Abstract
Objectives:
Pharyngocutaneous fistula (PCF) is the most frequent complication following total laryngectomy (TL). Its incidence increases in the presence of multiple risk factors related to the patient, the tumor, and the surgical procedure. Our study aimed to determine the incidence of PCF and its risk factors.
Methods:
We performed a retrospective study including patients who underwent primary TL (PTL) or salvage TL (STL) between 2007 and 2023. We studied the factors that increased the risk of PCF occurrence, thar are related to the patient, the disease, and the received treatment.
Results:
We included 132 patients with a male-to-female ratio of 32, who received PTL (91%) or STL (9%). The tumor was staged as T4a in 62% of patients. PCF occurred in 17 patients (13%) after a median of 14 days (5-36). PCF was managed conservatively in 13 patients (76%), while a surgical re-suture was required in four patients (24%).
In the univariate analysis, preoperative anemia, STL, widened forward TL, wound infection, and preoperative hypoproteinemia were significantly associated with PCF. In the multivariate analysis, preoperative anemia (P = .012), wound infection (P = .049), and STL (P = .005) were risk factors of PCF. At the multivariate analysis, preoperative anemia [P = .012, odds ratio (OR) = 6.9], wound infection (P = .049, OR = 6.5), and STL (P = .005, OR = 18.45) were independent risk factors of PCF.
Conclusion:
We demonstrated that preoperative anemia, wound infection, and STL were independent risk factors of PCF following TL. Avoiding these circumstances can help prevent PCF occurrence.
Introduction
Pharyngocutaneous fistula (PCF) is defined as a dehiscence in the suture of the neopharynx leading to an abnormal communication between the neopharynx and the neck skin, which causes the leakage of saliva through a cutaneous fistula. 1 It is the most frequent complication following total laryngectomy (TL). Its incidence varies between 3% and 65%.2-4
PCF alters the quality of life and increases treatment costs. PCF not only delays nasogastric tube removal and oral feeding but also increases the length of hospitalization and the rate of wound infections. These consequences delay vocal rehabilitation and adjuvant radiotherapy (RT) and/or chemotherapy (CT).1,4,5
The occurrence of PCF is linked to multiple risk factors that are related to the patient, the disease, or the received treatment. The most-previously-reported factors included RT, chemoradiotherapy (CRT), anemia, comorbidities, advanced tumor stages, and hypopharyngeal invasion.1,2,6,7 The type of pharyngeal closure has also been reported as a factor influencing PCF occurrence; mechanical stapler closure and horizontal closure reduced PCF rates.6,8,9
Our study aimed to determine the risk factors of PCF occurrence and its management modalities.
Materials and Methods
Study Design
We conducted a retrospective analytical study based on the medical records of patients who underwent TL in our otorhinolaryngology head and neck surgery department between 2007 and 2023.
We included all patients who underwent either primary TL (PTL) or salvage TL (STL) for a laryngeal squamous cell carcinoma (SCC). We did not include patients who had a primary hypopharyngeal carcinoma. We excluded patients who underwent TL for another non-laryngeal malignancy. We also excluded patients with incomplete medical records that lacked the necessary data, including demographic and disease features, surgical details, and data concerning PCF diagnosis and management.
Patients and Data Collection
We collected data related to the demographic features of the studied population and the disease characteristics. We also extracted data regarding the received treatments before surgery, and the histopathological features of the disease, and the postoperative complications.
For patients who developed a PCF, we collected the details concerning the timing of its occurrence, its localization, the required conservative or surgical treatment, its duration, and the timing of oral feeding and nasogastric tube removal.
Surgical Procedure and Postoperative Period
All the studied patients unerwent TLs by experienced seniors. Based on the tumor’s local and regional extension, we performed an extended TL by the resection to the pharyngeal mucosa, the skin, and the base of the tongue. In case of a cutaneous extension requiring anterior neck skin excision or a massive pharyngeal extension, the defect was reconstructed using a pectoralis major myocutaneous flap.
The closure of the neopharynx was performed in 2 layers. First, we conducted mucosal suturing using a T-shaped technique followed by muscular suturing. We performed interrupted sutures using Vicryl 3/0 (polyglactin 910). For patients who underwent STL following RT or CRT, we used a pedicled pectoralis major myofascial (PMMF) flap or a local sternocleidomastoid (SCM) muscle flap to reinforce the suture line of the neopharynx. In this group of patients, the choice between primary closure, PMMF flap, and local SCM flap depended on the preferences of the surgeon. At the end of the procedure, we placed at least 2 drains at the neck, each one external to the carotid sheath away from the pharyngeal closure.
Postoperatively, we administered intravenous antibiotics using Cefotaxime and Metronidazole and provided regular wound care provided enteral feeding through the nasogastric tube. We also performed regular blood sampling to detect postoperative anemia or hypoproteinemia to place the patients concerned on the required treatment. We performed a blue-dye swallowing test on days 10 and 14 postoperatively for PTL and STL, respectively. If no blue or saliva was recorded at the level of the wound or the tracheostomy, we repeated the test after a 2 day interval. If both tests were negative, we initiated oral feeding using bee honey and a pasty diet.
PCF Diagnosis and Management
The diagnosis of PCF was established if we noted a blue leakage at the blue-dye test or if we recorded saliva leakage at the wound (Figure 1). In these cases, we maintained the nasogastric tube and suspended oral feeding. The first-line treatment was conservative through antibiotics, daily-wound care, compressive dressing, and the drainage of fluid collection. In the absence of regression under conservative treatment, we conducted surgical management of the PCF; the choice of the surgical modality depended on the local condition, the size of the fistula, and the size of the pharyngeal defect.
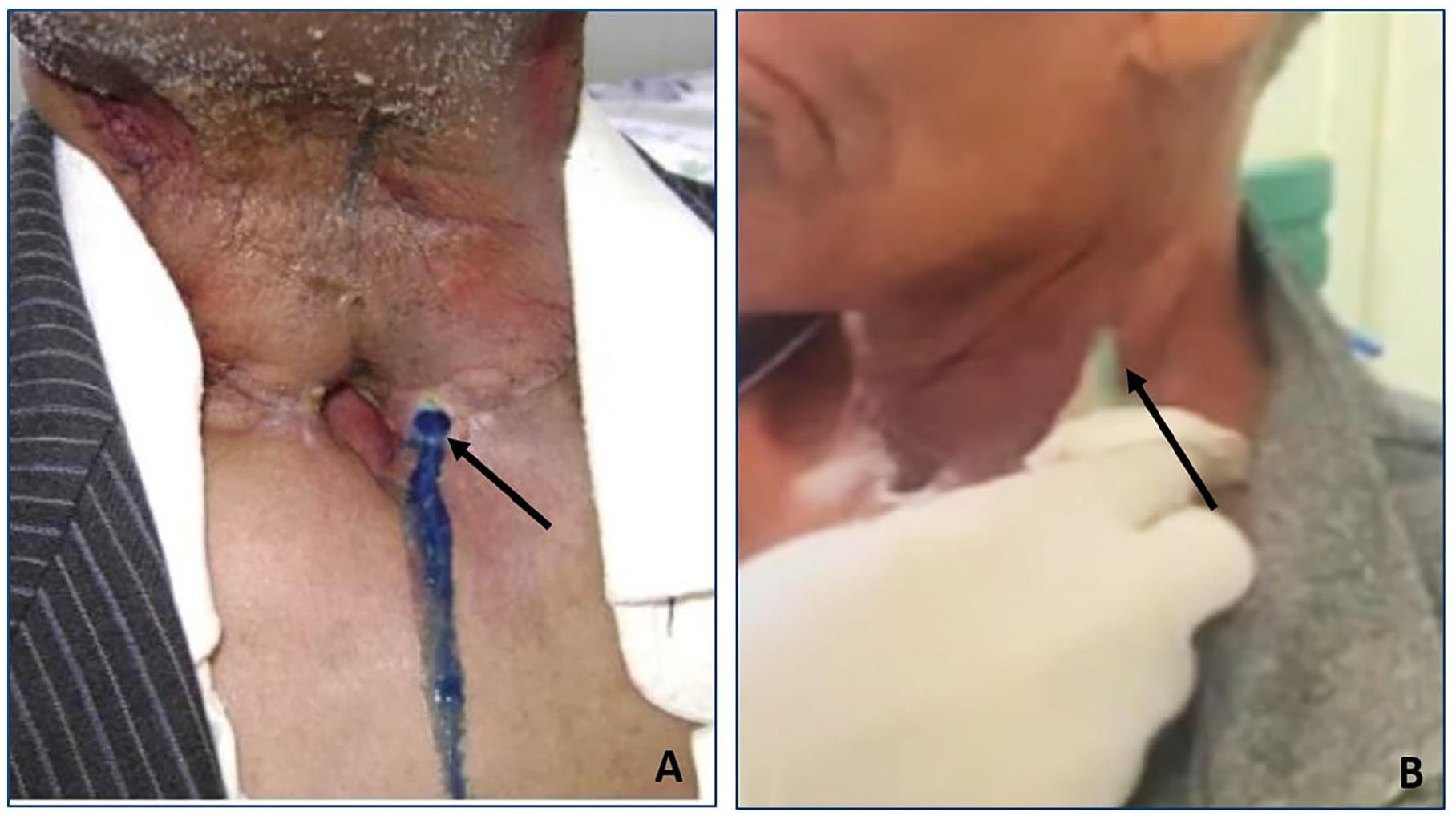
A: Blue-dye test showing the leakage of the blue product near the orifice of tracheostomy. B: Pharyngocutaneous fistula manifesting by saliva and pus leakage at the left lateral wound.
Risk Factors of PCF Occurrence
We studied risk factors of PCF occurrence. These factors were divided as follows:
Patient-related factors, including age, diabetes, cardiovascular comorbidities, and nutritional status based on biological parameters such as anemia and hypoproteinemia. Anemia was defined as a level of hemoglobinemia below 12.5 g/dl. Hypoproteinemia was considered if the serum protein levels were <70 g/l. 10 Similarly to other previously-published studies, we used protein levels and not albumin levels.10,11 We made this choice due to the lack of the latter in multiple medical records.
Disease-related factors, including tumor staging, close or positive surgical margins, vascular embolism, and perineural metastasis.
Treatment-related factors, including preoperative tracheostomy, preoperative RT or CRT, surgery extent, postoperative anemia, and hypoalbuminemia.
Statistical Analysis
We used IBM Statistical Package for the Social Sciences (SPSS), version 25 (IBM Corp., Armonk, NY, USA) to conduct the statistical analysis.
For descriptive analysis, depending on the normality of the distribution verified by Kolmogorov-Smirnov, we presented quantitative variables as mean values or median values and expressed the range as (minimum-maximum). We represented qualitative variables as absolute and relative frequencies (percentages).
For the univariate analysis of qualitative variables, we used either the chi-squared or Fisher’s exact test. To compare quantitative variables, we rather used the Student t-test or the Mann-Whitney U test depending on their normal distribution. The multivariate analysis was conducted using the logistic binary regression for factors that showed P < .2 at the univariate analysis. Any P value < .05 was considered as significant.
Results
Patients and Tumor Characteristics
We included 132 patients with a male-to-female ratio of 128:4. Their mean age was 63.5 years old (±10.6). We recorded tobacco consumption in 121 patients (93%). Four patients (3%) had a history of diabetes.
Surgery and Postoperative Period
Surgery consisted of PTL or STL in 91% and 9% of patients, respectively. TL was extended in 24 patients (19%) to the pharynx (9%), the anterior neck skin (3.8%), or the base of the tongue region (5%). At the final histopathological examination, the tumor was staged as T4a in 82 patients (62%).
Details of the demographic and tumor characteristics of our patients are summed up in Table 1.
Patients and Disease Characteristics.
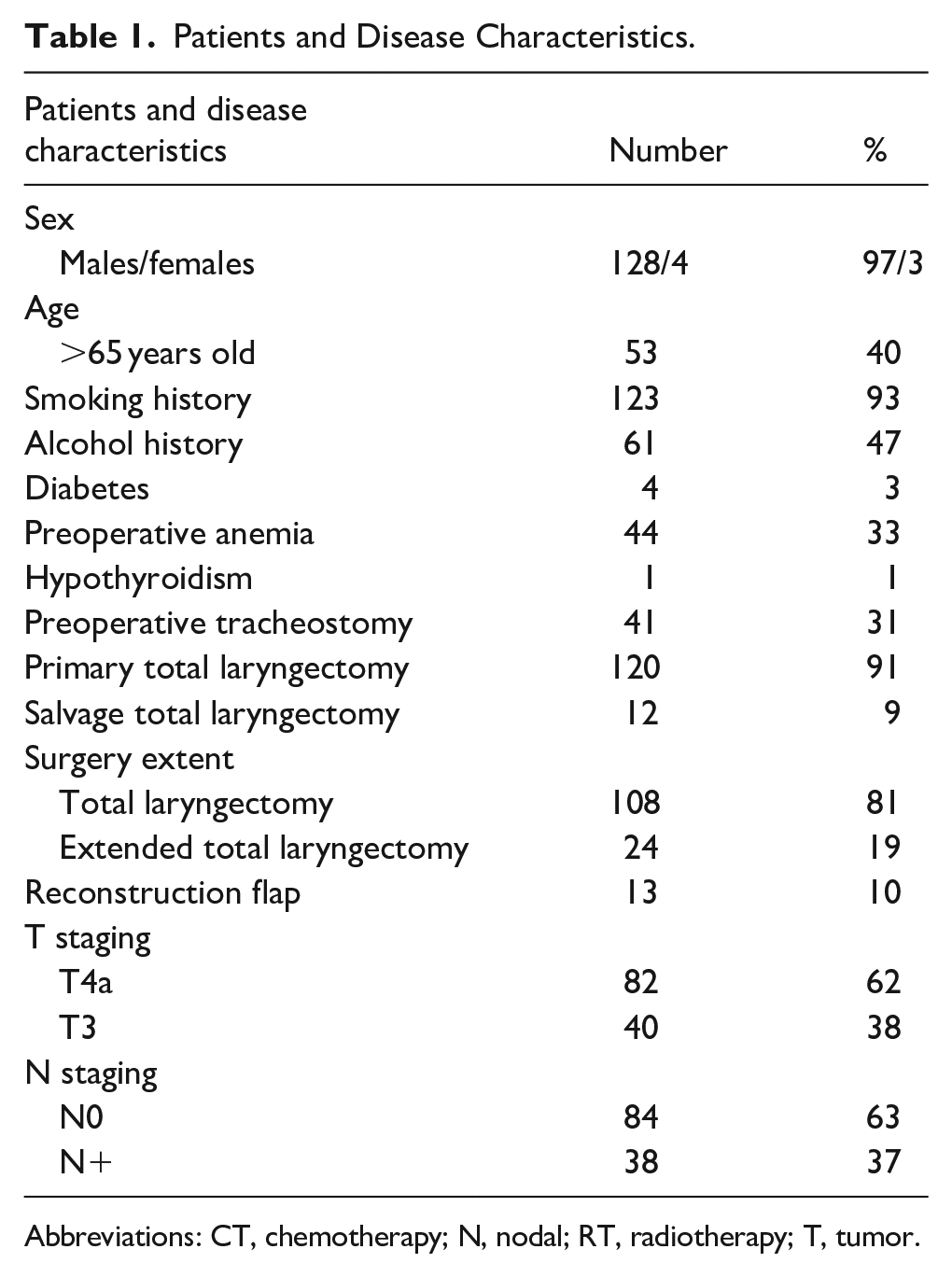
Abbreviations: CT, chemotherapy; N, nodal; RT, radiotherapy; T, tumor.
Postoperatively, all patients were placed under antibiotics for a median period of 10.5 days (10-14). Anemia and hypoalbuminemia were recorded in 104 (78%) and 38 (24%) patients, respectively. The median levels of hemoglobin and albumin were respectively 10.28 g/dl (8.4-12.6) and 28 g/dl (19.3-35). A respiratory or wound infection was noted in 7% (9 patients) and 11% (14 patients), respectively. Oral feeding was started after a median postoperative period of 12 days (10-17).
PCF Occurrence and Management
The incidence of PCF was 13% (17 patients). It occurred after a median period of 14 days (5-36). It manifested by a lateral fistula in 11 of the 17 patients (65%) and a fistula at the level of the tracheostomy in 4 patients (24%).
All patients underwent a compressive dressing. We used honey through both oral ingestion and direct application on the fistula in all patients; it resulted in the fistula healing in 13 patients, as shown in Figure 2. Four patients required a surgical suturing of the pharyngeal dehiscence along with the use of biological glue in the fistula path in two patients and the use of a reconstruction flap via the PMMF flap in two patients. The mean duration of antibiotics in these patients was 16.2 ± 12 days. We removed the nasogastric tube and started oral feeding after a mean period of 23 ± 12 days.
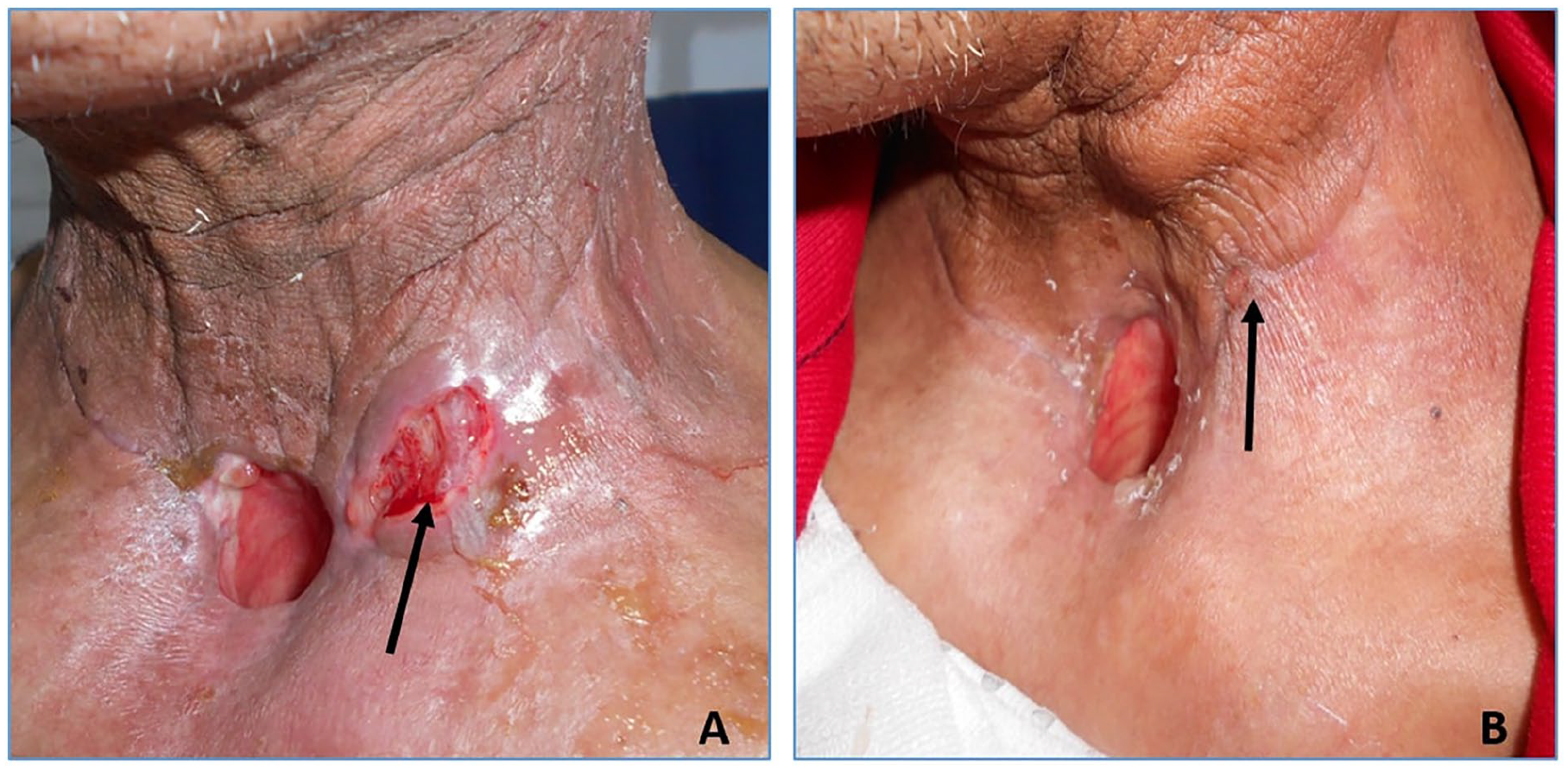
A: Saliva leakage through a lateral pharyngocutaneous fistula. B: Total disappearance of the fistula following conservative treatment.
Predictive Factors of PCF Occurrence
Factors Related to the Patient
We did not record a significant association between PCF and the following factors related to the patient: age >65 years old, tobacco consumption, and comorbidities. On the other hand, we noted that preoperative anemia (P = .035) and preoperative poor nutritional status (P = .004) were significantly associated with PCF as shown in Table 2 and Figure 3.
Univariate Analysis of Demographic and Preoperative Factors Associated With PCF.
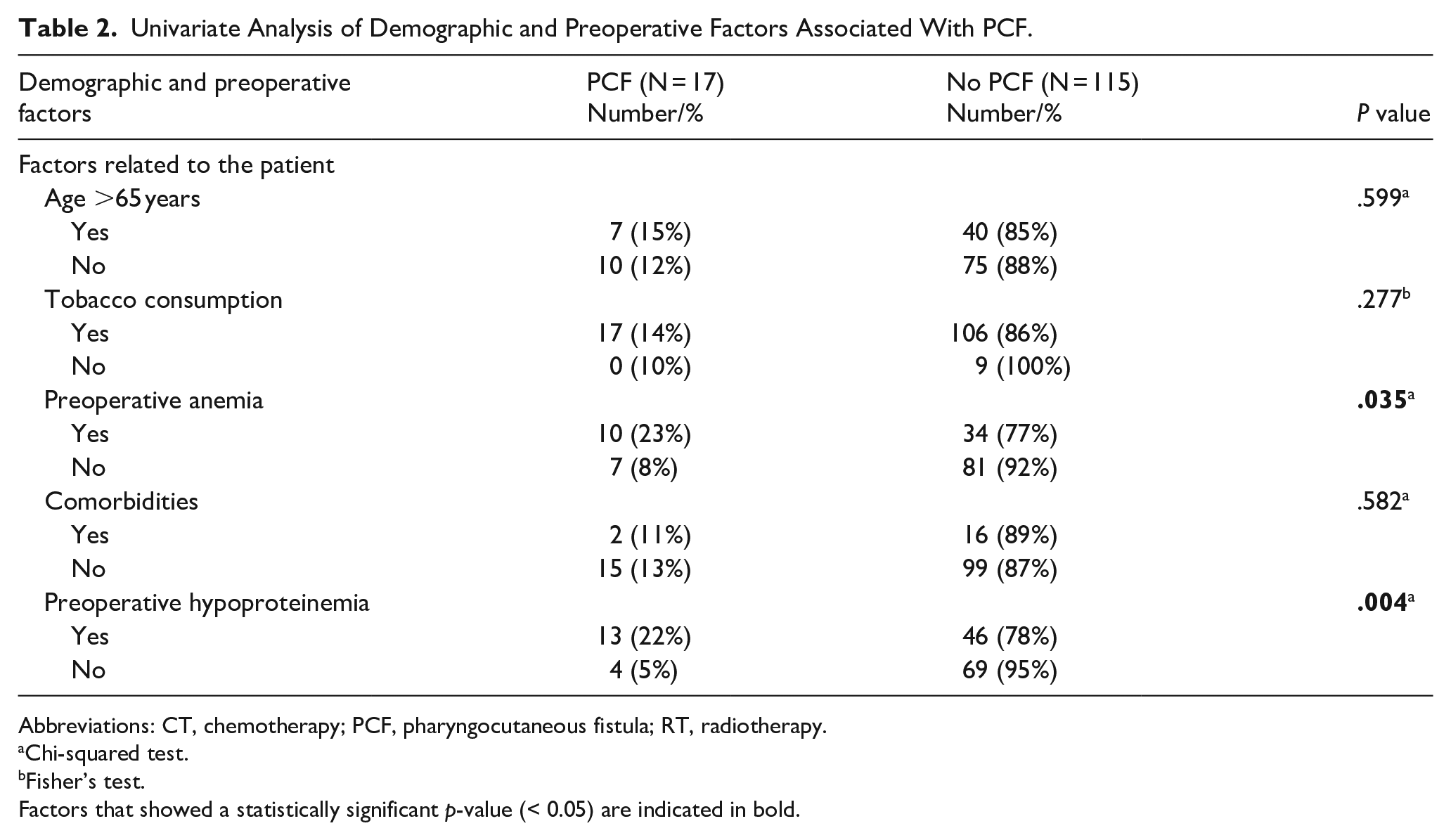
Abbreviations: CT, chemotherapy; PCF, pharyngocutaneous fistula; RT, radiotherapy.
Chi-squared test.
Fisher’s test.
Factors that showed a statistically significant p-value (< 0.05) are indicated in bold.
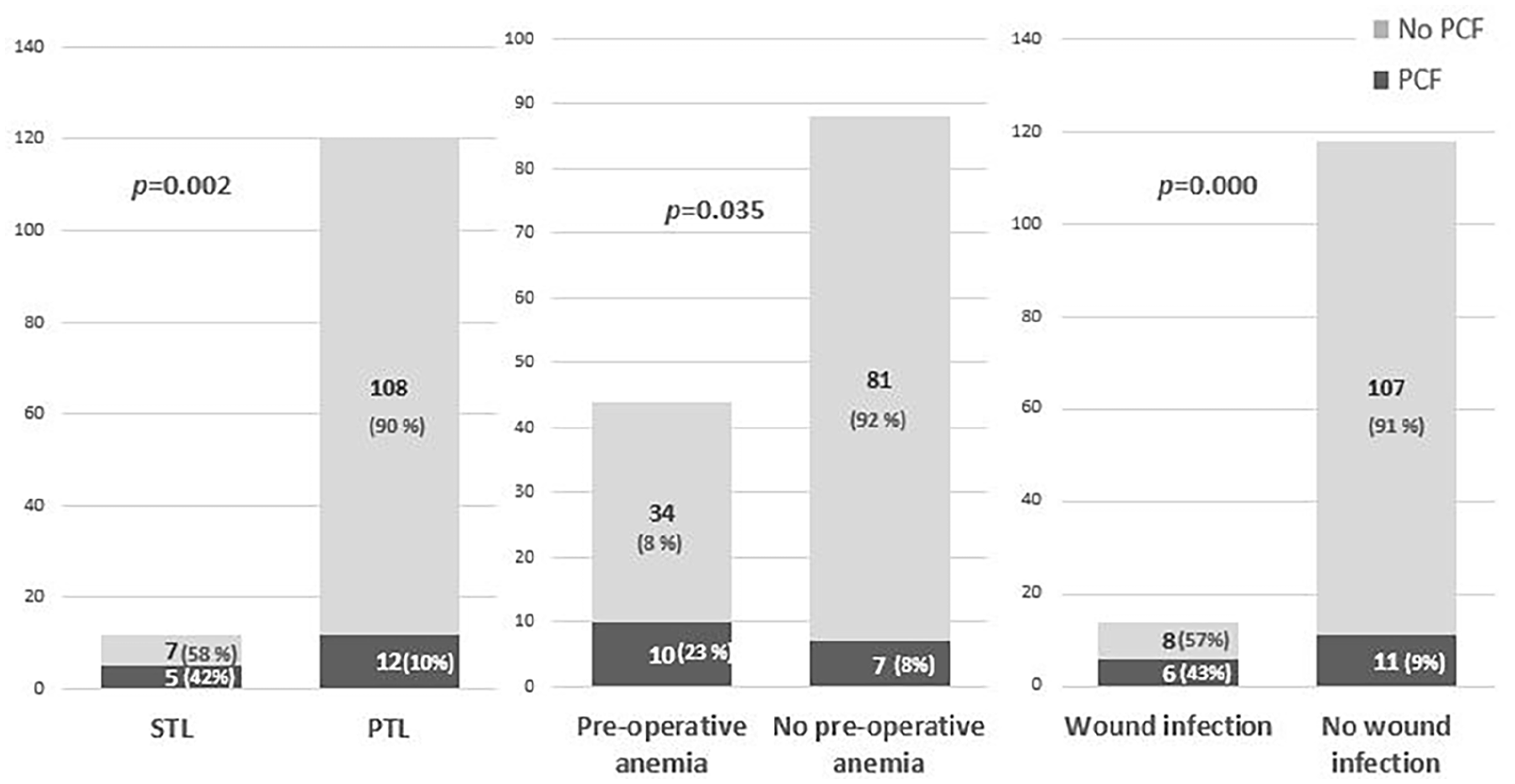
Graph showing factors associated with PCF occurrence at the univariate analysis. PCF, pharyngocutaneous fistula; STL, salvage total laryngectomy; PTL, primary total laryngectomy.
Factors Related to the Received Treatment and Its Complications
In the univariate analysis, we noted that PCF was significantly associated with STL (P = .002) and specifically CT-RT (P = .000). In addition, widened TL was also significantly associated with PCF (P = .006). Conversely, extended TL and preoperative tracheostomy were not significantly associated with the occurrence of PCF. For the group of patients that received an STL, we did not observe a significant association between the occurrence of PCF and the interval of time separating surgery from RT, the use of flap, and the use of CT. Regarding postoperative complications, we noted that wound infection increased the risk of PCF significantly (P = .000). On the other hand, we did note an association between PCF and postoperative protein and hemoglobin levels (Table 3 and Figure 3).
Univarite Analysis of the Disease and Surgical Risk Factors of PCF Occurrence.
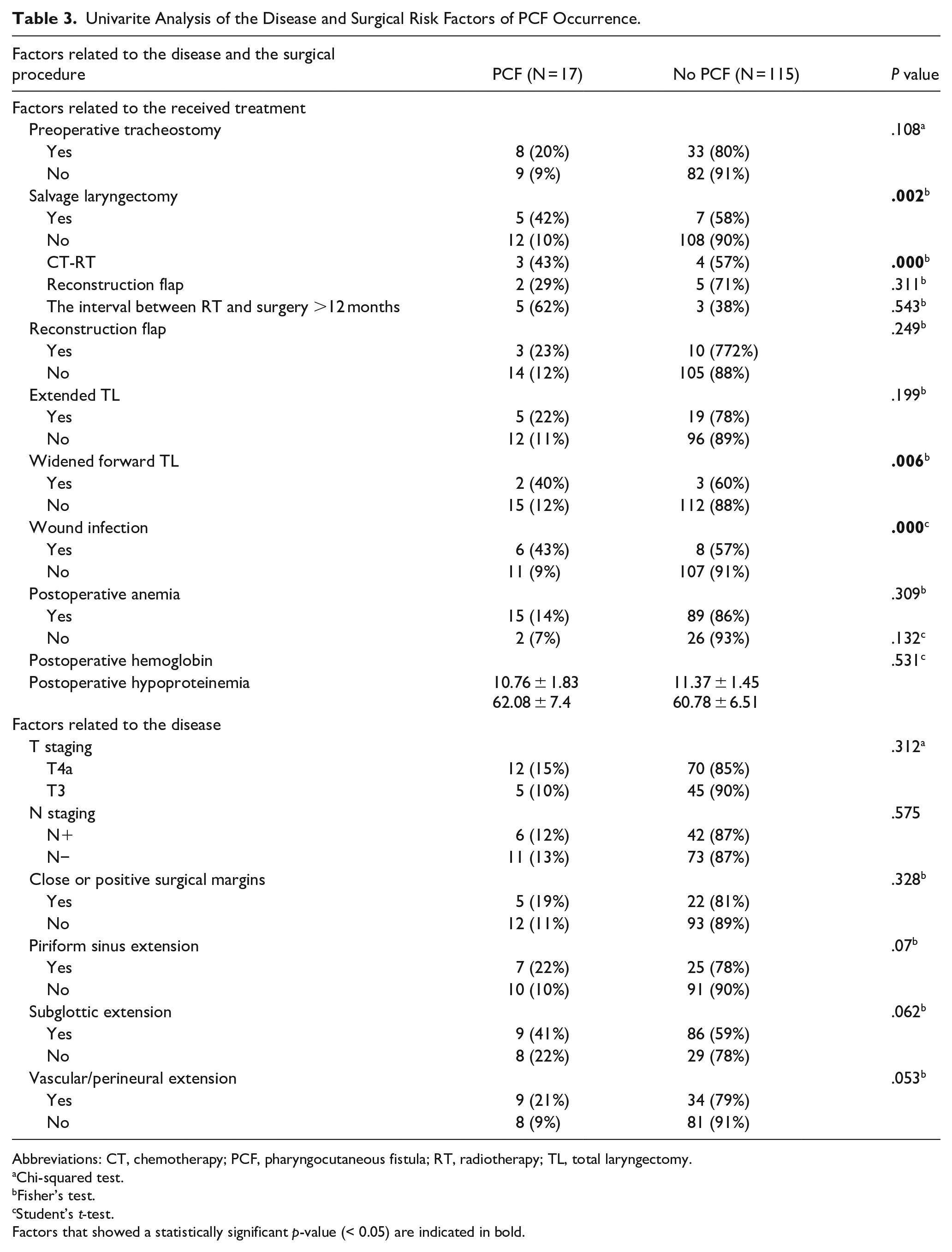
Abbreviations: CT, chemotherapy; PCF, pharyngocutaneous fistula; RT, radiotherapy; TL, total laryngectomy.
Chi-squared test.
Fisher’s test.
Student’s t-test.
Factors that showed a statistically significant p-value (< 0.05) are indicated in bold.
Factors Related to the Disease
Factors related to the disease that tended to significantly increase the risk of PCF were subglottic and piriform sinus extension, along with vascular and/or perineural extension. Details are summed up in Table 3.
Multivariate Analysis of Factors Predicting PCF Occurrence
In the multivariate analysis, preoperative anemia, STL, and wound infection were independent risk factors of PCF. SL increased the risk of PCF occurrence by 18.48 as detailed in Table 4.
Multivariate Analysis of Risk Factors of Pharyngocutaneous Fistula Occurrence.
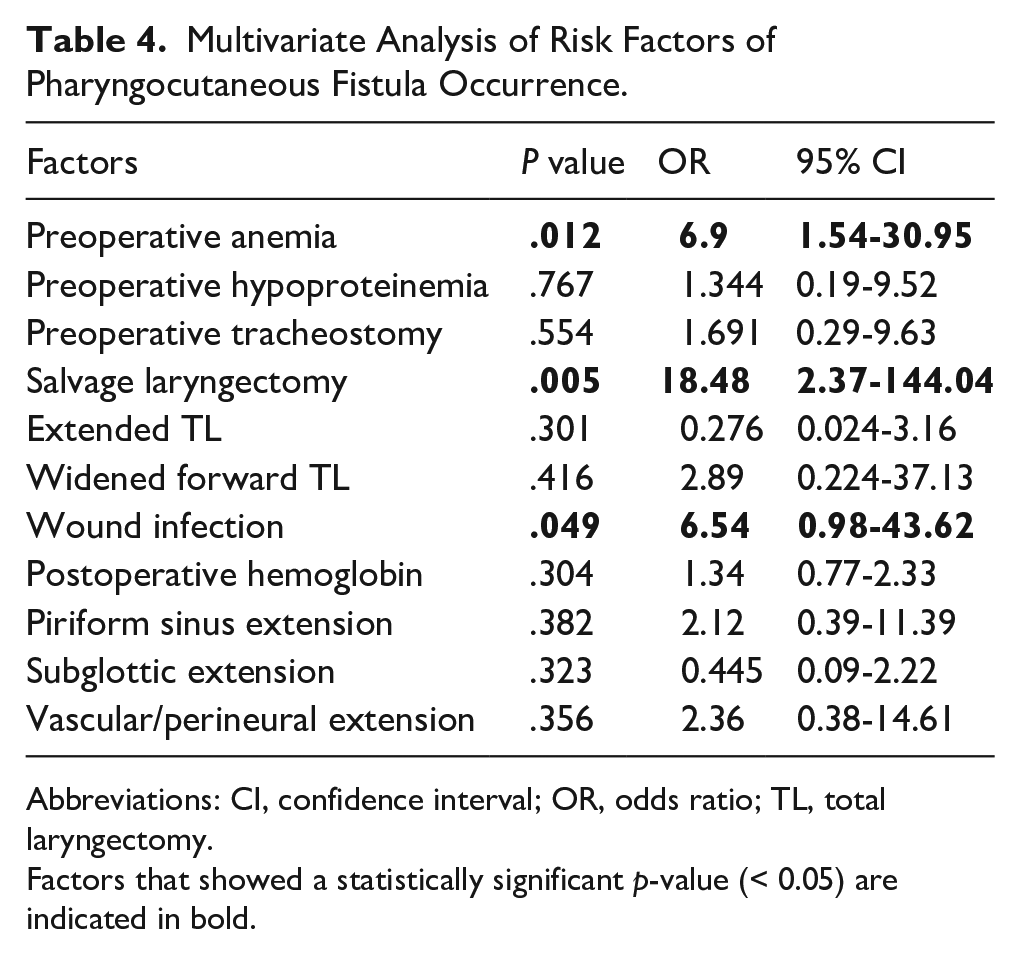
Abbreviations: CI, confidence interval; OR, odds ratio; TL, total laryngectomy.
Factors that showed a statistically significant p-value (< 0.05) are indicated in bold.
Discussion
Our study focused on determining risk factors of PCF occurrence following TL. The overall incidence of PCF in our patients was 12.88%. We recorded that preoperative anemia, wound infection, and STL were independent risk factors of PCF occurrence.
PCF is the most-frequently-encountered complication following TL for SCC. The incidence of PCF is highly variable; in the previously-published data, the rates of PCF vary between 3% and 65%. PCF alters the quality of life, lengthens the hospitalization stay, and increases health care costs. Thus, multiple previous studies focused on determining the risk factors of PCF to prevent its occurrence and to avoid its sequelae.7,11-13
Previous RT or CRT is one of the main factors that increase the rate of PCF; this complication occurs between 8% and 25% of cases in PTL and in between 20% and 80% of patients who underwent STL.7,14-18 In agreement with previous studies, in our series, the rate of PCF was more increased in patients who received a STL in comparison with PTL with respective PCF rates of 47% and 10%. We also noted that STL was the main independent risk factor of PCF. Previous RT has been widely reported as a risk factor for PCF due to the effects of RT on wound healing, which include local fibrosis, obliterative endarteritis, chronic injury to the local microvascular circulation, and hypoxia.2,16 SLT not only increases PCF occurrence but also interferes with the severity of PCF and decreases the rates of spontaneous PCF closure.1,5,11,12,19,20 In addition, higher doses of RT have been reported to increase the rates of PCF. However, the cutoff dose that increases the risk of PCF differed between 50 and 68 Gy.2,17,21 The timing between RT is also a factor that can influence PCF occurrence. Nevertheless, the optimal and safest timings remain not consensual; they vary in the literature between 4 and 12 months.22,23 CT has also been reported as a factor that increases PCF since it potentiates the toxic effects of RT on wound healing and also worsens the nutritional status of the patients.20,22,24
Thus, to reverse the toxicity of RT and its effects on pharyngeal healing, the use of vascularized reconstruction flaps from nonirradiated tissues to reinforce the suture line of the neopharynx has been widely studied to decrease PCF rates.16,21,22,25 The use of flaps also decreases the severity of PCFs, their duration, the timing of oral feeding, and the rate of their surgical management. 26
In our study, we used reconstruction flaps to cover the neopharynx in 58.3% of patients who underwent STL; they did not decrease the rate of PCF significantly. Similarly, other authors did not record that reconstruction flaps decreased PCF occurrence.23,26 On the other hand, Patel et al recorded that reconstruction flaps significantly decreased fistula occurrence. 21 In their meta-analysis, Williamson et al concluded that vascularized flap-assisted closure decreases PCF rates significantly. 27 The ultimate types are also controverted; the PMMF flap is the most used in previous studies; however, it can be associated with esthetic prejudice due to the more important muscular bulk, more altered vocal results, and shoulder dysfunction. 25 Other used flaps included free radial forearm or anterolateral thigh flaps. 28
Wound infection was an independent risk factor of PCF in our study. Similarly, Šifrer et al reported that wound infection was the most important risk factor of PCF. This complication is related to the exposure of neck tissues to the bacterial flora of the pharynx. Its prevention is mainly based on the use of antibiotics that cover aerobic, anaerobic, and Gram-negative bacteria. 2 However, the types of antibiotics were highly variable. We used prophylactic antibiotics in all patients, mainly based on Cefotaxime in association with Metronidazole, during a median period of 10 days. The recommended length of antibiotics is also not consensual, and it varies between 24 hours and 8 days postoperatively.29,30
The decrease in preoperative hemoglobin and albumin levels is an indicator of poor nutritional status, which can result in microvascular changes that compromise the wound healing process. Thus, anemia and hypoalbuminemia were widely reported as risk factors of PCF.1,6,7,11,19 Kim et al noted that postoperative hypoproteinemia showed the highest accuracy in predicting postoperative PCF and that anemia was also a risk factor for PCF. Similar findings were noted in the systematic review by Cecatto et al.1,11 In agreement with previous findings, preoperative anemia was an independent risk factor of PCF. In light of these findings, in patients presented with malnutrition, a preoperative supplementation can be helpful to avoid postoperative PCF.10,31
Pharyngeal reconstruction is an important factor involved in developing a PCF. 4 Pharyngeal closure can be performed using mechanical or regular manual closure. However, the superiority of one technique over another in reducing PCF appearance is still debated.6,21,32,33 In our study, we only performed manual suturing using a T-shaped technique. Manual closure can be performed using continuous or interrupted sutures with vertical or T-shaped closure. Findings concerning the better modality to ensure less PCF risk yet with a wider neopharynx and better swallowing outcomes remain controversial.4,9,34,35
In our study, extended TL was significantly associated with PCF, while advanced tumor stages did not increase PCF rates. Piriform sinus invasion rather tended to increase the risk of PCF significantly. Dedivitis et al, in their systematic review, considered that hypopharyngeal extension increased the risk of PCF by 9%. 20 The role of the advanced tumor stage and hypopharyngeal extension in PCF occurrence can be attributed to a more extensive mucosal pharyngeal resection leading to a high tension at the pharyngeal suture, to the impairment of blood circulation, and then to poor wound healing, suture dehiscence, leading subsequently to PCF. The advanced tumor stage can also be associated with positive margins. These factors lead to worse wound healing and PCF formation.2,3,7,11,14
Other factors were not associated with PCF in our study and are still debated in the literature. Previous tracheostomy was not considered a risk factor for PCF appearance. However, it may increase the risk of local infection at the tracheal orifice, leading to wound infection and then to PCF.7,20,23,36 Diabetes mellitus was considered a risk factor for PCF due to the microvascular changes and poor wound healing.6,14 Other factors included longer surgery duration, age >60 years old, smoking, neck dissection, hypothyroidism, blood transfusion during the operation, and the surgeon’s experience.1,5,7,19,20,23
The recognition of factors predicting PCF is important to avoid its occurrence and thus to avoid its consequences, which are mainly related to the increase in hospitalization stay, the delay in oral feeding, and the delay in postoperative adjuvant treatment and rehabilitation, along with the psychological sequels.
The management of PCF is either conservative or rather surgical. Conservative treatment is indicated in PCF at its acute phase, which can reach two weeks postoperatively. 37 It is based on compressive wound dressing, the excision of necrotic tissues, and the treatment of wound infections with adapted antibiotics, along with avoiding oral feeding.37,38 Oral feeding should be avoided and replaced with enteral feeding via a nasogastric tube or parenteral nutrition. 37 Negative pressure also results in faster PCF healing and improves the wound condition to prepare for surgical management. However, its application in PCF is challenging mainly due to the difficulty of obtaining an airtight seal if the fistula communicates with the tracheostomy.37,39-41 Surgical management is indicated in large PCFs and fistulas unresponsive to conservative management. This can be performed either through a direct suture of the remaining pharyngeal mucosa or through the use of a vascularized reconstruction flap to increase the pharyngeal circumference.38,42-44 In our study, we used a PMMF flap for a PCF that was irresponsive to the conservative treatment.
The present study has several limitations: They include its retrospective design and the relatively-reduced number of the included patients. In addition, the fact that different surgeons performed the surgical procedures could have influenced the results. Not including previously-studied factors such as albumin levels, transfusion, body mass index, and the type of pharyngeal closure is also another limitation of our study.
Conclusion
In summary, multiple risk factors can increase PCF occurrence. We concluded that preoperative anemia, STL, and wound infection were independent risk factors of PCF. The recognition of preoperative and postoperative risk factors of developing PCF is important to take preventive measures; they mainly include preventing infections and malnutrition, along with the use of a reconstruction flaps in STL to avoid PCF.
Footnotes
Ethical Considerations
Our institution does not require ethical committee approval for retrospective studies that report data anonymously.
Consent for Publication
Written informed consent was obtained from the included patients for publishing their information anonymously.
Author Contributions
Rania Kharrat: data curation, formal analysis, methodology, resources, supervision, validation, and writing—original draft preparation. Moncef Sellami: data curation, formal analysis, methodology, resources, supervision, validation, and writing—original draft preparation. Mariam Ben Ayed: data curation, formal analysis, methodology, and writing—original draft preparation. Nesrine Sellami: methodology, and writing—original draft preparation. Ghada Youssfi: methodology, and writing—original draft preparation. Malek Mnejja: supervision, validation, and writing—original draft preparation. Mohamed Amin Chaabouni: conceptualization, methodology, and writing—original draft preparation. Ilhem Charfeddine: validation, and writing—original draft preparation.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The analyzed data during the current study are available upon reasonable request.
