Abstract
Objective:
This study aimed to investigate the influence of cancer type on radiotherapy-induced otitis media with effusion (RTOME) occurrence and to assess the impact of anatomical region-specific radiation exposure on both RTOME incidence rates and associated pathogenic processes.
Methods:
A retrospective analysis was conducted on 1046 head and neck cancer (HNC) patients who underwent radiotherapy at Peking Union Medical College Hospital between 2016 and 2023. The incidence of RTOME was assessed across various cancer types, and the radiation exposure probabilities of different head and neck regions were compared between patients with and without RTOME using the chi-squared test (χ2) test and the Mann-Whitney U test.
Results:
The overall incidence of RTOME in patients with HNC was 5.83%. The incidence varied significantly by cancer type, with nasopharyngeal carcinoma showing the highest rate (17.40%), followed by ocular and orbital cancers (6.25%), nasal cavity and paranasal sinus cancer (4.35%), oral cavity and oropharyngeal cancers (4.32%), laryngeal and hypopharyngeal cancers (0.32%), and thyroid cancers (0%). Furthermore, patients with RTOME exhibited significantly-higher radiation exposure frequencies in five specific anatomical regions: the skull base and intracranial, parotid and periauricular, nasal cavity and paranasal sinuses, nasopharynx, and parapharyngeal space, than those without RTOME.
Conclusions:
The incidence of RTOME is strongly associated with both cancer type and radiation exposure to specific anatomical regions. Radiation targeting certain areas may increase the risk of developing RTOME.
Introduction
Radiotherapy is a key treatment for head and neck cancers (HNCs) but is often associated with complications, including otitis media with effusion (OME). The incidence of OME after radiotherapy varies widely between different studies, ranging from 8% to 29%. 1
The management of radiotherapy-induced otitis media with effusion (RTOME) is clinically challenging, frequently requiring prolonged intervention, including repeated myringotomy or grommet insertion. Despite interventions, recurrent RTOME remains difficult to manage, significantly reducing the quality of life of patients undergoing radiotherapy. 2
In previous studies on HNCs and RTOME, it has been suggested that radiotherapy-induced eustachan tube dysfunction (ETD) is a common mechanism leading to RTOME, depending on the radiotherapy dose and irradiation range. For example, the incidence of OME in patients with nasopharyngeal carcinoma (NPC) and parotid carcinoma is significantly higher than that in other patients with HNC,3-7 which may be related to the higher dose received by the eustachan tube (ET) and middle ear during radiotherapy. However, previous studies could not explain the difference in the incidence of OME in the same tumor with the same radiotherapy dose and site. Furthermore, in tumors distant from the ET and middle ear, the influence of the original tumor location and radiation-targeted anatomical regions on the incidence of RTOME remains to be illustrated.
This study aimed to compare the RTOME incidence across HNC types and radiation-targeted anatomical regions, to explain the possible pathogenesis of RTOME.
Patients and Methods
Patient Selection
The clinical data of 1046 patients with HNC who underwent RT at Peking Union Medical College Hospital between January 2016 and November 2023 were retrospectively analyzed.
Inclusion criteria:
Patients aged ≥18 years.
Completion of clinical evaluation and imaging studies for tumor assessment prior to radiotherapy.
Availability of complete clinical, pathological data and radiological records.
Diagnosis of OME confirmed by senior otolaryngologists based on standardized diagnostic criteria (see below).
Exclusion criteria:
History of OME, middle ear diseases, or ear surgeries prior to radiotherapy.
Previous radiotherapy or history of other malignancies.
Malignant tumors involving multiple organ systems or distant metastases.
Interruption or incomplete course of radiotherapy.
Diagnostic criteria for OME (based on reference 8 ):
Clinical symptoms: Presence of tinnitus, ear fullness, hearing loss, or related complaints.
Physical examination and otoscopy: Findings of congested or retracted tympanic membrane, limited mobility, and/or visible middle ear effusion.
Pure tone audiometry: Evidence of conductive or mixed hearing loss.
Acoustic immittance testing: Demonstrated type B or type C tympanometric curves.
Statistical Analysis
Statistical analysis was performed using SPSS 26.0 (IBM SPSS Statistics). Continuous data were compared using the Mann-Whitney U test, and categorical data were analyzed using the chi-squared test. A P value < .05 was considered statistically significant.
Ethics Statement
This study was approved by the Ethics Committee of Peking Union Medical College Hospital. All procedures were conducted in accordance with relevant guidelines and regulations. The ethics approval number for this study is KYC-IEC-2022-II-05-V1.0.
Results
The Incidences of RTOME in HNCs
In this study, a total of 1046 patients receiving radiotherapy in our hospital were enrolled, with the distribution of cancer types and RTOME cases summarized in Table 1, a total of 61 patients developed RTOME. The overall incidence of RTOME in HNC was 5.83%, ranging from 17.40% in NPC to 0% in thyroid cancer.
Incidence of RTOME in HNC.

Abbreviation: RTOME, radiotherapy-induced otitis media with effusion.
Significant differences in RTOME incidence were observed among different tumor types (P < 0.05). The incidence of RTOME decreased as the primary tumor location shifted from upper to lower head and neck regions (Figure 1).
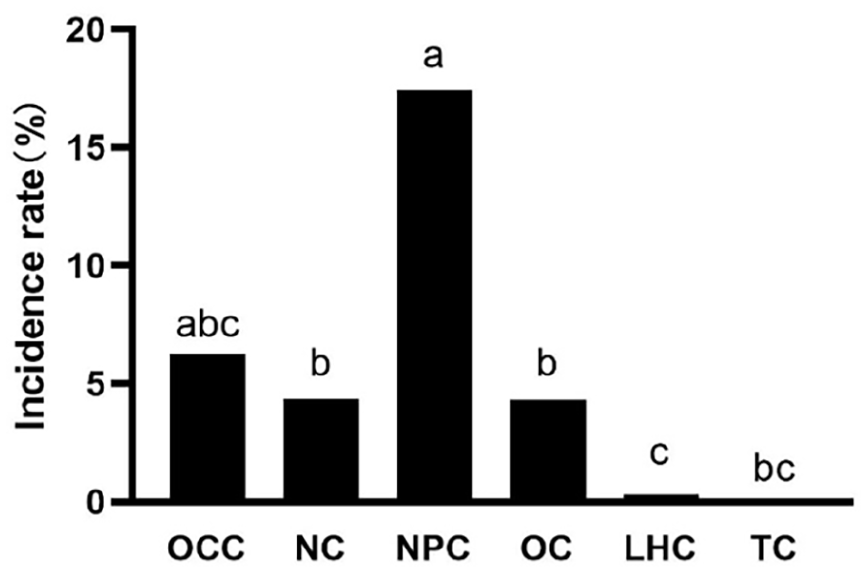
Incidence of RTOME in different types of head and neck cancer.
Radiation target regions of HNC
To determine factors driving RTOME incidence variability in patients with HNC, we systematically mapped 11 anatomical radiation target regions in the head and neck district (Figure 2, blue-shaded areas).
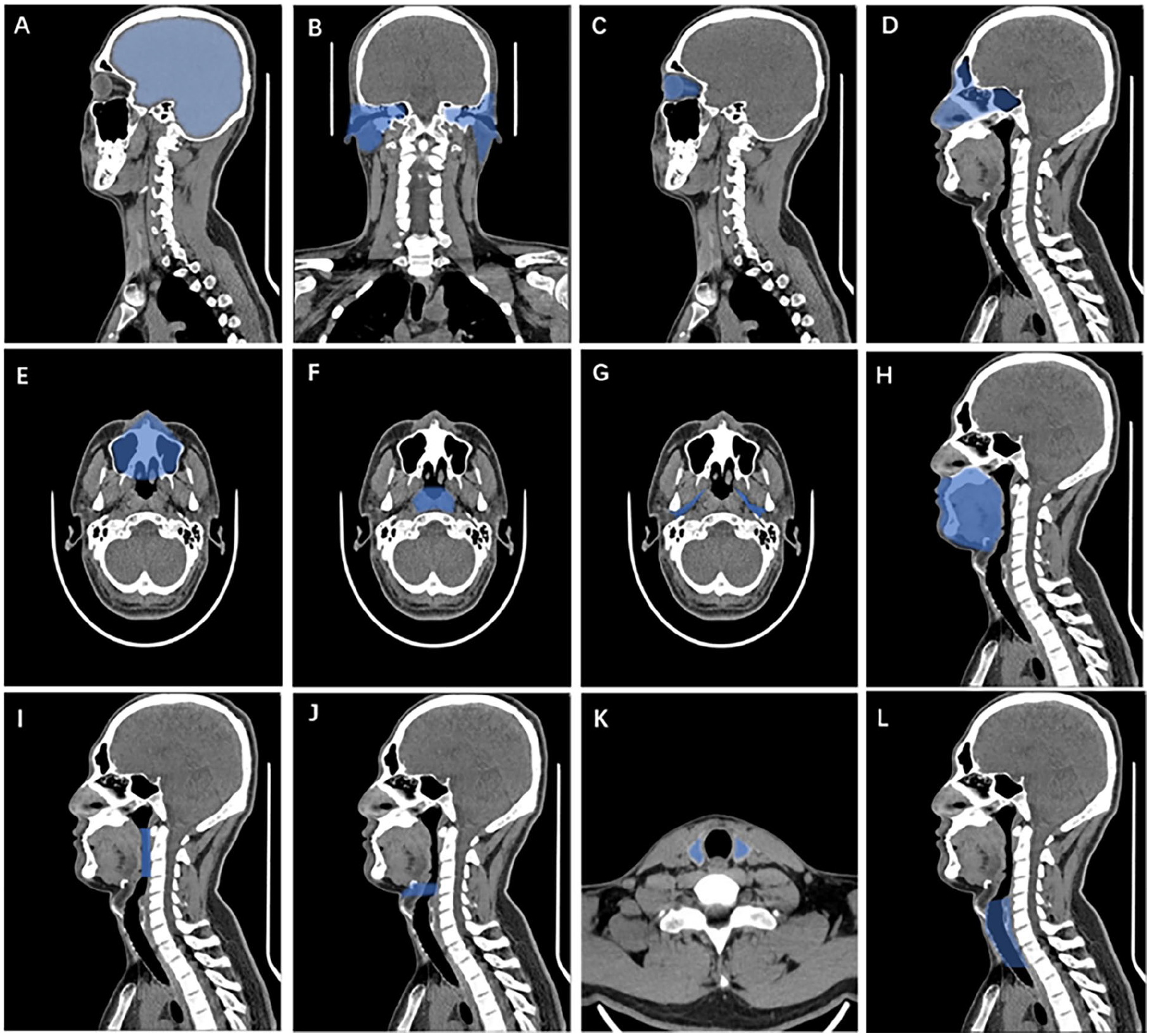
Eleven anatomical radiation target regions within the head and neck district. (A) skull base and intracranial, (B) parotid and periauricular, (C) orbit, (D) and (E) nasal cavity and paranasal sinuses, (F) nasopharynx, (G) parapharyngeal space, (H) oral cavity, (I) oropharynx and retropharynx, (J) larynx, (K) thyroid, (L) trachea and esophagus.
Through a comprehensive analysis of radiotherapy treatment plans from a cohort of 1046 patients, we quantitatively determined the radiation exposure probability for each anatomical region, with detailed results presented in Tables 2 and 3 and graphically illustrated in Figures 3 and 4. Our analysis revealed that the primary tumor site consistently demonstrated the highest radiation exposure probability across all HNC subtypes. A representative example was observed in NPC cases, where the nasopharyngeal region exhibited a 100% radiation probability. Furthermore, our investigation identified several anatomical regions, including the parapharyngeal space, that represented common high-risk radiation areas shared among multiple cancer types.
Radiation Probability of 11 Anatomical Radiation Target Regions.

Radiation areas in patients with and without RTOME.

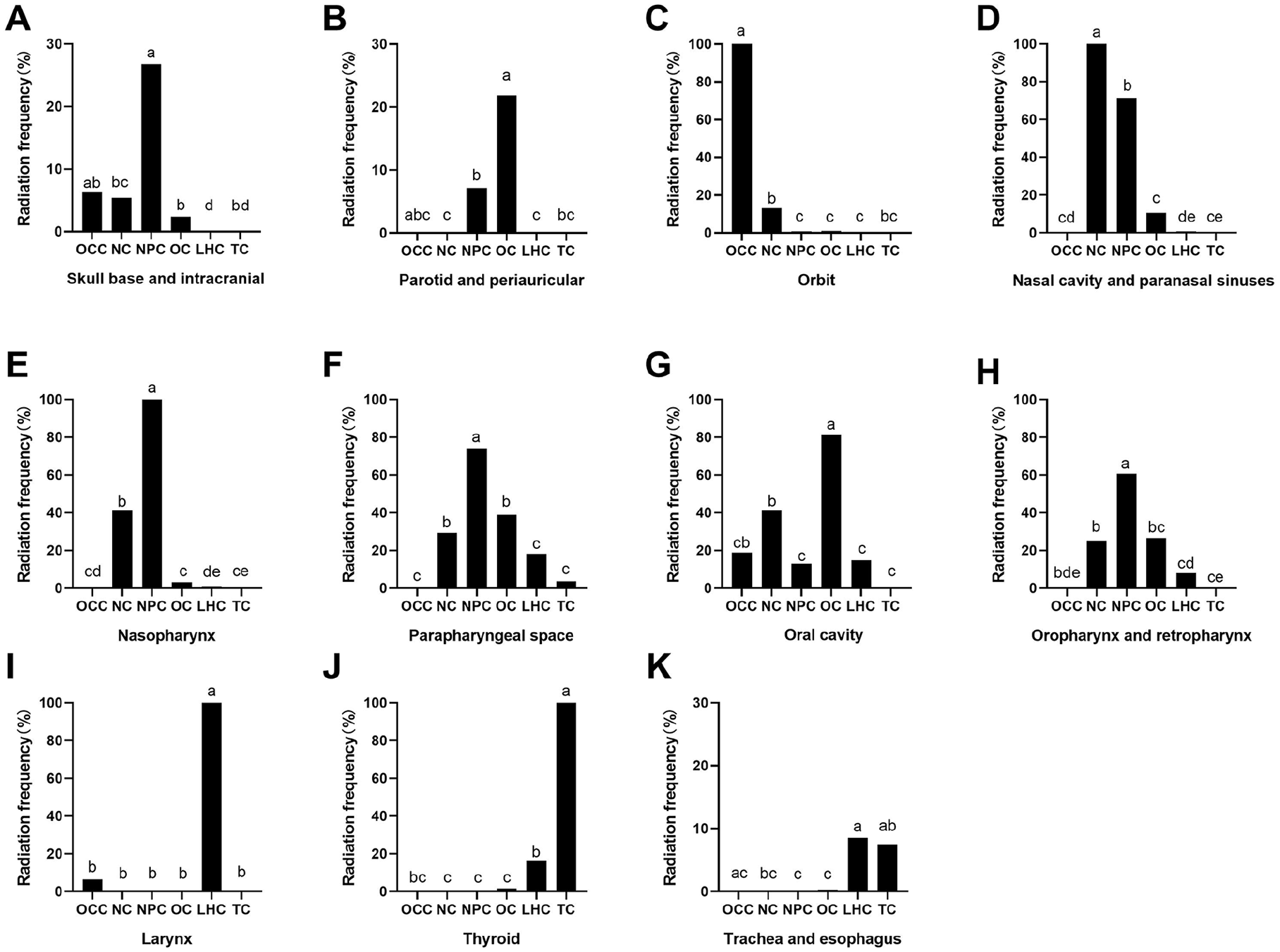
Radiation probability of 11 anatomical radiation target regions. (A) skull base and intracranial, (B) parotid and periauricular, (C) orbit, (D) nasal cavity and paranasal sinuses, (E) nasopharynx, (F) parapharyngeal space, (G) oral cavity, (H) oropharynx and retropharynx, (I) larynx, (J) thyroid, and (K) trachea and esophagus.
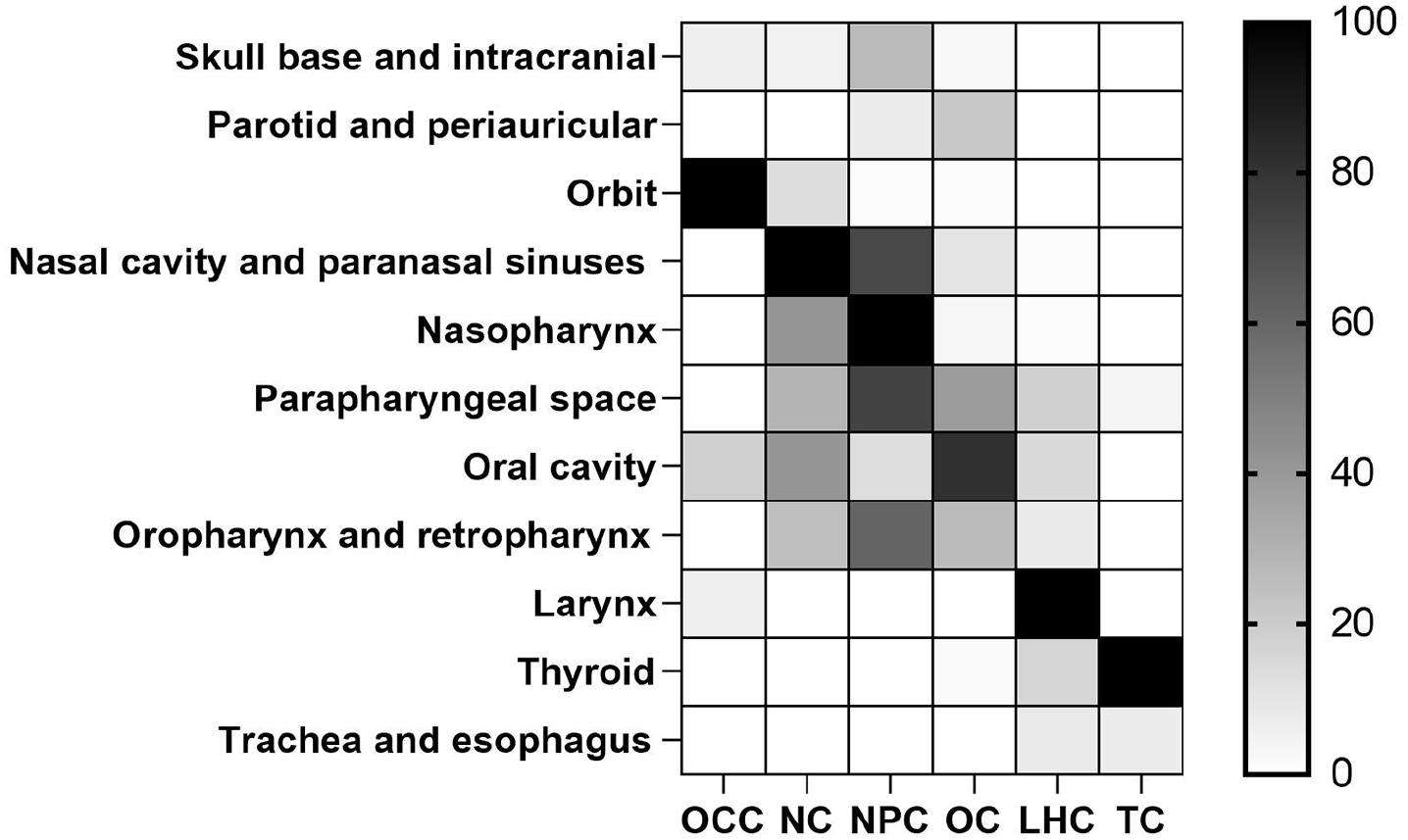
Heatmap visualization of 11 radiation probability areas. The color gradient represents the radiation probability intensity, ranging from white (0% probability) to black (100% probability). Each row corresponds to a specific cancer type, while columns represent anatomical regions.
Radiation Areas in Patients With and Without RTOME
Analysis between patients with and without RTOME revealed significantly-higher radiation exposure frequencies in six distinct anatomical regions: the skull base and intracranial region, parotid and periauricular region, nasal cavity and paranasal sinuses, nasopharynx, parapharyngeal space and larynx. These findings suggest a potential correlation between radiation exposure to these specific anatomical sites and the development of RTOME.
Disscusion
Pathogenesis of RTOME
The elevated incidence of RTOME is predominantly attributable to ETD induced by radiotherapy. 1 The pathological mechanisms involve multiple radiation-induced alterations: (1) Fibrotic changes in peritubal tissues, which represent one of the principal pathological manifestations of ETD, compromise the elastic properties and patency of the eustachian tube.9,10 (2) Radiation-induced mucosal edema, an early mark of postradiation OME, further impedes the physiological opening and closing mechanisms of the ET, disrupting middle ear pressure homeostasis. 11 (3) Radiation therapy, compounded by cancer-associated cachexia, may induce atrophy of adipose tissue, thereby exacerbating ETD. 12
The central pathophysiology of OME stems from eustachian tube dysfunction (ETD)-mediated impairment of middle ear pressure regulation, which generates negative pressure and subsequent fluid accumulation. This pathological cascade is further exacerbated by the impaired clearance of middle ear secretions due to ETD, creating a self-amplifying cycle of fluid retention and inflammation.
Importantly, radiation-induced ETD typically presents as an irreversible condition, inducing patients to persistent and recurrent episodes of RTOME.
Differences in the Incidence of HNC Types
The incidence of RTOME varies greatly in different HNC types, which shows a decreasing trend as the primary radiation site moves from upper to lower head and neck regions. Specifically, upper HNCs, including ocular and orbital cancers, nasal cavity and paranasal sinus cancer, NPC, oral cavity and oropharyngeal cancers, exhibit higher RTOME prevalence compared with lower HNCs such as laryngeal, hypopharyngeal, and thyroid cancers. This observation is consistent with previous research demonstrating greater ototoxicity in upper HNCs. For instance, Chang et al reported a significantly-higher incidence of sensorineural hearing loss following radiotherapy in patients with NPC than those with hypopharyngeal cancer.13,14 Correspondingly, Kaul et al observed that tumors originating in the oral cavity, oropharynx, and nasopharynx were more frequently associated with auditory complications than esophageal and thyroid cancers, which showed minimal hearing changes. 15
Influence of Special Radiation-Exposed Areas on RTOME Incidence
Targeted radiation exposure to specific high-risk anatomical regions may play a significant role in the development of RTOME. Our study identified six radiation-exposed areas that showed statistically-significant differences in frequency between RTOME and non-RTOME patients, including the skull base and intracranial region, parotid and periauricular region, nasal cavity and paranasal sinuses, nasopharynx, parapharyngeal space, and larynx. Notably, the laryngeal region demonstrated a unique negative correlation with RTOME incidence. Among 319 patients receiving laryngeal irradiation, no case developed RTOME, reinforcing our earlier finding that radiation exposure to lower head and neck regions exerts a minimal impact on RTOME pathogenesis.
In contrast, radiation exposure to five anatomical areas showed significant positive associations with RTOME development. Three of them—the parotid and periauricular, nasopharynx, and parapharyngeal space (PPS)—are anatomically and functionally connected to the ET and middle ear structures. The ET, which has its nasopharyngeal orifice situated in the nasopharyngeal region and its tympanic orifice located in the middle ear, is particularly vulnerable. Moreover, the tensor veli palatini (TVP) muscle, which regulates ET opening, is positioned within the PPS. Radiation-induced tissue damage in these critical areas can compromise ET functionality, leading to ETD, a primary etiology of OME.16,17 Additionally, radiation exposure to the temporal bone and middle ear cavity may directly induce radiation-related otitis media and mastoiditis, independent of ETD. Within the middle ear, radiation injury can result in structural alterations including ciliary loss, impaired ciliary motility, decreased cytoplasmic volume, and widened intercellular spaces.18,19 Clinical observations indicate that patients with HNC, particularly those with NPC, nasal cavity and paranasal sinus cancer, oral cavity and oropharyngeal cancer, show closer anatomical proximity to these three high-risk regions. The elevated incidence of RTOME in these specific cancer types further substantiates the important involvement of these anatomical areas in the pathogenesis of RTOME.
Our study identified two additional anatomical regions potentially associated with RTOME development: the skull base and intracranial region, the nasal cavity and paranasal sinuses area. Analysis of radiation exposure patterns revealed that NPC patients (excluding those with primary intracranial or skull base tumors) displayed the highest radiation frequency in the skull base and intracranial region, followed by patients with nasal cavity and paranasal sinus cancer. This observation aligns with the known propensity of these malignancies to invade skull base and intracranial structures.20,21 Such tumor invasion, particularly through bone defects or fractures, may predispose patients to various postradiation complications, including bone sclerosis, osteoporosis, osteolytic changes, radiation-induced osteomyelitis, and osteonecrosis.22-25
Although primary skull base and intracranial tumors were not included in our study cohort, we hypothesized that OME might be a secondary complication in these cases. This hypothesis is supported by previous radiographic findings demonstrating mastoid opacities in 40 out of 61 patients (65.6%) during post-radiotherapy follow-up, suggesting a potential association with OME development. 19
The nasal cavity and paranasal sinuses warrant special consideration, as their irradiation may predispose to OME through retrograde microbial translocation along the eustachian tube. Radiotherapy has been proved to influence the composition of resident microbial communities within the body, with much evidence of effects on the oral and nasal microbiota after radiotherapy.26,27 Contrary to the traditional view of the middle ear as a sterile, microbiota-free space,28,29 emerging evidence reveals commensal bacterial communities with biofilm-forming capacity in the tympanic cavities of asymptomatic individuals.30,31 Lee et al 32 demonstrated consistent biodiversity between eustachian tube nasopharyngeal orifice secretions and oropharyngeal-oral microbiota, a finding corroborated by similar results from Jacob and Reguera. 33 These observations suggest that radiation therapy may similarly affect the resident microbial communities within the middle ear.
Radiotherapy directly kills resident middle ear microbiota, establishing a sterile microenvironment. Under these conditions, RTOME primarily presents as sterile inflammation, a conclusion substantiated by Liang et al’s findings of culture-negative effusions in RTOME cases. 34 De Ru et al further indicated that such sterile inflammatory process constituted a defense mechanism against potential pathogens.
This point was supported by the detection of multiple inflammatory mediators (eg, cytokines, interleukins) in OME effusions that modulate local immune responses. 35 The protective mechanism functions as a protective response, representing a well-balanced reaction mediated by both innate and adaptive immune systems in response to potential infectious threats.36,37
Although most RTOME cases retain low microbial state post-radiotherapy, nasal-sinus irradiation could induce rhinogenic complications (rhinitis, sinusitis, nasopharyngitis) through radiation-associated mucosal injury, necrotic debris accumulation, and epithelial dehydration. Such inflammatory processes may propagate to the eustachian tube’s nasopharyngeal orifice. ETD in this context can facilitate retrograde microbial translocation,4,9 disrupting post-radiation sterility and potentially progressing to suppurative otitis media, tympanic membrane perforation, or other severe complications. Gorbunova et al reported that 30% of patients developed ETD and 10% experienced unilateral catarrhal otitis media, partially supporting the association between sinusitis and otitis media after radiotherapy. 26
The Limitation of the Study
This study has several limitations that should be acknowledged. First, the single-center retrospective design exists selection bias, particularly regarding tumor type and case demographics. Second, the skull base and primary intracranial tumors were not included, which limited comparative analysis of radiation effects within these anatomical regions. Furthermore, the analysis did not include audiological assessments, such as pure tone audiometry and tympanometric evaluations. In the next prospective study, we would conduct a multicenter study to reduce selection bias, and incorporate the results of audiology examination into statistics to improve its clarity and scientific rigor.
Conclusion
This study demonstrates that RTOME is a significant complication in HNC treatment, with an overall incidence of 5.83%. The anatomical distribution of radiation exposure plays a crucial role in RTOME development, compared with non-RTOME patients, RTOME patients received higher radiation frequencies in five critical regions—the skull base and intracranial, parotid and periauricular, nasal cavity and paranasal sinuses, nasopharynx, and parapharyngeal space. These findings have important clinical implications for radiotherapy planning and patient management.
