Abstract
Introduction
Respiratory epithelial adenomatoid hamartoma (REAH) is a histologically-benign lesion of the sinonasal region characterized by pseudo-glandular proliferation of respiratory epithelium and its growth into an edematous stroma, rich in inflammatory cells, especially mast cells. 1 In their 1995 retrospective study, Wenig and Heffner first described them in 31 cases. 2 According to the World Health Organization classification of head and neck tumors, REAH is classified as a benign tumor, along with seromucous and chondromesenchymal hamartoma. 3 However, the origin of REAH is still a matter of controversy. Although the name “hamartoma” indicates errors in the development of tissues and growths that occur due to these anomalies, there are also hypotheses about the tumor and inflammatory etiopathogenesis of REAH.4-6 Ozolek and Hunt 5 compared the genetic profile of REAH, sinonasal adenocarcinoma, and chronic inflammation of the nasal mucosa. The results showed similarities between REAH and adenocarcinoma in the percentage of fractional allelic losses and loss of heterozygosity, suggesting that REAH might even be a precursor of sinonasal adenocarcinoma. 5 Jo et al 6 described several REAH and low-grade sinonasal adenocarcinoma association cases. The origin of REAH has been described in different places in the nasal cavity, most often in the olfactory region, although the posterior-upper part of the nasal septum, the nasal turbinates, and the floor of the nasal cavity are also described as the origin.7-10 Unilateral or bilateral primary localization of REAH in the olfactory region leads to the widening of the olfactory cleft on the coronal and axial projection of computed tomography (CT).11,12 Previous studies indicate a high level of association of REAH with sinonasal chronic inflammatory entities: allergic rhinitis (AR), chronic rhinosinusitis with nasal polyps (CRSwNP), and aspirin-exacerbated respiratory disease (AERD).13-15 French authors report that in 35 to 48% of samples obtained during endoscopic sinus surgery (ESS) for CRSwNP, REAH tissue can also be found.16,17 On the other hand, an epidemiologic study from Brazil indicates that NPs are found in 47.4% of patients who underwent surgery for REAH. 18 Interestingly, in most of these patients, both REAH and NPs are most often located in the central parts of the sinonasal region, which closely resembles the endoscopic and radiological findings in central compartment atopic disease (CCAD).19,20 REAH is difficult to diagnose at the clinical and radiological findings level, given the high association level with CRSwNP. Symptoms are nonspecific and almost identical to those of CRSwNP. That is why, in most cases, REAH is discovered accidentally, during the analysis by a pathologist.1-7 Although their yellowish-pink color and rough cerebriform appearance is somewhat characteristic, a definitive diagnosis is made based on a detailed pathohistological or even immunohistochemical (IHC) analysis.6-8
Previous studies have not examined the influence of comorbidities on the clinical characteristics of REAH, and our study is the first of its kind. Therefore, the aim was to examine the frequency of comorbidities in patients with REAH and whether they influence the dimension of these lesions.
Materials and Methods
In this retrospective descriptive study, we evaluated the clinical characteristics of patients diagnosed with REAH who were surgically treated and followed up over 15 years. The study was conducted following the Declaration of Helsinki and after the decision of the Ethics Committee of the Faculty of Medicine of the Military Medical Academy in Belgrade, Serbia (IRB Approval 21/2022). Patients gave their informed consent for using data from their medical histories. The STROBE reporting guideline was followed to present the methodology and results.
Patients with REAH were treated and monitored in 2 university institutions, the Military Medical Academy in Belgrade and the Clinical Center of Vojvodina in Novi Sad, from January 2010 to December 2024. Given that REAH could not be clinically distinguished from CRSwNP based on symptoms (nasal obstruction, increased nasal secretion, pressure, pain in the face, weakened sense of smell), as well as based on endoscopic and radiological findings, the definitive diagnosis was established based on pathohistological findings. All patients had a histologically-confirmed diagnosis of REAH based on the examination of tissue samples after ESS. Two pathologists examined the specimens independently of each other. The samples were first analyzed after staining with the hematoxylin and eosin technique. After that, REAH tissue samples were analyzed by IHC, including examination for (i) cytokeratin 7 (CK7) to evaluate the distribution of keratin, characterized for pseudostratified respiratory epithelium; (ii) Ki67 to evaluate proliferative activity in the epithelium; (iii) Alcian Blue to evaluate the presence of mucin in the glandular goblet cells of the epithelium. Leica Biosystems, Deer Park, IL, USA, manufactured the antibodies for all 3 markers.
We recorded the side of the nasal cavity from where the REAH was removed and the primary localization. All patients were evaluated for allergies, based on previous AR symptoms, skin prick tests, and serological tests for inhalant allergens. Data on asthma were obtained from the medical history. The association with CRSwNP was evaluated based on endoscopic, radiological, and histological findings, considering the specific radiological finding of the enlargement of olfactory cleft in REAH situated in this region, and the specific histological findings of REAH, different from inflammatory polyps. Patients were recognized as suffering from AERD if they had information about (i) the presence of bilateral NPs on endoscopic examination, such as CRSwNP; (ii) the presence of asthma diagnosed by a pulmonologist; (iii) worsening of respiratory symptoms after taking aspirin or one of the nonsteroid anti-inflammatory drugs (NSAIDs).
We compared patients with and without comorbidities based on demographic and clinical characteristics, including duration of symptoms and REAH size expressed as virtual volume in cubic centimeter, and follow-up period after the surgical treatment. Pathologists estimated the sizes of REAHs after surgically removing lesions, depending on their unilateral or bilateral appearance. Based on the multiplication of 3 dimensions: anteroposterior, superoinferior, and medial-lateral diameter. Pathologists separated the lesions in cases of association with inflammatory NPs, based on differences in macroscopic appearance and histological findings. While NPs were multiple, semitransparent, edematous, smooth, and poorly blood-stained on the surface, REAHs were yellowish-pink or pale grayish in color, weakly transparent, and well-blooded, uneven, and cerebriform surfaces. There were also significant histological differences between REAHs and NPs.
Statistical Analysis
The Mann-Whitney U test and the analysis of variance test with Dunn-Bonferroni corrections were used to analyze statistical significance between groups. Pearson’s correlation test was used to analyze correlations between clinical parameters. A statistical significance was set at the P-level of less than .05.
Results
A total of 20 previously-unoperated patients with REAH, 12 men and 8 women, were treated and followed up in our 2 institutions. Demographic and clinical characteristics are shown in Tables 1, 2, and 3. The largest number of REAH, 11 (55%), grew from the olfactory cleft. On coronal and axial CT scans, 8 patients showed bilateral and 2 unilateral enlargements of the olfactory cleft (Figure 1a). In 5 (25%) patients, REAH grew from the nasal septum (Figure 1b), 2 (10%) from the inferior, and 2 from the middle turbinate. Comorbidities were observed in 14 (70%) patients. In 6 (30%), it was perennial AR (PAR), in 5 (25%) CRSwNP, and in 3 (15%) AERD. In 57% of comorbidity cases, there were bilateral lesions. Two patients with PAR, 2 with CRSwNP, and all with AERD had asthma. Also, 2 patients with CRSwNP were sensitized to perennial allergens. In 87.5% of cases of bilateral REAH, the lesion was associated with inflammatory NPs, in 80% of patients with CRSwNP as a comorbidity, and in 100% of patients where it was AERD. When we divided the patients into groups with and without comorbidities, the duration of symptoms was longer in those with associated sinonasal chronic inflammation (P < .038) (Table 2). We found the highest average volume of REAH in patients with AERD (59.33 ± 88.91 cm3), far greater than in cases with PAR (14.83 ± 9.80 cm3), and CRSwNP (7.60 ± 2.19 cm3) (P < .001; P < .001, respectively) (Table 3). Finally, a positive correlation was found between the duration of symptoms and the lesion volume (R = .700; P = .001). The follow-up period was slightly longer in patients with than in patients without comorbidities [29.57 ± 10.34 months (from 10 to 47 months) vs 19.83 ± 6.27 months (from 10 to 26 months), respectively] (P < .028) (Tables 1 and 2). Recurrences of REAH were noted only in 2 patients in whom the lesion was associated with AERD.
Demographic and Clinical Characteristics of Patients With REAH.
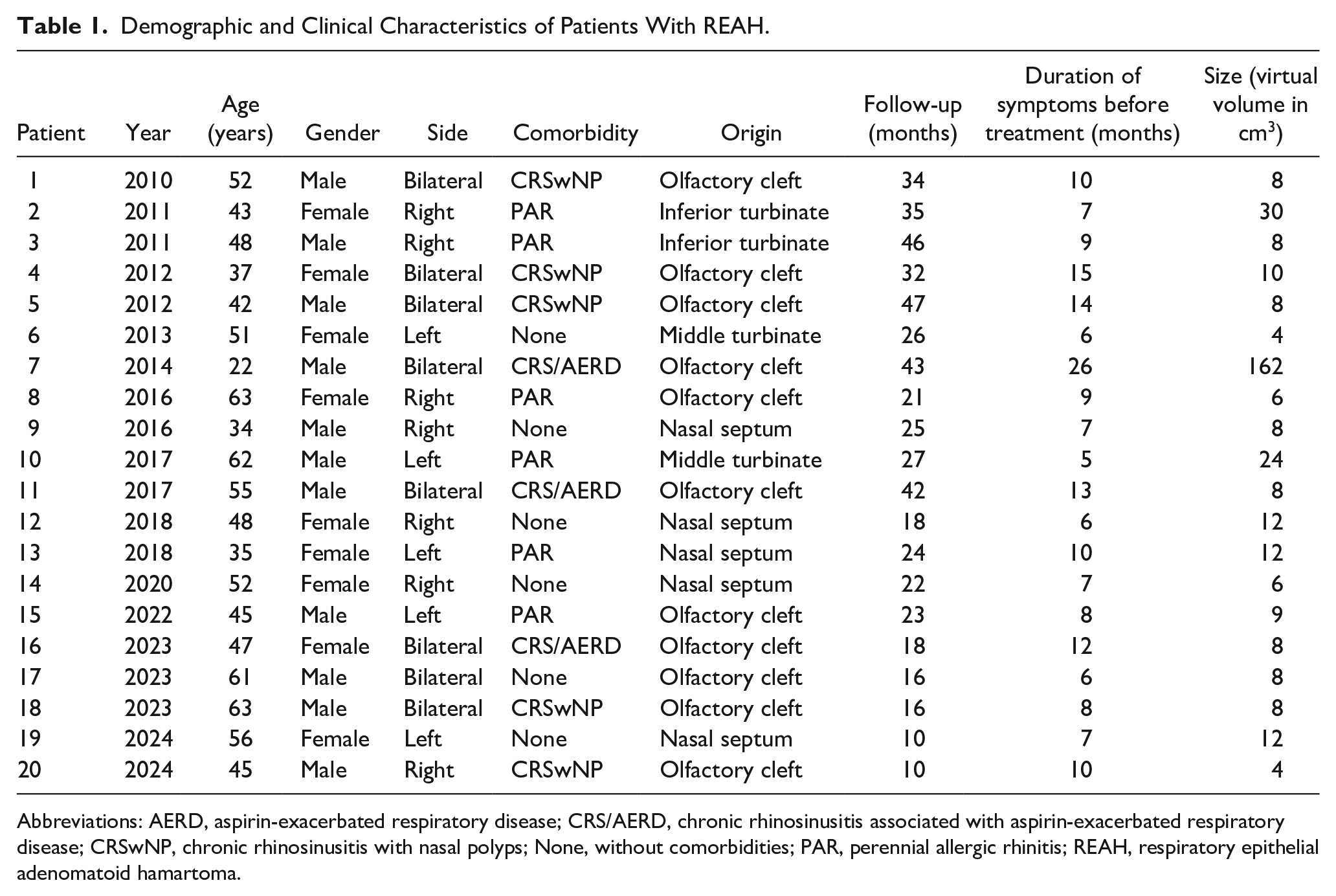
Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRS/AERD, chronic rhinosinusitis associated with aspirin-exacerbated respiratory disease; CRSwNP, chronic rhinosinusitis with nasal polyps; None, without comorbidities; PAR, perennial allergic rhinitis; REAH, respiratory epithelial adenomatoid hamartoma.
Comparison between Demographic and Clinical Parameters in Patients With REAH With and Without Comorbidities.
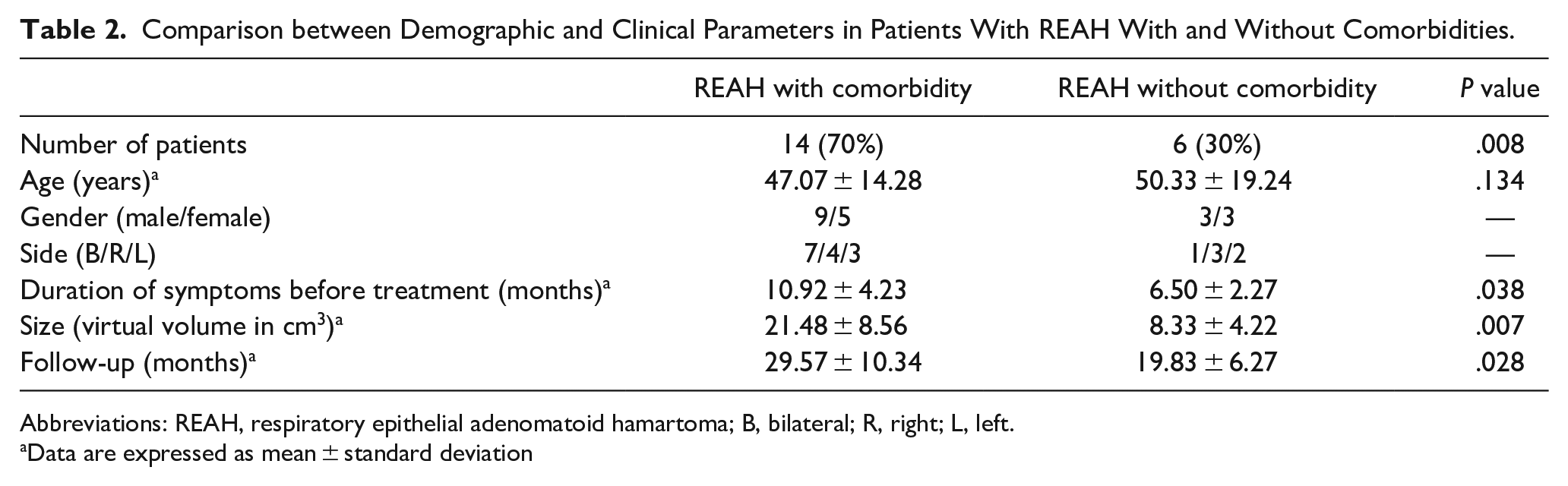
Abbreviations: REAH, respiratory epithelial adenomatoid hamartoma; B, bilateral; R, right; L, left.
Data are expressed as mean ± standard deviation
REAH Volumes Depending on Comorbidities.
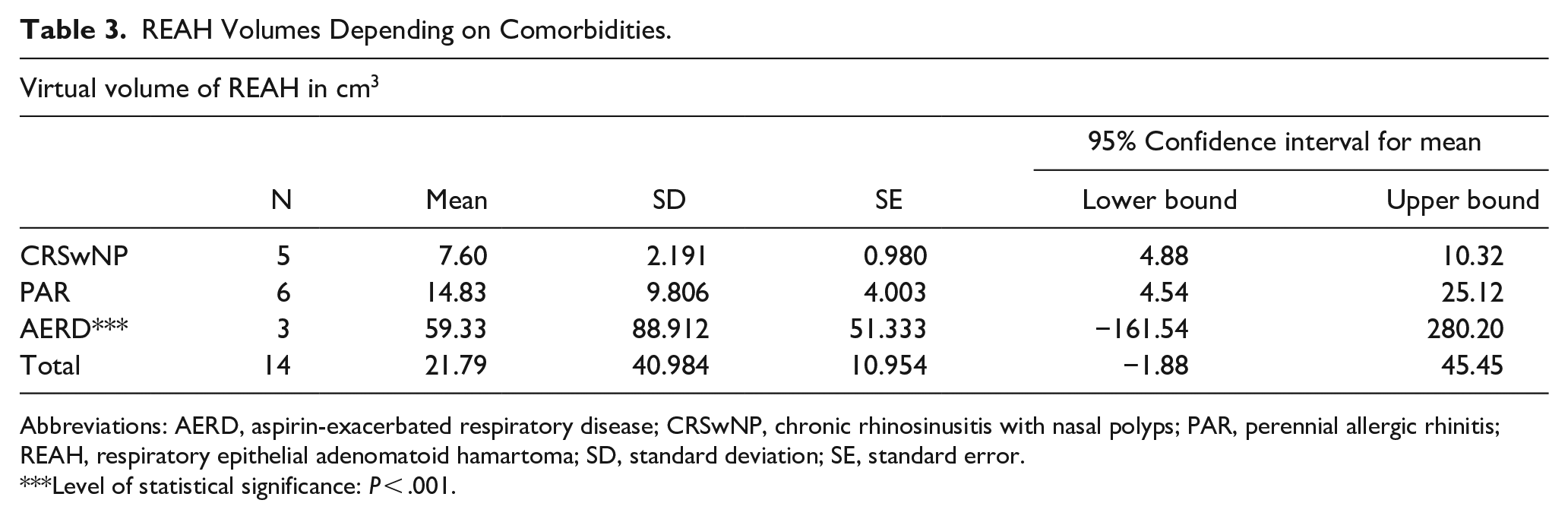
Abbreviations: AERD, aspirin-exacerbated respiratory disease; CRSwNP, chronic rhinosinusitis with nasal polyps; PAR, perennial allergic rhinitis; REAH, respiratory epithelial adenomatoid hamartoma; SD, standard deviation; SE, standard error.
Level of statistical significance: P < .001.
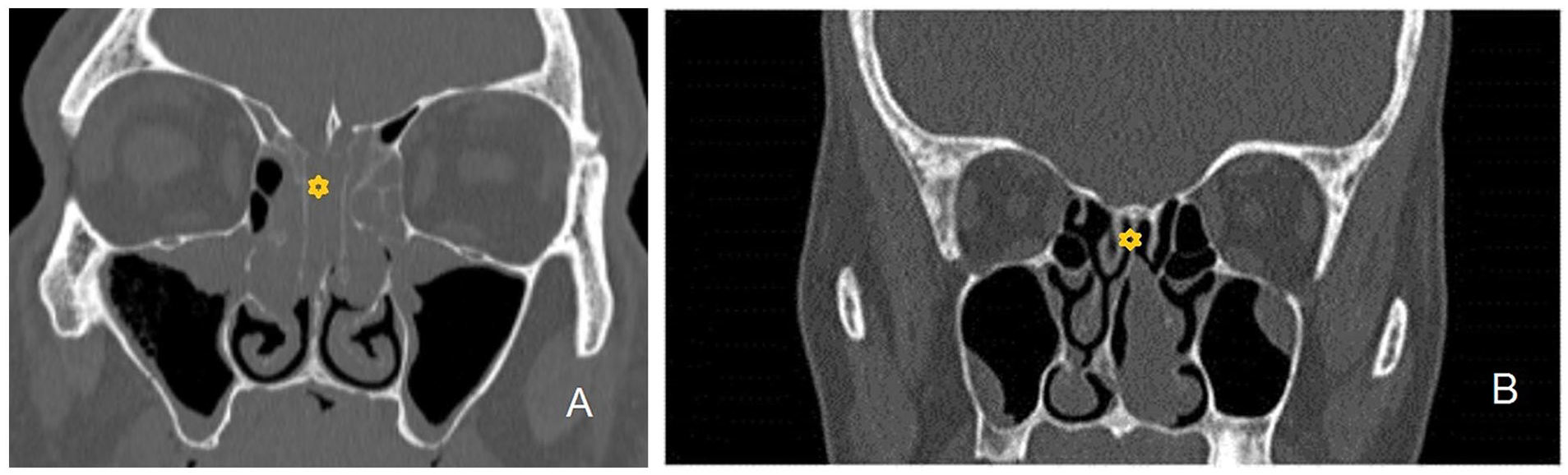
(a) Coronal CT scan shows the olfactory cleft’s right-sided enlargement due to the association between REAH and inflammatory NPs. (b) Coronal CT scan showing the left-sided lesion attached to the upper part of the nasal septum. CT, computed tomography; NP, nasal polyp; REAH, respiratory epithelial adenomatoid hamartoma.
The macroscopic appearance of REAH indicates yellowish-pink or pale grayish masses, with uneven surfaces and a cerebriform appearance (Figure 2a and b). Histologically, we found the polypoid lesion with pseudo-glandular proliferation. The pseudo-glands were widely spaced, small to medium sized, round or oval in shape, lined with pseudostratified ciliated columnar epithelium with mucin-secreting goblet cells. The stroma was edematous in some areas. “Glands” were surrounded by a thick eosinophilic basement membrane. There was a mixed inflammatory infiltrate in the stroma with domination of mast cells. Nuclear atypia and mitoses were absent (Figure 3a). Normal CK7 immunoreactivity (Figure 3b), low Ki67 immunoreactivity (Figure 3c), and high Alcian blue expression (Figure 3d) were found in all patients with REAH.
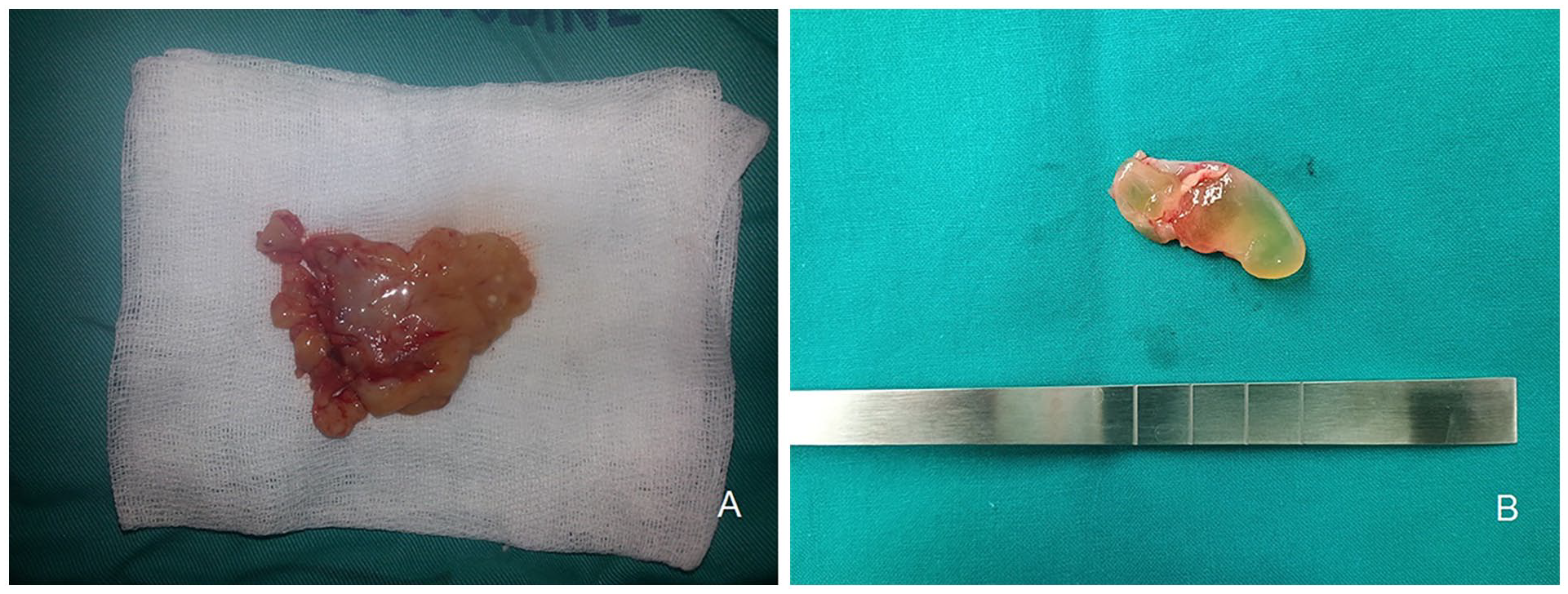
(a) The macroscopic appearance of REAH indicates yellowish-pink or pale grayish masses, with uneven surfaces and a cerebriform appearance; (b) the macroscopic appearance of the REAH removed from the nasal septum. REAH, respiratory epithelial adenomatoid hamartoma.
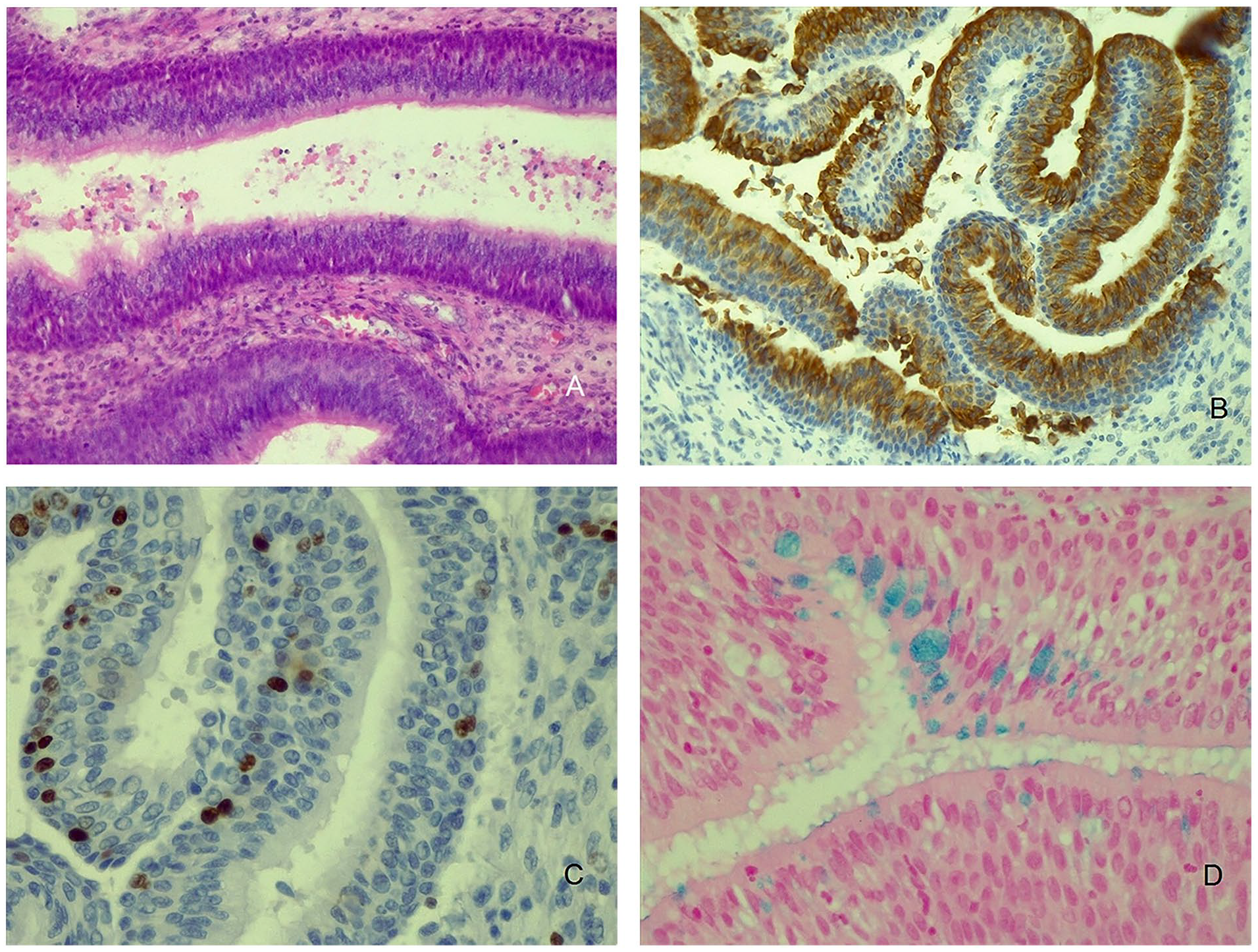
(a) The histological appearance of REAH. This is the polypoid lesion with pseudo-glandular proliferation. The pseudo-glands were widely spaced and lined with pseudostratified ciliated columnar epithelium with mucin-secreting goblet cells. The stroma was edematous in some areas. “Glands” were surrounded by a thick eosinophilic basement membrane. There was a mixed inflammatory infiltrate in the stroma with domination of mast cells. Nuclear atypia and mitoses were absent (hematoxylin and eosin staining, magnification ×200). (b) Normal CK7 immunoreactivity in the REAH tissue indicates an expression of keratin characterized for pseudostratified respiratory epithelium (magnification ×200). (c) Low Ki67 immunoreactivity indicates low mitotic activity in the tissue of REAH (magnification ×400). (d) High Alcian blue expression in the REAH pseudo-glands indicates the high expression of mucin inside of the goblet cells (magnification ×400). CK7, cytokeratin 7; REAH, respiratory epithelial adenomatoid hamartoma.
Discussion
The main differential diagnostic problems concerning REAH are inflammatory NPs, inverted (Schneiderian papilloma), and low-grade sinonasal adenocarcinoma. Inflammatory NPs are characterized by edema of the stroma with strong inflammatory infiltrate, composed mainly of eosinophils and lymphocytes, a proliferation of the respiratory epithelium, and stromal fibroblasts, mainly without glandular (pseudo-glandular) proliferation.21-25 Inverted papillomas are characterized by thickening and folds of modified epithelium, without glands.1,3,4 Low-grade sinonasal adenocarcinoma is composed of glandular proliferation, with pleomorphism, and numerous atypical mitoses with varying degrees of cellular atypia.1,3,5,6
Similar to previous studies,7,8,11,12,14,15 the most common location of REAH is the olfactory cleft, while the second most common location is the upper part of the nasal septum. In as many as 87.5% of cases, bilateral REAH was associated with inflammatory NPs. In 7 out of 20 patients, asthma was present, and in 8 patients, we had sensitization to perennial allergens, including dust mites, house dust, and animal hair. Given the above, it is suggested that REAH is one of the manifestations of airway inflammation driven by T2 mediators. However, it seems that the narrow space of the olfactory cleft is an ideal place for accumulating inflammatory cells that produce high amounts of inflammatory mediators. Also, the duration of symptoms is directly correlated with the volume of REAH. We assume that longer exposure of respiratory epithelium and stroma to such high levels of mediators leads to more intense growth of this lesion. T2 mediators, including those released from mast cells and eosinophils eventually lead to tissue remodeling with a predominance of glandular components. This could explain the high mucin immunoreactivity in the REAH tissue. Dense stromal infiltration by mast cells supports the hypothesis that REAH is one of the manifestations of strong local tissue response to high levels of inflammatory mediators. 16 The REAH is made up of pseudo-glands that are in continuity with the superficial respiratory epithelium. These “false glands” are predominantly composed of respiratory ciliated epithelium with numerous goblet cells that produce a large amount of mucin. 21 The richness of the stroma with mast cells that secrete the enzyme tryptase, including chymase, gelatinases (matrix metalloproteinases), and cathepsin, explains the degree of disruption of the tissue structure in REAH. 16
Our results suggest that AERD was the most powerful provoking factor for the growth of REAH. In patients with AERD, the lesions were by far the largest in volume, and in 2 out of 3 patients, we only had recurrences in our case series. This confirms the fact that CRS associated with AERD is the most difficult-to-control chronic inflammatory disease of the upper respiratory tract. 22 According to previous studies, the activity of mast cells and eosinophils is much higher in AERD than in other forms of CRSwNP.23,24 AERD is also one of the main provoking factors for the metaplasia of respiratory epithelium in NPs to transitional epithelium. 25 An interesting result is that patients with PAR have, on average, twice as large REAHs compared with patients with CRSwNP, outside of AERD. A possible explanation is that the narrow space of the olfactory cleft hindered the comparative growth of REAHs and NPs, which was overcome in patients with AERD by a large growth potential in severe chronic inflammation.
The retrospective nature of the study and the relatively-small number of patients are the main limitations. On the other hand, this is the largest series of REAH cases in Southeast Europe. Also, in this study, we looked at the clinical characteristics of the lesion, but we did not compare the histological and IHC characteristics of REAH depending on whether the lesion was associated with comorbidity. This will certainly be the subject of a future study with a larger number of patients.
Conclusion
The results of our study indicate a high degree of association between REAH and chronic inflammation of the nasal mucosa and suggest that inflammation in AERD is the strongest provoking factor for the growth of REAH. A direct correlation was observed between the duration of nasal symptoms and the size of the lesion, which implies the conclusion that the growth of the lesion is a direct consequence of the intensity and duration of chronic sinonasal inflammation.
Footnotes
Acknowledgements
This study was performed as a part of the scientific project of the Faculty of Medicine of the Military Medical Academy, University of Defence, Belgrade, Serbia (MFVMA02/23-25/).
Data Availability Statement
Data from the study are available upon request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
The study was approved by the Ethics Committee of the Military Medical Academy, Belgrade, Serbia (approval number 21/2022).
Informed Consent/Patient Consent
Written informed consent was obtained from all participants for using data from their medical histories.
Trial Registration Number
Not applicable.
