Abstract
Clear cell sarcoma (CCS) of the parotid gland is a rare and aggressive cancer, predominantly affecting the deep soft tissues of the lower extremities, and is more frequently observed in the elderly. Its occurrence in the head and neck area, particularly in the parotid gland, is exceedingly rare. This study presents a 75-year-old male who exhibited a substantial, swiftly-enlarging mass on the left side of the mandible, which was diagnosed as CCS of the parotid gland. Morphologically and immunohistochemically, CCS shares characteristics with melanomas, and Ewing sarcoma, complicating differential diagnosis. The primary treatment for localized CCS typically involves surgical excision with clear margins. However, due to its resistance to conventional chemotherapy, there is ongoing research regarding the efficacy of adjuvant therapies such as chemoradiation, lymphadenectomy as well as immunotherapies such as immune checkpoint inhibitors. Further research is imperative to enhance our understanding of CCS and to develop more effective treatment strategies.
Introduction
Clear cell sarcoma (CCS) was initially described by Dr. Franz Enzinger in 1965. Regarded as malignant melanoma of soft parts, it is a rare aggressive tumor that accounts for less than 1% of all soft tissue sarcomas. 1 CCS has historically been termed “malignant melanoma of soft parts” because of its melanocytic differentiation and the challenge of distinguishing it from malignant melanoma using immunohistochemistry. 2 Affecting mainly adolescents and young adults, CCS is associated with local recurrences and late metastasis. CCS preferentially affects the lower extremities, 3 and its presence in the head and neck is rare. 4 CCS originating from the salivary glands are unusual, although they have been previously reported in the parotid gland. 5 Accurate diagnosis is vital for correctly classifying and treating CCS, particularly in atypical locations. In our study, we present a case of CCS arising from the parotid gland. The aim of the study was to increase the awareness of this rare presentation, also we examineed the clinicopathological characteristics of CCS in the head and neck cases that were previously reported in the literature.
Case Presentation
A 74-year-old male presented with a history of a stuffy nose and nasal discomfort for 3 months. Physical examination revealed an irregular mucosal nodule on the left nasal septum, just above the left nostril. A computed tomography scan (CT scan) showed mucosal irregularity at the left aspect of the nasal septum close to the left nostril, with no distant metastasis. Excisional biopsy was performed, and histopathology revealed a high-grade spindle cell tumor with brisk mitotic activity.
The patient was recommended to start radiotherapy along with chemotherapy; however, he refused systemic chemotherapy and was given radiotherapy alone. He received 60 gray in 30 fractions of radiotherapy.
During follow-up, the patient complained of an enlarging lump on the left side of his face. A non-tender, firm, and rounded neck mass measuring 2 * 2 cm was noted upon examination. A PET-CT scan showed a hypermetabolic left lower parotid nodule. Additionally, a hypermetabolic lung nodule was observed (Figure 1).
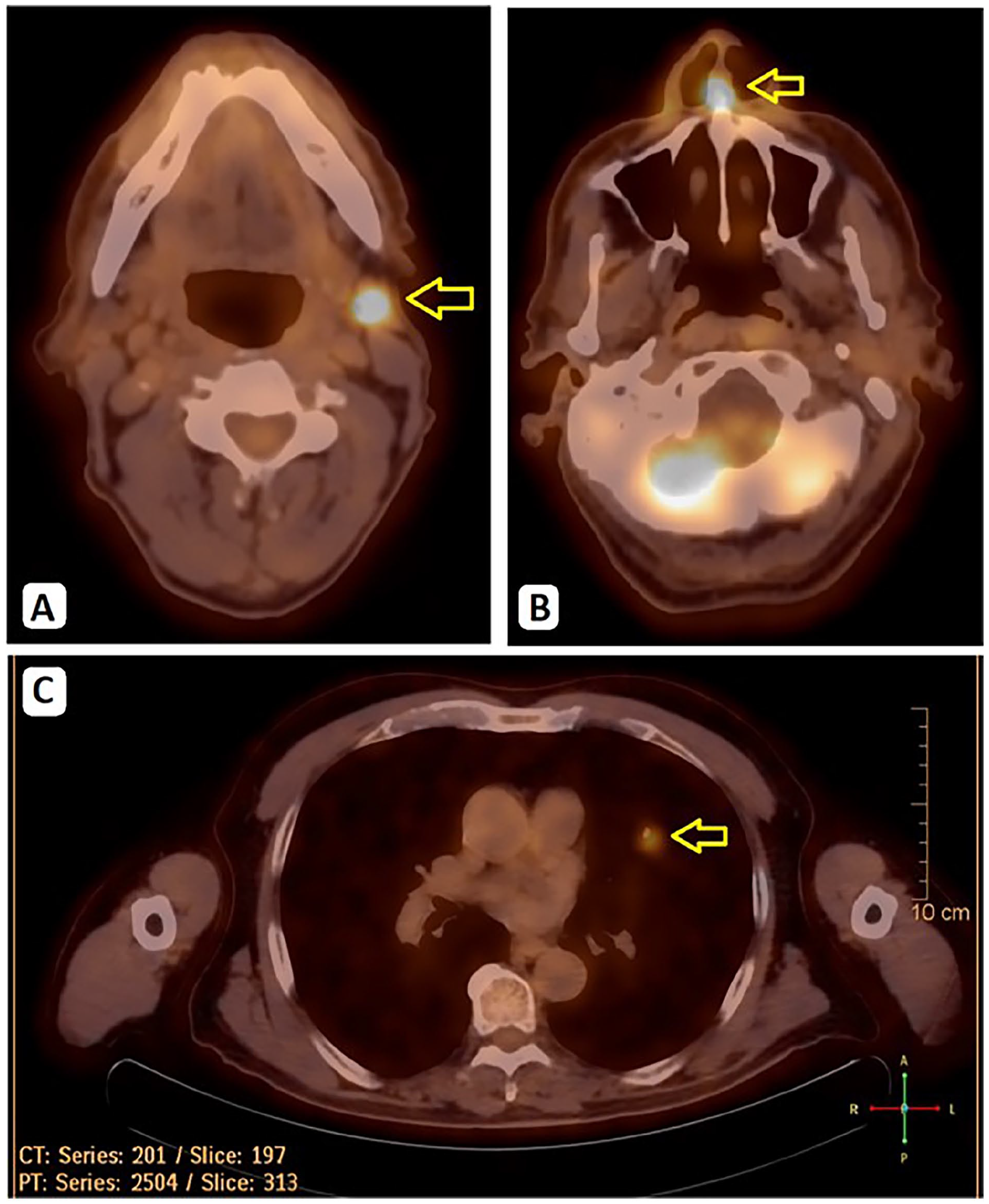
PET SCAN. (A) FDG-positive left lower parotid nodule measuring 1 cm with SUVmax 8.1. (B) Minimal FDG uptake in the left aspect of the nasal septum, and (C) A hypermetabolic lung nodule was observed.
The patient underwent an ultrasound of the left parotid gland, followed by fine needle aspiration, which revealed cellular clusters of atypical cells with prominent mitosis. An excisional biopsy was performed. Histopathologic examination showed proliferation of monotonous epithelioid and scattered spindle cells with clear-to-eosinophilic cytoplasm and prominent nucleoli infiltrating the parotid gland, and immunohistochemical analysis of the tumor cells was positive for SOX10, MelanA, and HMB45, with patchy immunoreactivity for S100 (Figure 2). In contrast, CK-pan, CK7, CK5/6, P63, and Desmin were negative. The histopathologic findings were consistent with CCS or melanoma of soft tissue. Molecular profiling indicated wild-type BRAF, intact MSI, and negative PDL-1. Fluorescence in situ hybridization (FISH) revealed positive EWSR1-ATF1 rearrangement. Based on these findings, a diagnosis of CCS was made.
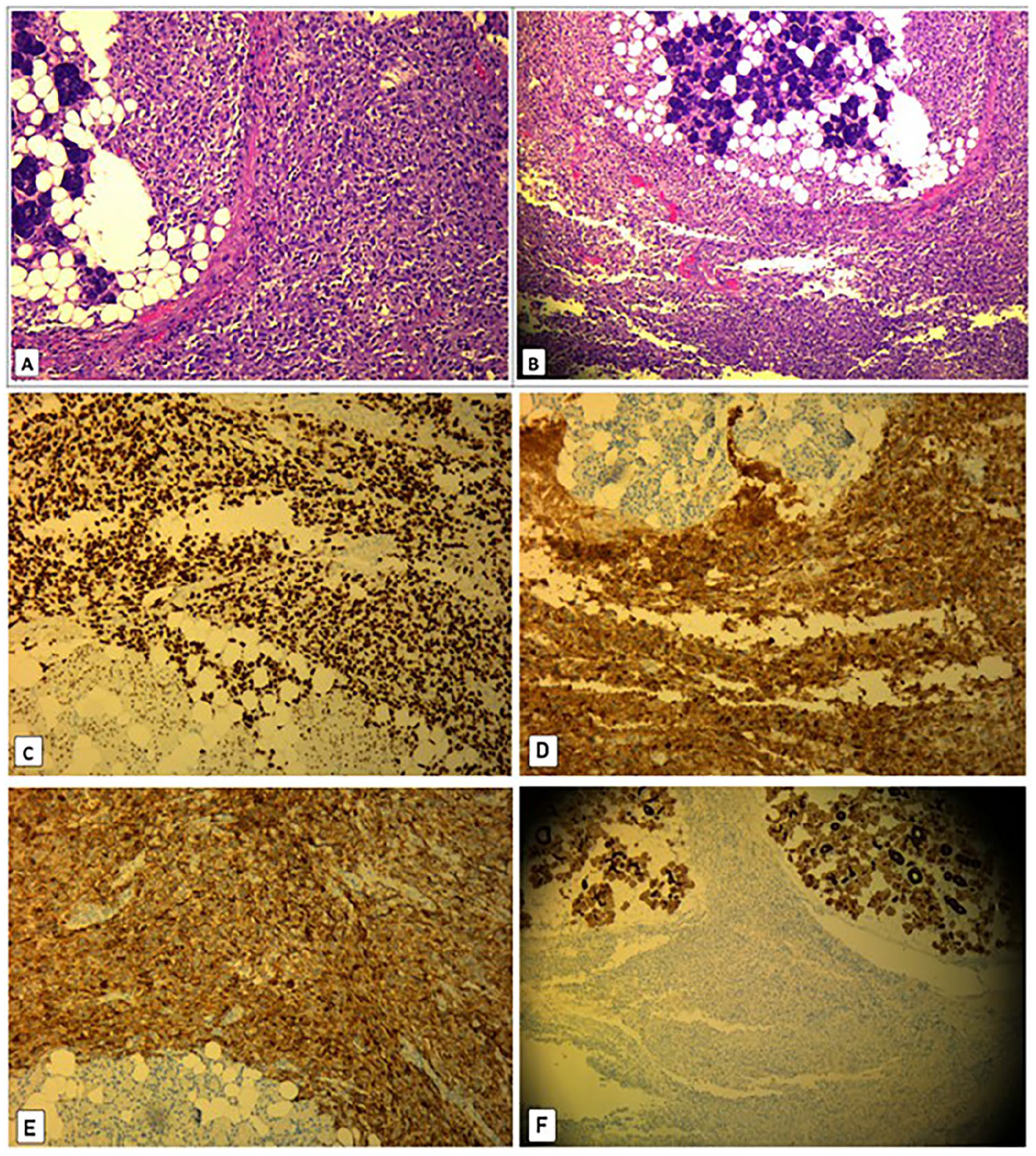
Histopathologic examination showed proliferation of monotonous epithelioid and scattered spindle cells with clear-to-eosinophilic cytoplasm and prominent nucleoli infiltrating the parotid gland. (A) High magnification 10×, (B) low magnification 4×. Histological findings of clear cell sarcoma. (C) SOX 10 immunostaining, (D) MELAN A immunostaining, (E) HMB 45 immunostaining, and (F) CK-pan immunostaining, all were positive in neoplastic cells.
As the mass grew rapidly, a new clinical assessment for the patient was made, and on physical examination, a non-tender, firm mass at the left angle of the mandible was found to be increased in size up to 3 * 4.5 cm (Figure 3). A CT scan was performed, which showed left parotid gland mass, and mucosal irregularity at the left aspect of the nasal septum same in size due to postoperative changes, and bilateral multiple metastatic pulmonary nodules, the largest measuring about 3.5 cm (Figure 3). The multidisciplinary team decided to start the patient on Pazopanib (VOTRIENT 200 mg) tyrosine kinase inhibitor (four 200 mg tablets) 800 mg orally once daily and be on a regular follow-up in the oncology clinic.
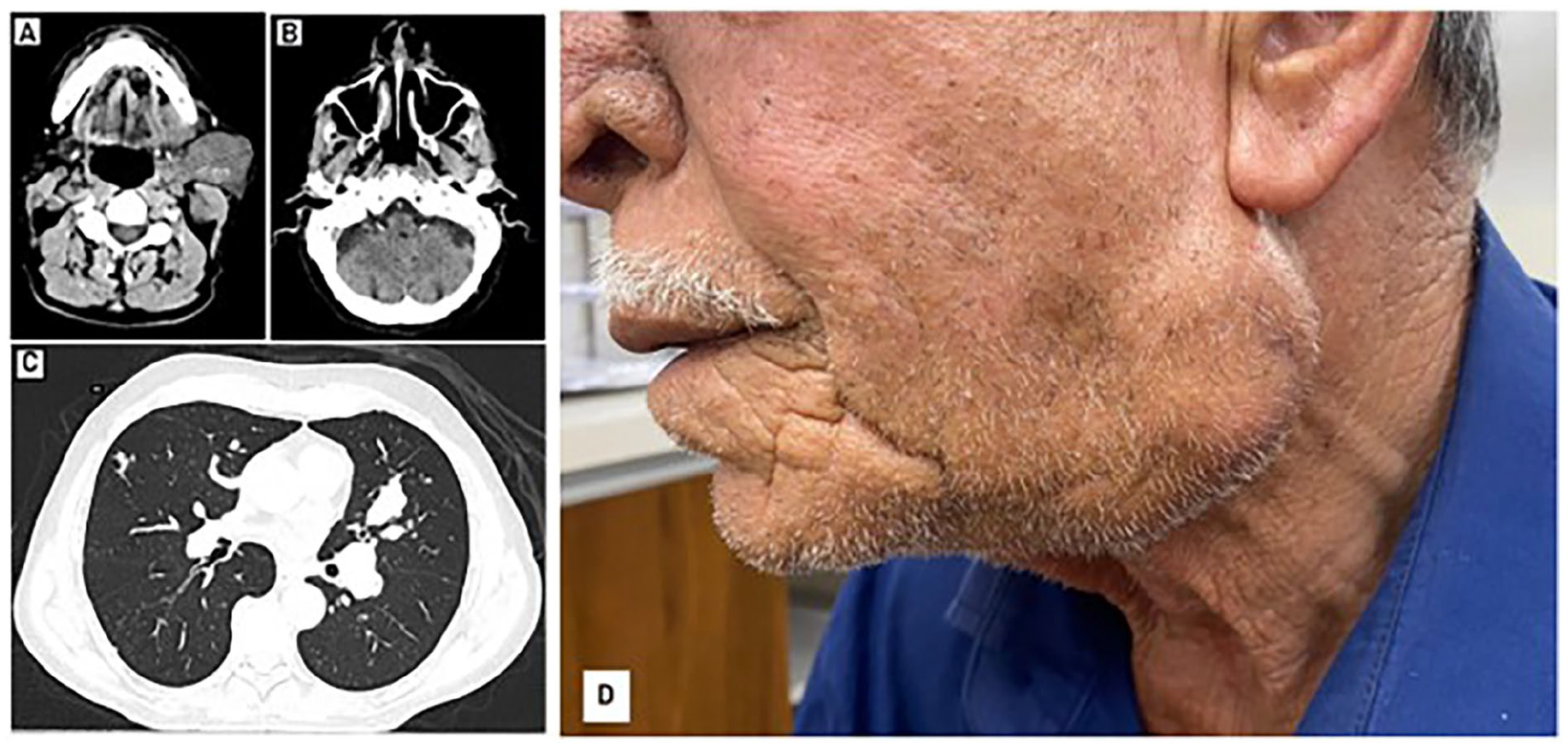
After 1 month follow-up, CT scan showed (A) left parotid gland measuring about 4.5 cm and a few pathological looking left cervical lymph nodes up to 2 cm in short axis. (B) Mucosal irregularity/ulceration is noted at the left aspect of the nasal septum close to the left nostril, stable in comparison with the previous examination. (C) A new bilateral multiple metastatic pulmonary nodules are noted, the largest measuring about 3.5 cm. (D) A non-tender, firm, and rounded neck mass measuring 3 cm × 4.5 cm at the angle of the left mandible.
Discussion
CCS of the parotid gland is an exceptionally-rare and aggressive malignancy, characterized by diagnostic challenges due to its histopathologic resemblance to malignant melanoma. 6 It typically occurs in young adults between 20 and 40 years old and is slightly more common in women than men. However, cases have been documented in both young children and elderly adults. 7 In the context of approximately 500 documented cases of CCS, a mere 1.2% have manifested in the head or neck regions, within this subset, the parotid area was affected in a scant 3 documented cases. 8 Characteristically, CCS exhibits a gradual and asymptomatic progression, typically identified when the growth is less than 5 cm in diameter. CCS shows little to no signs or symptoms in its primary stages. However, in its later stages, it can become aggressive and metastasize to the lymph nodes, lungs, or bone. 3
While CCS and melanoma share phenotypic similarities, they are distinct entities at the molecular level. EWSR1 rearrangements are a hallmark of CCS, most often presenting as EWSR1-ATF1 resulting from t (12; 22) (q13; q12) or, less frequently, as EWSR1-CREB1, and the detection of such rearrangements has been identified using FISH. 9 These translocations have not been observed in melanoma, and the absence of BRAF gene mutations readily differentiates CCS from cutaneous melanoma. 5 CCS frequently recurs locally and metastasizes later, often targeting regional lymph nodes, which is atypical for sarcomas. CCS strongly tends to metastasize via the lymphatic system, with lymph node metastases present in 6% to 23% of patients at diagnosis. 10 Local recurrence rates can reach up to 84%, late metastases can occur in up to 63% of cases, and metastases at the time of presentation can be found in up to 30% of patients. 11 Five and 10 year survival rates are approximately 47% and 36%, respectively. Larger tumors over 5 cm or those showing necrosis have poorer prognoses. 1 The most effective treatment approach currently includes comprehensive surgical excision followed by radiotherapy. Nevertheless, the limited number of cases makes the benefits of neck dissection and systemic adjuvant therapies unclear. Given the high risk of local recurrence and distant metastasis often years post-diagnosis, rigorous and ongoing monitoring is advised. 12 Targeted treatments such as sunitinib and MET inhibitors could yield promising outcomes. Immunotherapy, especially with immune checkpoint inhibitors like nivolumab, is expected to be a promising treatment for CCS and has proven effective in malignant melanoma. 13 Early diagnosis and complete surgical removal are critical for better outcomes, and further research into targeted therapies and immunotherapies is needed.
Several case reports have highlighted the clinical presentation, diagnostic process, and treatment outcomes of CCS in this region. Poignonec et al. 14 reported the first case involving the parotid region, which was treated by complete parotidectomy while ensuring the facial nerve was preserved. Manoel et al. 8 described a case in the parotid region, initially misdiagnosed as malignant melanoma, and later confirmed postoperatively through molecular testing, 8 months following the surgery, the neck tumor reoccurred, leading to the patient undergoing a radical neck dissection along with adjuvant radiotherapy. These reports emphasize the crucial role of molecular diagnostics in identifying CCS and the aggressive nature of the tumor, requiring comprehensive treatment (Table 1).
Conclusion of Clear Cell Sarcoma Cases in the Head and Neck Region.
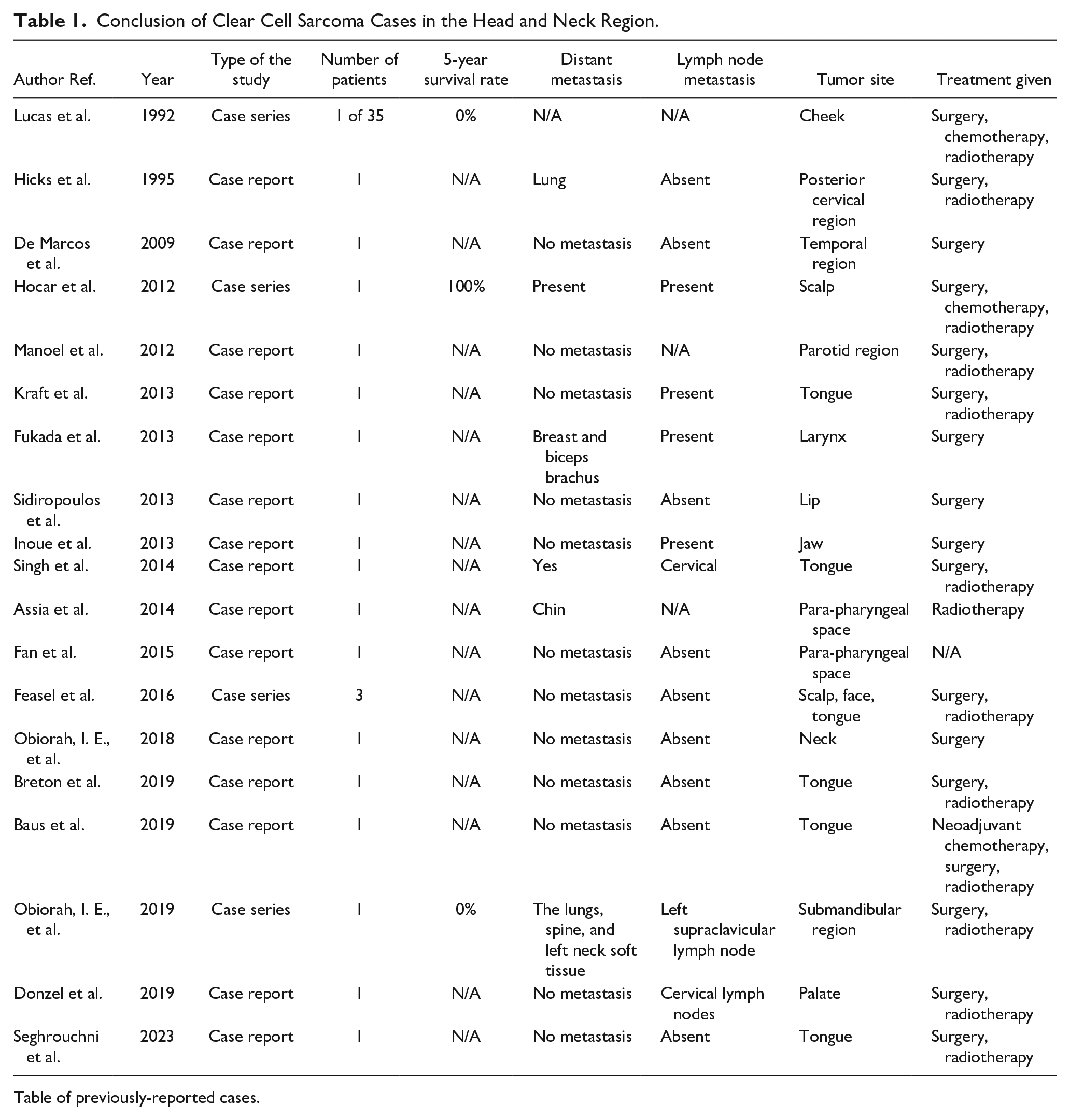
Table of previously-reported cases.
Conclusion
In conclusion, we report a scarce head and neck CCS focusing on the parotid gland, and even though they are rare, CCS may involve the head and the neck and are frequently mistaken for malignant melanomas. Developing novel therapeutic approaches, such as ICI and molecular targeted therapies, is a promising step toward improving the survival of patients with CCS. Molecular analysis is recommended for a conclusive diagnosis independent of the tumor’s location. This approach underscores the importance of comprehensive diagnostic strategies in evaluating rare tumors.
Footnotes
Acknowledgements
The authors are grateful to the doctors in the radiology and pathology departments at Augusta Victoria Hospital for their assistance in collecting patient data.
Data Availability Statement
The data used to support the findings of this study are included in the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Ethics approval to report this case was obtained from the Research Ethics Committee at Al Quds University.
Patient Consent Statement
The patients gave written informed consent for the publication of this study. The editor-in-chief of this journal can review a copy of the written consent upon request.
