Abstract
Sinonasal tumors represent a small percentage of head and neck malignancies. Advances in endonasal surgery have revolutionized the management of these tumors, offering improved oncological outcomes and reduced morbidity for selected cases compared with traditional open approaches. However, this kind of approaches often leads to large defects resulting in complications, including cerebrospinal fluid leaks or meningitis. Cerebral herniation is a rare, often unknown, and underreported complication in the literature. This article presents the case of a 33-year-old male patient with no medical history who underwent endoscopic resection of an olfactory esthesioneuroblastoma, followed with a frontal lobe herniation. We aimed to highlight the possible pathophysiology and several management challenges. The case underscores the importance of considering multifactorial causes of herniation. For our patient, we found that elevated intracranial pressure due to meningitis is a possible contributing factor. Even though cerebral herniation is a rare complication after endoscopic anterior skull base resection and reconstruction, its understanding is mandatory to identify several contributing factors preoperatively to optimize patient management and ensure a better outcome.
Keywords
Introduction
Sinonasal tumors are a rare entity, accounting for 3% to 5% of head and neck malignant neoplasms and 0.2% to 0.8% of all tumors. 1 Since the introduction of craniofacial resection for the treatment of malignant sinonasal lesions in the 1960s, huge strides have been made, notably with the introduction of endoscopic endonasal surgery for the treatment of tumors of the anterior skull base. 2 Initially controversial, especially for the treatment of malignant diseases, this approach gradually proved its worth in terms of oncological resection and respect of the margins, as well as in significantly improving morbidity and mortality by avoiding facial incisions, craniotomies, and manipulation of the cerebral parenchyma.3,4 Today, tumors such as ethmoid adenocarcinomas, adenoid cystic carcinomas, and esthesioneuroblastomas can be treated using a fully-expanded endoscopic skull base (EESB) approach, producing oncological results similar to those of traditional open methods.5,6
Most of these tumors are attached to the anterior part of the skull base; the defect resulting from this type of resection is variable depending on the extent of the tumor attachment and/or its intracranial extension. Often, an osteodural defect is observed that extends from the posterior table of the frontal sinus to the sphenoidal planum and from orbit to orbit. Reconstruction of such defects represents a real challenge for the surgeon. Different materials have been used for this purpose, such as fat grafts, multilayer fascia lata, and other synthetic materials. The most feared and commonly-observed complication was cerebrospinal fluid (CSF) leakage. The advent of the vascularized pedicled nasoseptal flap for the repair of large anterior skull base defects after endoscopic skull base surgery has significantly reduced the rate of postoperative CSF leaks.7,8 Another likely result of these large defects after endoscopic craniectomy is ptosis of the cerebral parenchyma after reconstruction, but this has not been considered a significant complication. 9 We only found 2 cases in the English literature. 10
We report here a case of frontal lobe herniation after endoscopic resection of the anterior skull base and discuss its probable pathophysiology and treatment.
Case Report
This is a 33-year-old man with no significant medical history who consulted with us due to a 1 year history of left nasal obstruction associated with 3 months of intermittent epistaxis. He reported anosmia, which he thought had developed since his last Covid-19 infection 2 years earlier. Examination revealed a blackish hemorrhagic tumor that filled the entire left nasal cavity. The biopsy revealed a Hyams grade III olfactory esthesioneuroblastoma. On imaging, the tumor occupied the entire left nasal fossa, the ethmoid, extending to the right through the septum with invasion of the cribriform plate and slight extension into the left anterior fossa (Figure 1A). The patient underwent extended endoscopic endonasal resection with craniectomy involving the ethmoid, left lamina papyracea, septum, dura, and olfactory bulbs bilaterally with a frozen section margin mapping of the tumor bed. The resulting defect extended from the medial table of the frontal sinus anteriorly to the sphenoidal planum posteriorly and from orbit to orbit. The surface area of the defect was measured to be 8.5 cm² (Figure 2A). For skull base reconstruction, we used the multilayer technique with 3 layers of fascia lata (intradural, epidural, and extradural) held together with Surgicel and biological glue, followed by Merocel. Postoperatively, the patient was maintained in the 30 degree position and covered with third-generation cephalosporin 3 with stool softeners. The nasal packing was removed on day 2 without incident. On postoperative day 4, the patient presented with a maximum fever of 38.8 with rhinorrhea; his neurological examination was normal, and nasal endoscopy showed signs of CSF leakage but could not confirm the status of the reconstruction. The emergency computed tomography scan showed no evidence of intracranial complications except for mild frontal pneumocephalus.
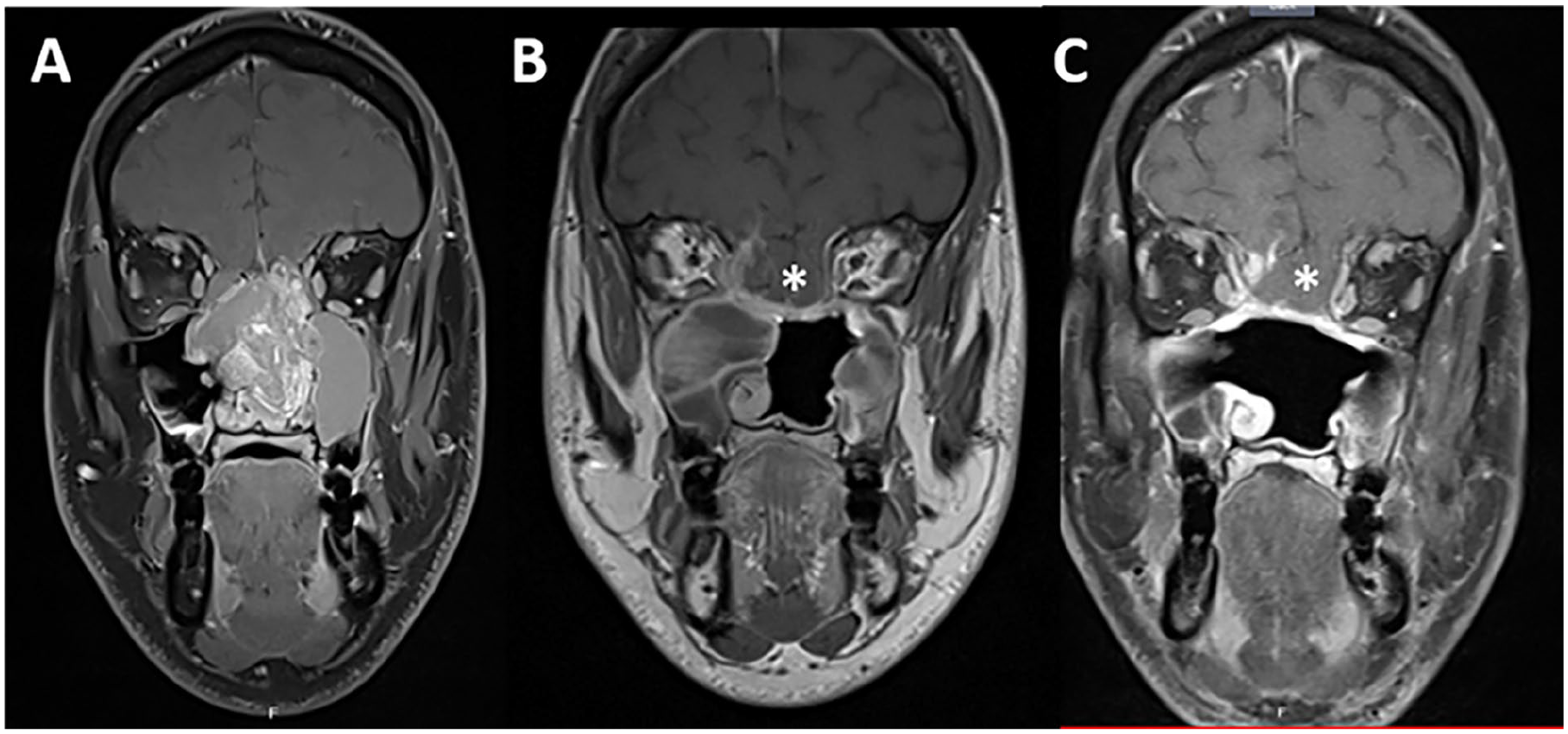
Preoperative contrast-enhanced MRI in coronal view (A), showing a large esthesioneuroblastoma, involving the nasal septum, both ethmoids and encroaching the left anterior skull base with a slight intracranial extension. Three month postoperative and postradiation MRI in coronal view (B) showing frontal lobe sagging (*). Contrast-enhanced MRI at the 18 month follow-up (C) showing stability of the brain herniation (*). MRI, magnetic resonance imaging.
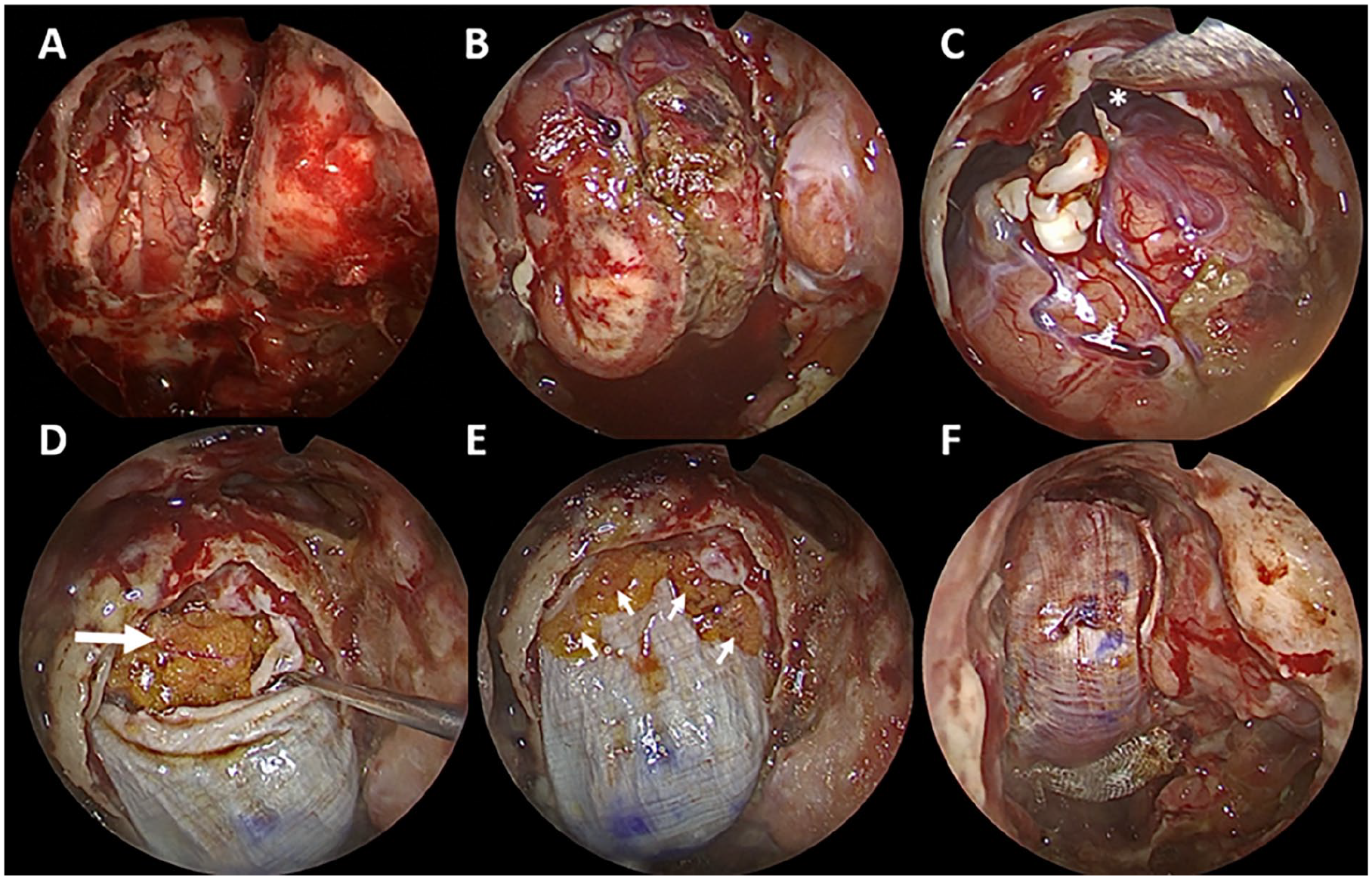
Initial skull base defect after tumor resection measured at 8.5 cm2 (A). Brain herniation after the removal of the 3 layers of fascia lata during revision surgery (B). Important dead space in the prefrontal region (*) (C). Large piece of fat graft (large arrow) placed in the dead space to support the first intradural layer of fascia lata (D). Fat pellets (small arrows) placed circumferentially to fill any remaining dead spaces between dura and fascia lata (E). Final appearance after the placement of extradural layer and before fibrin glue application (F).
A lumbar puncture was performed, revealing 2300 white blood cells/mm³ with neutrophil predominance, elevated protein concentration, and a 0.23 blood glucose ratio from CSF. Direct examination and CSF culture were negative. Empirical broad-spectrum intravenous antibiotic therapy was started with vancomycin and meropenem. After 48 hours of antibiotic therapy, the patient’s temperature returned to normal, but CSF leakage persisted, requiring a revision surgery on the sixth postoperative day. A migration of the fascia graft was observed, with denudated cerebral parenchyma toward the sphenoidal planum region. The 3 layers of the fascia lata were removed, and we were surprised by the significant herniation of the brain through the defect, with a large dead space in the prefrontal region (Figure 2B and C). After debridement and lavage of the nasal cavity with saline solution, fat was placed to fill this dead space (Figure 2D), providing a good base for the intradural fascia lata graft; The second epidural layer was reinforced with fat pellets placed circumferentially to fill any remaining dead spaces (Figure 2E); a final large layer of fascia was placed extradurally (Figure 2F). All held together with Surgicel and fibrin glue. The postoperative course was favorable, and the patient was discharged on the 10th day. He underwent volumetric-modulated arc therapy without incident. The patient is asymptomatic and disease-free during 2 years of follow-up with stability of brain herniation at the last magnetic resonance imaging (MRI) (Figure 1B, C).
Discussion
The ongoing development of endoscopic endonasal instrumentation, significant advances in cameras and endoscopes quality, and the growing collaboration between ENT and neurosurgeons to develop minimally-invasive approaches have revolutionized the management of skull base disorders in recent decades. 9 However, endoscopic access to increasingly-large tumors undeniably generates consequential defects, with an increased risk of CSF leakage and cerebral parenchymal ptosis, particularly of the frontal lobe, after the resection of the anterior skull base. The advent of pedicled flaps, led by the nasoseptal flap, has considerably reduced postoperative CSF leakage, and this has been reported in several studies since its beginnings in 2006. 7 On the other hand, very few studies address cerebral herniation after the endoscopic resection of the anterior skull base, and even fewer consider it a real complication that must be taken into account in subsequent treatment (Table 1). 10
Characteristics of the Reported Cases of Brain Herniation After Endoscopic Surgery.
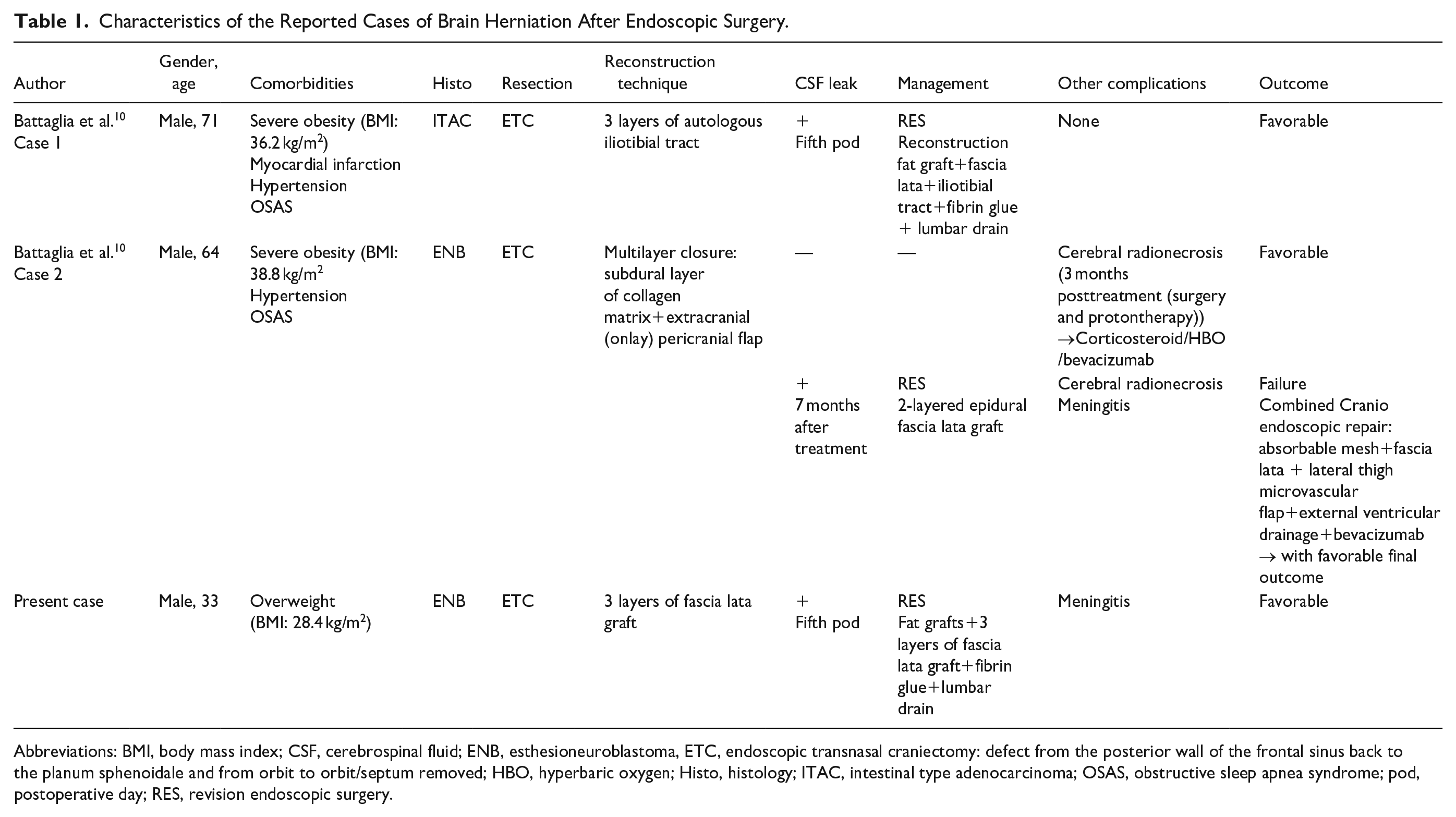
Abbreviations: BMI, body mass index; CSF, cerebrospinal fluid; ENB, esthesioneuroblastoma, ETC, endoscopic transnasal craniectomy: defect from the posterior wall of the frontal sinus back to the planum sphenoidale and from orbit to orbit/septum removed; HBO, hyperbaric oxygen; Histo, histology; ITAC, intestinal type adenocarcinoma; OSAS, obstructive sleep apnea syndrome; pod, postoperative day; RES, revision endoscopic surgery.
Eloy et al. did not show significant sagging of the frontal lobe after endoscopic repair of large cribriform defects using a triple-layer reconstruction technique that included fascia lata, acellular dermal allograft, and a pedicled nasoseptal flap. This suggests that a rigid structural graft, such as cartilage, bone, or alloplastic materials (eg, titanium plate), may not be necessary to reinforce the repair of the anterior skull base. 9 In this same path, Battaglia et al. support the idea that frontal lobe herniation into the sinonasal cavities is not caused by the size of the anterior skull base defect, the surgical technique, or the materials used in reconstruction. 10
In the 2 cases reported by Battaglia et al., the authors incriminate increased intracranial pressure secondary to obesity and obstructive sleep apnea syndrome. One patient showed an empty sella, lateral sinus collapse, and buckling of the optic nerves with increased perineural fluid on the preoperative MRI. Several studies have documented an increase in intracranial pressure during periods of apnea. 11 It is important then not only to measure body mass index and explore sleep apnea in patients at risk and possibly perform lumbar punctures for diagnostic purposes but also to reduce intracranial pressure before surgery. 10
The other hypothesis is the appearance of meningitis, as in the case of our patient. This may be directly responsible for an increase in intracranial pressure that results in cerebral herniation through the surgical defect at the skull base. In a systematic analysis of the literature, the incidence of meningitis after EESB surgery indicated an overall risk of 1.8% postoperative meningitis. The presence of a postoperative CSF leak was, not surprisingly, considered by many as an important factor predisposing to the subsequent development of meningitis. The incidence of meningitis without an associated CSF was very low 0.1%. 12 The course of ABM is accompanied by elevated intracranial pressure in up to 93% of patients. 13 The rise in intracranial pressure is multifactorial, with main elements being cerebral edema of vasogenic, cytotoxic, or interstitial types. 14 Although the clinical presentation of meningitis was not striking, and no germs were isolated in our patient, the hypothesis of increased intracranial pressure secondary to meningitis remains plausible.
A rarer etiology of elevated intracranial pressure after endoscopic skull base surgery is tension pneumocephalus. It is a life-threatening condition, favored by a progressive trapping of air through a small osteodural defect generally encountered as a complication of endoscopic sinus surgery. Its occurrence during extensive endoscopic skull base surgery is exceptional. On the contrary, simple pneumocephalus is common after EESB surgery, seen in 39.5% of cases. 15 Its finding is quite common on postoperative imaging. The mere presence of pneumocephalus, especially in the frontal or intraventricular regions, does not necessarily indicate a postoperative CSF leak. However, certain patterns of air, such as pneumocephalus in convexity, interhemispheric fissure, sella, parasellar, or perimesencephalic areas, are strongly related to a postoperative CSF leak. 16
Hence, the appearance of cerebral herniation after endoscopic skull base resection could be multifactorial, where several etiologies may be intertwined. Therefore, it is essential to assess the operative risk on a case-by-case basis to take adequate preventive measures.
Surgical revision of hernias can be considered for symptomatic or progressive forms or in the case of CSF leakage. External approaches may be indicated for the reduction or resection of the herniated parenchyma. 10 Whatever the surgical approach, the ultimate goal is to ensure watertight closure between the sinonasal cavities and the cerebral parenchyma. For our patient, we had opted to redo the endoscopic approach and repeat the same technique, adding fat to fill all the dead spaces. We preferred to keep the pericranium for a possible later external approach in case of failure. The use of rigid grafts such as cartilage, bone, or other heterologous materials is not recommended due to the high risk of infection, rejection, or necrosis, as we are often dealing with malignancies, and adjuvant radiotherapy is indicated for most cases. This radiation therapy must be planned with caution in cases of brain herniation due to the risk of radionecrosis and cerebral edema, as the location of the herniation is often in the area of the target volume. 10 These cases should be discussed within the tumor board to ensure optimal overall patient management.
Conclusion
Cerebral herniation is one of the less well-known complications associated with endoscopic skull base surgery. However, it requires particular attention, given the potentially-serious consequences it can have. Preoperative and postoperative identification of risk factors and their management is essential to ensure optimal outcomes. Close monitoring of a cerebral hernia is crucial, given its potential evolution and the emergence of late complications.
