Abstract
Plain language summary
The vestibular system uses information from vision, body sensation, and the inner ear to help maintain balance and coordinate movement. Structures in the inner ear provide information about which way is up and how the body is moving. Damage to the inner ear can lead to dizziness and unsteadiness and can increase the risk of falls.
The brains of people with this sort of injury use visual cues and body sensation (eg, pressure and position of the feet and ankles), in addition to information from any remaining inner ear function, to maintain their balance function. This is called vestibular compensation and happens spontaneously. However, strong reliance on vision and body sensation can cause problems when these senses are absent (such as in the dark) or provide false information (such as when objects are moving around a stationary person, or on slippery, uneven, or soft surfaces). Without a functioning inner ear to provide information about which direction is up and how the body is moving, people may be fooled by misleading information, and this can lead to a fall.
In this study, participants did exercises, prompted by a wrap-around computer screen, while standing on a weight-sensing platform. The participants had to maintain their balance when presented with misleading information, such as tilting a virtual horizon on the screen, or tilting the platform beneath their feet. After 12 sessions of exercises, we measured changes in the way participants dealt with unreliable visual, or body sensation information. We found that participants’ balance improved, even with their eyes closed, or with a tilting visual cue, or the tilting platform. We believe this improvement was due to learning to use parts of the inner ear not affected by the injury, in conjunction with vision and body sensation to keep their balance even in complex, moving situations.
Introduction
Vestibular dysfunction can result from absent, corrupted, or asymmetric vestibular afferents. Acute static symptoms, such as nausea, vertigo, and imbalance, often resolve quickly, but many individuals experience long-term dynamic symptoms, including dizziness and postural unsteadiness, due to impaired coordination of vestibular reflexes1-3 and impairments may increase the risk of falling. 4
There is a need for new treatments for individuals that continue to experience symptoms after receiving vestibular rehabilitation or who have functional impairment in dynamic sensory environments.
We have previously reported durable improvement in objective posturography as well as in participant-reported disability, balance confidence, and fear of falling for those with stable unilateral vestibular deficits.5,6 In this report, participants received computerized vestibular retraining therapy (CVRT), which challenges participants by systematic disruption of somatosensory and/or visual input or presentation of conflicting stimuli. Our hypothesis was that CVRT would lead to changes in sensory ratios of the sensory organization test (SOT), specifically increases in the visual (VIS) and vestibular (VEST) ratios, suggestive of learned reweighting toward effective use of residual vestibular senses.
Material and Methods
Study Design
This prospective, single-group, cohort study was conducted in a tertiary otolaryngology clinic. It was approved by the Clinical Research Ethics Board at the University of British Columbia (study # H20-04045), and all experiments were performed in accordance with relevant guidelines, regulations, and the Declaration of Helsinki. The study was performed in a tertiary otolaryngology office in British Columbia, Canada. The study has been registered (https://clinicaltrials.gov/study/NCT04875013; 04/27/2021). Recruitment took place between April 23, 2021, and June 10, 2021. All participants provided written informed consent.
Interventions
Participants in the CVRT group completed 12 twice-weekly sessions of CVRT in the clinic. The CVRT training was performed on a Bertec Balance Advantage computerized dynamic posturography system (Bertec, Columbus, OH, USA). 7 During the exercises, participants were challenged to volitionally shift their weight along the lateral and anteroposterior axes as directed by an interactive display or to maintain their balance, while the visual display and support surface either gave congruent sensory feedback or incongruent feedback (ie, created the illusion of rotation). The exercises grew progressively-more difficult over the course of the treatment protocol by changing several parameters: (1) the gain between the measurement of the center of pressure by the platform and the movement of the cursor on the display, (2) the degree to which the platform tilts forward and backward, (3) the time allowed for participants to complete an exercise or the speed at which they had to respond to visual stimuli, and (4) the complexity of the visual environment. The protocol included repetition to consolidate learning. The exercise programs were predetermined, and each participant received the same protocol, except to account for the laterality of their deficit.
Main Outcome Measures
Consenting participants were invited to the clinic for their baseline assessment where they completed a SOT and a limits of stability test on a computerized dynamic posturography instrument. The SOT test comprises 6 conditions, each performed in triplicate. The instrument software calculates a composite score as well as scores for each condition out of 100. From the condition scores, we calculated 4 ratios commonly included in SOT analysis (Table 1), as well as the ratio of condition 6 to condition 1 (6:1). 8 We also added a ratio that we have termed the dynamic stability ratio (DSR), which consists of the average scores for conditions 4, 5, and 6 (in which the support surface is mobile and sway-referenced) divided by the average scores for conditions 1, 2, and 3 (in which the support surface is fixed). During the posturography tests and all retraining exercises, the participants were supported by a harness as a precaution against falls. These assessments were administered upon enrollment and after the completion of the retraining intervention.
Description of Sensory Organization Test Conditions and Ratios.
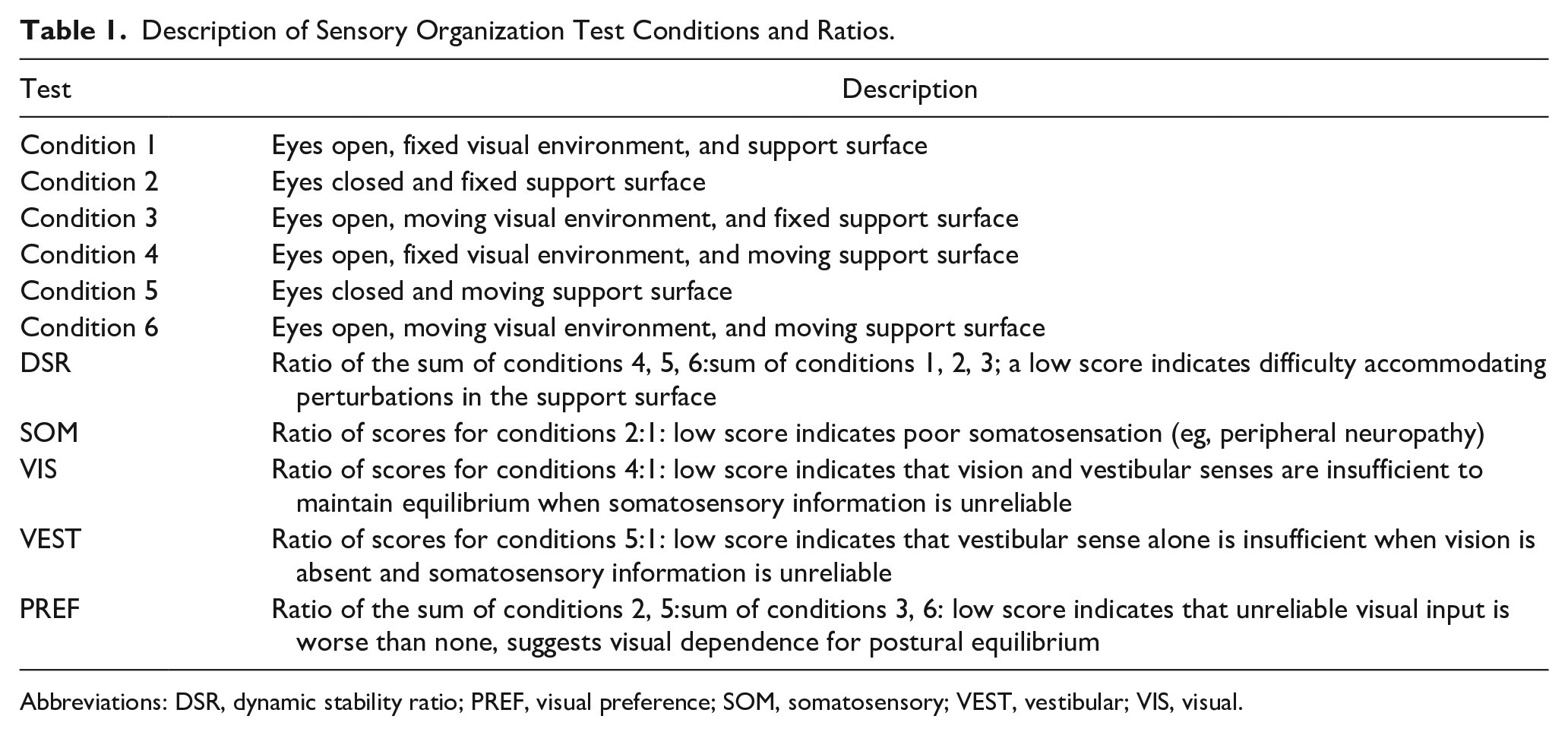
Abbreviations: DSR, dynamic stability ratio; PREF, visual preference; SOM, somatosensory; VEST, vestibular; VIS, visual.
Scores are reported as median (range) and change in scores are reported as the median change and 95% confidence interval (95% CI). Pretreatment and posttreatment scores were analyzed using the Wilcoxon matched-pairs signed rank test. As an exploratory analysis, participants were stratified according to the initial Dizziness Handicap Inventory (DHI) to those with moderate-to-severe disability, (scores >30) and those with mild disability (DHI ≤30). 9 Correlation with age, as a potential confounder, was analyzed by the Spearman correlation. Analysis was performed using Prism 9, version 9.3.1 (GraphPad Software, San Diego, CA, USA).
Participants
Candidate participants were identified from patients referred to the primary investigator’s otolaryngology practice: Eligible participants were aged between 18 and 80 and reported symptoms of imbalance present for more than 6 months that negatively affected their day-to-day activities. To be included in the study, the symptoms were clinically assessed to be caused by a stable, non-fluctuating vestibular deficit rather than an active or irritative vestibulopathy based on the criteria of the Barany Society International Classification of Vestibular Disorders consensus classification of vestibular symptoms. 10 Objective determination of unilateral peripheral vestibular deficit required at least one of the followings: (a) unilateral weakness during videonystagmography (VNG), as defined by a 25% or greater difference between ears using bithermal caloric testing; (b) significant cervical or ocular vestibular evoked myogenic potential (VEMP) interaural asymmetry, or absent cervical or ocular VEMP responses in one ear with intact responses in the other ear. 11 We excluded individuals who exhibited fluctuating symptoms of an active vestibulopathic cause within the last 6 months, such as active Menière’s disease (characterized by fluctuating hearing loss, tinnitus, and vertiginous exacerbations lasting >20 minutes according to the American Academy of Otolaryngology-Head and Neck Surgery criteria); had a concurrent diagnosis of benign paroxysmal positional vertigo; or had clinical and audiometric evidence of a perilymphatic fistula, or otosyphilis. We also excluded those with a deficit that precluded providing informed consent or completing the rehabilitation exercises, such as orthopedic or neurological deficits. Those meeting the eligibility criteria were contacted by telephone and invited to enroll in the study.
Results
This study enrolled 13 participants with stable unilateral vestibular deficits, which were confirmed by either demonstration of greater than 25% unilateral asymmetry during bithermal VNG testing or a significant interaural ratio difference during cervical or ocular VEMP testing. The median age was 51 years (range 18-67) and 5 were female. Seven showed a vestibular deficit by bithermal caloric testing with normal VEMPs, 1 had abnormal cervical VEMP and ocular VEMP but normal videonystagmogram, and 5 had abnormal VEMP and videonystagmogram results (Table 2). All 13 completed the full course of retraining sessions and all follow-ups, and there were no adverse events.
Participant Demographics and Vestibular Test Results.
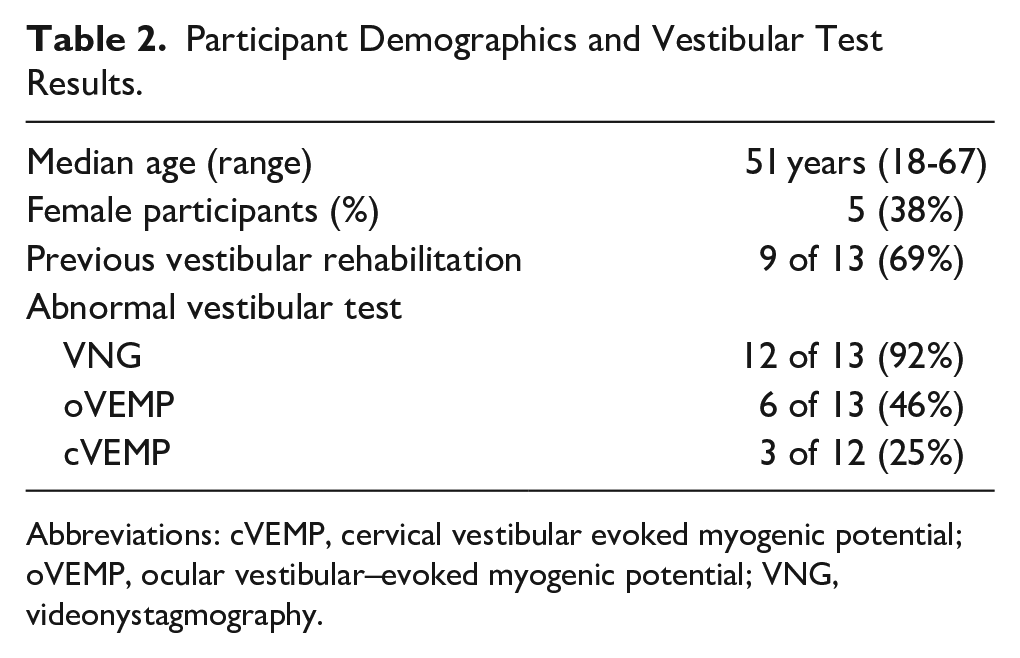
Abbreviations: cVEMP, cervical vestibular evoked myogenic potential; oVEMP, ocular vestibular–evoked myogenic potential; VNG, videonystagmography.
Before CVRT, the participants in this study had a median somatosensory (SOM) ratio of 1.00 (range 0.85-1.30), indicating intact ability to use somatosensory information to maintain equilibrium. SOM showed a negligible change of 0.01 (95% CI −0.06 to 0.04, P = .2402) after CVRT. The initial VIS [0.78 (0.46 to 1.05)], VEST [0.68 (0.32 to 0.98)], and 6:1 [0.62 (0.3 to 0.79)] ratios indicated significant difficulty maintaining equilibrium on the moving, sway-referenced platform prior to retraining. After CVRT, VIS increased by a median of 0.12 (−0.09 to 0.30; P = .0498), VEST increased by 0.10 (−0.060 to 0.25; P = .0122), and 6:1 improved by 0.15 (0.02 to 0.3, P = .0061) (Figure 1A). We also measured the change associated with CVRT for 2 composite ratios: visual preference (PREF) and DSR. The PREF ratio was 0.96 (0.64 to 1.09) prior to CVRT and changed negligibly by 0.02 (−0.04 to 0.12; P = .1843), and DSR was 0.66 (0.52 to 0.78) prior to CVRT and improved by 0.15 (0.03 to 0.24, P = .0012) (Figure 1B).
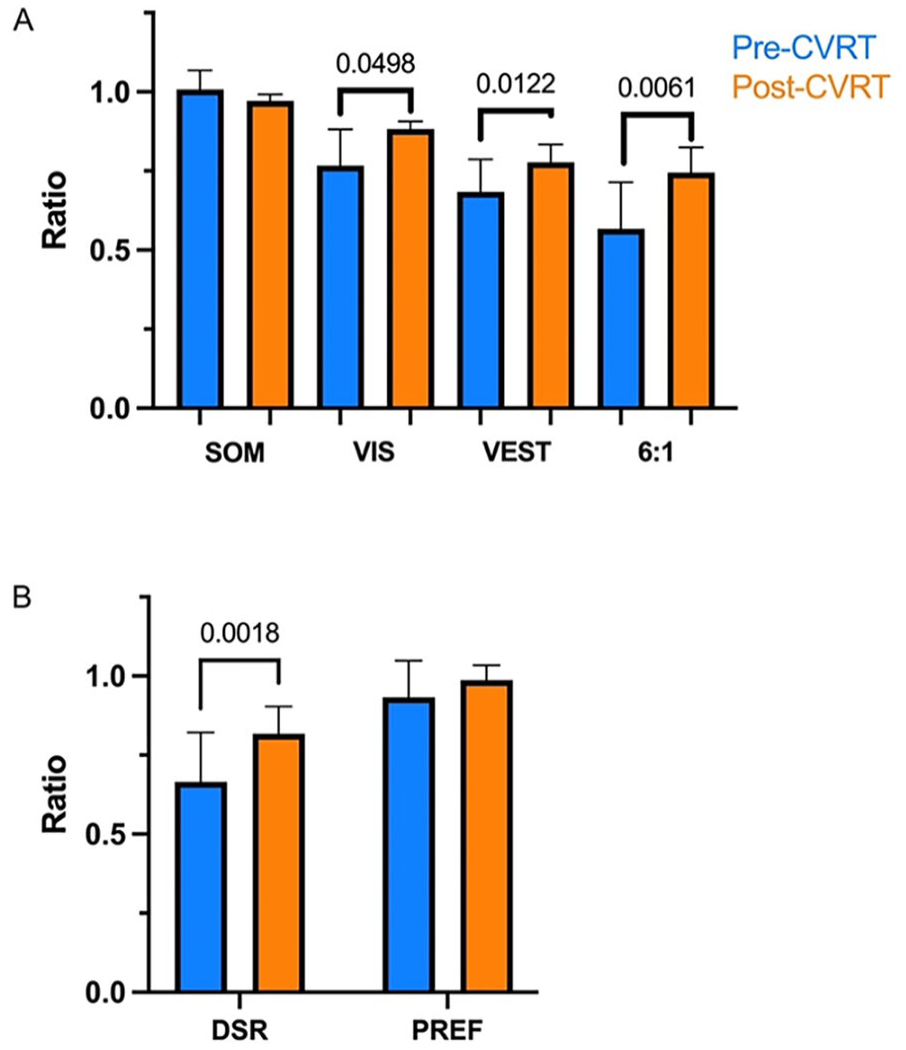
SOT SOM, VIS, VEST, condition 6:condition 1 (6:1), PREF, and DSR ratios before and after CVRT (A) simple pairwise ratios of SOT equilibrium scores and (B) composite ratios before (blue) and after (orange) 12 sessions of CVRT. Chart shows median values and error bars indicate 95% confidence intervals. SOM, somatosensory; VIS, visual; VEST, vestibular; PREF, visual preference; DSR, dynamic stability ratio; CVRT, computerized vestibular retraining therapy.
In post hoc stratification of participants with mild disability (DHI ≤30, n = 6) and those with moderate-to-severe disability (DHI >30, n = 7), we found that the moderate-to-severe group demonstrated significant improvements in the VIS ratio [0.16 (0.09 to 0.39)] and VEST ratio [0.12 (0.08 to 0.28)] after CVRT while the SOM and PREF ratios did not change (Figure 2). No changes were observed in the mild disability group. Accordingly, regression analysis found a higher initial DHI scores correlated with greater changes in VIS ratio (P = .022) and DSR (P = .012) (Table S1).
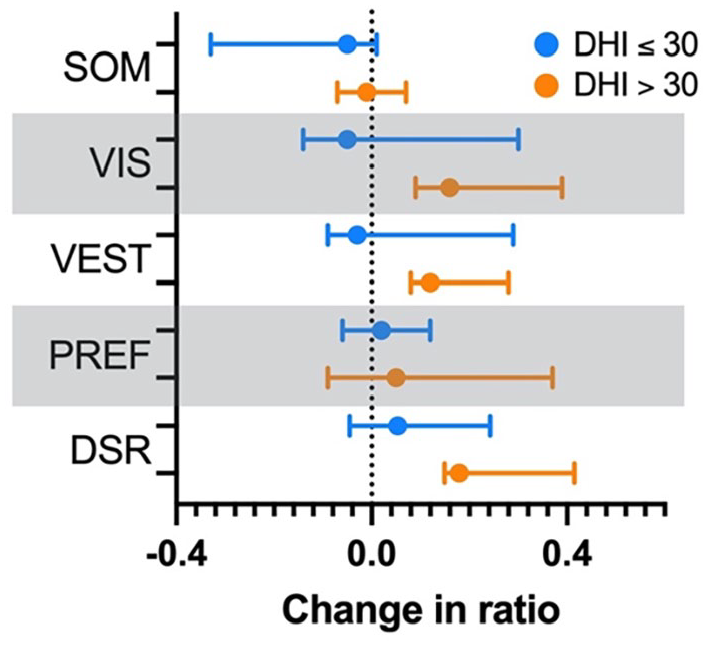
Comparison of changes in the SOT SOM, VIS, VEST, and PREF ratios for participants with mild disability (DHI ≤30; n = 6, open circles) to moderate-to-severe disability (DHI >30; n = 7, close circles). Points indicate the median change, and bars indicate 95% confidence intervals. SOM, somatosensory; VIS, visual; VEST, vestibular; PREF, visual preference; DSR, dynamic stability ratio; DHI, Dizziness Handicap Inventory.
Regression analysis found no association between change in sensory ratios and age. Neither male or female sex, nor history of vestibular physiotherapy was associated with a difference in sensory ratio response to CVRT (Table S1).
Discussion
Sensory Ratios of the SOT
This study used the SOT test to measure changes in postural control following 12 sessions of CVRT. The SOT employs precise posturographic measurement of sway, while participants attempt to maintain their balance during 6 different testing conditions. These conditions challenge the participant to maintain equilibrium while systematically removing or creating conflict with the somatosensory and visual frames of reference. Disruption of visual and somatosensory information is accomplished by “sway referencing”—that is, tilting of the support surface and/or visual field such that their orientation remains constant in relation to the sway angle. 8 In this way, as the participant leans in the anteroposterior axis, there is no visual and/or somatosensory cue of their change in angle relative to vertical.
The sensory ratios (Table 1) offer insight into the participants’ sensitivity to the loss of sensory input and weighting of these inputs. 8 The SOM (somatosensory) ratio describes the decrement in postural control when the 3 sources of sensory input—visual, somatosensory, and vestibular—are reduced to 2 by closing of the eyes. The VIS (visual) ratio describes the decrement when somatosensory information is lost through activation of the sway-referenced platform, leaving only visual and vestibular senses. The VEST (vestibular) ratio “isolates” the vestibular sense through activation of the sway-referenced platform and by closing of the eyes. The 6:1 ratio, which is associated with an elevated risk of falling,12,13 is similar in that the vestibular sense is the only veridical sense but, rather than removing vision by closing the eyes, a sway-referenced visual is presented. We included 2 composite ratios in our analysis, which measure sensitivity to conflicting information. The PREF ratio compares postural stability when participants are provided with conflicting, sway-referenced, visual information to performance with absent visual information (eyes closed). Finally, the DSR measures postural stability in 3 conditions, which each employ a sway-referenced platform against 3 conditions using a fixed platform.
A low SOM ratio would indicate uncompensated static balance deficits indicative of the very early stages of vestibular injury. The participants in this study had stable symptoms for greater than 6 months and had achieved static compensation; accordingly, they all had SOM ratios near 1. The VIS and VEST ratios both incorporate testing on the sway-referenced platform. After CVRT, participants displayed improved postural stability on the sway-referenced platform compared with the fixed platform, both in the presence of visual cues (VIS) and in their absence (VEST). There was no change in PREF, suggesting that participants were not so dependent on their visual sense that a conflicting visual reference is worse than none (Figure 1B). The DSR was included because this measure highlights the participants’ susceptibility to perturbation of their balance and to discordant somatosensory information.
It well established that both healthy participants14,15 and those with vestibular deficits 16 perform significantly worse on the sway-referenced platform than on the fixed platform. This reflects the importance of somatosensation through the feet and ankles during quiet stance but, further, is a consequence of the mobile platform causing a perturbation that tends to exacerbate small amplitude sway, thus requiring larger coordinated responses to stay balanced. We posit, therefore, the improvement in DSR indicates an improved ability to discern sway and improved capacity to integrate remaining sensory information (visual in condition 4 only and vestibular in conditions 5 and 6) to coordinate the appropriate motor response to remain standing.
Improvement in performance on the SOT associated with practice has been reported previously, including for sensory ratios. 17 The changes we observed after CVRT were approximately double (0.12 vs 0.059 for VIS; 0.10 vs 0.035 for VEST) what Bernstein and Burkard described for repeated administrations of the SOT in their cohort. Another study estimated that an 8-point improvement on the SOT composite score exceeded the 95% confidence interval of a practice effect 18 and, as we have previously reported, mean improvement in the SOT composite score after CVRT exceeded this threshold (8.8 points). 19 As described in Results, the improvements were even greater in those with more severe vestibular-associated disability.
Collectively, the improvements we observed for VEST, 6:1, and DSR, in excess of what would be expected from practice and independent of visual cues, suggest that improved postural stability arose from a gain in vestibular function.
Rationale for CVRT
In the days after unilateral vestibular injury, spontaneous nystagmus is resolved and static balance symptoms are greatly ameliorated through acute compensation. 20 Improvements in dynamic balance, through complex and diverse electrophysiologic and behavioral changes, continue to take place for most individuals over the ensuing weeks.
Commonly-recommended exercises are effective for improving participant-reported dizziness, visual acuity, postural stability, and gait,21,22 in part by promoting greater reliance on somatosensory or visual cues along with down-weighting of vestibular information.
However, compensatory strategies that are set during this period may rely too much on visual and somatosensory information. This may not be optimal for those with unilateral deficits, as individuals often have intact vestibular function on the contralateral side and may have some residual function on the affected side. Yet, many struggle to use their remaining intact function and continue to suffer impaired dynamic balance.23-25
Individuals with vestibular deficits adopt diverse motor-learning strategies to navigate the requirements of daily life, 26 and these strategies vary in their dependence on vision. 27 Horak suggested that residual, possibly-distorted vestibular information may be worse than none at all; however, those who learned to use remaining vestibular information from the intact ear performed better than those who relied heavily on visual and somatosensory cues. 25 Consistent with this, a high dependence on visual cues for postural control can lead to persistent postural-perceptual dizziness, a condition in which individuals become sensitive to discrepancies between predicted movement and actual motion. 28
We observed improved global balance performance, even in the absence of a somatosensory reference and of visual cues, coupled with the reduction in the between-participant variability, suggesting that compensation by use of remaining vestibular function, as described by Horak, was taking place for the participants in this study.
There is growing evidence that brain plasticity, which describes functional or structural changes to the brain, mediates changes in the processing of postural information that arise from elite athletic training or injury.29,30 This includes plasticity of the brain regions responsible for visual, vestibular, and somatosensory sensation, as well as motor control.31-33
Experiments in animal models have found that compensation after vestibular loss may involve neurogenesis, regrowth of hair cells, 34 and synaptic repair. 35
Neither hair cell regrowth nor adult neurogenesis in the vestibular nucleus has been demonstrated in humans to date; however, substitution to intact organs on the contralateral side is known to be an important mechanism of compensation for patients with unilateral deficits 25 and restoration of function of injured semicircular canals has been reported after vestibular neuritis. 36 If these responses to vestibular injury are conserved between these mammalian model species and humans, it suggests that in the weeks and months following vestibular injury, there are ongoing neuroplastic events occurring that may confer adaptive or maladaptive changes in postural perception. Indeed, structural changes in the brain have been observed in humans after unilateral 37 and bilateral 30 vestibular loss. Researchers have drawn parallels between changes associated with vestibular loss and adaptations observed in highly-trained athletes and dancers. 32
One consequence of continuous and evolving compensation is that early-compensatory behavioral and electrophysiological changes take place while neurological healing and restructuring is ongoing. Tighilet and Chabbert wondered, in their 2019 review, how sensory input from re-afferentiated vestibular organs would be reintegrated and whether repaired synapses that do not exactly recapitulate the pre-injury state could lead to aberrant sensory input during movement. 2 For some patients, early-compensation strategies that are “good enough” may fail to reintegrate retained or restored vestibular function into their postural maintenance strategy. Training protocols that call upon vestibular input may promote reweighting from an over-reliance on vision and somatosensation to a more balanced integration of sensory information that makes use of what vestibular senses are retained by the patient.
There is some evidence in the literature that training that incorporates incongruent sensory information may help improve dynamic balance. Telescopic glasses and virtual reality headsets, which introduce visual conflict, have been shown to benefit compensation.38-41 Likewise, tilting platforms that introduce somatosensory conflict may be superior to standard vestibular exercises for both subjective outcomes and gait. 42 Conversely, one study found that optokinetic stimulation without specific training for sensory conflict increased visual dependence, while a computerized dynamic posturography-based intervention resulted in improved postural stability. 43
CVRT seeks to promote the use of remaining vestibular function, whether visual and somatosensory information is congruent or not, in a manner that more closely replicates the integration of sensation by individuals with no deficit. By challenging participants to maintain their balance on an unsteady surface and in visually-complex environments, participants in this study improved their postural control in a variety of conditions. In fact, following retraining, their balance scores and sensory ratios compared well with those of healthy controls. 15
Conclusions
In this single-group pilot study, CVRT was associated with improvement in global balance and changes in the way sensory information is weighted to achieve postural control, in particular when vision and somatosensation are unreliable or absent. Changes were consistent with increased weighting of vestibular information over vision. These findings will be used to inform the design of a randomized controlled trial of this intervention.
Limitations
This single-group study did not include a no treatment or alternative treatment control and enrolled a small sample size of 13 participants, which limited statistical power. No sample size calculation was performed a priori. We enrolled participants with persistent, stable symptoms to minimize variability; however, we cannot rule out symptom variability unrelated to treatment. Individuals with mild impairment showed no benefit, but we cannot determine whether this was because of a ceiling effect of the SOT or whether those with mild impairment do not respond to treatment. It would be of interest to investigate whether CVRT is useful in other patient groups that experience postural unsteadiness and whether CVRT might be a useful adjunct to other treatments, such as vestibular physiotherapy. Results of this study will inform the design of a randomized controlled trial.
Supplemental Material
sj-docx-1-ear-10.1177_01455613251321978 – Supplemental material for Computerized Dynamic Posturography-Guided Vestibular Rehabilitation Improves Vestibular Sensory Ratios
Supplemental material, sj-docx-1-ear-10.1177_01455613251321978 for Computerized Dynamic Posturography-Guided Vestibular Rehabilitation Improves Vestibular Sensory Ratios by Eytan A. David, Navid Shahnaz, Isabel Wiseman, Yael David and Chris L. Cochrane in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
The authors acknowledge Alex Gouvea, M.Aud, for technical help with participant assessments.
Data Availability Statement
The dataset generated during the current study is not publicly available to protect the privacy of participants. Following publication, the data that underlie the results reported in this article are available from the corresponding author to researchers who provide a methodologically-sound proposal and sign a data access agreement, the conditions of which protect participant confidentiality.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
Consent to Participate
Participants in this study provided written informed consent.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
