Abstract
Importance:
Osteoradionecrosis (ORN) has been shown to affect up to 20% of all head and neck cancer patients receiving radiation therapy. Despite its prevalence, a standard definitive therapy to treat this condition has yet to be available. A novel treatment modality with pentoxifylline, tocopherol, and clodronate (PENTOCLO) was first reported in the literature in 2005. There has since been a steady rise in the number of studies exploring the efficacy of PENTOCLO in treating ORN in head and neck patients.
Objective:
This article investigates the efficacy of PENTOCLO in treating ORN in head and neck cancer patients.
Evidence Review:
PubMed, Scopus, Cochrane Library, and Medline were used to review articles on PENTOCLO use for head and neck ORN from 2005 to the present. Two reviewers independently screened titles and abstracts from 114 articles, and inclusion was based on pair consensus. The main inclusion criteria were human clinical studies using PENTOCLO for at least 6 months in treating ORN in head and neck cancer patients. The selected main outcomes to determine the efficacy of PENTOCLO therapy were wound healing status after treatment, reduction in the percentage of exposed bone, and symptom surveys post-PENTOCLO therapy. Eight studies were selected for final statistical analysis.
Findings:
The primary site of ORN in most studies was mandible (77%). Of the 141 patients encompassed in this review, 66 (46.8%) patients had complete resolution, and 61 (43.3%) patients had significant wound healing. Total wound healing was achieved on an average of 10 months. The mean area of exposed bone decreased by 96% and the symptom survey scores decreased by 93% after treatment.
Conclusion and Relevance:
These results suggest PENTOCLO may be an effective treatment option in patients with head and neck ORN.
Key Points
Question: Is PENTOCLO, pentoxifylline, tocopherol, and clodronate treatment effective in the treatment of osteoradionecrosis of the head and neck?
Findings: This systematic review investigates the efficacy of PENTOCLO by utilizing 8 clinical studies of patients with head and neck osteonecrosis (n = 141). Results show an improvement in symptom scores, wound healing status, and mean area of exposed bone after an average of 10 months of PENTOCLO therapy.
Meaning: This systematic review suggests that PENTOCLO effectively treats osteoradionecrosis of the head and neck.
Introduction
Osteoradionecrosis (ORN) of the mandible is a significant complication of radiation therapy for the treatment of head and neck cancers. 1 Current literature suggests that up to 20% of patients undergoing head and neck radiation therapy may succumb to ORN. 2 Despite its severity and prevalence, there is currently no consensus on the most effective therapy for the treatment or prevention of ORN. Treatment options for ORN include surgical interventions such as debridement to remove infected or necrotic tissue, free flap reconstruction to transplant tissue, bone, blood vessels, and arteries from another body site, and full bone removal and replacement using bone-containing flaps. Drug treatments include antibiotics for infection, vasodilators for refractory cases, a pentoxifylline-vitamin E combination to reduce fibrosis and stimulate healing, and clodronate to limit bone destruction.3-6 Hyperbaric oxygen therapy (HBOT) enhances wound healing by addressing hypoxia, hypovascularity, and hypocellularity and is effective for early-stage ORN.7-9 Additional approaches include frequent saline irrigation, steroid use pre- or post-radiation, sequestrectomy, and vascularized free tissue transfer. This condition arises due to radiation-induced tissue damage, leading to a feared complication in patients receiving radiation to the head and neck region. 10
Various treatment modalities are available for managing ORN; however, a consensus on the most effective approach is yet to be established. Among the emerging therapeutic options, a combination of pentoxifylline, tocopherol, and clodronate (PENTOCLO) has garnered considerable attention in recent years. PENTOCLO is a novel treatment modality based on the fibro-atrophic theory of ORN pathophysiology. 4
Within this proposed model, an initial stage of inflammation after radiation is followed by fibrosis and remodeling due to dysregulation of fibroblasts. 4 This ultimately leads to tissue necrosis and its sequela including recurrent infection, persistent pain, and poor functionality of affected structures. The PENTOCLO regimen includes 3 drugs, each targeting distinct aspects of the fibro-atrophic model. Pentoxifylline promotes vasodilation, reduces platelet aggregation, inhibits inflammation, and enhances erythrocyte membrane flexibility. 11 By improving blood flow to the irradiated tissue, pentoxifylline may reduce ischemia and facilitate tissue repair, thus mitigating the effects of radiation-induced damage and preventing further fibrosis: Its role in improving microvascular circulation is critical in counteracting the reduced oxygenation and nutrient supply that contribute to ORN.11,12 Second, tocopherol, or vitamin E, functions as an antioxidant. By reducing oxidative damage to cellular membranes, collagen, and other extracellular matrix components, tocopherol helps to preserve the structural integrity of the affected tissues, potentially preventing further fibrosis and tissue degeneration. 13 Third, clodronate, a bisphosphonate, is a drug that is designed to decrease osteoclastic bone destruction, which plays a significant role in the progression of ORN.14,15 By inhibiting osteoclast activity, clodronate reduces the resorption of bone tissue, thereby preventing the exacerbation of bone necrosis and promoting bone healing.14,15 This action is crucial in the context of ORN, where osteoclast-mediated bone destruction plays a significant role in the loss of bone structure and function. 3 Together, these 3 agents work in concert to target the multifactorial pathogenesis of ORN, including inflammation, fibrosis, oxidative damage, and bone destruction, offering a comprehensive approach to managing this challenging condition. Figure 1 illustrates the effects of PENTOCLO on ORN.
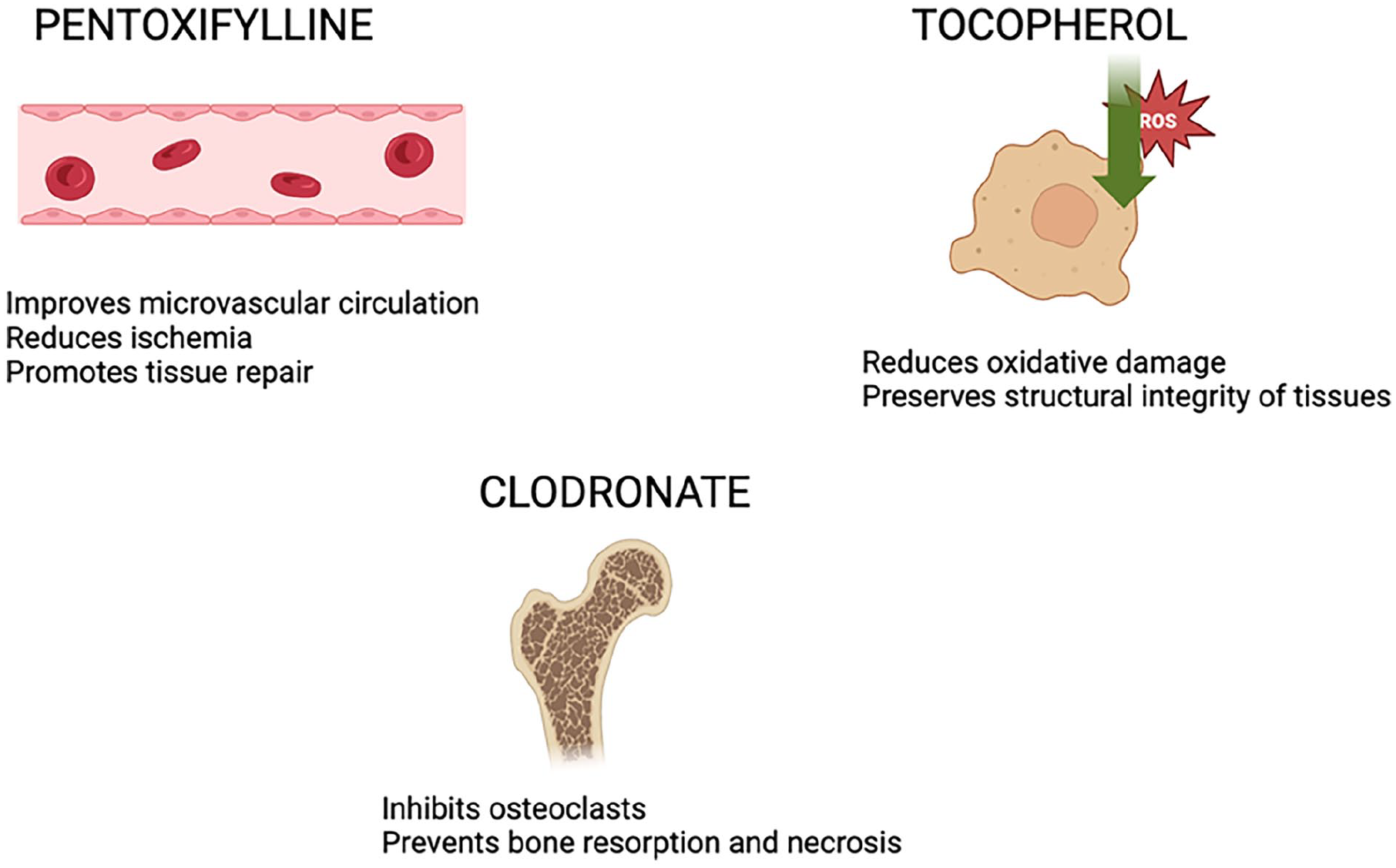
PENTOCLO and its effects on ORN. ORN, osteoradionecrosis; PENTOCLO, pentoxifylline, tocopherol, and clodronate.
This systematic review aims to provide insight into the efficacy of PENTOCLO as a treatment modality for head and neck ORN. The findings of this study may contribute to the identification of a more effective and standardized approach for managing this challenging condition, ultimately improving the quality of life for patients with ORN.
Methods
Study Design
A systematic review approach was used to synthesize and analyze existing literature on PENTOCLO therapy for treating ORN in head and neck cancer patients. This review followed established guidelines from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) to ensure a sound and transparent methodology. 16
Information Sources
The following databases were searched for relevant articles published from January 2005 to January 2023: PubMed, Scopus, Cochrane Library, and Medline. These databases were chosen to ensure a comprehensive coverage of the available literature on PENTOCLO therapy for ORN.
Search Strategy
The search strategy included specific terms related to PENTOCLO therapy and ORN. The search terms used are as follows “PENTOCLO,” “pentoxifylline + tocopherol + clodronate,” and “head + and + neck + osteoradionecrosis.”
Screening Process and Eligibility Determination
Two independent reviewers conducted the initial screening by evaluating the titles and abstracts of the retrieved articles from the search. In cases of discrepancies, a third reviewer acted as a mediator to reach a consensus. An initial search yielded 97 unique studies. Figure 2 shows the flowsheet of inclusion at each stage of screening. After screening, 2 reviewers assessed full-text articles similar to the inclusion criteria.

PRISMA flow diagram of the study selection process.
Inclusion Criteria
The inclusion criteria for the study were as follows: human clinical studies (clinical trials, cohort studies, case series and reports, systematic reviews, meta-analyses), PENTOCLO as a therapeutic intervention, minimum treatment duration of 6 months, minimum follow-up period of 12 months, and treatment of ORN at any site within the head and neck. Our methodology focused exclusively on studies involving PENTOCLO. While papers evaluating pentoxifylline and tocopherol alone were identified during screening, they were excluded, as our aim was not to compare different regimens but to evaluate the outcomes of the established PENTOCLO protocol.
Data Extraction and Synthesis
The included studies that met the selection criteria were subject to data extraction by the reviewers. Information regarding study design, patient demographics, intervention details, follow-up duration, and outcome measures were extracted.
Outcome Measures
Four outcome measures were selected to help determine the efficacy of PENTOCLO therapy. The first outcome measure was wound healing status after treatment categorized into fully healed, partially healed, or progression of disease. Second, the time to complete healing was assessed. The third outcome measure, the mean area of exposed bone before and after treatment, served as a quantitative measure. Finally, improvement in symptoms was determined by a reported reduction in SOMA (subjective, objective, management, analysis) scores. These questionnaires included several common symptoms of ORN such as trismus and pain.
Data Analysis
Data synthesis and statistical analysis using descriptive statistics were performed to assess the overall efficacy of PENTOCLO treatment for ORN. The study resulted in 8 relevant studies that met the inclusion criteria and were eligible to be included in the statistical analysis.
Results
On initial search, 97 unique articles were retrieved after the removal of duplicates. These articles were then screened using the titles and then the abstracts, resulting in 26 subsequent studies. These articles then underwent review to determine whether they met the inclusion criteria. All references from systematic reviews were gathered and further screened for inclusion.17,18 This resulted in a total of 8 studies that were ultimately used for quantitative analysis. The articles selected included 3 prospective clinical trials, 2 retrospective studies, 3 case series, and 1 case report.4,19-25
In all, 141 patients collectively underwent PENTOCLO treatment for over 6 months. The mean patient age was 61 years old (SD = 10.8) with a male predominance of 76%. All patients had primary head and neck malignancies requiring varying degrees of radiation therapy. The most common location for primary tumor site was the oropharynx at 48.4%, followed by oral cavity primary tumors at 36.5%. The remaining 15% of patients had other primary tumor locations including (in order from most to least common) temporal bone, parotid/salivary gland, unknown primary, nasopharynx, larynx, and thyroid. All but one of the studies focused on the treatment of mandibular or maxillary ORN, with one case series by Glicksman et al investigating temporal bone ORN. 19 Of the studies included, only 5 reported radiation doses. The radiation range received was 50 to 130 Gy, with most patients receiving 60 to 80 Gy before treatment. Prior treatment of ORN, either medical or surgical, varied throughout the study’s populations. Prior treatment was not commonly reported in these studies.
All studies administered pentoxifylline 800 mg daily, tocopherol 1 g daily, and clodronate 1600 mg every other day or twice daily in 1 study by Burns and Patel. 20 In addition, 5 studies utilized steroids (prednisone or methylprednisone) and ciprofloxacin twice weekly on the days that clodronate was not given.4,19,22,24,25 While the actual PENTOCLO regimen was fairly standard among studies, pre-treatment phases, and their respective regimens were widely varied. The majority (5 of 8) of studies implemented a pre-treatment phase that consisted of a combination of antibiotics, antifungals, steroids, and proton pump inhibitors.4,19,22,24 The timing and duration of these pre-treatment regimens also widely varied (Table 1).
Osteoradionecrosis Treatment Regimen From Studies Included.

Abbreviations: BID, twice daily; IU, international units; N, none; qAM, once in the morning; QD, once daily; QOD, once every other day; qPM, once in the evening; SD, standard deviation.
Of the 141 patients included in this review, 66 (46.8%) patients had complete resolution, and 61 (43.3%) patients had significant wound healing described as near-complete mucosal covering of exposed bone. The remaining 10.1% of patients had some degree of progression of their ORN (Table 2). Patients who had complete resolution of their ORN disease did so in an average of 10 months on PENTOCLO therapy (SD = 7.6).
Results of Treatment.

Abbreviations: EOT, end of treatment; NA, not available; SD, standard deviation; SOMA, Subjective Objective Medical management and Analytic evaluation of injury.
Five out of 8 studies reported the mean area of exposed bone. The pre-treatment mean area of exposed bone in these studies was 4.7 cm2 (n = 109). The post-treatment mean area of exposed bone was 0.2 cm2, resulting in a 95.6% reduction in the mean area of exposed bone (Table 2). Finally, SOMA (subjective, objective, management analysis) questionnaires were utilized by 3 out of 8 studies. This questionnaire considered several reported symptoms including trismus, pain, and mastication ability. At the end of treatment time, there was a 93% decrease in SOMA symptom surveys indicating improvement in patient’s symptoms relating to their ORN.
Discussion
Despite the prevalence of ORN as a severe and debilitating complication of head and neck radiation therapy, the underlying pathophysiology and subsequent treatment modalities remain poorly understood. This study found that the mandible is the most common location for ORN of the head and neck, followed by the maxilla. Radiation to the oropharynx and oral cavity are the most common sites resulting in ORN, indicating collateral radiation damage to surrounding structures in anatomical sites that are difficult to access within the head and neck. Although not fully reported in these selected studies, comorbidities such as active alcohol use, tobacco use, tooth extractions, and other conditions that lead to poor wound healing further increase the risk of ORN. 26 As such, patients with these specific primary tumors or additional comorbidities should be carefully monitored.
This systematic review involved screening 97 journal articles, with 8 selected that contained qualitative and quantitative outcomes from the use of PENTOCLO in treating head and neck ORN. Most of the included studies were prospective clinical trials without control or placebo groups, along with case reports, case series, and retrospective studies (Table 3). The strength of each article was assessed based on study type, sample size, and methodology, with most studies categorized as level 4 evidence (Table 4) according to the 2011 Oxford Center for Evidence-Based Medicine criteria. 27 This classification was primarily due to the lack of control groups. Importantly, no randomized controlled study has yet evaluated the efficacy of PENTOCLO in treating ORN. Nevertheless, the included studies demonstrate solid sample sizes, minimal loss to follow-up, and the use of both qualitative and quantitative measures to assess ORN resolution.
Studies Included and Patient Demographics.

Abbreviations: F, female; M, male; NA, not available; ORN, osteoradionecrosis.
Study Level of Evidence.

Abbreviation: SOMA, Subjective Objective Medical management and Analytic evaluation of injury.
All studies reported final wound healing outcomes after PENTOCLO treatment. However, methods for determining wound status varied across studies. While easy to assess at regular follow-up visits, categorizing wound status can be prone to subjective bias. Several studies, such as Robard et al and Delanian et al, used radiographic evidence for a more objective determination of ORN resolution.4,25 Even more objective was the use of a mean area of exposed bone to provide a quantitative and more objective measure of wound healing. Five out of 8 studies reported the mean area of exposed bone before and after PENTOCLO therapy.4,21,22,24,25
This systematic review suggests that PENTOCLO is effective in treating ORN of the head and neck, as shown by most patients within this review experiencing significant clinical improvement in their wound status within a reasonable amount of time (average of 10 months). It is important to also consider improvements in ORN symptoms such as trismus, pain, and the ability to masticate, as these symptoms profoundly affect the quality of life. Three included studies attempted to quantify the patient experience after treatment and showed improvement in symptoms post-PENTOCLO.22,24,25
Currently, there is no consensus on what constitutes the optimal treatment regimen or prophylaxis for ORN of the head and neck. Treatment modalities for ORN range from conservative measures, such as antibiotics, to major surgical interventions like resection of devitalized bone and subsequent reconstruction. HBOT is another commonly cited therapy in treating ORN. Based on the principle of hypocellularity, hypoxia, and hypo-vascularity, HBOT has been proposed to aid in angiogenesis and increase oxygen delivery to hypoxic tissue, such as irradiated bone in ORN.7,28 Literature regarding HBOT has recently been mixed. A 2011 meta-analysis and a 2010 systematic review suggested insufficient evidence to support the use of HBOT in treating mandibular ORN.8,29 Similarly, a 2004 randomized clinical trial found no difference between HBOT and placebo in treating ORN. 9
New treatment modalities have since appeared that target alternative models of ORN pathophysiology. The fibro-atrophic model is currently favored in explaining ORN pathophysiology. This model shifts focus from hypoxia-driven tissue necrosis to dysregulation of fibroblasts, which leads to increased fibrosis, atrophy, and eventual necrosis of irradiated tissue. 30 As a result, treatments for ORN now aim to target this dysregulation. Synergistic treatment with pentoxifylline and tocopherol (PENTO), an antifibrotic and antioxidant agent respectively, has emerged as the most common and promising therapy in the literature. 4
The use of PENTOCLO for head and neck ORN was first introduced in 2005 by Delanian et al. 22 This clinical trial included a subset of patients with refractory ORN. Clodronate was added to pentoxifylline and tocopherol, leading to a complete healing rate of 89% within this subset. Clodronate has increasingly been included in studies investigating PENTO therapy. Clodronate, or clodronic acid, is a bisphosphonate with anti-fibroblast properties, specifically targeting osteoblasts to increase bone formation.14,15 While clodronate currently lacks US FDA approval, ongoing randomized controlled trials may change this status in the future. However, the lack of FDA approval should not overshadow the demonstrated effectiveness of PENTOCLO and its various combinations in treating ORN.
PENTO has shown promising outcomes in managing medication-related osteonecrosis of the jaw (MRONJ), offering a conservative and well-tolerated alternative to surgical interventions. Epstein et al demonstrated a 74% mean reduction in exposed bone area and symptomatic improvement in 5 out of 6 patients without adverse effects, supporting the potential utility of PENTO in MRONJ management. 31 Similarly, Magremanne and Reychler reported complete mucosal healing, pain resolution, and no further bone spicule expulsions in a patient with stage 3 MRONJ treated with PENTO, antibiotics, and chlorhexidine rinses for 12 months. 32 Owosho et al observed symptom relief in all 7 patients treated with PENTO over an average of 16.8 months, with evidence of new bone formation and complete resolution of exposed bone in some cases. 33 Seo et al further highlighted a 21.25% increase in radiographic bone density and clinical improvements with PENTO in patients with long-term bisphosphonate exposure. 34 Varoni et al noted a 92% success rate in long-term healing among patients treated with PENTO for an average of 3.81 months, with minimal adverse effects. 35 Finally, Epstein et al described the complete resolution of bone exposure and mucosal coverage within 9 to 13 months of treatment in 3 complex cases involving the maxillary alveolus and sinus, emphasizing PENTO’s effectiveness in promoting bone remodeling and mucosal healing. 36 Collectively, these findings suggest that PENTO therapy offers a promising pharmacological approach for MRONJ management, warranting further investigation to optimize treatment protocols.
The prophylactic use of PENTO in head and neck cancer patients undergoing invasive dental procedures has also shown promising results in reducing the incidence of ORN. Studies suggest that PENTO is associated with a lower ORN rate compared to traditional approaches like antibiotic prophylaxis or HBOT, offering cost-effectiveness and accessibility. 37 Notably, Lombardi et al observed that only 13.8% of patients who underwent dental surgeries while receiving PENTO developed ORN, primarily among high-risk individuals. 37 Similarly, Patel et al highlighted favorable outcomes, including a 0% ORN rate in a small cohort. 38 This efficacy is supported by findings from Samani et al, who reported lower ORN incidences over time: 6% at 1 year, 12% at 2 years, and 16% at 5 years post-radiotherapy, emphasizing the extended risk period despite preventive measures. 39 Aggarwal et al further corroborated PENTO’s potential in significantly reducing ORN risk, positioning it as a valuable intervention for irradiated patients requiring dental extractions. 40 Overall, the evidence underscores PENTO’s efficacy, safety, and economic benefits, though careful consideration of patient selection and contraindications is essential.
At the University of Texas Medical Branch Department of Otolaryngology, our current clinical approach to managing ORN does not include clodronate due to its lack of approval by the US FDA. While studies have demonstrated clodronate’s anti-fibroblast and bone-healing properties, its unapproved status limits its clinical use in the United States. Instead, we employ a two-phase treatment regimen using FDA-approved therapies. Phase 1 focuses on controlling infection, inflammation, and fungal overgrowth with a combination of Augmentin (amoxicillin-clavulanic acid), ciprofloxacin, fluconazole, prednisone, and omeprazole, typically for 4 to 6 weeks, as determined by the treating physician. Phase 2 transitions to long-term therapy aimed at reducing fibrosis and promoting healing, utilizing pentoxifylline, vitamin E, and weekend-only prednisone, continuing until complete healing is achieved.
While our results indicate that PENTOCLO is an effective regimen in treating ORN, there are limitations to this review. As PENTOCLO is a newer therapy, the associated literature remains limited in scope. Most importantly, there are yet to be true randomized and blinded control trials comparing PENTOCLO to either a placebo or a true control group. While the current studies using PENTOCLO therapy maintain a homogeneous regimen in their dosing, there is wide variation in the use of pre-treatment phases and the medications included in these phases. Further studies are needed to fully understand the use of PENTOCLO therapy for ORN, particularly regarding its safety profile and long-term effectiveness.
Future studies on the treatment of ORN in head and neck cancer patients should focus on several key areas. Randomized controlled trials (RCTs) are crucial to establish the definitive efficacy of PENTOCLO therapy in comparison to other treatment modalities such as HBOT, antibiotics, and surgical interventions. Larger sample sizes with longer follow-up periods are necessary to strengthen the evidence base and assess the long-term benefits and potential side effects of PENTOCLO. Exploration into the role of patient comorbidities—such as tobacco and alcohol use—should be incorporated to better understand their impact on treatment outcomes. In addition, investigating the optimal timing, dosing, and duration of the PENTOCLO regimen is needed, as variations in treatment protocols across studies may contribute to inconsistent outcomes. Finally, utilizing more objective and standardized outcome measures, including advanced imaging techniques for wound healing and functional assessments of quality of life, will provide a more comprehensive evaluation of the therapeutic impact of PENTOCLO on ORN.
Conclusion
ORN remains one of the most debilitating complications of radiation therapy in the head and neck, severely impacting patients’ quality of life and pushing the boundaries of current medical management. As our understanding of ORN evolves—recognizing the pivotal roles of inflammation and fibrosis—treatment strategies must advance accordingly. PENTOCLO has emerged as a promising therapeutic approach, demonstrating significant success in promoting wound healing, restoring bone coverage, and alleviating the devastating symptoms of ORN. This systematic review underscores its potential to transform ORN management by directly targeting the underlying pathological mechanisms. However, despite encouraging outcomes, critical gaps persist. The absence of large-scale, RCTs hinders definitive conclusions regarding its long-term efficacy and optimal use. Future research must bridge these gaps, refining treatment protocols and establishing standardized guidelines to maximize patient benefit. Advancing this field is not merely a scientific necessity but a crucial step toward improving survival and quality of life for those suffering from ORN.
Footnotes
Acknowledgements
We would like to thank Efstathia Polychronopoulou PhD from the University of Texas Medical Branch, Galveston TX Department of Biostatistics for providing us with statistics support in this project.
Author Note
For Peer Review Meeting information: Combined Otolaryngology Spring Meeting 2023 American Broncho-Esophagological Association. Boston, Massachusetts, United States of America. May 6, 2023.
