Abstract
Introduction
Sjogren’s syndrome (SS) is the second most common autoimmune rheumatic disease, with a global incidence of approximately 7/105. 1 Lymphocyte infiltration is the primary pathological feature, leading to progressive damage to exocrine glands. 2 Glucocorticoids and immunosuppressants are the main treatments for controlling and delaying tissue and organ damage caused by autoimmune responses. 3 However, due to impaired immune function, infections are a major cause of poor prognosis and mortality in patients with SS. 4 Mucormycosis is a rapidly progressive and often fatal invasive fungal infection that primarily affects immunocompromised patients with underlying conditions such as diabetes and hematologic disorders. 5 Epidemiological studies have shown an increasing trend in the incidence of mucormycosis, rising from 0.7 per 1,000,000 to 1 per 1,000,000 between 1997 and 2006. 6 Based on the site of infection, mucormycosis can be classified into rhino-orbito-cerebral mucormycosis (ROCM), cutaneous mucormycosis, gastrointestinal mucormycosis, pulmonary mucormycosis, and disseminated mucormycosis. Among these, ROCM is the most common. 7 ROCM can be further categorized into acute and chronic forms. Acute ROCM progresses rapidly, spreading from the nasal cavity to the paranasal sinuses, eyes, orbital apex, and intracranial structures through vascular dissemination. Within days, it can invade the brain, leading to severe complications such as black eschar formation on the nasal palate, blindness, embolism of critical adjacent blood vessels, and cerebral ischemia with necrosis. The mortality rate exceeds 80%, yet it is frequently misdiagnosed. 8 This article reports a case of Sjogren’s syndrome complicated by rhinocerebral mucormycosis, detailing its diagnosis, treatment, and prognosis. The aim is to enhance understanding of this rare and severe condition, emphasize the importance of early diagnosis, and promote accurate and timely treatment.
Medical Records
General Data
On May 14, 2024, a patient presented to the hospital with complaints of “nasal pain and swelling persisting for 20 days.” The patient reported the spontaneous onset of a nasal lesion 20 days prior, which progressively enlarged and eventually ruptured. A paranasal sinus CT scan performed on May 2 revealed bilateral maxillary sinusitis, ethmoid sinusitis, nasal septum deviation, and bilateral inferior turbinate hypertrophy. Upon admission, physical examination identified black eschar necrosis on the nasal surface accompanied by skin defects (Figure 1a). The patient also reported associated symptoms, including nasal pain, obstruction, gum discomfort while eating, and nasal reflux when drinking. The patient’s medical history was notable for Sjogren’s syndrome diagnosed 6 years prior, managed with prednisone acetate, aspirin, hydroxychloroquine, and mycophenolate mofetil. Other significant comorbidities included a 2-year history of hypertension controlled with valsartan, steroid-induced diabetes managed with subcutaneous insulin for 2 years, aseptic necrosis of the femoral head for 5 years, primary Sjogren’s syndrome-associated renal impairment for 2 years, and a history of hypokalemia, tuberculosis, and hyperlipidemia.
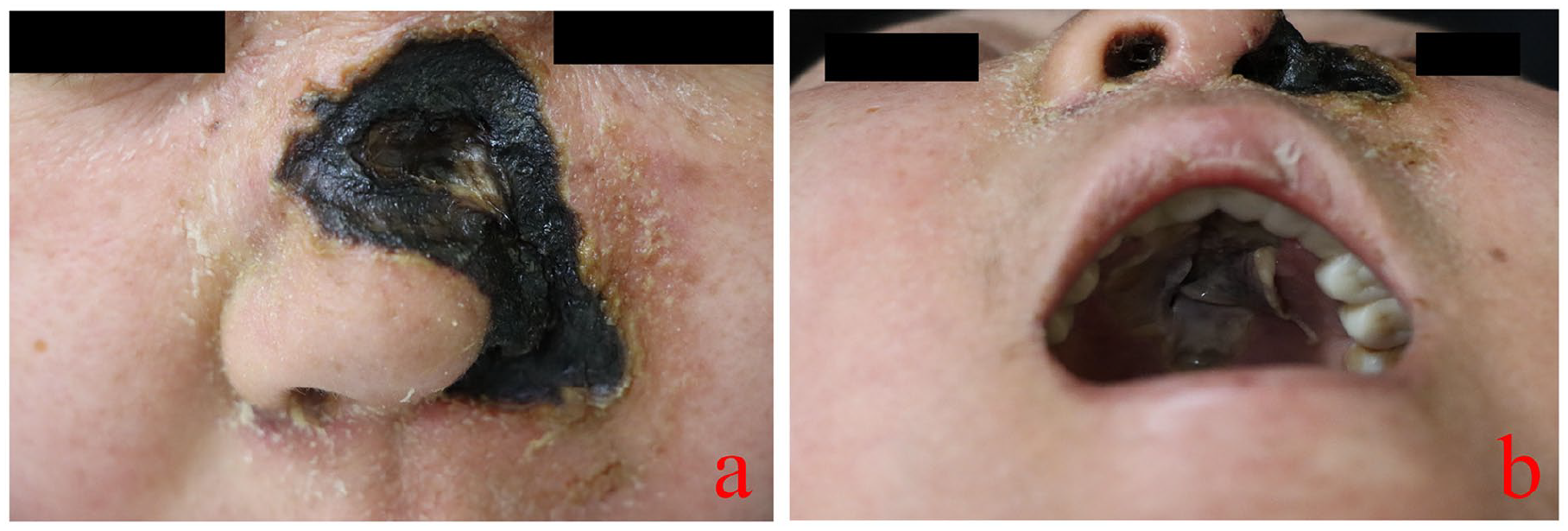
(a) Black eschar necrosis with skin defects on the nasal surface; (b) 1.5 × 4.5 cm mucosal necrotic cavity-like ulceration in the maxilla, appearing yellow-white.
Physical examination
Temperature: 36.2℃, Pulse: 109 bpm, Respiratory Rate: 18 breaths/min, Blood Pressure: 107/63 mmHg. The patient is well-developed, well-nourished, and appears natural in posture. They are conscious, articulate, and cooperative. There is eyelid edema present; the conjunctiva appears normal without jaundice or scleral icterus. Pupils are equal and reactive to light. A black eschar is observed on the facial skin on the left side of the nose. A blood scab is adherent to the inside of the left nasal cavity. Nasal secretions are present in the right nasal cavity. The patient has missing teeth and is wearing dentures. A hard palate defect is noted, which communicates with the nasal cavity (Figure 1b).
Imaging examinations
Cardiac Doppler ultrasound revealed right atrial and right ventricular enlargement, severe pulmonary artery hypertension, and significant tricuspid regurgitation. Doppler ultrasonography of the liver, gallbladder, pancreas, spleen, and kidneys showed increased echogenicity of the bilateral renal parenchyma along with kidney stones. Thyroid and cervical lymph node Doppler ultrasound revealed no apparent abnormalities in the thyroid gland. Nasal and paranasal sinus MRI indicated a defect in the left maxilla, indistinct visualization of the left alar cartilage and part of the nasal bone, bilateral maxillary sinusitis, ethmoid sinusitis, nasal septum deviation, and hypertrophy of the right inferior turbinate with unclear delineation of the structure (Figure 2). A chest CT scan of the lungs showed bilateral localized interstitial lesions with lung inflammation compared to 3 months prior. Multiple nodular shadows, some with partial cavitation, were observed in both lungs, suggestive of potential infective lesions or large granulomas. Other findings included mild cardiomegaly with enlarged pulmonary arteries, pericardial effusion, bilateral pleural thickening, and a low-density shadow in the spleen. The electrocardiogram revealed sinus tachycardia, mild ST-segment depression, abnormal T waves, an R/S ratio >1 in lead V1, and right axis deviation.
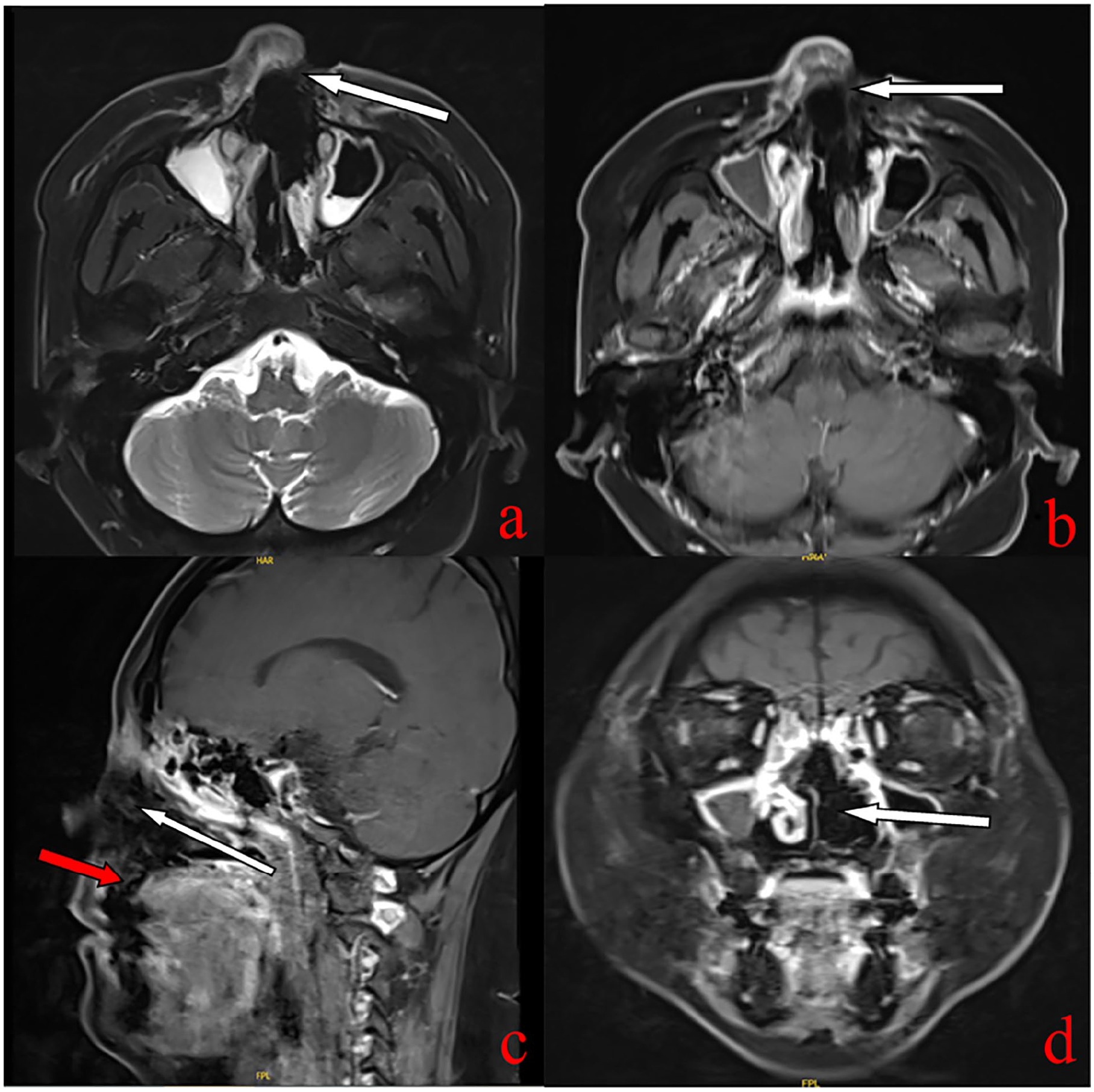
Nasopharyngeal MRI scans with the following findings: (a) T2W1 (horizontal) showing a missing portion of the left maxillary anterior wall. (b) T1W1 (horizontal) showing a missing part of the left maxillary anterior wall. (c) T1W1 (sagittal) the white arrow indicating wide erosion inside the nasal cavity; the red arrow indicating the absence of the palate communicating the nasal cavities. (d) T1W1 (coronal) showing the absence of the left nasal structure.
Laboratory test results
Blood routine inspection showed the following: white blood cells 8.27 × 10⁹/L, neutrophils 5.17 × 10⁹/L, red blood cells 4.26 × 10¹²/L, hemoglobin 120.0 g/L, platelets 143 × 10⁹/L, lymphocytes 2.26 × 10⁹/L, monocytes 0.51 × 10⁹/L, eosinophils 0.30 × 10⁹/L, and basophils 0.03 × 10⁹/L.
Clotting tests indicated an activated partial thromboplastin time (APTT) of 50.50 s, an international normalized ratio for APTT of 1.48%, and fibrinogen concentration of 8.14 g/L.
Biochemical tests revealed: blood glucose 4.08 mmol/L, potassium levels 3.35 mmol/L (↓), chloride levels 117.1 mmol/L (↑), CO₂ levels 13.5 mmol/L (↓), cholinesterase activity 4330 U/L (↓), albumin concentration 26.2 g/L (↓), globulin concentration 36.1 g/L (↑), alkaline phosphatase activity 387.1 U/L (↑), γ-glutamyl transpeptidase 365.9 U/L (↑), lactate dehydrogenase 486.3 U/L (↑), lipoprotein (a) 676.5 mg/L (↑), sialic acid 10,625 μg/mL (↑), procalcitonin 0.31 ng/mL (↑), C-reactive protein 65.6 mg/L (↑), anti-mycobacterium tuberculosis antibody IgG positive, and erythrocyte sedimentation rate 70 mm/h (↑). Immunoglobulin M was 0.355 g/L (↓).
Urinalysis showed proteinuria (+), white blood cells 95.20/μL (↑), red blood cells 206.40/μL (↑), and epithelial cells 47.28/μL (↑).
Tumor markers showed: carbohydrate antigen 125 (CA125) 56.4 U/mL (↑), neuron-specific enolase 167 ng/mL (↑).
Antinuclear antibody (ANA) spectrum revealed ANA-IgG (+), nRNP/Sm-IgG (+++), SSA-IgG (+++), Ro-52-IgG (+++).
Biopsy findings
The biopsy showed chronic inflammation with infection and necrosis, as well as squamous epithelial hyperplasia with a few large nuclei in the stroma. Tissue bacterial and fungal cultures identified Klebsiella pneumoniae, Staphylococcus epidermidis, and Candida species. Fungal immunofluorescence examination showed hair fungal hyphae with wide septa and right-angle branches. Microscopic examination revealed fungal spores, as shown in Figures 3 and 4.
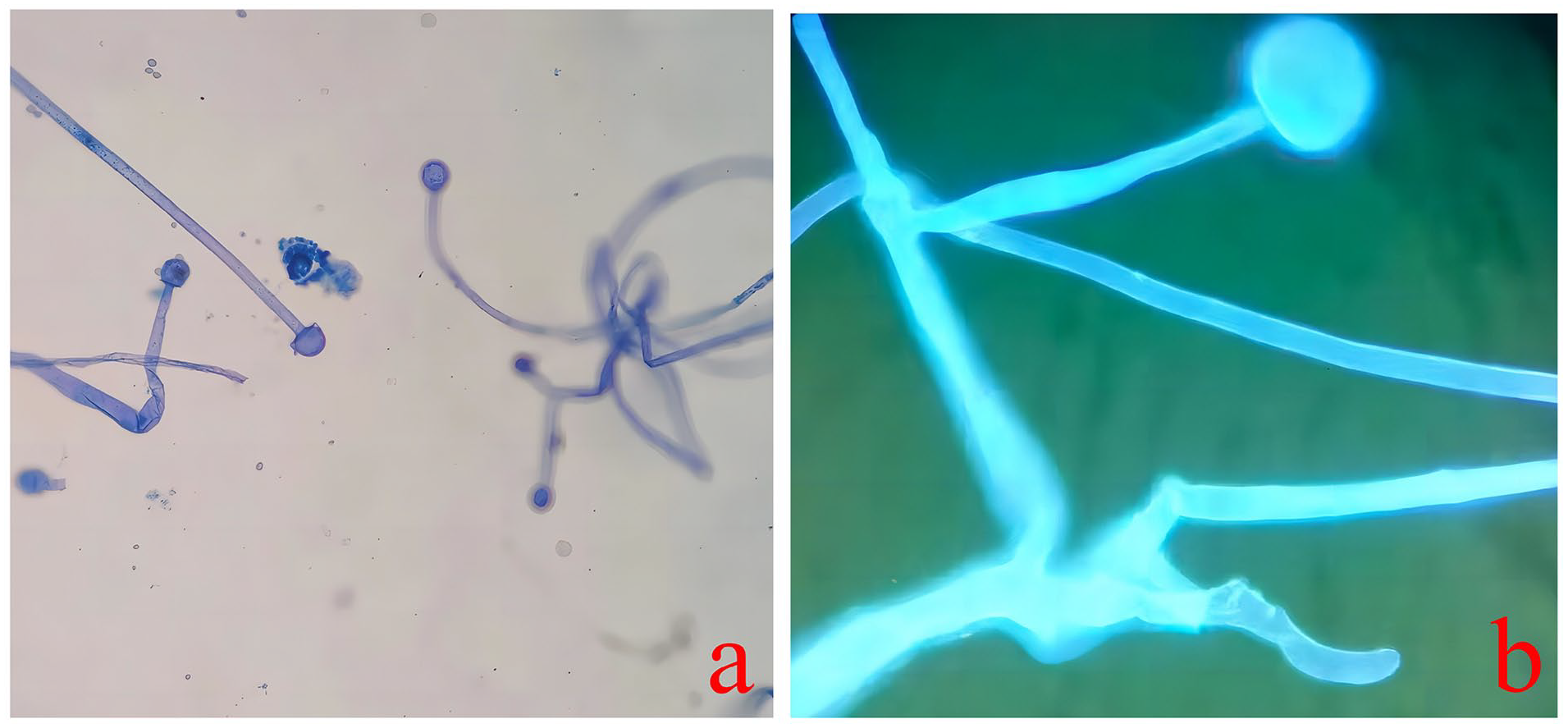
Immunofluorescence and phenol medan staining shows mucormycosis hyphae with wide, nonseptated, right-angled branches. (a) Immunofluorescence staining at 400x magnification. (b) Lactic acid phenol medan dyeing at 400× magnification.
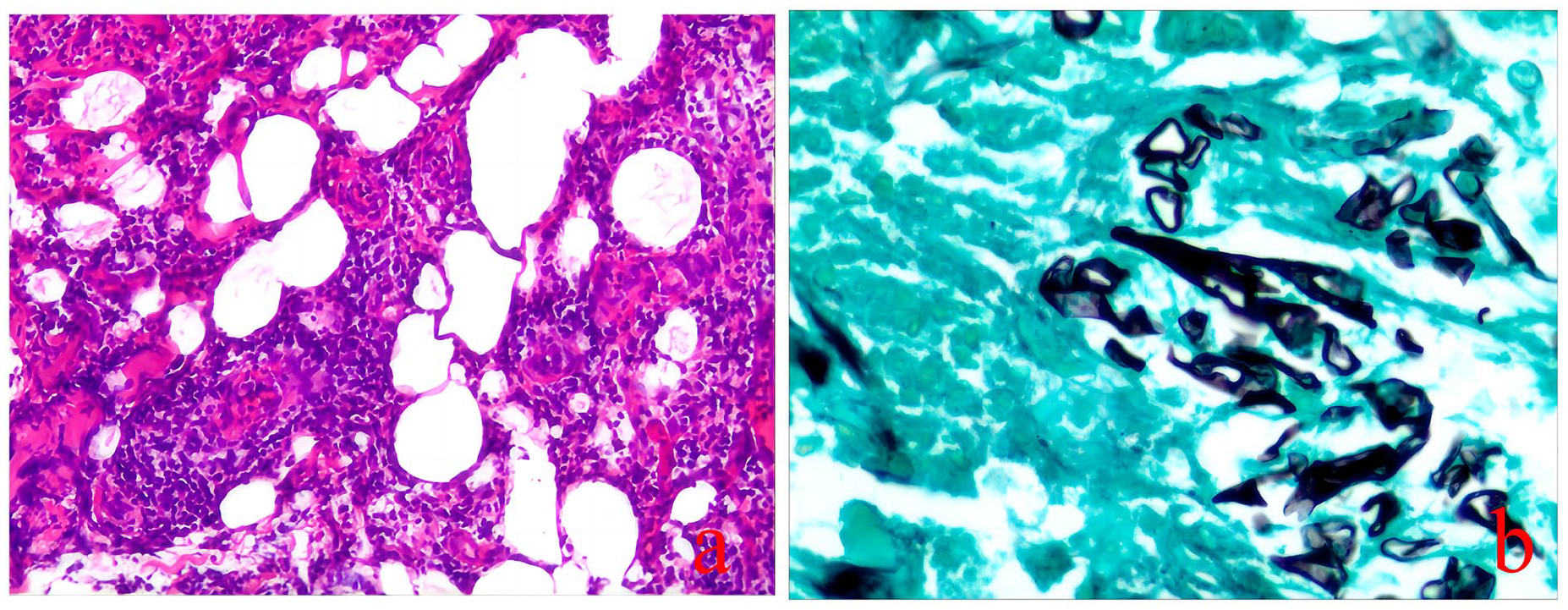
Histopathological examination of the lesions. (a) H&E staining at 100× magnification reveals chronic inflammation with infection and necrosis, along with squamous epithelial hyperplasia and a few large nuclei in the stroma. (b) Silver hexamethonium stain at 200× magnification, black arrows indicate mucor infection.
Treatment
Upon admission, the patient received symptomatic treatment and supportive care, including management of blood glucose and blood pressure, anti-infection therapy with piperacillin-tazobactam sodium 4.5 g TID, and fluid infusion. An immediate paranasal sinus MRI was performed, and samples from the paranasal and hard palate lesions were sent for bacterial culture, fungal culture, and histopathological examination. Fungal immunofluorescence testing revealed the presence of Mucor hyphae. The patient underwent a partial maxillectomy under general anesthesia, along with open sinus surgery and facial soft tissue debridement, followed by antifungal therapy postsurgery. During the procedure, a nasofacial incision was made along the decomposed edge (approximately 5 mm). Black eschar was removed, and full-thickness skin necrosis was observed in the right nasal alar and surrounding skin. Necrotic tissue was excised down to the bone, revealing erosion and blackening of the bilateral maxilla, nasal bone, ethmoid bone, and frontal bone (Figure 5). Necrotic bone was also excised, including the affected tissue extending into the frontal sinus and down to the level of the hard palate at the palatine bone plate. The mucous membrane necrosis of the hard palate bone was preserved after thorough removal of the nasal bones, bilateral maxillary portions, maxillary sinus walls, and the frontal, sphenoid, and ethmoid sinuses. Hemostasis was achieved, and the surgical cavity was repeatedly irrigated with normal saline. A gastric tube was inserted through the right nostril, and iodoform gauze was placed within the surgical cavity, followed by pressurized bandaging. The patient was then transferred to the intensive care unit. Unfortunately, on May 20th, 2024, the patient succumbed to a brain infection.
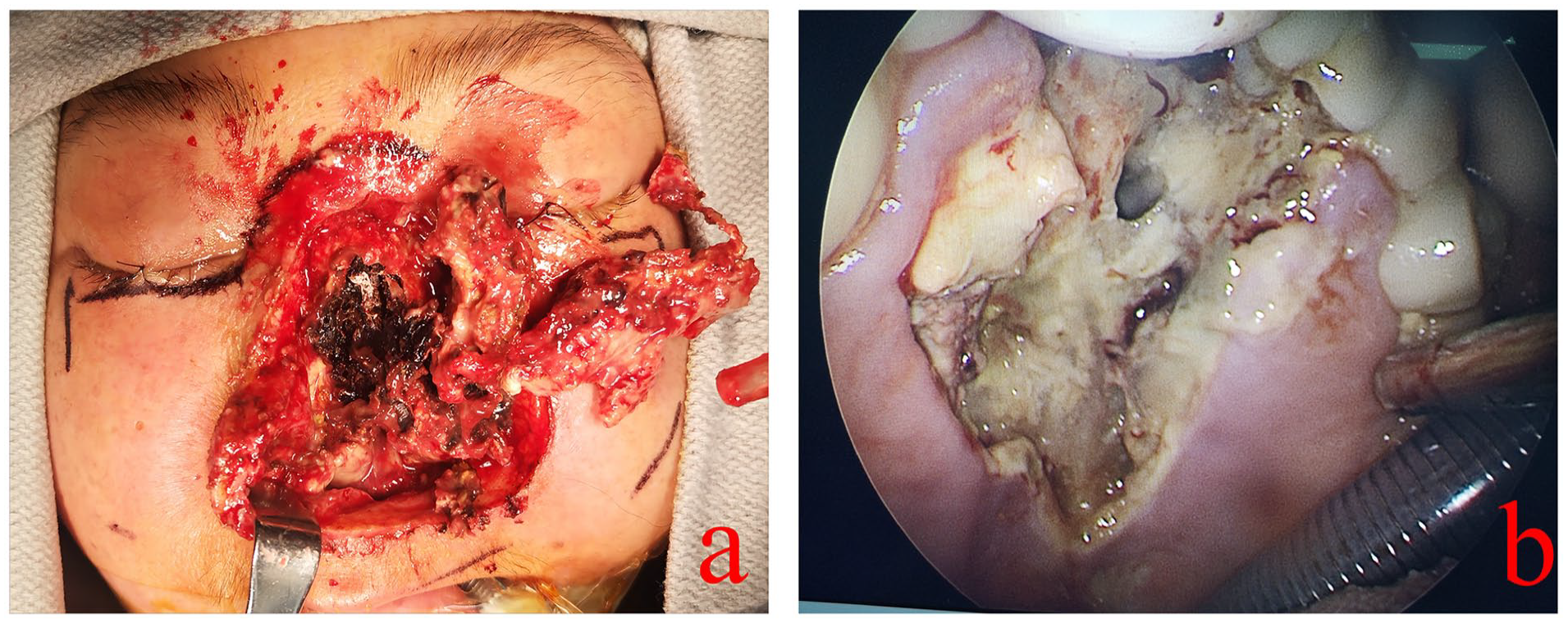
(a) Extensive necrosis is observed in the nasal cavity, with black corrosion of the bone; (b) mucosal necrosis of the hard palate with exposed bone (as arrow shows), communicating with the nasal cavity.
Discussion
Mucormycosis is a vascular-invasive infection. When the fungal hyphae invade blood vessels from the lesion site, they disrupt the integrity of the vascular intima, triggering a fibrin response. This leads to vascular embolism or aneurysm formation, causing tissue ischemia and infarction. Vascular obstruction further exacerbates tissue hypoxia and acidosis, creating a more favorable environment for the reproduction, growth, and spread of Mucormycosis, thus initiating a vicious cycle. The clinical presentations of mucormycosis are diverse, depending on the type of infection. ROCM occurs when pathogenic fungi enter through the nose or conjunctiva, spreading to the sinuses, oral mucosa, eyes, and ultimately reaching the brain. Key symptoms include facial edema, nasal sinus empyema, headache, fever, and blurred vision. A hallmark feature of ROCM is the presence of black eschar necrosis on areas such as the nose, mouth, or face. 9 Pulmonary mucormycosis results from the inhalation of pathogens into the respiratory tract, leading to pneumonia and bronchitis. The main clinical manifestations include coughing, chest pain, dyspnea, and fever. 10 Cutaneous mucormycosis typically arises from local infections caused by trauma or damage to the skin barrier. It presents as skin ulcers, blisters, redness, with visible red rings around the lesions. 11 Gastrointestinal mucormycosis most commonly affects patients with chronic digestive system diseases, presenting with symptoms such as abdominal pain, diarrhea, nausea, vomiting, and gastrointestinal bleeding. 12 Blood-borne disseminated mucormycosis primarily occurs when the fungi invade the bloodstream, causing acute inflammatory embolic lesions across multiple organs. 13 Lesion-type mucormycosis in solid organs such as the kidneys, liver, bladder, and uterus exhibits distinct presentations based on the affected organ. 14
Early clinical manifestations of ROCM lack specificity, which can lead to delays in diagnosis and treatment. 14 Acute ROCM progresses rapidly and has a high mortality rate, highlighting the importance of prompt and accurate diagnosis. Diagnosis is primarily based on nasal endoscopic findings, clinical manifestations, imaging studies, pathogen detection, and histopathological examination. Nasal endoscopy is a crucial diagnostic tool for early ROCM detection, allowing for direct observation of nasal mucosal lesions, along with histological and fungal assessments. Imaging plays a pivotal role in identifying invasive sinus and nasal bone infections, which can cause extensive destruction of nasal cavity structures and may potentially erode the skull base. Biopsy and fungal culture remain the gold standard for confirming mucormycosis diagnosis. 15 Clinicians should maintain a high level of suspicion for mucormycosis in patients with long-term immune dysfunction, diabetes, hyperlipidemia, and other risk factors, especially when presenting with nasal endoscopic ulcers, scabby nasal mucosa, peri-orbital swelling, black eschar necrosis or perforation on the turbinates or hard palate. Imaging may reveal erosive bone defects in the sinuses or nasal bones. Currently, the primary treatment for mucormycosis involves a combination of antifungal therapy and surgical intervention. Endoscopic debridement of the nasal cavity and paranasal sinuses is the standard approach for ROCM treatment, with the goal of removing necrotic tissue and preventing further spread of the hyphal infection. 16 Although Amphotericin B (AmB) and liposomal Amphotericin B (L-AmB) are the preferred antifungal agents, they are associated with significant nephrotoxicity and a risk of other adverse effects, including refractory hypokalemia, nausea, vomiting, and other toxicities. The antifungal mechanism of AmB is not fully understood, but the most widely accepted hypothesis is that AmB forms transmembrane channels by binding to ergosterol in the fungal cell membrane. This binding induces the efflux of potassium ions, ultimately leading to cell lysis and a bactericidal effect. 17 Studies have shown that in the treatment of childhood trichophytosis, the mortality rate was 5% higher in the AmB monotherapy group compared to the combination drug and surgery group, suggesting that antifungal drugs combined with surgery are the most effective strategy for reducing mortality in childhood trichophytosis. 18 AmB has been found to exert an oxidative killing effect on fungal cells, while also inducing the production of pro-inflammatory factors such as TNF-α, IL-6, and IL-1β, along with chemokines and prostaglandins, through a signaling pathway mediated by Toll-like receptors and co-receptor CD14 in host cells. This action can be both bactericidal and damaging to host cells, which is one reason why AmB causes nephrotoxicity. Additionally, this mechanism contributes to persistent hypokalemia, nausea, and vomiting, along with other toxic side effects. 19 According to the Expert Consensus on Diagnosis and Management of Mucormycosis in China (2022), 20 for patients with normal renal function, the recommended dosage of AmB is 0.5 to 0.7 mg/kg per day, with the maximum daily dose for adults not exceeding 1 mg/kg per day. L-AmB), a derivative of AmB, offers several advantages over AmB, including a significantly lower incidence of renal damage. Additionally, high-dose L-AmB demonstrates a stronger antifungal effect compared to the same dose of AmB, making it the preferred drug in combination regimens. 21 Both the European Confederation of Medical Mycology (ECMM) 22 and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) 23 recommend L-AmB as the drug of choice for antifungal therapy. L-AmB shows significant dose dependence, with the recommended daily dose ranging from 1 to 10 mg/kg/day. However, higher doses may cause nephrotoxicity and hypokalemia, though these reactions are typically reversible. In a retrospective analysis of 120 patients with trichotillomania, the mortality rate was 25% (eight patients) in the sequential treatment group with AmB and L-AmB, and 37.5% with L-AmB alone, both of which were superior to AmB monotherapy. 21 It is important to note that L-AmB requires a 5% glucose solution for infusion. For diabetic patients receiving L-AmB, continuous insulin administration is necessary to control blood glucose. Poorly controlled blood glucose levels may facilitate the growth of Trichophyton rubrum, which could undermine the efficacy of L-AmB. 24 For patients who cannot tolerate AmB or L-AmB, triazole antifungal drugs such as esaconazole, posaconazole, and voriconazole are alternative options. These drugs work by inhibiting the fungal enzyme cytochrome P450-dependent 14-α-sterol demethylase, which prevents the conversion of lanosterol to ergosterol. This inhibition disrupts the synthesis of fungal cell membranes, compromising their integrity, permeability, and fluidity, ultimately leading to cell rupture and apoptosis. 25 Studies have indicated that voriconazole has a high probability of crossing the blood–brain barrier (up to 50%) and demonstrates significantly better efficacy in treating invasive fungal infections compared to amphotericin B. As a result, the survival rate of patients with intracranial infections secondary to ROCM can increase to approximately 35%.26,27
Since the combination of two or more antifungal drugs with synergistic effects offers complementary pharmacokinetics or pharmacodynamics, it can improve antifungal efficacy, drug effectiveness, and safety, while also alleviating the emergence of drug resistance. As a result, antifungal combination therapy has gained increasing attention from the academic community in recent years. 28 A study of 32 hematological patients with invasive Trichoderma infections treated with L-AmB in combination with azole antifungal drugs and followed up for 3 months revealed that 56% of patients treated with L-AmB and posaconazole showed improvement in clinical symptoms. However, 28% of patients did not respond to the treatment and ultimately died, suggesting that the combination of L-AmB with voriconazole may offer a promising treatment approach for invasive Trichoderma infections. 29 In a prospective multicenter study involving 232 patients with suspected invasive mycoses and 13 patients with confirmed or suspected diagnoses of trichomycosis, the results showed that serum Trichothecene qPCR had a sensitivity of 85.2% and a specificity of 89.8%. Notably, the median time to the first positive qPCR sample occurred four days prior to the first positive mycological or histological sample, and one day before the first imaging test. 30 Among the 33 patients who received L-AmB treatment two days after their first positive serum qPCR, 21 became qPCR-negative within four days. Patients who became qPCR-negative within seven days of treatment initiation had significantly higher 30-day and 6-month survival rates compared to those who remained qPCR-positive. 30 These findings suggest that serum Trichophyton qPCR is a noninvasive technique that can aid in the early diagnosis of trichophytosis and trigger timely, targeted antifungal therapy.
The patient in this case study, diagnosed with Sjogren’s syndrome, had been undergoing prolonged treatment involving high doses of corticosteroids and immune inhibitors, which contributed to the development of steroid-induced diabetes and hyperlipidemia. These factors are recognized as risk factors for mucormycosis. Twenty days prior to hospital admission, the patient experienced swelling of the nasal furuncle without an obvious cause, which gradually worsened and eventually ruptured. A paranasal sinus CT examination performed 7 days later did not show any significant abnormalities. Despite taking anti-inflammatory drugs at home, the treatment was ineffective, as the skin lesions continued to enlarge and eventually ruptured. The patient sought medical attention 20 days after symptom onset. Upon examination, visible black eschar necrosis was observed on the nasal and facial regions, along with hard palate necrosis, which had perforated into the nasal cavity and was accompanied by a putrid odor. MRI revealed erosion and loss of basal bone in the nasal region. Fast histopathological and fungal examinations confirmed the diagnosis of mucormycosis. Under general anesthesia, the patient underwent partial maxillectomy, sinus surgery, and facial soft tissue debridement. Given the patient’s history of renal damage due to Sjogren’s syndrome and the presence of electrolyte disturbances (hypokalemia), amphotericin B was not selected for treatment due to its nephrotoxic potential. Instead, voriconazole was administered for antifungal therapy. However, despite these interventions, the patient’s condition rapidly deteriorated, and they succumbed to a brain infection on May 20th, 2024.
Conclusion
In conclusion, ROCM is a rare and often fatal infection that predominantly affects immunocompromised individuals, particularly those on long-term immunosuppressive or corticosteroid therapy, as well as patients with underlying conditions such as diabetes and hyperlipidemia. Early clinical and imaging manifestations are often nonspecific, which can lead to misdiagnosis or delayed diagnosis. This case study underscores the importance of early detection and timely intervention in improving the chances of survival. By enhancing clinicians’ awareness of this condition, we aim to promote earlier diagnosis and more effective treatment, ultimately improving patient outcomes.
Footnotes
Acknowledgements
We would like to express our gratitude to the patient and his family for their cooperation and consent.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Consent for Publication
All authors approved the submitted version.
Consent to Participate
We obtained written informed consent from the patient’s guardian to publish this case report. The study was carried out in accordance with the principles of the Declaration of Helsinki.
