also complained of tinnitus, which was resolved, and otalgia for 1 week. On physical exam, a right-sided facial nerve palsy with a House–Brackmann (HB) grade III/IV was noted due to complete paralysis of the forehead and mouth with preserved right eye closure. Right cranial nerve VI palsy was also noted by impaired abduction of the right eye. Dix–Hallpike was performed and the patient complained of vertigo with leftward head movement. Rinne test showed air conduction greater than bone conduction on the left, and air conduction equal to bone conduction on the right. Weber test lateralized to the left. Overall results were consistent with right-sided sensorineural hearing loss. A computed tomography (CT) scan of the head was ordered which demonstrated right-sided opacification of mastoid air cells. Antibiotics were ordered for presumed mastoiditis, prednisone taper for facial palsy and sudden hearing loss, and CT as well as magnetic resonance imaging (MRI) IAC for further evaluation.
CT and MR IAC demonstrated an infiltrative and inflammatory mass centered on the right petrous apex and extending into the right skull base and nasopharynx. The mass was noted to be circumferential to the carotid artery extracranially (Figure 1A). Dural involvement is noted with bony destruction in the right petrous apex. Additionally, a hypointense enhancing lesion is seen in the right IAC (Figure 1B). Nasal endoscopy demonstrated asymmetric fullness of the right fossa of Rosenmüller. Otoscopy at this time revealed a red mass behind the tympanic membrane and a middle ear effusion. The patient subsequently developed worsening right-sided facial nerve palsy (HB VI/VI) and decreased sensation of right cranial nerve V in the distribution of V1 to V3. A right-sided transcanal middle ear biopsy was performed, with pathology results demonstrating atypical small blue cells (Figure 2) and immunohistochemistry (IHC) positive for CK5/6 and patchy pankeratin, but negative for P63, chromogranin, and synaptophysin. Additional testing showed the mass to be a basaloid neoplasm with IHC p16 negative, p40 negative, mixed cytokeratin (AE1/AE3/Cam52) positive, INSM1 negative, CD117 positive, Sox 10 negative, high risk Human Papilloma virus in situ hybridization negative, and Ki-67 <5%. Due to the indeterminate nature of the mass, a plan for additional nasopharyngeal biopsy was made.
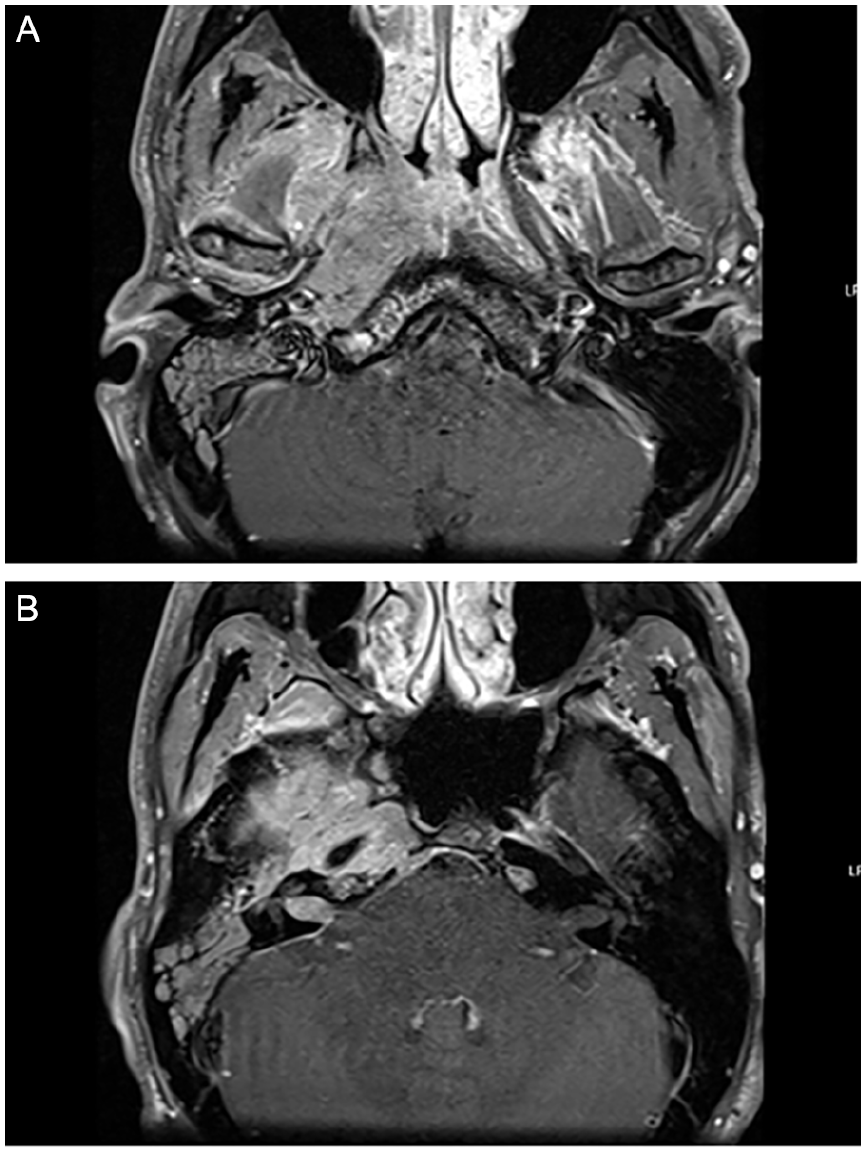
(A, B) MRI IAC demonstrating right nasopharyngeal mass. Axial view T1-weighted MRI with contrast of IAC demonstrating a heterogeneous mass centered at the right petrous apex with extension into the nasopharynx surrounding the carotid canal (A) and a hypointense enhancing lesion in the right IAC with dural involvement causing bony destruction in the right petrous apex (B).IAC; internal auditory canal; MRI, magnetic resonance imaging.
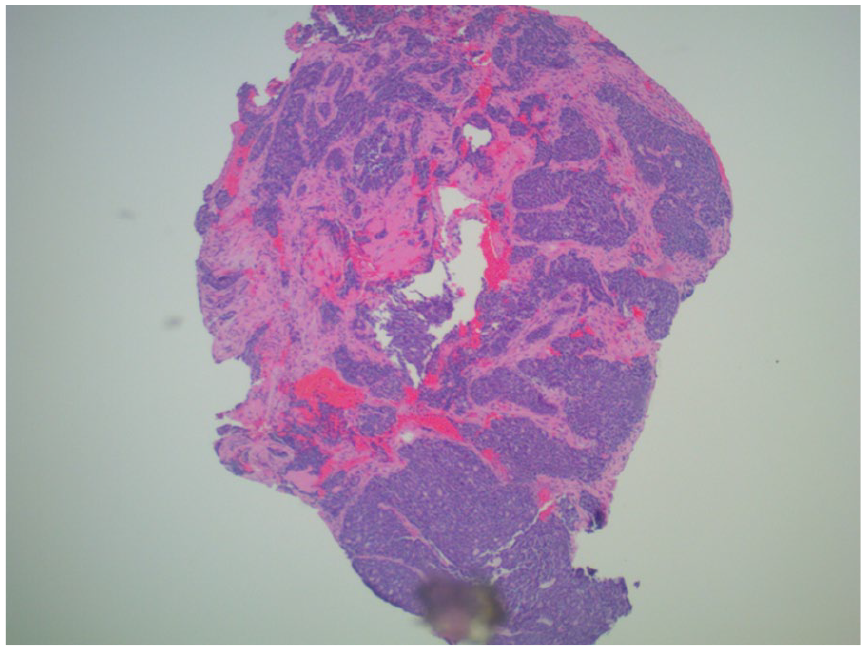
H&E staining of transcanal biopsy specimen. H&E staining of transcanal biopsy specimen demonstrating fibrous connective tissue with infiltrative malignant glands comprised of small blue cells, forming irregular cystic spaces. H&E, hematoxylin and eosin.
Histology results of the right nasopharyngeal biopsy demonstrated fragments of respiratory mucosa with subepithelial proliferation of nerve bundles and lymphocytic infiltration. Final pathology demonstrated an epithelial neoplasm with cribriform and tubular growth patterns. IHC was performed with differential diagnosis including ACC and polymorphous adenocarcinoma, low grade. Given the pathology findings, tumor markers, and involvement of multiple cranial nerves, a presumed diagnosis of ACC was made.
Postbiopsy, the patient complained of ear pain and drainage. The mass was noted to have grown out of the middle ear and into the EAC. Fluid was suctioned and cortisporin drops were prescribed to prevent superinfection. Given the primary site location in the nasopharynx and intracranial involvement, surgical management was not recommended. Chemoradiation was recommended. The patient completed a course of definitive radiation therapy, receiving 60 Gray in 30 fractions. Posttreatment, restaging facial MRI demonstrated significantly decreased size of the right skull base/right nasopharyngeal mass with improved involvement of the right sphenoid bone, right cavernous sinus, right foramen ovale, and right middle cranial fossa. There was also improved involvement of the right orbital apex and interval resolution of vasogenic edema involving the right temporal lobe. At a follow-up visit 3 months after treatment, the patient reported resolution of pain, improvements in eye mobility and diplopia, and decreased symptoms in the distribution of the trigeminal nerve.
Discussion
ACC is an aggressive malignancy with high rates of both local recurrence and distant metastasis. Delayed metastases are also common and may occur up to decades after treatment of the primary tumor or establishment of loco-regional control. 5 This may be explained by the tumor’s slow growth and propensity for perineural invasion, with tumor cells lying dormant beyond surgical margins.2,3 Singh et al 6 analyzed patterns of spread among various ACC of the head and neck, characterizing commonly implicated routes of perineural invasion. Tumors located in the palate tend to spread along the greater and lesser palatine nerves to the pterygopalatine fossa (PPF) and foramen rotundum. Tumors located in the nasal cavity and maxillary sinus tend to spread along the infraorbital nerve to the PPF and foramen rotundum. When located in the retromolar trigone, it may spread along the inferior alveolar and lingual nerves to the foramen ovale. Parotid tumors invade the facial nerve and auriculotemporal branch of V3. 6 Distant metastases may also occur via hematogenous spread of ACC to the lungs and bone. Rarely, ACC exhibits local lymph node involvement via lymphatic spread. Spread to the IAC is highly unusual and has not been thoroughly documented. A thorough literature search was conducted and only 1 paper by Nibhanupudi et al was identified reporting involvement of the IAC. The tumor is described as having epicenter at the petrous temporal bone, eroding through squamous, petrous, and mastoid temporal bone as well as the greater wing of sphenoid, parietal, and occipital bones. There is involvement of the external and IACs, facial canal, and intracranial extension. 7 The degree of IAC involvement is not documented nor readily apparent on imaging. This article may be one of the first reports of ACC with clear extension to the IAC.
Establishing an accurate diagnosis of ACC is imperative—as early recognition can greatly improve survival—and involves ruling out competing diagnoses. The differential diagnosis for ACC also varies with tumor location, but common pathologies with similar presentation include pleomorphic adenoma, basal cell adenoma, basaloid squamous cell carcinoma (BSCC), mucoepidermoid carcinoma, and polymorphous low-grade adenocarcinoma (PLGA). For most competing diagnoses, there are key differentiating features. Pleomorphic adenoma is not invasive and does not demonstrate perineural invasion. BSCC is not as frequently found in salivary glands, demonstrates squamous differentiation on histology, and consistently stains p63-positive. 8 Mucoepidermoid carcinoma is more frequently in the major salivary glands and contains intermediate and goblet cells. 9
Distinguishing PLGA from ACC can pose significant challenges due to similarities in histological appearances but is imperative with regards to management. While ACC exhibits aggressive behavior with frequent recurrence and metastasis 10 as well a 15 year survival rate of only 29.4%, PLGA only infrequently demonstrates recurrence or metastasis and rarely leads to death. 11 As such, the management of ACC is typically much more involved than that of PLGA and may incorporate neck dissection and adjuvant chemoradiation. Differentiating the 2 relies on a combination of factors including histopathology, growth patterns, and IHC. Histopathology and growth patterns are useful differentiators of ACC from PLGA. Historically, ACC was first termed “cylindroma” due to the histopathologic presence of cylinders of epithelial/secretory cells and a hyaline stroma. 1 ACC is typically described as biphasic due to the presence of both epithelial and myoepithelial components. These components are arranged in 1 of 4 growth patterns: tubular, cribriform, solid, or mixed. This helps differentiate ACC from PLGA, as PLGA typically only demonstrates 1 cell type. Furthermore, the biphasic nature of ACC results in patchy as opposed to diffuse staining for S100 and CK7 on IHC. Although there is controversy in the literature surrounding the overall utility of immunohistochemical markers in distinguishing PLGA from ACC, studies have demonstrated that ACC exhibits stronger expression of c-kit (CD117), Ki-67, smooth muscle actin, and muscle-specific actin.12-14 In this case, the biopsy of the nasopharyngeal mass demonstrated tubular and cribriform growth patterns, patchy staining, and strong expression of CD117, findings favoring a diagnosis of ACC over PLGA.
Conclusion
ACC is a rare, slow-growing malignancy of the secretory glands with a tendency for perineural invasion and hematogenous spread. Though ACC has been shown to spread to distant peripheral sites such as the lungs, bone, and liver, it more frequently exhibits centripetal spread toward the central nervous system. This can result in involvement of sites such as the PPF or foramen rotundum, which can serve as a hub for further spread. There are also numerous reports of ACC involving the EAC. However, despite its expansive reach, there are minimal to no reports of ACC involving the IAC. Surgery followed by radiation is the typical paradigm for ACC. However, a nasopharynx primary presents a significant surgical challenge. Its indolent characteristics make it notoriously difficult to treat with definitive radiation. Early detection and ruling out competing diagnoses are essential for minimizing associated morbidities. This case highlights an unusual presentation of ACC and is likely the first report of nasopharyngeal ACC spreading into the IAC. Recognition of the IAC as a potential site for spread may expedite management and prevent complications.
Footnotes
Ethical Considerations
Our institution does not require ethical approval (ie, IRB) for case reports outside of obtaining informed consent for publication.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report.
Author Contributions
Taimur Khalid: Preparation of manuscript including literature review, chart review, and writing introduction, case presentation, discussion, and conclusion. Emily S. Sagalow: Contributions to case presentation section and editing/review of entire manuscript. Nathan Lloyd: Editing and review of manuscript. Jo-Lawrence Martinez Bigcas: Editing and review of manuscript. Javi Hartenstine: Acquisition of pathology slides. Matthew Ng: Verification of patient information, assistance with MRI images, review of manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is archived in the electronic health record system and not publicly available as this would constitute a breach of patient privacy.
