Abstract
Introduction
Laryngopharyngeal reflux (LPR), also known as supra-esophageal reflux, is a general term for extra-esophageal symptoms and signs caused by gastroesophageal reflux disease (GERD).1,2 In the past, laryngopharyngeal reflux disease (LPRD) was often treated as the manifestation of GERD in the throat, but an increasing number of studies have shown that LPRD and GERD are significantly different in the reflux mechanism, clinical manifestations, diagnosis, and treatment and that LPRD should be regarded as an independent disease in otolaryngology-head and neck surgery (OHNS) clinics.3-5 GERD is the result of decreased lower esophageal sphincter tonicity, that is, lower esophageal sphincter incompetence, while patients with LPRD have a normal lower esophageal sphincter function but with upper esophageal sphincter incompetence, also with fewer signs of heartburn but more reflux symptoms. 6
Studies have shown that LPRD is associated with a variety of respiratory symptoms and diseases, such as sore throat, foreign body sensation, hoarseness, throat clearing, dysphagia, chronic cough, and postnasal drip.7-9 Many reports on the correlation between LPRD and benign and malignant diseases of the throat have emerged in recent years. LPRD may be involved in the pathological process of vocal cord leukoplakia, as evidenced by the immunohistochemical detection of pepsin expression in leukoplakia. 10 Parsel et al. 11 noted in their meta-analysis that LPRD was significantly associated with vocal cord malignancies. Due to the laryngopharyngeal symptoms caused by LPRD, the diagnosis of LPRD in OHNS clinics is increasing in recent years, but there is still a lack of research on the incidence, clinical characteristics, and diagnosis and treatment of patients with LPRD in OHNS clinics. 12
There is no single way to diagnose LPRD. The diagnosis of LPRD depends on the score of symptoms and signs, the efficacy of the proton pump inhibitor (PPI) experimental treatment, and the objective examination results. 13 Belafsky et al. 14 introduced the Reflux Symptom Index (RSI) scale in 2002 as a clinical tool to evaluate symptomatic improvement after treatment in patients with LPRD, and he concluded that RSI > 13 as abnormal with increased incidence of LPRD. Li et al 15 validated the Chinese version of RSI and confirmed its good reliability and validity. Subsequently, the RSI scale has been used in a number of studies, including the posttreatment monitoring of LPRD and the clinical diagnosis of LPRD in different patient populations. In addition, the RSI scale combined with the reflux finding score (RFS) is also widely used in the diagnosis of LPRD. 16 Most reports indicate that RSI > 13 points or RFS >7 points are thresholds for suspected LPRD.14,17 While most studies on LPRD in China focus on incidence, symptoms, and risk factors, only some limited reports are available for prevalence. There is a limitation of follow-up investigations, and feasibility studies of RSI as an alternative diagnostic tool for LPRD in OHNS clinics.
In the present study, RSI as an LPRD diagnostic tool has been used to analyze the overall incidence, clinical characteristics, and diagnostic status of LPRD in OHNS clinics. Follow-up observation was carried out for 6 months in patients with suspected LPRD (RSI > 13), and the questionnaire based on RSI was completed again after treatment to analyze the treatment status of LPRD. The aim of the present study was to prove the RSI tool as a reliable diagnostic tool for LPRD in OHNS clinics.
Materials and Methods
Participants and Sampling Method
Using centrally-located systematic sampling, from the total number of 50 (N) patients treated by each physician in the OHNS clinic of the Affiliated Hospital of Southwest Medical University, 5 patients were selected, the sampling interval was k (N/n), and patients with treatment serial numbers ending in k/2, k/2+k, k/2+2k, k/2+3k, . . ., k/2 +(n-1) k were selected successively, that is, 5,15,25,35,45. The survey time was the second week of each month, from Monday to Friday. If the survey time falls on the Spring Festival and National Day holidays, the time was changed to the first week after holiday.
The inclusion criteria were as follows: (1) age ≥ 18 years; (2) acceptance to participate in the survey, a sufficient understanding of the content of the questionnaire, and a good cooperation; (3) no serious medical conditions, especially irritable bowel syndrome. If the selected patients did not fulfill the inclusion requirements, they were excluded from the study.
Data Collection and Definitions
The data for this study were collected using the RSI questionnaire. The content of the questionnaire covered 9 symptoms listed on the Chinese Version of the RSI scale (Table 1). RSI questionnaire items were scored using a range from 0 (no symptoms) to 5 (very severe), and the total score ranged from 0 to 45. General information (eg, sex, age, previous diagnosis of LPRD, previous use of acid-suppressing and gastrointestinal motility drugs, smoking history, and alcohol consumption history) of the respondents was also collected. The initial RSI > 13 patients were followed up for 6 months, and anti-reflux therapy history and the RSI score after treatment were collected as well. Age groupings were determined using the current Chinese standard: young age group (18-40 years); middle age group (41-65 years); old age group (>65 years). Smoking history was defined as continuous daily smoking (unlimited amount), and alcohol consumption history was defined as daily-alcohol consumption (unlimited amount). In this study, suspected LPRD was defined as positive when RSI > 13.
Reflux Symptom Index Questionnaire.

Statistical Methods
Data were analyzed using the IBM SPSS software, version 25 (SPSS Inc, Chicago, IL, USA). Chi-square, odds ratios (ORs), and independent
Results
Basic Characteristics of Participants
In this study, 1405 patients were included, while in 1187 patients, valid data could be gathered, with a nonresponse rate of 15.52%. Among the responders 679 patients (57.20%) were female and 508 patients (42.80%) were male; the ages ranged from 18 to 100 years, with an average age of 41.4 ± 13.8 years. A total of 169 patients (14.24%) were smokers, 135 patients (11.37%) consumed alcohol regularly. Among the 1187 patients, 94 participants showed RSI > 13 while the LPRD-positive rate was 7.92%. There were significant differences in the LPRD-positive (RSI > 13) rate between patients with smoking and without a smoking history, and smoking significantly increases the LPRD-positive rate (
Demographic Characteristics of the Participants [n (%)].

Abbreviations: LPRD, laryngopharyngeal reflux disease; RSI, Reflux Symptom Index.
Clinical Risk Factors Associated With LPRD
Logistic regression analysis was used to investigate the association between the variables shown in Table 2 and LPRD. The results indicated that smoking significantly increased the risk of LPRD (OR: 2.140; 95%CI: 1.058-4.331;
Multivariate Logistic Regression Analysis of Association Factors for Patients With LPRD.

Abbreviation: LPRD, laryngopharyngeal reflux disease.
Characteristics of the 9 Symptoms Scores Distribution Listed on RSI
Among the symptoms listed on the RSI scale evaluated in all patients, “foreign body sensation in the throat” (symptom 8) had the highest incidence (58.30%), while “coughing after meals or lying down” (symptom 5) had the lowest incidence (20.64%). In patients with LPRD positive, the incidence of “frequent throat clearing” (symptom 2) was the highest (94.68%). There were significant differences in the distribution of scores for the 9 symptoms listed in RSI between patients with LPRD positive and LPRD negative (all
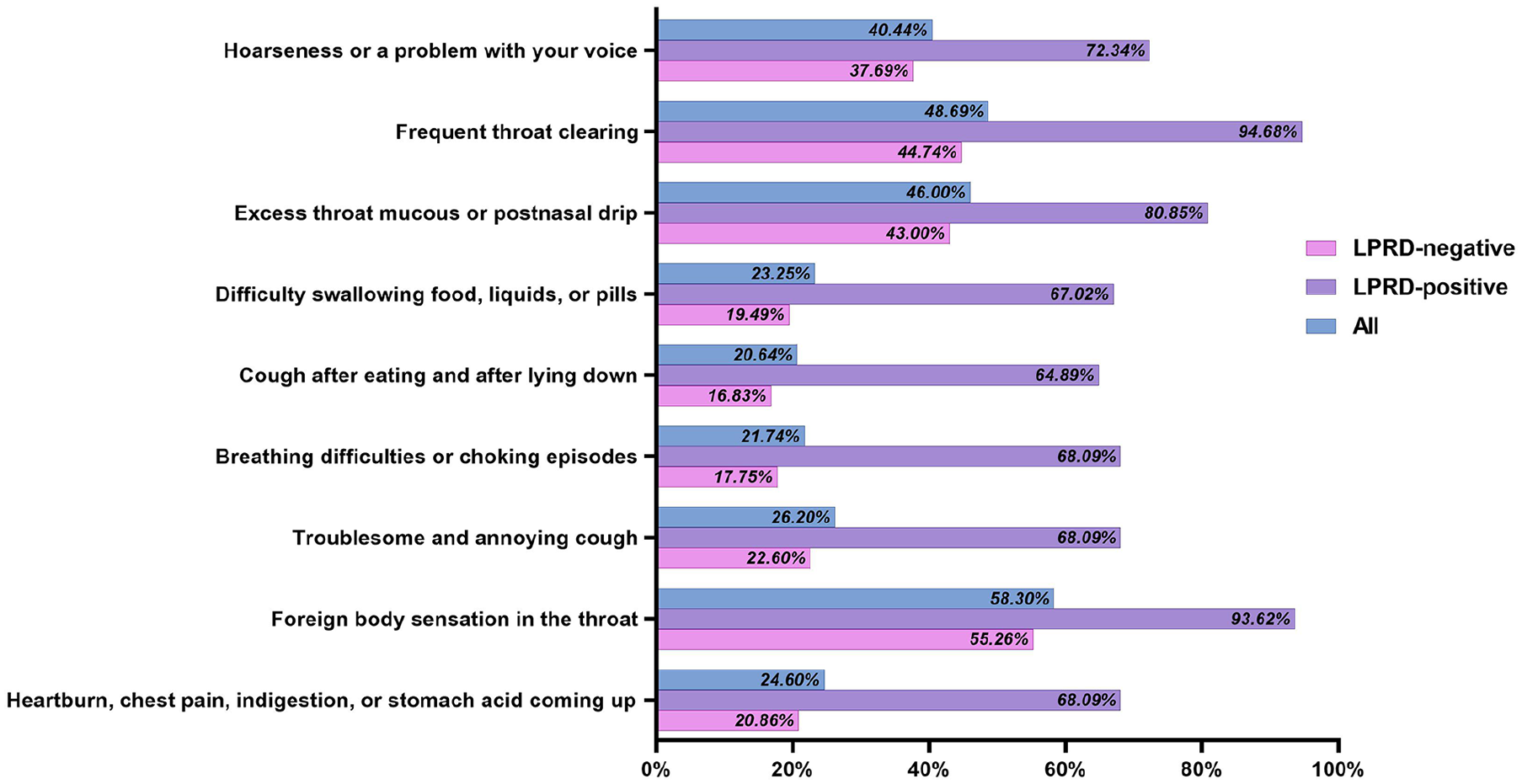
Distribution and scores of 9 symptoms in the RSI of the participants, and LPRD positive is defined as RSI > 13.
Correlation Analysis Between the RSI and Each Symptoms Score
Spearman correlation analysis indicated that in all patients, symptom 8 score (
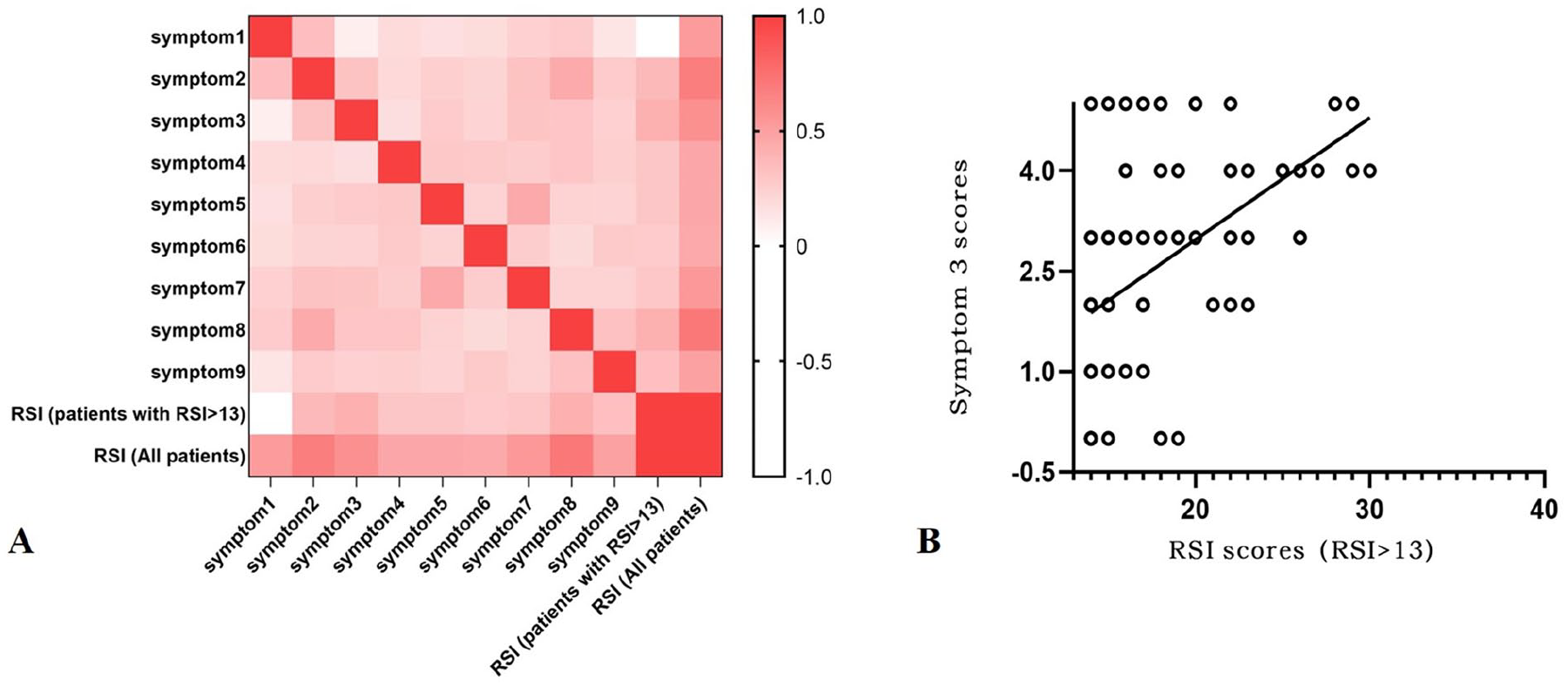
(A) Spearman correlation analysis was used to determine the significance of the correlation between the RSI and each symptom scores, and among each symptoms. (B) Showing the scatter of RSI scores and symptom 3 (excess mucus in the throat or postnasal drip) scores in patients with LPRD positive, and LPRD positive is defined as RSI > 13.
The Sensitivity and Specificity of RSI in the Diagnosis of LPRD in OHNS Clinics
Among the 94 suspected patients with LPRD positive, there were 65 patients subsequently with anti-reflux therapy, and the treatment rate was 69.14% (65/94); 56 patients out of 65 had good respond to anti-reflux therapy, and the RSI score was found significantly decreased compared with pretherapeutic score (
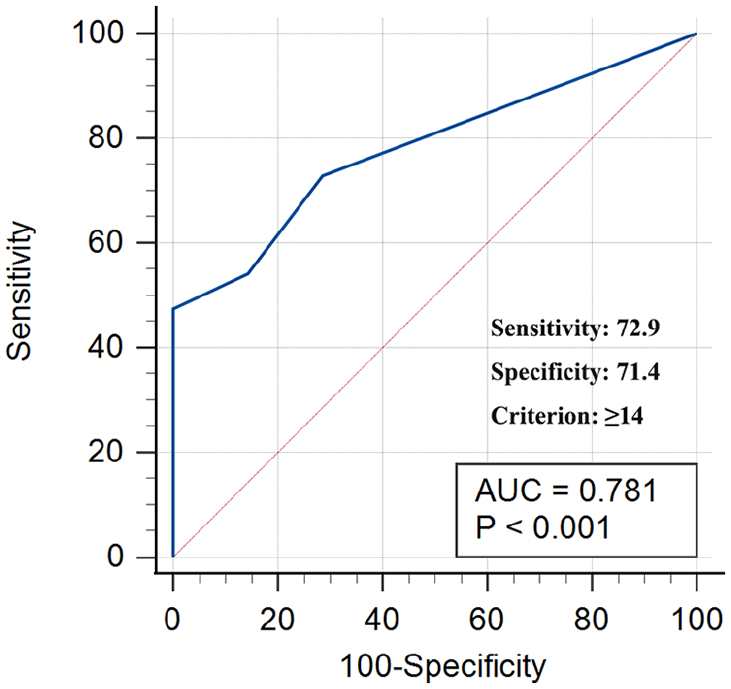
ROC fitting curve showing the sensitivity and specificity of RSI > 13 for the diagnosis of LPRD.
Discussion
Due to the lack of a gold standard for the diagnosis of LPRD, the objective examination is 24 hour multichannel intraluminal impedance-pH (MII-pH) monitoring, which is an invasive procedure and not convenient for large samples screening. Most otolaryngologist prefer to use PPI experimental treatment in patients with suspected LPRD. 19 PPI experimental treatment has high sensitivity and is suitable as a primary screening test for clinical suspicion of LPRD. In the present study, 94 patients with an initial RSI > 13 were followed up for 6 months, of which 65 patients were treated with PPI for 8 to 12 weeks. Fifty-six out of sixty-five showed a positive PPI experimental treatment, with all the RSI scores reduced more than 6. We found when RSI ≥ 14 (ie, RSI > 13), the specificity and sensitivity of RSI for the diagnosing LPRD is 71.4% and 72.9%, respectively. This study suggests that RSI can be used as a reliable tool for screening LPRD in OHNS clinics due to its noninvasive and easy to application, as well as used as a larger sample epidemiological investigation tool.
The results of this study suggested that the overall incidence of LPRD in OHNS clinics was 7.94% according to the RSI questionnaire. Most patients did not have any diagnosis and treatment history. Our results show that there is a high incidence of LPRD in OHNS clinics but a relatively-low rate of specific diagnosis and standardized treatment. Currently, most Chinese otolaryngologists have heard of LPRD, but according to a recent national survey, the deep understanding of risk factors, main clinical symptoms, diagnostic methods, and treatment of LPRD is not present. 20 This situation requires an improved clinical orientation of otorhinolaryngologists in understanding LPRD and to facilitate a better management of this disease. Compared with the results of the national LPRD epidemiological survey during the same period time, 12 the prevalence of LPRD in this study was low, suggesting that there are geographical differences in the prevalence of LPRD. Studies have shown that dietary habits, such as high-fat diets, high-sugar diets, spicy diets, and chocolate and caffeine intake, can exacerbate gastric acid reflux;21,22 therefore, adding items such as dietary habits to the questionnaire when collecting personal histories would be advisable.
Regarding to the risk factors, the LPRD-positive (RSI > 13) rate among patients who smoked and consumed alcohol was significantly higher than that among patients who did not smoke and did not consume alcohol. In addition, the present study results suggested smoking increases the risk of LPRD. Studies by Spantideas et al, 23 Lin et al, 24 and Xiao et al 12 support smoking as a risk factor of LPRD, consistent with the present study, although some reports show conflicting results. Kamani et al 25 reported there is no significant association between smoking and alcohol consumption with LPRD. Studies have shown that alcohol consumption promotes gastric acid secretion, strengthens gastric peristalsis and esophageal contractions, and aggravates throat reflux; additionally, smoking can lead to decreased esophageal sphincter tonicity and salivary secretion.26,27 Anyway, the current recommendations for the conservative treatment of LPRD include lifestyle habit changes. 28 So, patients with LPRD should avoid or minimize smoking and consuming alcohol. A few studies in China have pointed out that the LPRD-positive rate among middle-aged male patients is high; maybe, in this age group they are the backbone of society and families and are more likely affected by factors such as the pressure of life, mental pressure, smoking and drinking; therefore, the incidence of LPR increases.29,30 However, in the present study we found no relationships between the RSI score and age and sex, reflecting the general applicability of the RSI scale for patients of different ages and sexes. Further research is needed to determine whether social psychological factors will exacerbate LPR.
In this study, we found the incidences of “foreign body sensation in the throat” (symptom 8) and “frequent throat clearing” (symptom 2) were the highest among OHNS outpatients. If these 2 symptoms appear concurrently and no obvious organic lesions are found in an examination, LPRD should be suspected, and if necessary, RSI should be completed and combined with other LPR tests, such as the RFS and 24-hour MII-pH monitoring, to reduce the misdiagnosis rate of LPRD.31,32 Notably, this study analyzed the correlation between the total RSI score and each symptom score in patients with LPRD positive, suggesting that “excess mucus in the throat or postnasal drip” (symptom 3) had the highest correlation, followed by “foreign body sensation in the throat” (symptom 8). Studies have shown that the reflux of pepsin, bile salts, and other gastrointestinal proteins into the upper aerodigestive tract mucosa leads to mucosal modifications, including mucosal injury, inflammation reaction, mucus dryness, epithelium thickening, and microtrauma. 33 Accumulation of sticky mucus induces postnasal drip and globus sensation, throat clearing, and cough. And indirect injury from vagally-mediated reflexes may also favor the development of LPR symptoms, as the stimulation of gastroduodenal content into the low esophagus may stimulate the mucosa chemoreceptors, leading to excess mucus in the throat or postnasal drip. 34
This study has several limitations. To facilitate a large-scale epidemiological survey, this study only used RSI to define LPRD; there are no invasive tests and procedures such as pH monitoring and endoscopic scoring, for example, RFS used in the investigation. The RSI scale is not an objective diagnostic tool of LPRD, and its accuracy can be easily affected by subjective factors of the implementers, which is a limitation of our study. Using different diagnostic methods, such as pH probe monitoring or RFS, rather than a single diagnostic tool, may lead to differences in some indicators and ultimately affect the LPRD-positive rate; however, some studies have reported the positive correlation of RSI with the results of the PH value monitoring and dynamic multi-probe esophageal impedance.35,36 The main limitation of RFS tool is due to the apparent subjectivity of otolaryngologists in performing RFS scores based on laryngoscope findings. Chang et al 37 concluded that the use of RFS by general otolaryngologists in the diagnosis of LPRD is not reliable as the otolaryngologists of different subspecialties had different rates of laryngeal features detection. Another limitation of this study is that we only revaluated the RSI score of the patients who treated with PPI. PPI is the first choice for the experiment treatment of LPRD. A meta-analysis of randomized controlled trials reported patients with LPRD who received a PPI treatment had significantly-higher response rates and improved RSI scores than those who received placebo. 38 Furthermore, other studies have found that PPI was given twice daily for 3 months, and the response rate of patients with LPRD reached 41% to 100%.39,40
A recent study found that pH probe monitoring of the esophagus did not predict improvements in RSI scores after PPI therapy, 41 and another study used pharyngeal impedance monitoring and found that RFS scores did not correlate well with proximal esophageal acid exposure and that, interestingly, RSI scores were better correlated. 35 Although some studies have found that the RSI score is well correlated with MII-pH monitoring results, 35 the symptoms included in RSI lack specificity and are affected by subjective factors, such as regional differences and education levels of patients, 42 resulting in differences in LPRD-positive rates.
The diversity of LPRD clinical features and symptoms complicates the diagnosis and treatment. RSI is a simpler and reliable method, which can be used by otolaryngologists as an initial screening and diagnostic tool for LPRD in OHNS clinics in China. In future research, diagnostic tools that are more in line with national conditions should be designed and validated. For example, the Polish-designed Warsaw A-E scale has been confirmed to have a better value than other scales in diagnosing LPRD in Poland, 43 and the self-administered patient-reported outcome questionnaire reflux symptom score. 44 In addition, LPRD also present in children, and there is an urgent need to develop effective, reliable, and noninvasive methods for the clinical diagnosis of LPRD in children. It is expected that RSI could be improved as an adjunct to the screening and diagnosis of LPRD in children as well.
Footnotes
Acknowledgements
None.
Authors’ Note
Yilin Bao designated as the point of contact responsible for all communication with the journal about the manuscript and article.
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Southwest Medical University Fund Project (Grant No. 2017-ZRQN-003) and Doctoral Research Initiation Fund of Affiliated Hospital of Southwest Medical University.
Ethical Considerations
This study was approved by the Ethics Committee of the Affiliated Hospital of Southwest Medical University. Consent for study participation was obtained from patients recruited to participate in the study (project number: ky2017004).
Consent for Publication
Not applicable.
