Abstract
Objective
The role of lifestyle habits in patients with laryngopharyngeal reflux disease (LPRD) is comparatively underexplored. We aim to examine the specific lifestyle habits in patients with LPRD.
METHODS
Systematic sampling was applied to select respondents aged 18 through 80 years in otorhinolaryngology-head and neck surgery (OHNS) clinics in Nan Fang Hospital during August 2017–July 2018, 1658 eligible participants were included by a systematic sampling method. Subjects with RSI score>13 were considered as LPRD patients. The risk of reflux symptoms was estimated and multivariate calculated as odds ratios in relation to exposure to tobacco smoking, alcohol, coffee, tea, carbonated drinks, chocolate, spicy food, night sleep time, dinner-to-bed time, subjective sleep quality, and physical exercise.
Results
There was a significant dose-response association between carbonated beverage and LPRD. Among people who had drinking carbonated drinks the odds ratio was 1.76 (OR 1.77, 95% CI 1.24–2.50, P = .002) compared with non-carbonated drinker. A similar positive association was found for poor subjective sleep quality and shorter night sleeping time, the odds ratio for reflux was 1.58 (95% CI 1.14 to 2.18) among those who always have poor subjective sleep quality compared with those whose have good subjective sleep quality. The odds ratio for reflux was 2.29 (95% CI 1.23–4.28, P = .015) among those who always sleep 3–5 hours every night compared with those who sleep more than 8 hours every night. Beyond that, we found high BMI may have a negative correlation with LPRD, the odds ratio for reflux was .61 (95% CI 0.39 to .95, P = .054) among those whose BMI >25 kg/m2 compared with those BMI ≤ 20 kg/m2.
Conclusions
Patients with LPRD may have certain lifestyle habits, avoid carbonated beverage, poor subjective sleep quality, and lack of sleep should be advised in treatment of LPRD.
Keywords
Introduction
It is generally assumed that certain behavioral or dietary habits, such as alcohol consumption or smoking, induce LPRD. 1 Lifestyle modifications have been a first-line therapy for patients with LPRD. According to the treatment used in the UK District General Hospital, lifestyle behavior modification is considered to be very effective for the management of LPRD. 2 Much of the lifestyle modification suggestions came from guidelines for gastroesophageal reflux disease (GERD) because of a scarcity of published evidence on lifestyle habits of patients who suffer from LPRD. Although LPRD and GERD both involve retrograde reflux of the gastric contents, the two disorders have been found to have different pathophysiologies, symptomatology, and patient characteristics. 3 Lifestyle modification suggestions from the guidelines of GERD may not be suitable for LPRD patients, and lifestyle-related risk factors in patients with LPRD are largely unexplored. The need to cover this gap of knowledge has been recently emphasized.
The LPRD development may be associated with weight, 4 high-protein diet, 5 or alcohol, tobacco,6,7 exercise, 8 or depression, 9 anxiety. The overweight and obesity are well-known favoring factors of GERD, while the results were conflicting in research of LPRD.4,10 Indeed, the customs, eating habits, and climate in are varied in different country, such as tea consumption, lifestyle-related risk factors other than carbonated drink, spicy food, chocolate and tea consumption, as well as poor subjective sleep quality have not been investigated in previous research.
Epidemiological data of LPRD represent an important reference for clinical diagnosis and treatment. Recently, a multi-center cross-sectional study with 72 tertiary comprehensive hospitals in China using the Reflux Symptom Index (RSI) suggested that the incidence of LPRD-related symptoms in outpatients at the ENT department was up to 10.15%. 6 Using the RSI in 2012, Athanasia et al showed that the prevalence of LPRD in Greek is 8.5%, 7 In 2002, Belafsky et al developed the RSI, a self-administered nine-item questionnaire designed in an attempt to standardize patient symptoms and improve initial diagnoses. 11 This tool is now widely used clinically in the management of LPRD and in epidemiological studies. The scale for each individual item ranges from 0 (no problem) to 5 (severe problem), with a maximum total score of 45. A RSI >13 is considered abnormal and strongly indicative of LPRD. Since its introduction, the RSI has been translated into several languages, the Chinese version of the RSI had confirmed its good reliability and validity. 12
This study aimed to analyze the lifestyle habits of patients with LPRD and whether the frequent consumption of potential reflux-provoking food and habits such as smoking, alcohol, tea, coffee, chocolate, carbonated drink, and spicy food consumption, night sleep time, dinner-to-bed time, and physical exercise may be associated with the risk of LPRD in outpatients at the otorhinolaryngology department.
Methods
Ethics approval was obtained from the Nan Fang Hospital Research Ethics Committee (REGISTRATION NUMBER: NFEC-2017-104). In the Nan Fang Hospital of China, an extensive public health survey was performed in OHNS clinics of Nan Fang Hospital during August 2017–July 2018, from August 2017 to July 2018. The survey questionnaire was administered by trained interviewers in the clinic’s temporary research center. The total amount of the exact questionnaires is 1751 respondents and 1658 eligible participants were included by a systematic sampling method. The method yielded a sample that was self-weighted to provide an estimated yearly number of clinical patients.
Those whose registration number ended in five were selected to fill out questionnaires during the second week of every month in the OHNS clinics of Nan Fang Hospital. Those excluded were patients outside the age range, patients with upper respiratory tract surgery or tumors and any acute illness, patients with severe mental disorders, and patients who refused to fill out the informed consent.
For the purpose of this study, LPRD diagnosis was based on an RSI score > 13, as proposed by Belafsky et al.
Sleep status was defined according to Pittsburgh sleep quality index (PSQI), 13 for item of subjective sleep quality, each asking “During the past month, how would you rate your sleep quality overall?” Subjects were allowed to choose either one of the following answers: (1) Very good, (2) Fairly good, (3) Fairly bad, and (4) Very bad.
Definitions of smoking and drinking in WHO.

Drinking status was defined according to World Health Organization classifications 15 (Table 1): abstainers were defined as those who reported no drinking in the reference period; general or ever-drinkers included persons who had ever consumed at least one alcoholic drink of any kind; current drinkers were defined as persons who consumed one or more alcoholic drinks in the year preceding the interview; and former drinkers were persons who had not drank an alcoholic beverage during the past 12 months. Beverage type was that which was typically drunk, and volume was estimated by the frequency method.
The definition of physical exercise was at least 30 min of moderate physical activity a week. 16
Questionnaire
The questionnaire was developed to evaluate the risk factor of OHNS clinics with LPRD. The survey questionnaire was administered by trained interviewers in the clinic temporary research center. All participants completed extensive written questionnaires covering a wide variety of exposures. In our study, we specifically analyzed variables that were related to lifestyle. These variables included tobacco smoking, alcohol consumption, coffee (caffeinated) intake, tea (total types) beverage, chocolate beverage, carbonated beverage, spicy food intake, night sleep time, subjective sleep quality, dinner-to-bed time, as well as physical exercise. In addition, age, sex, ethnic education, and body mass index (BMI; body weight in kilograms divided by the square of body height in meters) were assessed as potential confounders. The dietary habits of lifestyle habits contained 6 items, each asking the number of days per week one certain kind of food or beverage was consumed within the prior 3 months.
Items of lifestyle habits in the questionnaire.
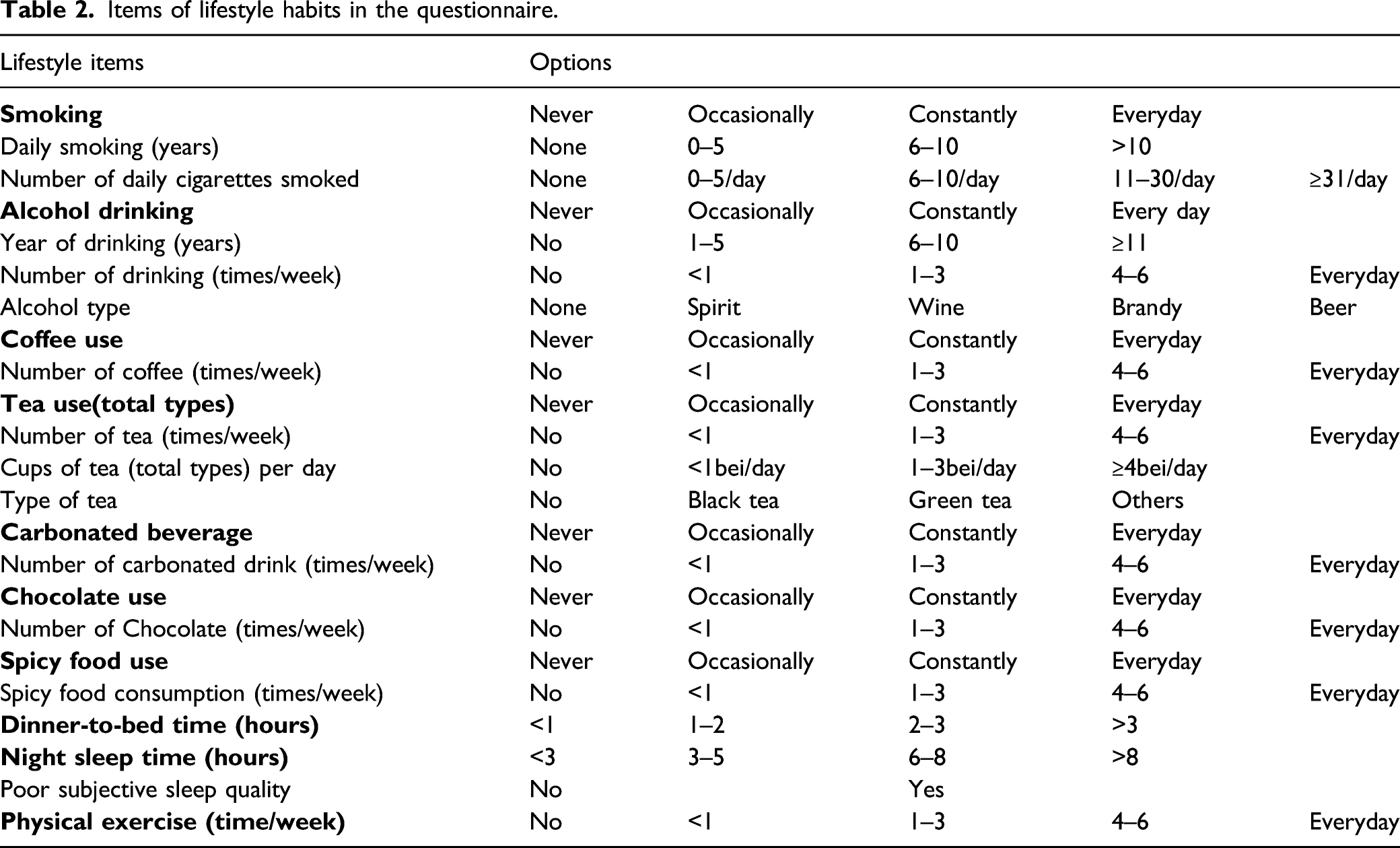
The questionnaire was pretested amongst 25 randomly selected hospital staff members before the study commenced, to identify any problem areas in the questionnaire and to make appropriate alterations, as adjudged necessary. The questionnaire was usually completed within five min.
Statistical Analyses
Some study variables were categorized to facilitate analysis. The categories for the number of years of tobacco smoking (cigarette, pipe, or cigar) were none (reference), 1–5 years, 6–10 years, and ≥11 years. The daily number of cigarettes smoked was categorized as 0 cigarettes (reference), 0–5, 6–10, 11–30, and ≥31 cigarettes. The categories for the number of years of alcohol drinking were none (reference), 1–5 years, 6–10 years, and ≥11 years. The categories for BMI were classified as ≤20 kg/m2, 20–22 kg/m2, 22–25 kg/m2, and >25 kg/m2. The categories for age were 18–20 years, 21–34 years, 35–49 years, 50–64 years, and 65–79 years.
All data including demographic data (age, sex, BMI, education) and lifestyle habits (smoking; alcohol, coffee, tea, carbonated drink, chocolate, and spicy food consumption; poor subjective sleep quality; and physical exercise) were compared between subjects with and without LPRD. The chi-square test was used to analyze categorical data, and Student’s t test was used to analyze continuous data. Using unconditional logistic regression, we calculated the odds ratios (ORs) as estimates of the relative risk and related 95% confidence intervals (CIs) to measure the association between frequent lifestyle habits and LPRD. Variables with a P <.25 in the univariate analysis were entered as candidate risk factors in the multivariate logistic regression analysis to identify independent risk factors of LPRD. Potential confounding effects such as age, sex, and BMI were tested by introducing them one by one into the model. All analyses were performed with IBM SPSS version 20. A 2-sided probability value of less than .05 was considered statistically significant.
Results
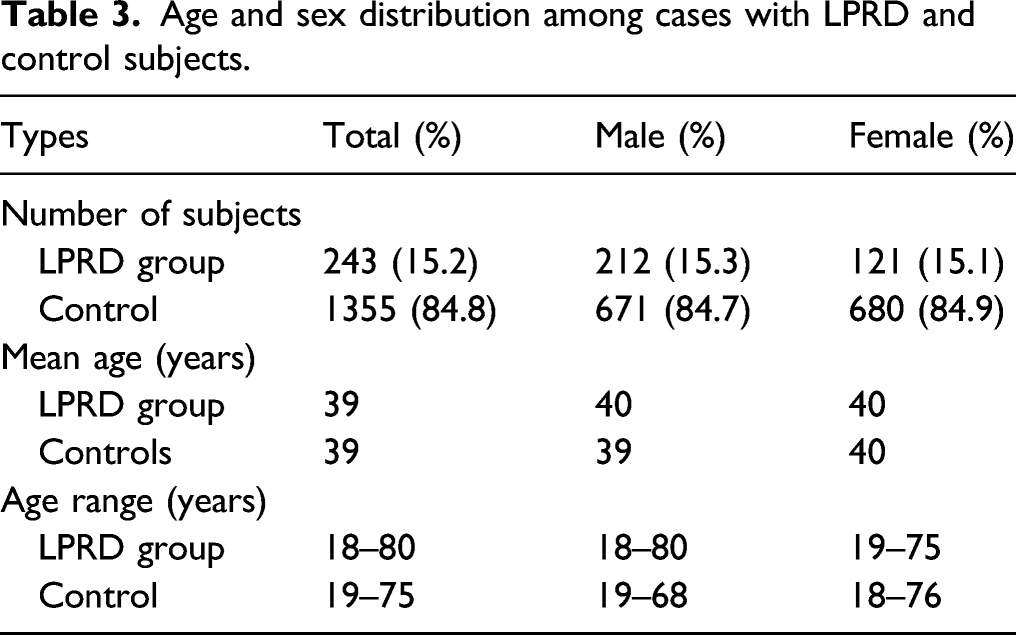
Age and sex distribution among cases with LPRD and control subjects.
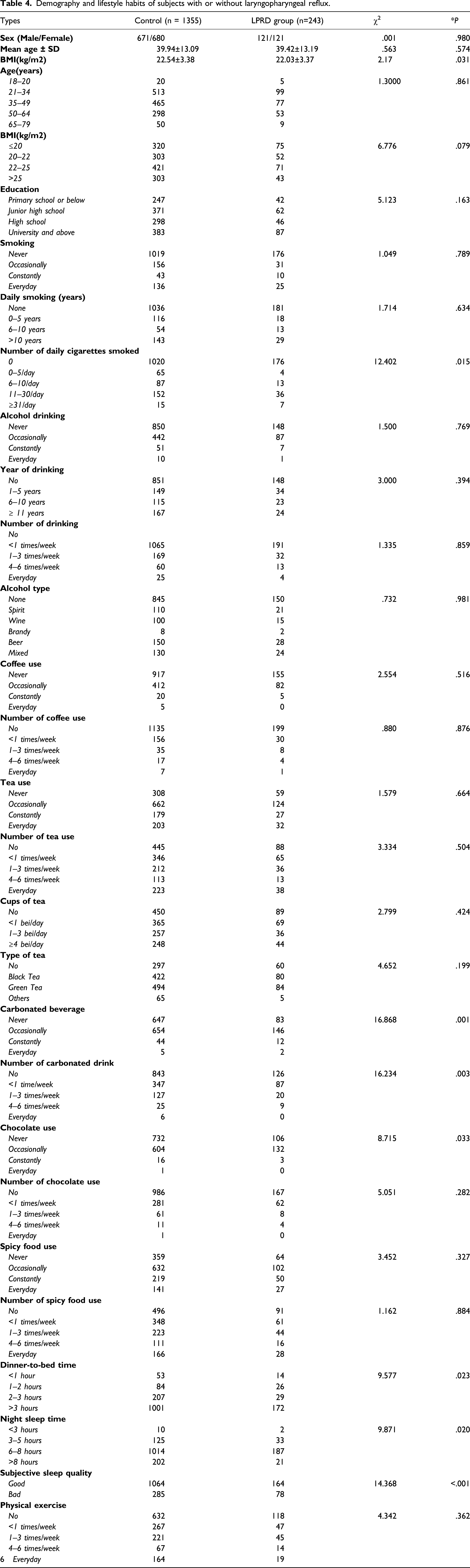
Demography and lifestyle habits of subjects with or without laryngopharyngeal reflux.
Lifestyle habits and risk of LPRD.
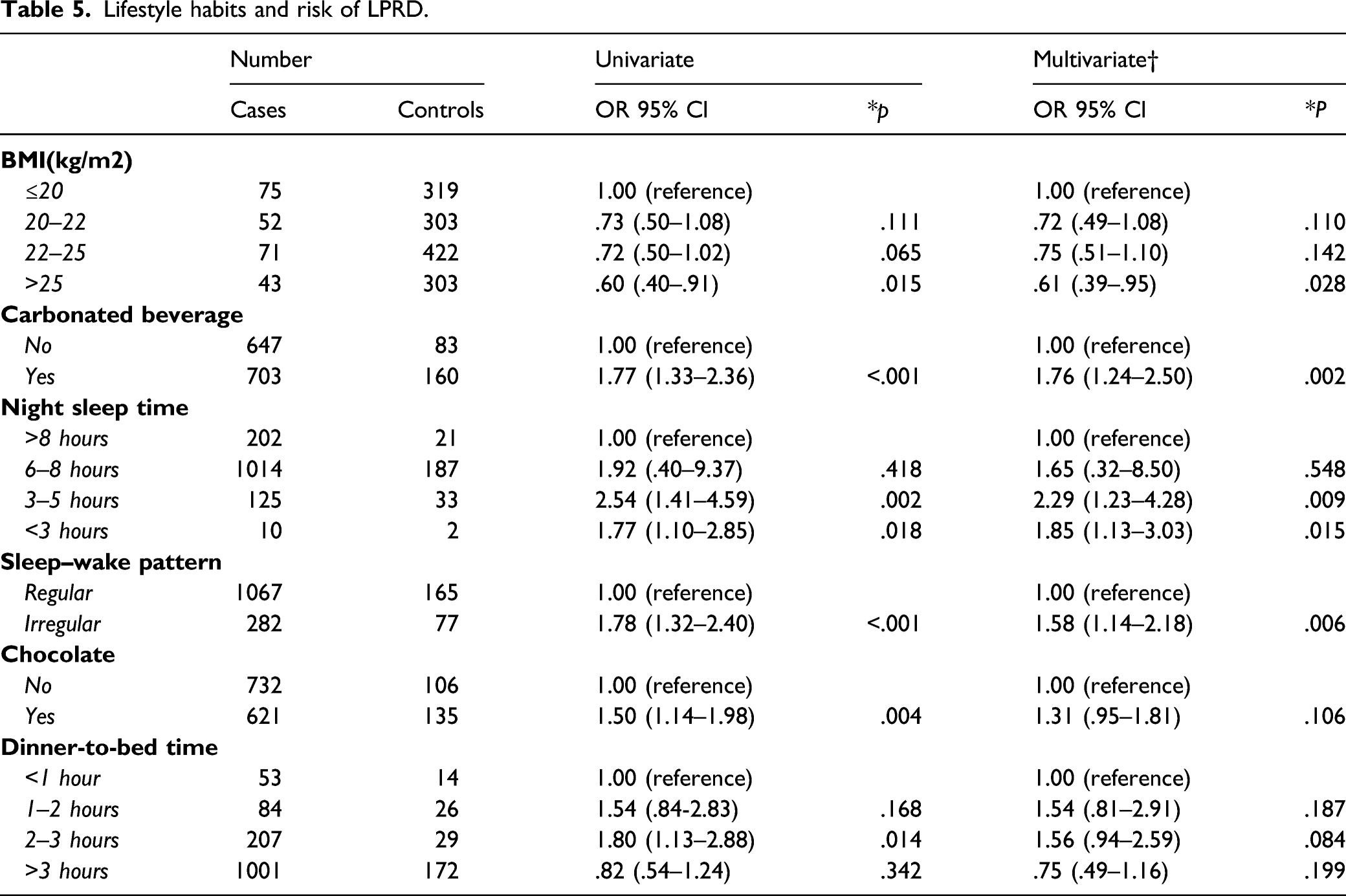
In the multivariate logistic regression analysis model, carbonated drink consumption (OR 1.76, 95% CI 1.24–2.50, P = .002), a short night sleep time (3–5 hours every night) (OR 2.29, 95% CI 1.23–4.28, P = .015), and an poor subjective sleep quality (OR 1.58, 95% CI 1.14–2.18, P = .006) were all independently associated with LPRD (Table 5).
In the multivariate analysis, there was a negative association between night sleeping time and LPRD, with an approximate 129% increase in risk among people who sleep 3-5 hours every night compared with those who sleep more than 8 hours (OR 2.29, 95% CI 1.23–4.28, P = .015). Similarly, a poor subjective sleep quality was associated with an increased risk of LPRD (P value for linear trend <.01). The risk of LPRD was 58% higher among people who had poor subjective sleep quality than among those who had good subjective sleep quality (OR 1.58, 95% CI 1.14–2.18, P = .006) (Table 5). Dinner-to-bed time was not associated with any change in the risk of reflux. This finding differed from the univariate analysis of dinner-to-bed time, which showed a decrease in the risk of reflux when comparing the same groups as above (OR .75, 95% CI 0.49 to 1.16, P = .199). The analyses revealed that the decreased risk of reflux identified in the univariate data could be entirely explained by confounders. No strong confounding effects were identified in the analyses of night sleeping time and poor subjective sleep quality (Table 5).
There was a moderately strong and dose-dependent association between an increasing frequency of carbonated beverage consumption and the risk of LPRD (Table 5). The multivariate analysis revealed that among individuals who drink carbonated drinks, the risk of reflux was significantly increased by 76% compared with those who did not drink any carbonated drinks (OR 1.76, 1.76, 95% CI 1.24–2.50, P = .002). Chocolate consumption was not associated with any change in the risk of reflux. This finding differed from the univariate analysis of chocolate consumption, which showed an increase in risk of reflux when comparing the same groups as above (OR 1.31; 95% CI 0.95 to 1.81, P = .106). The analyses revealed that the increased risk of reflux identified in the univariate data could be entirely explained by confounders.
There was a negative association between BMI and LPRD, with an approximate 39% (OR .61, 95% CI 0.39-.95, P = .054) decreased risk among people with a BMI>25 kg/m2 compared with those with a BMI < 20 kg/m2 (Table 3). No strong confounding effects were identified in the analyses of BMI.
Discussion
This study explored the risk factors of laryngopharyngeal reflux disease (LPRD) among ENT clinic outpatients, focusing on their lifestyle habits. We discovered that lack of sleep, poor subjective sleep quality, and carbonated beverage consumption were associated with an increased risk of reflux, and the frequent consumption of carbonated drinks was independently associated with an increased risk of LPRD with an apparent dose-response relationship. Additionally, LPRD is not associated with increasing BMI or obesity.
It is generally agreed that obesity and GERD are strongly associated, in our study, patients with LPRD were not obese. Controversy exists as to the effect of obesity on LPRD as results of different studies are diverse and contradictory.4,10 Although, there is no prospective study have shown a positive correlation between increasing BMI and LPRD. More prospective studies are needed to identify mechanisms and causes.
Cessation of carbonated beverage consumption is commonly recommended as a lifestyle modification for patients with LPRD. 2 Several studies have suggested that carbonated beverages may exacerbate GERD. 17 Similarly, increased carbonated beverage consumption in our study was significantly different between the LPRD and non-LPRD groups. In our multivariate analysis, there was a consistent dose-response relation between levels of carbonated beverage consumption (OR 1.76, 95% CI 1.24–2.50, P = .002), suggesting that a frequent carbonated beverage consumption is indeed a risk factor for LPRD. Carbonated beverages have been shown to increase postprandial esophageal acid and have been associated with promoting GERD symptoms by decreasing the lower esophageal sphincter (LES) pressure; beverage consumption was also found to predict GERD symptoms in a multivariate analysis. 17 Presently, there are very few studies supporting the role of carbonated beverages in LPRD.
Sleep disturbance and poor quality of sleep have been shown in a significant portion of patients with GERD.18,19 The hypothesis is that LPRD and sleep disturbance create a “vicious cycle” whereby sleep disturbance enhances throat and esophageal acid sensitivity, which in turn further enhances sleep disturbance. In our multivariate analysis, patients with a lack of night sleeping time (3-5hours) were characterized with high rick of LPRD than those have more than 8 hours sleeping time (OR 2.29,95% CI 1.23–4.28, P = .015). Patients with shorter night sleeping time have been linked to the poor subjective sleep quality. Poor subjective sleep quality has in turn been associated with increased incidence of LPRD-related symptoms. In our study, multivariate analysis showed that patients with irregular sleep wake pattern was associated with LPRD, with an odds ratio for reflux of 1.58 (95% CI 1.14–2.18, P = .006).
Tea and coffee are generally thought to be potent stimuli of gastric acid secretion and thus have been associated with heartburn. 20 However, the results of the effects of coffee and tea on LES pressure and postprandial acid reflux time, or the number of acid reflux episodes, were conflicting.21-23 In a population-based cross-sectional study, coffee exposure data showed a reduced risk of reflux symptoms among coffee drinkers compared with non-coffee drinkers, with an approximate 40% decrease in risk among people who drank more than seven cups of coffee per day compared with those who drank less than one cup (OR .6, 95% CI 0.4 to .7). A recent survey in Taiwan described no association between coffee drinking and GERD incidence, 21 multivariate analysis from the study showed that drinking tea ≥4 days/week (OR 1.6, 95% CI 1.1–2.3) was an independent risk factor of asymptomatic erosive esophagitis (AEE) in men. A large epidemiologic study found that tea drinking was not associated with any effect on the risk of reflux symptoms. 22 Our study showed no relation between tea consumption, type of tea consumption, frequency of tea consumption, coffee consumption, frequency of coffee consumption and LPRD incidence. Other foods, including chocolate and spicy foods, have also been found to be associated with acid regurgitation or heartburn among patients with GERD; however, no association has been reported with LPRD.
Alcohol consumption and habitual smoking have been reported to be putative risk factors for LPRD. 6 Hence, the recommendation of lifestyle modifications for the treatment of LPRD include smoking cessation and limiting alcohol intake. 2 Previous studies, though limited in sample size and lacking multiple factor analysis, have still been controversial regarding the associations of smoking and alcohol consumption with LPRD symptoms. A case series study suggested that some items of the RSI were significantly associated with smoking or drinking. 23 Nikolaos Spantideas et al pointed out that alcohol drinkers have higher RSI scores than nondrinkers, and smokers have higher RSI scores than nonsmokers (t test, P < .001 and P = .006, respectively). 7 Controversially, pseudorandom sampling performed in an English population found no association between LPRD and smoking or alcohol consumption. 8 Our present cross-sectional study, which included smoking, duration of smoking, daily smoking, alcohol consumption, duration of alcohol consumption, daily alcohol consumption and alcohol type, found no association between the factors above and LPRD.
This study focused on the lifestyle-related risk factor of patients with LPRD, which has rarely been researched. Moreover, we collected a wide range of exposure data our questionnaire, making it possible to evaluate a wide range of potential confounding factors in the multivariate analyses. The present study indicated an important role for lifestyle-related factors in the etiology of LPRD. However, few robust data have been published.
There are several limitations in our study. First, the main tool of the epidemiological survey-RSI scale was not an objective diagnostic tool of LPRD, without any objective methods, such as pH measurements. However, there is not an easy and generally accepted diagnostic method available for large-scale outpatient studies in otorhinolaryngology clinics, and those that are available are invasive and costly. Another potential source of error in our study was confounding; however, this was thoroughly evaluated in our multivariate analyses. Third, we could not identify any causal relationship due to the cross-sectional design of the study. Finally, we examined dietary habits using self-reported questionnaires, which might have resulted in some level of inaccuracy in classifying participants into different categories.
In conclusion, our study on the lifestyle habits of outpatients with LPRD provides evidence that poor lifestyle habits, such as carbonated beverage consumption, a short night sleep time, and poor subjective sleep quality may contribute to LPRD. Future research to corroborate our hypotheses is warranted for these common lifestyle habits, which have been strongly embedded in our dietary culture.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received support by the Gungdong Basic and Applied basic Research Foundation, grant number 2020A1515110638.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Nan Fang Hospital Research Ethics Committee ( REGISTRATION NUMBER: NFEC-2017-104 ) approved protocols.
Informed Consent
Written informed consent was obtained from a legally authorized representatives for anonymized patient information to be published in this article.
