Abstract
Endolymphatic sac tumor (ELST) is a rare, aggressive hypervascular tumor of the posterior petrous bone. These tumors can manifest independently or in conjunction with Von Hippel–Lindau (VHL) syndrome. Currently, the primary treatment strategy involves complete surgical excision, though the role of radiotherapy remains debated.
We are reporting a case of a 35-year-old female patient affected by ELST associated with VHL syndrome, managed solely through stereotactic radiosurgery and monitored by time-resolved imaging of contrast kinetics magnetic resonance sequence.
Introduction
Endolymphatic sac tumor (ELST) is an uncommon and locally aggressive lesion, originating from the papillary epithelium of the endolymphatic sac and slowly growing into the postero-medial part of the petrous temporal bone. They are associated with Von Hippel–Lindau (VHL) disease in 11% to 30% of cases. Given that approximately 10% of VHL patients develop ELSTs, routine screening is advisable to detect these tumors early.1,2
Clinically, ELSTs typically present with progressive sensorineural hearing loss, and may also involve other cranial neuropathies depending on the extent of the lesion’s cerebral involvement.1,2
Diagnosis often employs digital subtraction angiography (DSA) to evaluate the hypervascular nature of these lesions, supported primarily by the external carotid arteries for tumors smaller than 3 cm (predominantly the ascending pharyngeal and the stylomastoid branches), and by both internal and external carotid arteries for larger lesions. 3
Time-resolved imaging of contrast kinetics magnetic resonance angiography (TRICKS-MRA) is a modern MR technique that improves temporal resolution compared to conventional contrast-enhanced MRA, providing images similar to DSA. Many authors highlighted the clinical significance of TRICKS-MRA in assessing lesion hemodynamic and treatment responses, with an excellent interobserver agreement when compared to DSA. 4
Surgical excision remains the preferred treatment for ELSTs, followed by long-term monitoring. However, complete removal of advanced tumors is challenging, and recurrence rates are high. While the use of radiotherapy is controversial, it may serve as an alternative for patients ineligible for surgery. 2
In this case report, we present an ELST in a 35-year-old female with VHL disease, treated with stereotactic radiosurgery (SRS) and evaluated with TRICKS-MRA over multiple follow-up examinations to assess treatment efficacy.
Case Report
A 35-year-old female patient affected by VHL disease presented to our hospital. She was suffering from flaccid paralysis resulting from previous bleeding of spinal hemangioblastomas 2 years before. Several infratentorial hemangioblastomas were also present. She referred to previous surgical treatments for renal cell carcinoma and retinal hemangioblastomas, 4 and 10 years before, respectively.
Recently the patient complained about dizziness and bilateral tinnitus; despite this, clinical examination showed normal audiometric tests.
The high-resolution CT study revealed a 17 × 13 mm, expansive, permeative mass lesion along the right retro-labyrinthine petrosal bone (Figure 1A-C). The lesion appeared as a locally aggressive tumor centered in the endolymphatic sac region. Calcifications were present both centrally and on the posterior edge. The mass minimally extended into the right cerebellopontine angle, sparing but skirting the internal auditory canal, the sigmoid sinus, the adjacent mastoid air cells, and the facial canal.
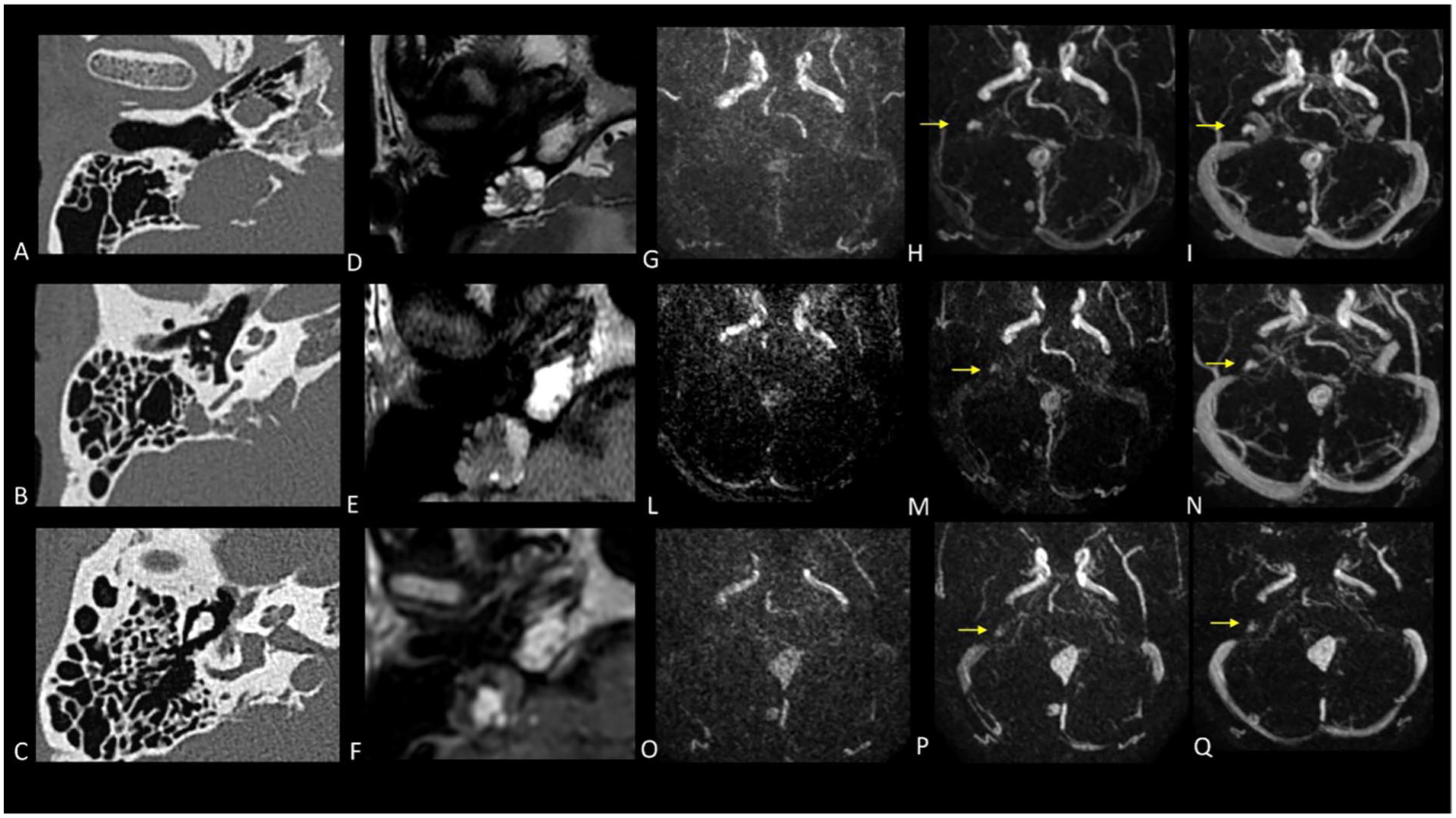
Multiparametric evaluation of radiotherapy effects CT head (A-C) demonstrated the presence of a lytic lesion of the right posterior cranial fossa, causing erosion of the right petrous bone where the vestibular aqueduct is present. There are no evident abnormalities in the semicircular canals or the vestibular-cochlear bony components. MRI examination revealed a lesion with heterogeneous hyperintense signal in T2-weighted images (D) and a mixture of iso-hyperintense signals in T1-weighted images (E), suggesting a cystic formation with hemorrhagic content. After gadolinium administration, early enhancement of the central solid component is evident (F), synchronous with the known multiple enlarging lesions located below the tentorium, as depicted in the dynamic angiographic study with TRICKS sequence (G-I). A follow-up MRI performed after 3 months (L-N) revealed a noticeable diminished enhancement during the initial acquisition phases of the dynamic contrastographic study compared to the prior examination, suggesting a potential post-radiation treatment effect. A subsequent MRI follow-up performed after 2 years (O-Q) showed a continued moderate decline in enhancement compared to previous examinations, supporting the hypothesis of probable post-treatment effects. TRICKS, time-resolved imaging of contrast kinetics; MRI, magnetic resonance imaging.
On magnetic resonance imaging (MRI), the lesion was characterized by a heterogeneous signal, predominantly hyperintense on T2-T2-weighted images and hypointense on T1-T1-weighted images with T1 hyperintensity foci within the lesion, suggestive of hemorrhage (Figure 1D and E). The solid component of the lesion was associated with intense enhancement after administration of gadolinium (Figure 1F). The MR examination revealed the presence of several infratentorial hemangioblastomas characterized by solid nodular enhancement after gadolinium with peripheric cystic lesions.
TRICKS-MRA was performed to assess retro-labyrinthine lesional vascularity. Three-dimensional TRICKS parameters were as follows: repetition time/echo time, 5.4/1.8 ms; flip angle, 45°; field of view, 28 cm; matrix, 320 × 192 pixels; slice thickness, 1.2 mm; and bandwidth, 41.67 MHz. A sagittal acquisition was performed to cover the entire lesion with a total of 15 dynamics within 2 min. A first phase of 3-dimensional data was obtained before injecting the contrast agent and then the scanning of subsequent phases began simultaneously with the contrast injection (bolus of a gadolinium-based agent at a rate of 3 ml/s, followed by a saline flush of at least 15 ml). The subtraction of the pre-contrast phase (mask) was performed automatically to maximally suppress signal from background tissues.
Thanks to this contrast-enhanced angio-MRI sequence an arterial enhancement of the solid component of the petrosal lesion was observed, which occurred nearly simultaneously with the enhancement seen in hemangioblastomas (Figure 1G-I).
Considering these features, the hypothesis of an ELST was made.
In our case, although complete resection is the standard of care for ELST, considering the high rate of recurrence, the risk of surgical complications, and the clinical absence of hearing loss, a single-fraction SRS (12 Gy) was preferred as the primary exclusive treatment. No new neurologic deficit was observed in the early post-radiosurgery period.
About 3 months after radiation therapy, MRI revealed substantial volumetric stability of the mass in the right petrous bone. Nevertheless, TRICKS-MRA showed a slower and lower vascularization of ELST compared to hemangioblastomas and pre-treatment levels, supporting the hypothesis of a partial treatment response (Figure 1L-N).
The patient was further monitored, first, every 3 months and later every 12 months, noting an additional progressive reduction of tumor vascularity concurrent with a late minimum down-sizing (14 × 11 mm vs 17 × 13 mm) at 2 years of MRI follow-up (Figure 1O-Q).
Discussion
The endolymphatic sac is a thin anatomic structure of the membranous labyrinth that lies along the posterior and medial petrous temporal bone, approximately midway between the internal auditory meatus and the sigmoid sinus. As mentioned before, ELST is an uncommon and locally aggressive lesion, originating from the papillary epithelium of the endolymphatic sac and slowly growing at first into the postero-medial part of the petrous temporal bone.
The endolymphatic sac tumor was first identified by Hassard et al during a sac decompression surgery in 1984, and this term was first used by Heffner in 1989.5,6 Since its first description in 1984, only approximately 200 cases have been reported in the literature. 7
This tumor can manifest independently or, in 11% to 30% of cases, in conjunction with VHL syndrome. VHL syndrome is a rare autosomal dominant disease (incidence about 1 in 36,000 live births) resulting from deletions or mutations in the VHL gene located on the short arm of chromosome 3. The normal VHL gene is a tumor suppressor gene, whose mutation leads to angiogenesis via hypoxia-inducible factor-1 (HIF-1) and to the various manifestations of the disease which may include unilateral or bilateral ELSTs, multiple hemangioblastomas (in the brain, spinal cord and retina), renal carcinoma, pheochromocytoma, pancreatic cysts, neuroendocrine tumors, and/or cystadenomas. 8
Genetic pathogenesis and molecular studies of sporadic and VHL disease-associated ELSTs reveal both a pivotal role of inherited and somatic VHL gene mutations. 9
Although the VHL gene regulates angiogenesis via HIF-1, both sporadic and VHL-associated ELSTs appear hypervascular despite normal HIF-1 levels. This is attributed to the retention of one functional VHL gene copy. Histologically, these types of neoplasms are papillary adenomatous tumors, occasionally accompanied by colloid-filled cysts with fronds lined by a single layer of cuboidal cells. 10 It is also reported that ELST typically stains positive for Cytokeratins, EMA, Vimentin, and Periodic acid Schiff and shows variable positivity for S100, NSE, Synaptophysin, Chromogranin A, and GFAP. 11
ELSTs behave as low-grade carcinomas. They have been reported as metastasizing in rare situations, but usually recur and invade adjacent structures such as the mastoid, internal and external auditory canals, middle ear, labyrinth, facial canal, cerebellopontine angle, the sigmoid sinus, and the bulb of the jugular vein. Consequently, the most common symptoms may include progressive hearing loss, vertigo, tinnitus, dizziness, ataxia, otalgia, otorrhea, and facial nerve paresis. In the late stage, fourth ventricular compression may lead to hydrocephalus and clinical progression may present as headache, vomiting, and pupillary dilatation. 12
To the best of our knowledge, no similar ELST cases that have been exclusively managed with SRS and monitored using TRICKS-MRA have been documented in the literature.
In general, primary SRS is commonly advised for patients who may not be suitable candidates for surgery or have been denied surgical treatment options. In a recent review, 3 patients with ELST who received primary SRS with a margin dose ranging from 15 to 18 Gy were assessed, and none of these patients showed signs of tumor progression. 10 In addition, regarding residual or recurrent ELSTs managed with single-fraction adjuvant SRS, 8 cases were identified, with the majority showing either tumor reduction or stability in clinical and MR follow-ups.13-16 Recently, also a case of complete regression after adjuvant gamma knife radiosurgery was reported. 16
In our patient, we experienced a similar SRS effect due to stable tumor dimensions and reduced vascularization as confirmed by TRICKS-MRA acquisitions. TRICKS-MRA sequences were useful in evaluating 2 hemodynamic conditions: the synchronous enhancement of ELST and hemangioblastomas at diagnosis, and the post-SRS treatment effects. The simultaneous enhancement of both ELST and hemangioblastomas might imply they share some histological features, however, there is no evidence in the current medical literature to justify this correlation. Indeed, in histological terms, hemangioblastomas are defined by neoplastic stromal cells surrounded by a dense network of delicate vessels.17,18 In contrast, ELSTs are characterized as papillary adenomatous tumors comprising cuboidal cells arranged in a papillo-tubular pattern, occasionally accompanied by colloid-filled cysts. 19
Despite these differences, ELST and hemangioblastomas showed similar hemodynamic behaviors at diagnosis. To our knowledge, to date, no comparative studies on the vascularization of ELST and hemangioblastomas in VHL syndrome are described in the literature.
Moreover, TRICKS-MRA acquisitions also provided a valid follow-up tool. In our case, post-treatment TRICKS-MRA detected a progressive reduction in the vascularization of the lesion along with a late minimum down-sizing. This study suggests that the combined use of TRICKS-MRA may be helpful for non-invasively evaluating ELST response to therapy and detecting residual tumors. Additional diagnostic tools to verify treatment efficacy are reported by Geerts B et al., suggesting that the pCASL sequence is reliable for detecting residual ELST post-surgery by assessing changes in cerebral blood flow within the lesion. 20
Conclusion
Our study demonstrates that TRICKS-MRA effectively identifies the hypervascularity of ELST and it can help evaluate treatment response as well as the reduction of lesion vascularization.
Footnotes
Acknowledgements
None.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Patient consent the patient signed an informed consent.
