Abstract
Background:
Previous research has shown that myofibroblasts in the nasal/sinus mucosa are not only structural cells but also a rich source of inflammatory mediators. Thus, they influence the immune response and play an essential role in the pathogenesis of chronic rhinosinusitis with nasal polyposis. Previous research did not provide a clear answer to the question of whether the number of myofibroblasts in nasal polyps (NPs) affects the extent of sinus disease. This cross-sectional study investigates the relationship between the number of active myofibroblasts, eosinophil count, and clinical parameters in patients with NPs with and without aspirin sensitivity.
Methods:
Patients diagnosed with NPs, with and without aspirin sensitivity, and healthy subjects were assessed according to the severity of their symptoms as well as endoscopic and radiological findings. The quantification of myofibroblasts present in the surgically excised polyp tissue/healthy nasal mucosa was conducted based on the expression of alpha-smooth muscle actin (α-SMA) in the subepithelial and perivascular compartments. Eosinophils were counted in the subepithelium of the tissue samples from all subjects.
Results:
Forty-nine subjects were included: 23 NP patients without, 14 NP patients with aspirin sensitivity, and 12 control participants. The highest levels of α-SMA expression and eosinophil count were observed in NP patients with aspirin sensitivity and the lowest in healthy subjects (P < .001; P < .001, respectively). A strong correlation was noted between α-SMA expression and clinical parameters, particularly with disease extent evaluated radiologically and endoscopically.
Conclusion:
Our results suggest a positive correlation between the quantity of myofibroblasts and the severity of chronic inflammation in the sinonasal region. The number of myofibroblasts in the NP tissue may serve as a reliable marker for assessing the clinical status of the disease.
Introduction
Chronic rhinosinusitis with nasal polyps (CRSwNP) is a heterogeneous, multifactorial disease characterized by persistent inflammation of the sinonasal mucosal membrane for more than 3 months, tissue remodeling, and the formation of inflammatory nasal polyps (NPs).1,2 The T2 endotype underlies the pathogenesis of NP in most European, North American, and Australian populations.1,2 This signaling pathway is characterized by elevated levels of T2 cytokines (IL-4, IL-5, and IL-13) and other inflammatory mediators that activate eosinophils, mast cells, macrophages, epithelial cells, goblet cells, and fibroblasts, all of which are responsible for tissue inflammatory responses.2-5
Aspirin-exacerbated respiratory disease (AERD) is characterized by a triad of clinical entities: non-allergic asthma, recurrent CRSwNP, and airway hypersensitivity to aspirin and other non-steroid anti-inflammatory drugs (NSAIDs).1,2 The T2 immune response in this clinical form of CRS differs from the inflammatory cascade in NSAID-insensitive patients. The inflammatory response is dominated by an eicosanoid imbalance resulting from various factors, including a disorder in arachidonic acid metabolism, a mixed inflammatory pattern, and genetic polymorphisms.6,7 Consequently, proinflammatory cysteinyl leukotrienes (cysLTs), especially their stable end metabolite LTE4 and prostaglandin D2 (PGD2), induce and activate innate lymphoid type 2 cells (ILC2) to secrete T2 cytokines.6,7
Crucial for the pathogenesis of CRS are persistent mucosal inflammation and tissue remodeling. Fibroblasts, as one of the primary supporting cells within sinonasal connective tissue, must be regarded not solely as structural cells but also as pivotal sources of inflammatory mediators.8-10 The activated phenotype of fibroblasts are myofibroblasts, which express alpha-smooth muscle actin (α-SMA). 8 They are formed in a process called epithelial-mesenchymal transition (EMT) via proto-myofibroblasts under the influence of platelet-derived growth factors and stem cell factors. 8 Final differentiation into myofibroblasts is regulated by inflammatory mediators such as transforming growth factor beta and IL-1β via inflammatory signaling pathways. 8 EMT also results in gaining mesenchymal markers (α‑SMA, vimentin, and fibronectin) that are expressed in the sinus epithelium. 10
Previous research has demonstrated that myofibroblasts play a crucial role in tissue remodeling and the size of NPs by synthesizing key extracellular matrix components, including collagen, fibronectin, and vimentin.9,10 The actin filaments (α-SMA) in the myofibroblast cytoplasm are essential for fibroblast migration and contractility. 10 However, the involvement of myofibroblasts in the innate and adaptive immune response is crucial as it initiates chronic inflammation and maintains its duration and intensity.11,12 Fibroblasts of NPs secrete chemokines that participate in migrating eosinophils, neutrophils, dendritic cells, macrophages, and cytokines, some of which mediate T2 immune response.11,12 Critical interactions occur between myofibroblasts and eosinophils. Recent research has identified a positive feedback loop involving eosinophils and myofibroblasts, which intensifies local inflammation and promotes the remodeling of the nasal mucosa. 12 However, previous studies indicate that we still lack sufficient knowledge about the relationship between myofibroblast activity and the clinical manifestations of CRS, particularly in patients with AERD.
The aims of this study were (i) to evaluate whether the degree of α-SMA immunoexpression in subjects with CRSwNP with and without NSAID hypersensitivity may be significant in assessing the severity of the clinical manifestation of disease and (ii) to investigate the association between eosinophilia and the α-SMA expression in the tissue of NPs.
Materials and Methods
Study Protocol
This cross-sectional study was conducted in our institution from January to December 2024. All procedures were performed according to the latest valid version of the Declaration of Helsinki. All patients gave their informed consent. The research was approved by the decision of the local Ethics Committee (Approval Number 21/2022). The STROBE reporting guideline was used to present methods and results.
Participants
The diagnosis of CRSwNP was made according to the criteria outlined in the European Position Paper on Rhinosinusitis and Nasal Polyps 2020. 2 The category of CRS patients selected for endoscopic sinus surgery was divided into 2 subgroups: those with and without hypersensitivity to NSAIDs. Patients with AERD were included in the study if they met 3 criteria: (i) diagnosis of CRSwNP; (ii) patient-reported history regarding the worsening of respiratory symptoms after taking one of the NSAIDs; and (iii) diagnosis of mild persistent asthma by a pulmonologist. 13 The last criteria was introduced to exclude patients with difficult-to-control asthma requiring daily oral and/or inhaled corticosteroids, which could affect the inflammatory status of the nasal/sinus mucosa.
The control subjects were participants with nasal obstruction but without symptoms and signs of chronic inflammation who had a pneumatized middle turbinate (concha bullosa). They were selected based on endoscopic examination and a cone beam computed tomography (CT) scan of the paranasal sinuses. To improve respiratory function, they recommended surgery to reduce the volume of the middle turbinate by its lateral resection, with or without correction of the nasal septum deformity.
Exclusion criteria
Subjects younger than 18 and older than 65 years, pregnant and lactating women, patients with systemic diseases affecting the nose/sinus, with chronic fungal sinusitis, choanal polyps, and hamartomas, subjects receiving topical and/or systemic corticosteroids, antibiotics, antihistamines, and leukotriene receptor antagonists within 4 weeks before the start of the study.
Clinical Assessment
Each subject assessed the severity of his symptoms (nasal obstruction, impaired sense of smell, nasal discharge, nasal itching, and sneezing) using the method according to Tsicopoulos et al. 14 The intensity of complaints is graded as follows: 0, absence of symptom; 1, mild symptom; 2, moderately severe symptom; and 3, extremely severe symptom. The maximum nasal symptom score (SS) is 15.
An endoscopic assessment of NP size was performed sitting, using rigid endoscopes 0° and 30°. Decongestant drops and anesthetics were not used. The extension of NPs was classified concerning anatomical landmarks, according to the scoring system of Meltzer et al 15 : 0, no NPs; 1, NPs limited to the middle meatus; 2, multiple NPs obstructing the middle meatus; 3, NPs extending beyond the middle meatus; and 4, NPs that obstruct the nasal cavity entirely. The maximum endoscopic score (ES) is 8, observed bilaterally. The extent of chronic inflammation on the CT scans was assessed based on the Lund-Mackay score (LMS), with a maximum of 24 points. 16
Tissue Sampling, Histological, and Immunohistochemical Analyses
All subjects were surgically treated with an endoscopic approach. NPs were removed from the ethmoid region while mucosal samples of the pneumatized middle turbinate were taken during their lateral resection. Considering that CRSwNP is a bilateral disease, we analyzed all the removed material from the ethmoid area on both sides. In addition, we examined the mucous membrane that lines the inner wall of the concha bullosa unilaterally or bilaterally, depending on whether this anatomical variation is present on one or both sides of the nasal cavity. The removed material was fixed in 10% formaldehyde, embedded in paraffin, cut with a microtome into 5 μm sections, and stained with hematoxylin and eosin. Histological analysis was performed using a digital optical microscope and a computerized image analysis system. The system was programmed by Image J (Java-based imaging processing program) (National Institute of Health, Laboratory for Optical and Computational Instrumentation, LOCI, University of Wisconsin, USA). Eosinophils were counted in the superficial layer of the mucosa to a tissue depth of up to 0.2 mm below the basement membrane at a magnification of ×200. We determined the mean value of eosinophils per field by examining 10 randomly selected fields of 1 section, as previously described. 17
Tissue samples were then subjected to immunohistochemical (IHC) analysis to assess the level of α-SMA expression and determine the number of myofibroblasts using commercial human antibodies (Bio-Techne R&D Systems, Minneapolis, MN, USA). The quantification of α-SMA-positive cells was performed in 2 ways: (1) the total number of α-SMA-positive cells in the subepithelium outside the walls of blood vessels and stromal glands in NPs/healthy nasal mucosa. IHC expression of α-SMA was evaluated by first calculating the average value of α-SMA-positive cells in the subepithelium by counting in 10 randomly selected fields at ×200 magnification. The degree of expression was assessed semi-quantitatively according to the following scale: 1, <30 positive cells; 2, from 31 to 50 positive cells; and 3, more than 50 positive cells, as previously described.11,12 (2) The quantification of α-SMA-positive cells in the perivascular space, outside the walls of blood vessels. In addition, the percentage ratio between the volume of blood vessels in the NP/healthy nasal mucosa stroma and the number of perivascular myofibroblasts in the same 10 randomly selected fields was determined.
Statistical Analysis
The data normality was tested using Kolmogorov-Smirnov and Shapiro-Wilk tests. Kruskal-Wallis H test and Mann-Whitney U test were used to explore the differences between patient groups relative to the values of their clinical parameter scores and number of eosinophils and α-SMA-positive cells. In addition, Fischer’s exact probability test was used to assess the difference in frequencies of examined variables, mainly the immunoexpression of α-SMA-positive cells. Correlation testing between these parameters was done using Spearman’s rank correlation coefficient. A P-value less than ≤.05 is considered statistically significant. Results in the figures are represented as medians with 25th and 75th percentiles.
Results
Forty-nine (n = 49) patients were included in the present study. Among these patients, 23 were diagnosed with CRSwNP without aspirin hypersensitivity, 14 had AERD, and 12 patients were without nasal inflammation but experienced nasal obstruction due to the pneumatization of the middle turbinate. All were selected for surgical treatment. The demographic characteristics of the participants are presented in Table 1.
Demographic Characteristics of Participants Included in Our Study.

Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRSwNP, chronic rhinosinusitis with nasal polyps.
Expressed as mean ± standard deviation.
For each participant, 3 clinical parameters were analyzed: SS, size of NP, that is, the ES and LMS. A significant difference was detected for all patient groups for SS: controls versus AERD (P < .001); controls versus CRSwNP (P < .001); CRSwNP versus AERD (P < .05; Figure 1). A similar statistically significant difference was detected between the CRSwNP and AERD groups for both ES (P < .01) and LMS (P < .001) parameters (Figure 2).
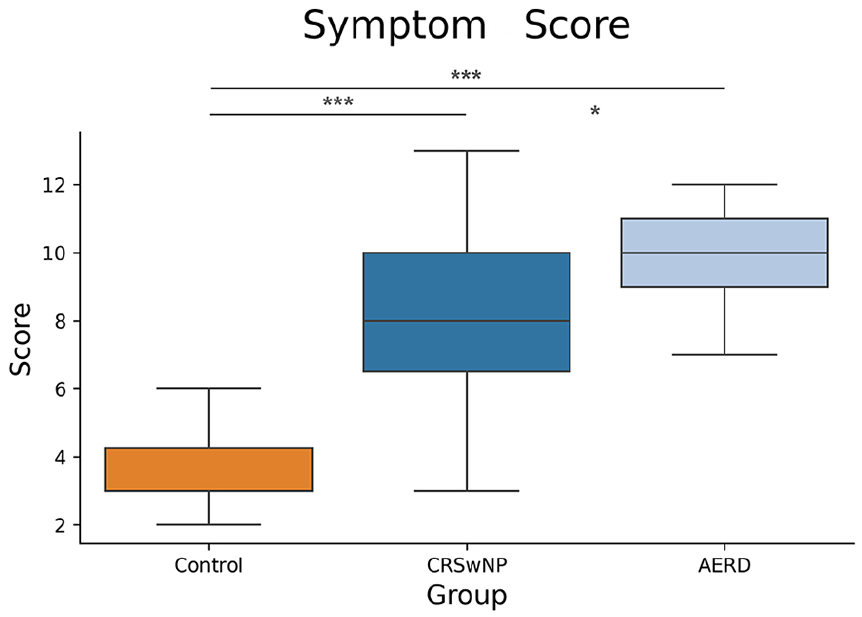
Comparison of TNSS between patient groups showed significantly higher values in CRSwNP and AERD groups compared to the control group. Results are represented as medians with 25th and 75th percentiles. SS, symptom score; CRSwNP, chronic rhinosinusitis with nasal polyps; AERD, aspirin-exacerbated respiratory disease.
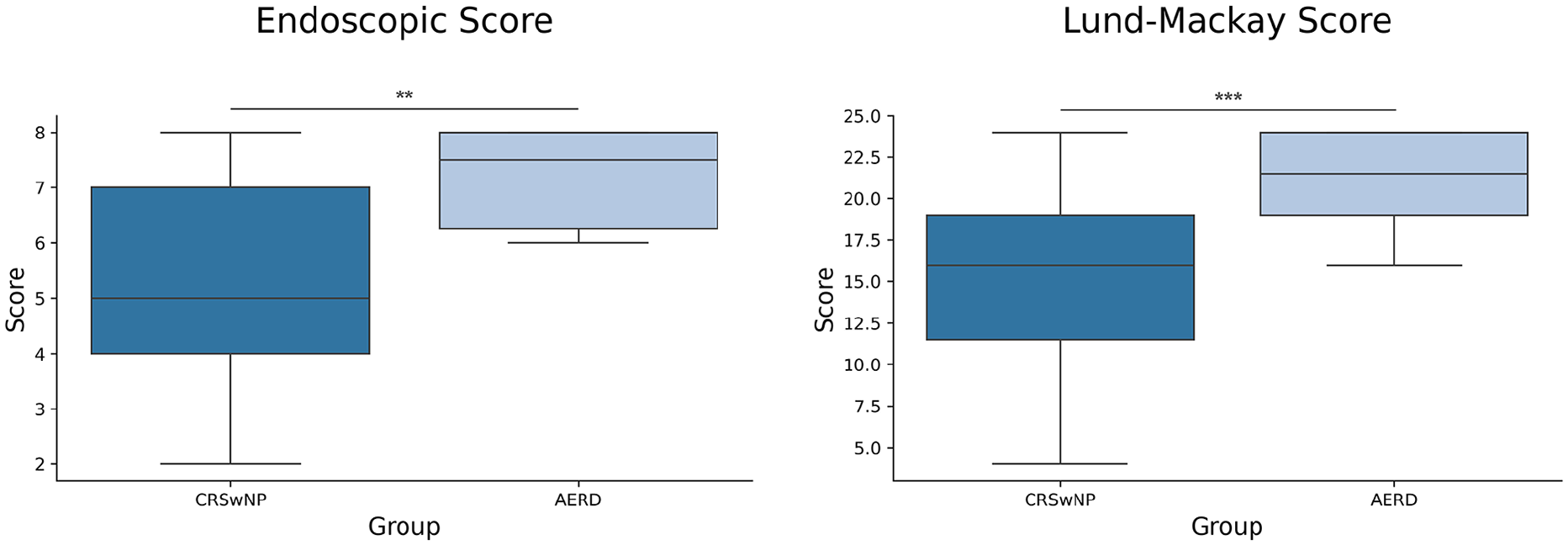
Comparison of ES and LMS. ES and LMS were only assessed in patients with nasal polyps. Significantly higher score values were observed in the AERD group for both ES and LMS. Results are represented as medians with 25th and 75th percentiles. ES, endoscopic score; LMS, Lund-Mackay score; CRSwNP, chronic rhinosinusitis with nasal polyps; AERD, aspirin-exacerbated respiratory disease.
Regarding the subepithelial eosinophil counts, a statistically significant difference was found between the control and CRSwNP group (P < .001) and between the control and AERD group (P < .001), while there was no difference between AERD and CRSwNP group (P > .05; Figure 3). Concerning the number of α-SMA-positive cells, the lowest number of those (grade 1) was detected in the control group, while patients from the CRSwNP and AERD groups had a higher average number of positive cells (ie, grades 2 and 3, respectively; Table 2). Representative examples of α-SMA expression in patients’ groups are presented in Figure 4. Regarding the percentage of perivascular α-SMA-positive cells, a difference is observed between the control and CRSwNP group (P < .001) and the control and AERD group (P < .001), but not between the CRSwNP and AERD groups (P > .05; Figure 5).
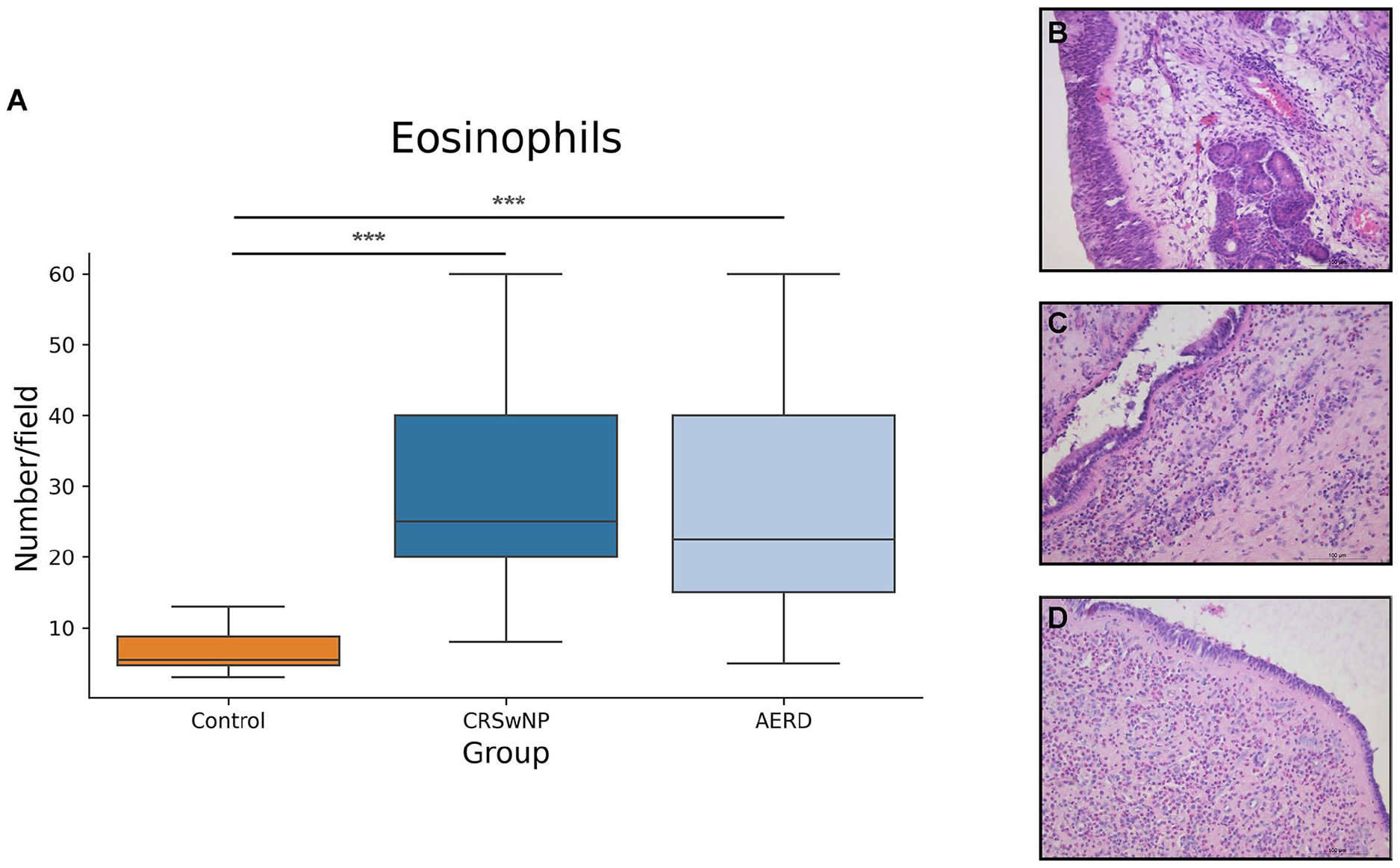
Comparison of average number of eosinophils between patient groups. (A) The difference in the number of eosinophils was shown between the control and CRSwNP group and control and AERD, but not between AERD and CRSwNP. Results are represented as medians with 25th and 75th percentiles. (B) Representative histological image of eosinophils in samples obtained from the control group. (C) Representative histological image of eosinophils in samples obtained from the CRSwNP group. (D) Representative histological image of eosinophils in samples obtained from the AERD group (hematoxylin and eosin stain, magnification ×200). CRSwNP, chronic rhinosinusitis with nasal polyps; AERD, aspirin-exacerbated respiratory disease.
The Frequency of α-SMA Expression Grade in 3 Groups of Patients.

Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRSwNP, chronic rhinosinusitis with nasal polyps; α-SMA, alpha-smooth muscle actin.
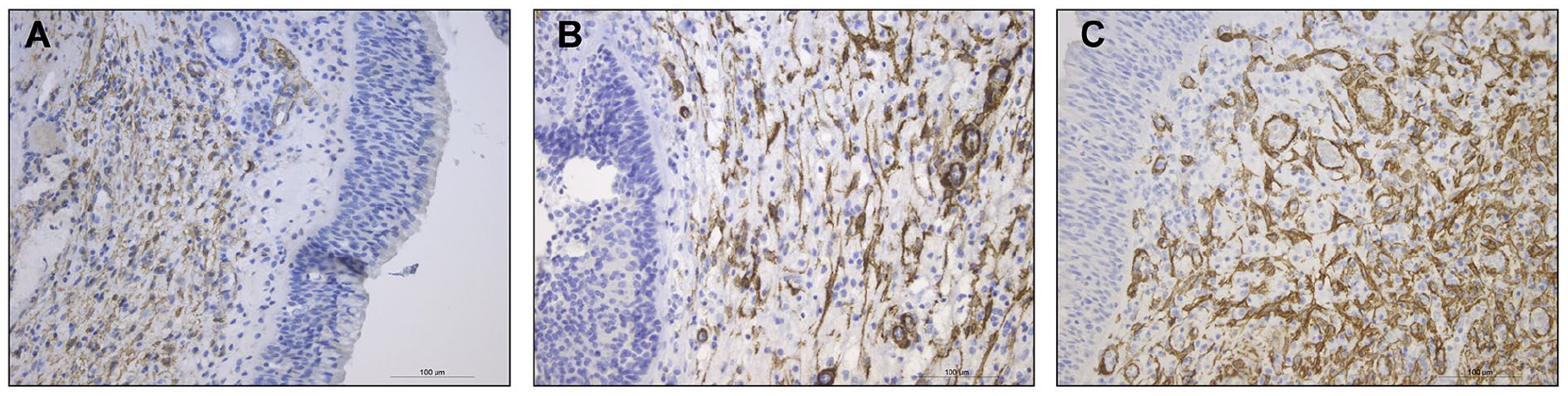
IHC expression of α-SMA in surgical samples obtained from 3 groups of patients. (A) Representative IHC image of α-SMA expression from the control group. (B) Representative IHC image of α-SMA expression from the CRSwNP group. (C) Representative IHC image of α-SMA expression from the AERD group (magnification ×200). AERD, aspirin-exacerbated respiratory disease; CRSwNP, chronic rhinosinusitis with nasal polyps; IHC, immunohistochemical; α-SMA, alpha-smooth muscle actin.
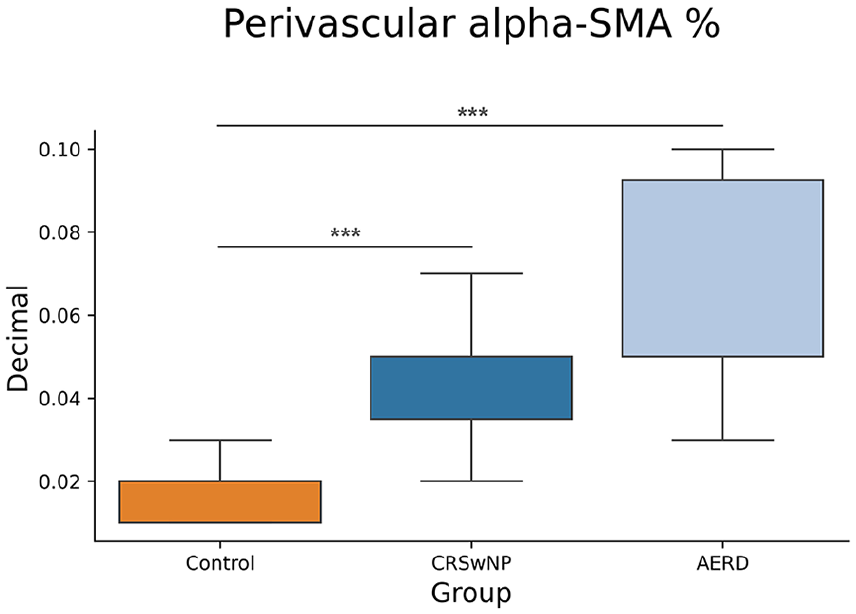
Comparison of percentage of perivascular α-SMA-positive cells between patient groups. The difference in the number of α-SMA-positive cells was shown between the control and CRSwNP and the control and AERD groups but not between the CRSwNP and AERD groups. Results are represented as medians with 25th and 75th percentiles. CRSwNP, chronic rhinosinusitis with nasal polyps; AERD, aspirin-exacerbated respiratory disease; α-SMA, alpha-smooth muscle actin.
Statistical analysis of the relationship between α-SMA expression, clinical parameter scores, and eosinophil counts revealed a positive correlation with high statistical significance (P < .001) for all parameters (Figure 6). α-SMA expression in NP tissue correlates strongly with ES (R = .8), LMS (R = .79), SS (R = .65), and the eosinophil counts (R = 0.54). However, the percentage of perivascular-positive α-SMA cells correlates slightly better with eosinophil counts (R = .63).
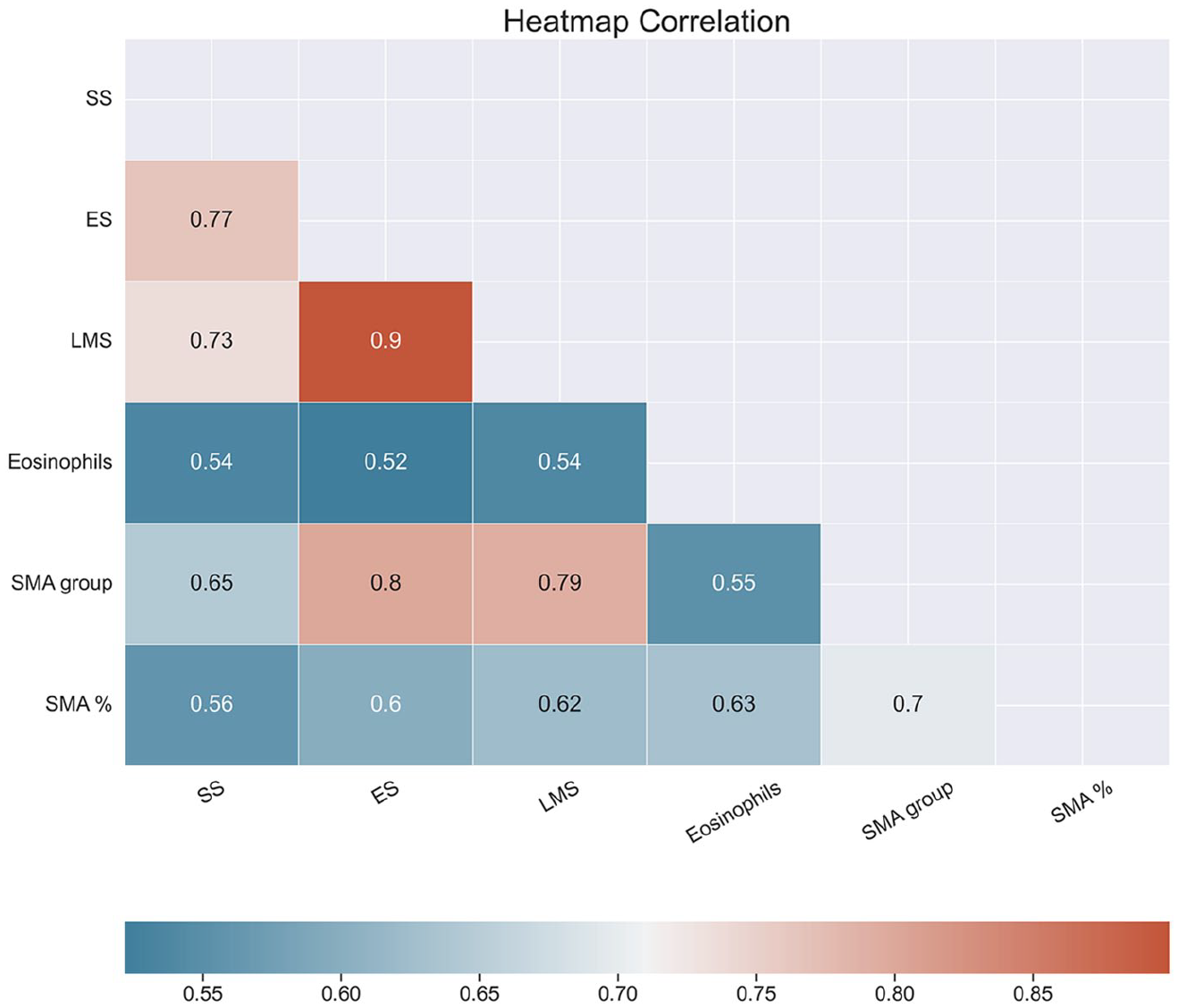
Heatmap of correlation of parameters analyzed in the present study. Colors in the heatmap correspond to the strength and direction of correlation, where a red color denotes a high positive correlation, while a blue color denotes a less positive (moderate) correlation. Values in the heatmap boxes represent Spearman’s correlation coefficient. SS, symptom score; ES, endoscopic score; LMS, Lund-Mackay score; SMA, smooth muscle actin subepithelial expression; SMA%, percent of perivascular-positive SMA cells.
Discussion
Considering that myofibroblasts are important factors in the pathophysiology of NP, we primarily assessed whether there is a difference in IHC expression of α-SMA among our participants. The results indicated significant differences in the extent of this immunoexpression. The fewest activated fibroblasts were found in patients with clinically healthy mucosa, while the highest IHC expression was observed in patients with AERD. The obtained differences can be interpreted as key roles of fibroblasts in initiating and maintaining the inflammatory process in inflammatory NPs. Zhang et al 10 demonstrated that myofibroblasts secrete numerous inflammatory mediators. Foremost among these are chemokines with potent eosinophilic chemotactic activity: CCL5, also known as regulated on activation, normal T cell expressed and secreted, eotaxin, CCL13, also known as monocyte chemoattractant protein 4 (MCP-4), as well as CCL20, known as macrophage inflammatory protein 3 alpha, which facilitates the migration of immature dendritic cells and T cells, and CCL2 (monocyte chemoattractant protein-1, MCP-1) which recruits macrophages. 10 The secreted cytokines support the hypothesis that fibroblasts in NPs are simultaneously important initiators of the T2 immune response—IL-33 and TSLP but also powerful modulators of the intensity of the inflammatory response—IL-4 and IL-32. Interestingly, fibroblasts in NPs produce IL-17A, a characteristic cytokine of Th17 cells, IL-6, and IL-8, which are important chemoattractants for neutrophils.10,18
Myofibroblasts of the nasal mucosa attract dendritic cells by chemokines and prepare them to initiate T2 inflammation.10,18 The interaction with eosinophils is particularly important. Yang et al 19 demonstrated in an in vitro study that co-culturing eosinophils with nasal fibroblasts leads to extensive production of cytokines and growth factors. Collectively, this results in the intensification of local inflammation and tissue remodeling. 19
The results of our study suggest that the level of myofibroblast activity is significantly the highest in the NP tissue of patients with AERD. We hypothesize that this is due to the increased production of pro-inflammatory CysLTs and PGD2, which induce excessive recruitment of eosinophils, basophils, and ILC2 from the bloodstream into the tissue. This contributes to significant local inflammation and promotes myofibroblast differentiation, creating a positive feedback loop that further amplifies the inflammation.6,7 In addition, Liu et al 20 identified COX-2 mRNA in NP fibroblasts using in situ hybridization, suggesting that these fibroblasts may also be responsible for part of the excessive PGD2 production.
Although the number of eosinophils was the highest in the NP tissue of patients with AERD, no statistically significant difference was found between the number of eosinophils in the NPs of these patients and those with CRSwNP. While these results initially seemed illogical to us, similar findings have been reported by other authors, such as Stevens et al. 6 The explanation is that activated eosinophils in whole numbers are difficult to demonstrate with the hematoxylin-eosin staining technique. Eosinophils should be quantified in the active phase, when they secrete a major basic protein (MBP), as shown in the study by Vukadinović et al. 21 That study clearly showed that patients with AERD have significantly higher immunoexpression of MBP compared to other patients with CRSwNP. 21
While examining the correlations between histological and clinical parameters, we found that both the tissue eosinophil number and the degree of IHC expression of α-SMA are positively correlated with clinical parameters. This correlation is the highest between the total subepithelial expression of α-SMA and the ES and between α-SMA expression and degree of disease extension on CT images of the paranasal sinuses. So, our results suggest a direct relationship between the number of myofibroblasts and the severity of chronic inflammation in the sinonasal region.
Special attention was given to evaluating the ratio between perivascular α-SMA cells and the diameter of blood vessels in the analyzed tissue samples. We assumed that their accumulation in the zone around blood vessels would be significantly higher due to the activity of vascular cell adhesion molecule 1 (VCAM-1). VCAM-1 is an endothelial vascular cell protein that has an affinity for circulating T2 cells, especially eosinophils, and allows them to enter directly into the tissue. VCAM-1, also expressed on the surface of nasal fibroblasts, attracts eosinophils and mast cells, contributing to the shift toward a T2 response in CRS. 22 Cytokines, secreted by nasal fibroblasts, cause tissue eosinophilia primarily by interacting with vascular endothelial cells but also by directly affecting the degree of VCAM-1 receptor expression at the surface of fibroblasts. 22 Our results indicate a significant difference in the percentage ratio of perivascular myofibroblasts between the control group and both groups with sinonasal polyposis. However, no difference was observed between patients with CRSwNP and those without AERD. An assumed limitation is the absence of double staining with both cytoplasmic and nuclear markers, which would make a clear distinction between myofibroblasts and surrounding cells and enable accurate perivascular counting. Such an estimation could be the focus of some future studies.
Our study has certain limitations. Apart from the fact that IHC did not detect eosinophils in the active phase, we did not estimate how myofibroblasts, as the main structuring cells, participate in the growth of NPs. For that type of research, it is necessary to determine the concentrations of growth factors, primarily basic fibroblast growth factor, either in nasal secretions or directly in the tissue of NPs, and this should be the subject of our next study.
Conclusion
Our results indicate that the overall IHC expression of α-SMA, a marker of active myofibroblasts, is highest in the NP tissue of AERD patients. Although the number of eosinophils, assessed by the hematoxylin-eosin technique, was highest in the AERD group, there was no significant difference compared to patients with CRSwNP without aspirin sensitivity. The total level of IHC expression of α-SMA is correlated well with the size of NPs and the extent of the disease on CT scans of the paranasal sinuses, suggesting that determining the number of myofibroblasts in the tissue of NPs could serve as a reliable marker for assessing the clinical status of the disease.
Footnotes
Acknowledgements
This study was conducted as a part of a scientific project of the Faculty of Medicine of the Military Medical Academy, University of Defence, Belgrade, Serbia (MFVMA02/23-25/). Thanks to Professor Ivan Zaletel for his help in the statistical analysis and graph making.
Ethical Considerations
The study was approved by the Ethics Committee of the Military Medical Academy, Belgrade, Serbia (Approval Number 21/2022).
Consent for Publication
Written informed consent was obtained from all participants for using data from their medical histories.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data from the study are available upon request.
