Abstract
Objectives:
To assess the impact of hypoglossal nerve stimulation (HNS) on neurocognitive, psychological, and sleep outcomes in older adults with obstructive sleep apnea (OSA).
Methods:
A retrospective analysis of 42 patients aged 50 years and older who underwent HNS for OSA was conducted. Patient-reported outcomes were collected using validated tools, including the Insomnia Severity Index (ISI), PROMIS measures (Sleep-Related Impairment, Cognitive Function, Depression, Anxiety, Anger, Physical Function), Epworth Sleepiness Scale (ESS), Functional Outcomes of Sleep Questionnaire (FOSQ-10), and NIH Toolbox Loneliness survey. Pre- and post-HNS scores were compared using paired t-tests and Wilcoxon tests.
Results:
Significant improvements were observed in apnea–hypopnea index (AHI) (−17.6, P < .0001), ISI (−7.02, P < .0001), ESS (−3.4, P < .0001), and FOSQ (4.05, P < .0001). PROMIS measures showed significant posttreatment improvements in sleep impairment, cognitive function, depression, anxiety, anger, and loneliness (all P < .01). Physical function and pain did not change.
Conclusion:
HNS significantly improves sleep characteristics, cognitive function, and psychosocial outcomes in older adults with OSA. Future studies should include larger and more diverse samples with longer follow-up, as well as a control condition.
Keywords
Introduction
Obstructive sleep apnea (OSA) is a sleep disorder characterized by repeated interruptions in breathing during sleep due to upper airway obstruction with a higher prevalence in older adults.1,2 OSA is linked to severe health issues such as chronic fatigue, heart disease, stroke, cognitive impairment, and mood disorders like depression and anxiety.3-9 OSA patients have significant cognitive and psychological burden, experiencing deficits in attention, executive function, memory, and processing speed, all of which can substantially impact daily functioning and quality of life. 10
Continuous positive airway pressure (CPAP) is first-line treatment for OSA, improves sleep characteristics, daytime alertness, and mood, and reduces cardiovascular issues such as hypertension, atrial fibrillation, and congestive heart failure. However, treatment adherence is challenging for many patients. 11 Recent meta-analyses of randomized controlled trials have shown that while CPAP significantly reduces daytime sleepiness, it has not consistently demonstrated improvements in cognitive function domains, 12 underscoring the need to examine cognitive outcomes of alternative treatments, particularly in older populations vulnerable to the effects of sleep disruption. 2
Hypoglossal nerve stimulation (HNS) is an FDA-approved treatment for OSA patients intolerant of CPAP that involves implanting a device to electrically stimulate distal branches of the hypoglossal nerve and activate the genioglossus muscle, enlarging the airway at the level of the palate and tongue base.13-20 Several large, multicenter trials have demonstrated the effectiveness of HNS for long-term improvements in both apnea–hypopnea index (AHI) and overall quality of life. However, its cognitive and psychological impacts remain understudied.
Older patients seeking HNS treatment in our cross-sectional study had significantly higher rates of insomnia, sleep-related impairment, and depression compared to the general population, 7 and previous research has consistently demonstrated neurocognitive dysfunction in older OSA patients. 21 Using validated patient-reported outcome measures, this longitudinal cohort study builds on previous work to determine if neurocognitive and psychosocial outcomes significantly improve in older patients after HNS implantation. We hypothesized significant improvements across sleep, neurocognitive, and psychosocial domains following HNS in older patients. By comprehensively examining these outcomes following HNS therapy, this study furthers our understanding of treatment options for CPAP-intolerant OSA patients.
Patients and Methods
Data were drawn from a registry of HNS patients approved by the IRB (HM20023041).
Procedures
Patients 50 years and older seen for HNS consultation completed questionnaires (Supplemental Material) assessing self-rated health, insomnia, pain, physical function, sleep-related impairment, cognitive function, depression, anxiety, anger, loneliness, social quality-of-life, functional outcomes, and sleepiness. Both patients from our prior cross-sectional study and additional patients seen in the practice since publication were included if they fulfilled inclusion criteria and had postoperative records. Many domains were assessed using the Patient-Reported Outcomes Measurement Information System (PROMIS), person-centered measures developed by the National Institutes of Health. PROMIS tools use modern measurement techniques to assess physical, mental, and social health in adults and children and have been validated across diverse populations and conditions.22,23 These measures, standardized to t-scores with a mean of 50 and a standard deviation of 10 in the general U.S. population, enable meaningful comparisons across different health states and populations. PROMIS research suggests a threshold of 3 T-score points is reasonable for most contexts comparing group-level change. 24
The same questionnaires were re-administered at post-HNS implantation follow-ups. Inclusion criteria required fully completed questionnaire packets, with one at least three-months postoperatively. Exclusion criteria included missing postoperative sleep studies and those defined by the registry (eg, younger patients, children, incarcerated, pregnant individuals). Intervals between surgery and postintervention surveys were calculated using appointment and surgery dates. Higher scores on disability or impairment instruments represent worse health states.
Demographics and Adherence
Participant demographics, sleep apnea severity, and treatment adherence were obtained from electronic medical records (EMR). Adherence data, obtained at the time of the postoperative questionnaires, included average nightly usage (hours) and percentage of nights usage exceeded 4 hours.
Self-Rated Health
Self-rated physical health was assessed using a 1-item visual analog scale adaptation of the PROMIS Global Health v1.2 instrument. 25 This item is scored utilizing a 10-point Likert scale from 0 (worst imaginable health) to 10 (best health).
Insomnia Severity Index
Insomnia symptoms were measured using the Insomnia Severity Index (ISI), a 7-item self-report measure that assesses the nature, severity, and impact of insomnia over the past month. 26 Items are scored utilizing 5-point Likert scales from 0 (none) to 4 (very severe) for a total score of insomnia symptomatology. The ISI demonstrated high reliability (Cronbach α = .91) in clinical samples. 27
PROMIS Pain
Pain intensity was measured using the PROMIS Pain Intensity 1-item short form which asks participants to rate their average pain over the preceding 7 days on a Likert scale from 0 (no pain) to 10 (worst imaginable pain). PROMIS pain measures demonstrated high reliability (Cronbach α = .99) in previous studies. 28
PROMIS Physical Function
Self-reported physical capability was measured using the PROMIS Physical Function (PF) v2.0 4-item short form. It measures the ability to perform activities of daily living (ADL) and instrumental ADL but not the actual performance. 29 Items are scored on Likert scales from 1 (unable to do) to 5 (without any difficulty) for a total score of physical function. The PROMIS PF short form has acceptable reliability (Cronbach α = .92) in a large diverse sample. 30
PROMIS Sleep-Related Impairment
Alertness, sleepiness, tiredness, and functional impairments due to sleep problems during waking hours were measured using the PROMIS Sleep-Related Impairment (SRI) v1.0 4-item short form. 31 Each item is scored on a Likert scale from 1 (not at all) to 5 (very much) for a total sleep-related impairment score. The PROMIS SRI has acceptable reliability (Cronbach α = .90). 32
PROMIS Cognitive Function Ability
Cognitive functioning was measured using the PROMIS Cognitive Function Ability (CFA) v2.0 6-item short form. Its 6-items assess different facets of cognitive functions, such as mental acuity, thought processes, concentration, and memory. Items are scored on Likert scales from 1 (not at all) to 5 (very much) for a total score of cognitive functioning. The PROMIS CFA demonstrated good reliability (ie, Cronbach α = .95).33,34
PROMIS Depression
Depressive symptoms were measured using the PROMIS Depression v1.0 4-item short form. This measure assesses self-reported negative mood, self-worth, social cognition, and decreased levels of positive affect. 35 Items are scored on Likert scales from 1 (never) to 5 (always) for a total score of depressed mood. The PROMIS Depression short form demonstrated acceptable reliability (Cronbach α>.90) in a large racially and ethnically diverse sample. 36
PROMIS Anxiety
Emotional distress and anxiety were measured using the PROMIS Anxiety v1.0 4-item short form. This measure assesses self-reported fear, anxiety, hyperarousal, and somatic symptoms associated with anxiety. 35 Items are scored on Likert scales from 1 (never) to 5 (always) for a total score of anxiety. The PROMIS anxiety short form has demonstrated Cronbach α of .91 across numerous demographic characteristics. 37
PROMIS Anger
Self-reported facets of anger were measured using the PROMIS Anger v1.1 5-item short form. This measure assesses angry mood, negative interpersonal cognitions, and ability to control one’s anger. 35 Each item is scored on a Likert scale from 1 (never) to 5 (always) for a total score of anger. PROMIS anger scale demonstrated a Cronbach α of .96. 35
Loneliness
Loneliness was measured using the NIH Toolbox Loneliness Survey 5-item short form. This measure assesses perceptions that one is alone, lonely, or socially isolated from others. 38 Each item is scored on a Likert scale from 1 (never) to 5 (always) for a total loneliness score. The NIH Toolbox Loneliness survey has demonstrated a Cronbach α of .94. 38
Ability to Participate in Social Roles and Activities (Neuro-QoL)
Participation in social roles and activities was measured using the Neuro-QoL Ability to Participate in Social Roles and Activities 8-item short form. This assesses the degree of involvement in one’s usual social roles, activities, and responsibilities, including work, family, friends, and leisure. Each item is scored on a Likert scale from 1 (never) to 5 (always) for a total score for social quality of life. Neuro-QoL has demonstrated a Cronbach α of .96 across diverse demographics. 39
Functional Outcomes of Sleep Questionnaire
The functional outcomes of sleep questionnaire (FOSQ) is the gold-standard, OSA-specific instrument designed to assess how sleepiness impacts the ability to conduct daily activities. The 10-item short form (FOSQ-10) assesses the same five domains as the long form: general productivity, activity level, vigilance, social outcomes, and intimate and sexual relationships. FOSQ-10 demonstrated similar internal consistency as the long form (Cronbach α = .87 vs .95, respectively). 40
Epworth Sleepiness Scale
Self-reported sleepiness was measured utilizing the 8-item Epworth sleepiness scale (ESS). This measure assesses the propensity to fall asleep during one of eight situations: sitting and reading, watching television, sitting inactive in public, being a car passenger, lying down to rest, sitting and talking, sitting quietly after a meal without alcohol, or in a car while stopped for a few minutes in traffic or at a light. Items are scored on an ordinal scale from 0 (would never nod off) to 3 (high chance of nodding off). The estimated ESS in a meta-analysis of 63 studies was shown to have a cumulative Cronbach α of .82. 41
Data Analyses
Sample characteristics were described through means and frequency distributions. Adherence data (nights >4 hours used) was imputed in one case with missing information using regression imputation with average usage per night as a predictor. Scores on all NIH HealthMeasures instruments, except for PROMIS pain, which does not have a conversion, were converted to t-scores using the HealthMeasures scoring tool. Figures were created for measures with t-score thresholds to assign clinical characteristics based on published normative data developed through large-scale calibration.42,43 Paired sample t-tests and Wilcoxon signed-rank tests were used to compare pre- and postintervention outcomes for normal and nonnormally distributed data, respectively, as determined by Shapiro–Wilk testing. Mean difference (MD) is reported for pre/post changes. Temporal cohort sensitivity analysis was conducted using Early (0-180 days), Mid-Term (181-365 days), and Late (365+ days) interval thresholds. Kendall’s Tau correlations, with Benjamini–Hochberg correction for multiple comparisons, were used to assess relationships between three variables: age, nightly adherence, and percentage of nights with usage exceeding 4 hours, and the outcomes. Data were analyzed using R 4.0.4. 44
Results
The final sample included 42 individuals 50 years and older (mean = 67.67 ± 7.51 years) who underwent HNS treatment for OSA and met inclusion criteria. Of these, 40 identified as white (95.2%) and 22 as female (52.4%). The median interval to postoperative survey completion was 185 (interquartile range: 238) days. Average nightly adherence was 7.05 ± 1.65 hours with 77.85 ± 23.58% of nights >4 hours. The sample had severe baseline OSA with a mean AHI of 31.2 ± 12.3, which significantly decreased postintervention to 13.7 ± 16.1 [MD = −18.62, 95% CI (−22.47, −14.77), P < .001].
In terms of clinical characteristics, mean self-rated health increased from 6.4 ± 1.6 to 7.0 ± 1.5 [MD = 0.57, 95% CI (0.20, 0.94), P = .004]. Insomnia severity significantly improved from a baseline average of 14.6 ± 4.5 to 7.6 ± 4.5 [MD = −7.02, 95% CI (−8.74, −5.31), P < .001]. Notably, the percentage of participants with clinically significant insomnia (ISI ≥ 10) dropped from 86% to 29%. 27
PROMIS pain raw scores and physical function t-scores did not significantly change from preintervention (mean = 3.0 ± 2.6 and 46.3 ± 7.7, respectively) to postintervention (mean = 2.9 ± 2.9 and 46.6 ± 8.3, P = .92 and .78, respectively). Sleep-related impairment showed significant improvement, with the t-score decreasing from 57.1 ± 8.5 to 46.7 ± 10.3 [MD = −10.39, 95% CI (−13.34, −7.44), P < .001]. Cognitive function improved, with t-scores increasing from 43.0 ± 9.3 to 51.0 ± 13.5 [MD = 7.98, 95% CI (3.43, 12.53), P = .001].
Both depressive and anxiety symptoms significantly improved, with depression t-scores dropping from 50.1 ± 9.6 to 46.3 ± 8.4 [MD = −3.81, 95% CI (−5.56, −2.05], P < .001] and anxiety t-scores decreasing from 48.4 ± 8.5 to 44.4 ± 7.1 [MD = −3.97, 95% CI (−5.93, −2.01), P < .001], respectively. Similarly, the mean t-score for anger significantly decreased from 48.4 ± 10.5 to 43.2 ± 10.5 [MD = −5.13, 95% CI (−7.51, −2.75), P < .001].
Loneliness t-scores significantly improved from 49.2 ± 7.7 to 43.7 ± 8.0 [MD = −5.52, 95% CI (−7.92, −3.13), P < .001] following HNS. Social participation also showed significant improvements, with mean t-scores increasing from 47.6 ± 7.7 to 52.0 ± 7.9 [MD = 4.36, 95% CI (2.39, 6.33), P < .001].
Finally, the FOSQ and the ESS both significantly improved from 15.7 ± 2.9 and 9.4 ± 4.9 to 17.6 ± 2.4 [MD = 2.03, 95% CI (1.36, 2.69), P < .001] and 6.0 ± 4.5 [MD = −3.43, 95% CI (−4.57, −2.29), P < .001]. See Figure 1 for a graphical depiction of pre- and post-HNS scores across a variety of outcome measures.
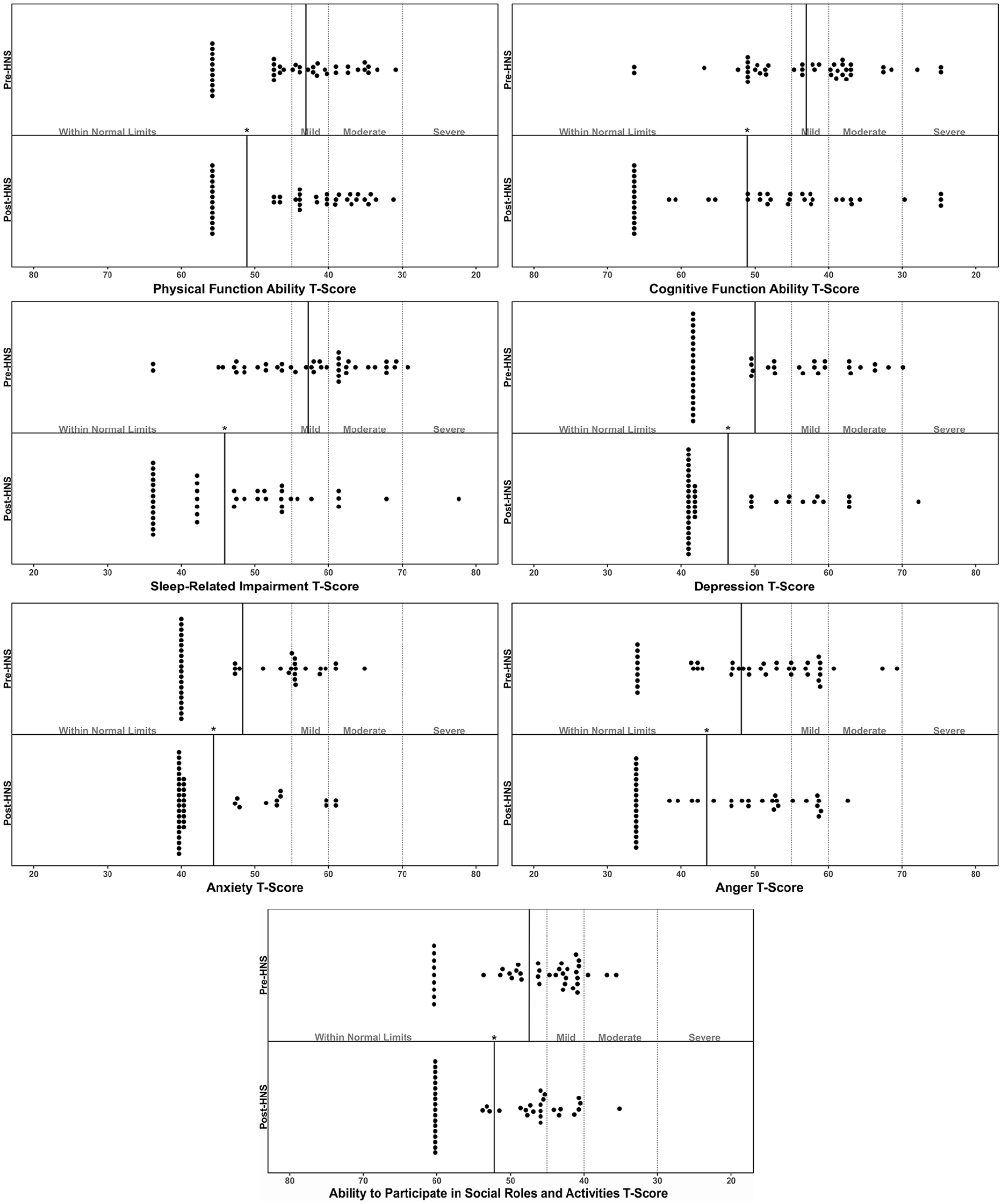
Clinical characteristics of older individuals before and after HNS treatment for obstructive sleep apnea for measures with clinically defined thresholds. HNS, hypoglossal nerve stimulation.
One-way ANOVA was conducted to assess the impact of follow-up intervals on changes in patient-reported outcomes. Among analyzed measures, only changes in PROMIS SRI demonstrated a significant difference between temporal cohorts (F(2, 39) = 4.60, P = .016). Post-hoc Tukey’s HSD revealed the early cohort (0-180 days, n = 20) had significantly greater improvements in PROMIS SRI compared to the mid-term cohort [181-365 days; n = 10; MD: 9.75, 95% CI (1.51, 17.98), P = .017]. There were no significant differences between the early and late cohorts (365+ days; n = 12; P = .150) or between the mid-term and late cohorts (P = .593).
Lastly, Kendall’s Tau correlation analysis identified a significant negative association between percentage of nights used >4 hours and ∆ISI (τ = −0.30, P = .007) and between age and ∆FOSQ (τ = −0.25, P = .026). After multiple comparison corrections, these associations were not significant.
Discussion
To our knowledge, this study is the first to comprehensively evaluate such an extensive array of psychosocial and neurocognitive characteristics in older OSA patients before and after HNS placement. Using validated questionnaires, HNS resulted in improvements in almost all psychosocial outcomes, including sleep-related impairment, anxiety, depression, anger, loneliness, cognitive function, and social participation. Pain and physical function did not significantly improve.
Following HNS, older patients experienced significant AHI reductions, improving average OSA severity from severe to mild, consistent with other studies.18,45,46 Sleep characteristics also improved, as evident through meaningful decreases in ISI (−7.0) and ESS (−3.4), and increased FOSQ-10 scores (+4.1), affirming the positive impact of HNS on sleep outcomes.15,47,48
As HNS gains popularity for CPAP-intolerant patients, its potential psychosocial and cognitive benefits have garnered interest. A prior study highlighted elevated rates of insomnia, sleep-related impairment, and depression, along with lower physical and cognitive function in older patients considering HNS. 7 The current study builds on these findings to evaluate similar postoperative outcomes with evidence for extensive benefits of HNS treatment.
Hofauer et al. reported similar AHI reduction (32.8/h to 12.6/h) in 102 patients and used an adaptation of the attitudes to CPAP treatment inventory survey, and patients strongly agreed HNS reduced OSA-related problems. 49 Another study, by Grieco et al. examined neurocognitive improvement in a small number of individuals with Down syndrome post-HNS with severe OSA. 50 Despite varying outcomes and populations, both studies highlighted improvements in patient well-being beyond OSA symptom relief. 50
Rosenthal et al. assessed anxiety, depression, and emotional distress using Generalized Anxiety Disorder-7 and Patient Health Questionnaire-9 surveys in relation to HNS adherence in a cross-sectional study of 33 patients. 51 Those using HNS ≥ 28 hours per week had significantly lower anxiety and emotional distress scores than the nonadherent group (<28 h/week). 51 This suggests that HNS treatment can positively impact daytime functioning, depression and anxiety symptoms, and aligns with our cohort’s results with an average nightly adherence exceeding 7 hours.
This retrospective study has some limitations. EMR data may contain entry errors and gaps. Sleep studies heterogeneity (home sleep tests, polysomnography, titration) potentially limits interpretation of AHI improvements. Excluding patients with incomplete questionnaires could introduce some selection bias if these patients were substantially different from the current cohort. While validated surveys were used to mitigate recall and social desirability biases, ISI—though significantly improved—has not been validated for OSA and includes noninsomnia-specific sleep quality items. The variable timeframe for questionnaire completion also introduces potential confounding, though temporal sensitivity analysis showed minimal temporal effects. Finally, negative findings should be interpreted cautiously due to lack of a priori power analysis.
Future studies could use behavioral indicators, such as wrist actigraphy and objective cognitive testing, to corroborate self-reported data. Given our limited sample size and diversity, further research should include larger, more diverse populations to improve comparison correlation calculations.
Conclusion
This study compared the clinical, psychological, and social effects of HNS in older patients using validated NIH PROMIS surveys. 30 Results suggest HNS improves sleep-related impairment, self-reported cognition, depression, anxiety, and anger in this population. As PAP-alternative treatments for sleep apnea become more accessible, it’s crucial to study various treatment outcomes. HNS may not only affect disease-specific outcomes, but it may also benefit cognitive and psychosocial functioning.
Supplemental Material
sj-pdf-1-ear-10.1177_01455613251320486 – Supplemental material for Neurocognitive and Psychosocial Outcomes in Older Adults with Obstructive Sleep Apnea Following Hypoglossal Nerve Stimulation
Supplemental material, sj-pdf-1-ear-10.1177_01455613251320486 for Neurocognitive and Psychosocial Outcomes in Older Adults with Obstructive Sleep Apnea Following Hypoglossal Nerve Stimulation by Arman Saeedi, Alyssa N. Calder, Andrew J. Belilos, Roberto Spilka, Ryan S. Nord and Joseph M. Dzierzewski in Ear, Nose & Throat Journal
Footnotes
Data Availability Statement
Deidentified datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Ryan Nord is a consultant for Inspire Medical Systems. Dr Joseph Dzierzewski served on an advisory panel for Eisai Pharmaceuticals and Apnimed and has received honorarium for presentation to the Nevada Psychological Association.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by a grant from the National Institute on Aging of the National Institutes of Health under Award Number K23AG049955 (PI: Dzierzewski). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or National Sleep Foundation.
Statement of Ethics
This study received ethical approval from the VCU Health IRB (approval #HM20023041) on September 12, 2024. This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
