Abstract
Introduction
Tympanostomy tube insertion (TTI) is one of the most common ambulatory surgical procedures performed in children. 1 Recently, pediatric TTI has successfully been performed in the office setting, with safety and effectiveness comparable with TTI performed in the operating room.2-8 Despite the benefits of avoiding risk of exposure to general anesthesia,9-11 increasing caregiver convenience and reducing procedure costs, 7 the real-time procedure-specific tolerability of in-office TTI in young children has not been clearly measured. Yet, in a 2021 survey of otolaryngologists, Schild and Rosenfeld 12 found that emotional pain and physical pain were 2 of 3 top barriers to the adoption of pediatric office TTI by otolaryngologists and that improved topical anesthesia would enhance the adoption of this technique.
Numerous office-based procedures are performed in children, including childhood vaccinations, venipuncture, gastrostomy tube exchange, and urodynamic studies. Assessment of how well a child tolerates a procedure that may be painful without sedation has been studied for many different treatments.13-16 In otolaryngology, cerumen removal, foreign body removal from ears and noses, and office laryngoscopy are accepted as routine practice. All are performed without sedation and often without topical anesthesia.
For in-office TTI, it is difficult to judge whether a young child is responding to immobilization, ear wax removal, application of topical anesthesia, or the tube insertion itself. Therefore, we attempted to evaluate tolerability of in-office TTI, utilizing only physical immobilization and topical application of phenol, in children aged up to 24 months using a novel objective measurement technique patterned after the Face, Legs, Activity, Cry, and Consolability (FLACC) scale 17 and 3 unique subjective assessments performed by (1) independent clinician experts, (2) the otolaryngology staff participating in the TTI procedure, and (3) the patient caregivers.
Methods
As part of a prospective, multicenter, single-arm clinical study designed to assess the safety and efficacy of in-office TTI using a single-pass TT delivery device [Hummingbird® Tympanostomy Tube System (H-TTS); Preceptis Medical, Golden Valley, MN, USA] with topical phenol tympanic membrane anesthesia, 5 30 patients, aged 6 to 24 months, were consecutively enrolled to have their intervention audio- and video-recorded at participating sites. These patients were a subset of the total study population of 211 enrolled in the H-TTS pivotal study, performed to gain market access in the United States by the U.S. Food and Drug Administration (FDA), and were among the last patients treated in the study. The sample size of 30, chosen with FDA input, is considered a reasonable number for what is considered a pilot study18,19 and contributes to a normal data distribution. 20 The study protocol was approved by the institutional review board of each of the 5 study sites, and informed consent for videotaping was obtained from caregivers/parents.
Objective Expert Video Review of Patient Responses
Investigators were provided digital recording equipment and were trained in equipment use and standardized scene capture. Clinicians and parents (if present and actively involved in distraction and consolation) were also to be visible in the recorded scene. The position of the video camera was kept as unobtrusive as possible to minimize any influence on child behavior. Recordings were standardized to begin when the patient entered the procedure room and continued until at least 3 minutes after the procedure endpoint (with the patient back in caregiver arms). If the parent chose not to be present for the procedure (2/30 procedures), the video would continue until 3 minutes after the parent entered the procedure room and is reunited with their child. The video field of view was also standardized to include head-to-toe visualization of the patient with facial expression visible.
The procedural methodology for assessing tolerability using the video recordings was codeveloped with FDA during the H-TTS device approval process. Recognizing that limited data supports the use of the FLACC scale for assessing procedural pain21,22 and that the papoose or swaddling used in the in-office TTI procedure would prevent an accurate assessment of the FLACC “Leg” activity element, a novel scale was developed to incorporate elements related to in-office visits. Defined patient responses (none, mild, moderate, and significant) at different stages of the recorded procedures were assessed independently by a panel of 3 clinician experts not involved in the trial, using standardized definitions (Table 1).
Behaviors and the Patient Response Categories Used to Evaluate Patient Experience During In-Office Ear Tube Insertion.
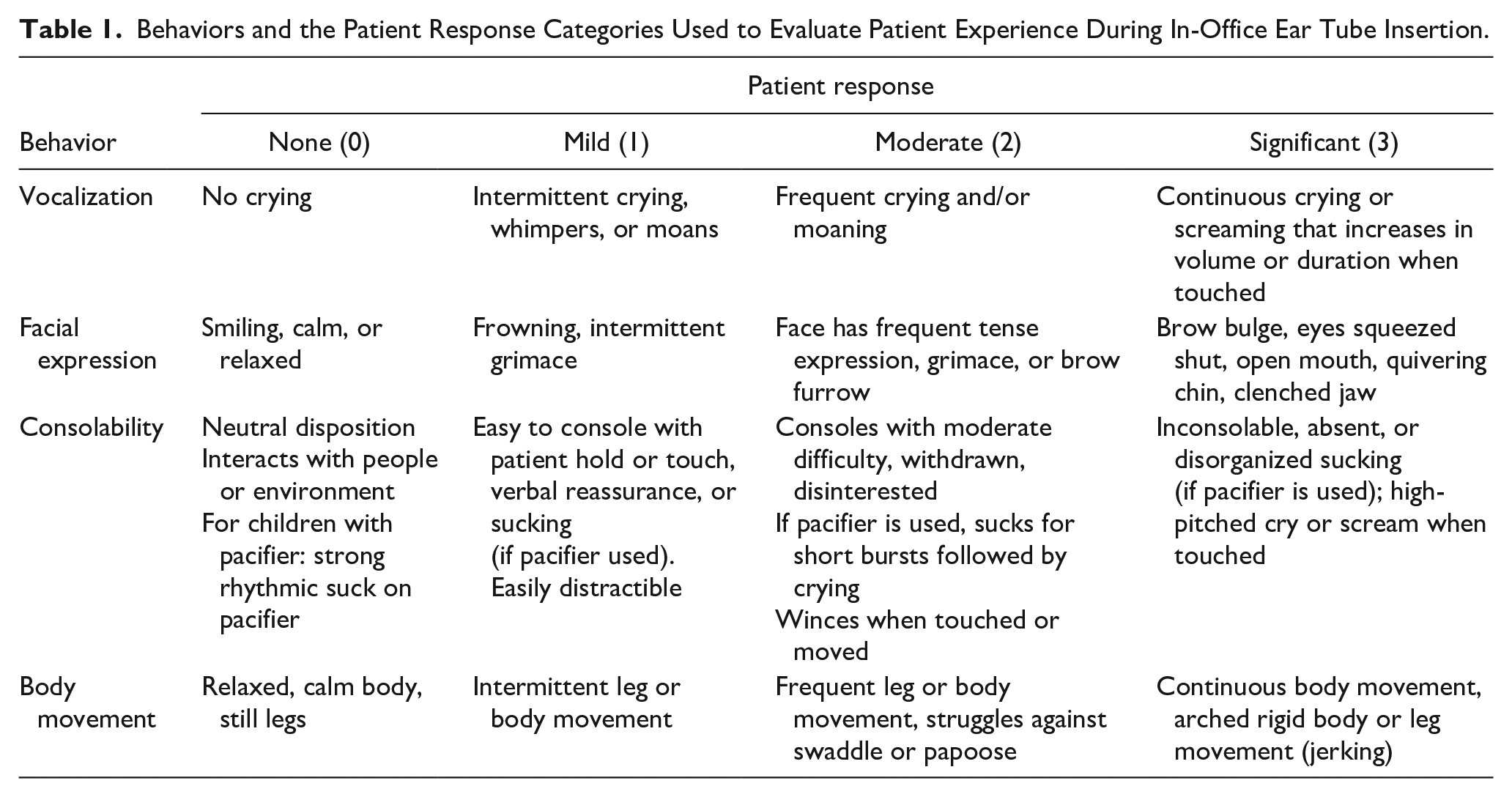
Clinician experts included a neurotologist, a pediatric intensivist, and a pediatrician. Training on behavior response definitions and evaluation methods prior to viewing the study videos was provided for these expert observers. Observers rated patient behavior at 5 procedural phases: (1) when the patient enters the room and during pre-procedural otoscopy (as applicable), (2) after papoose or swaddling, (3) during wax removal (as applicable), (4) during topical anesthesia application/ear tube insertion, and (5) 3 minutes after the procedure is completed.
For analysis purposes, the patient responses of none, mild, moderate, and significant were assigned numerical values of 0, 1, 2, and 3, respectively. Median and 25th/75th percentile scores were calculated for each expert as well as for the combined overall assessment. Comparisons of results between phases were performed using the Wilcoxon signed-rank test for difference with 0.05 as the significance threshold. In addition, evaluation of inter-rater reliability using this scale was also performed using the intra-class correlation coefficient (ICC). The analyses were performed with the Stata 17 statistical software package (StataCorp LLC, College Station, TX, USA).
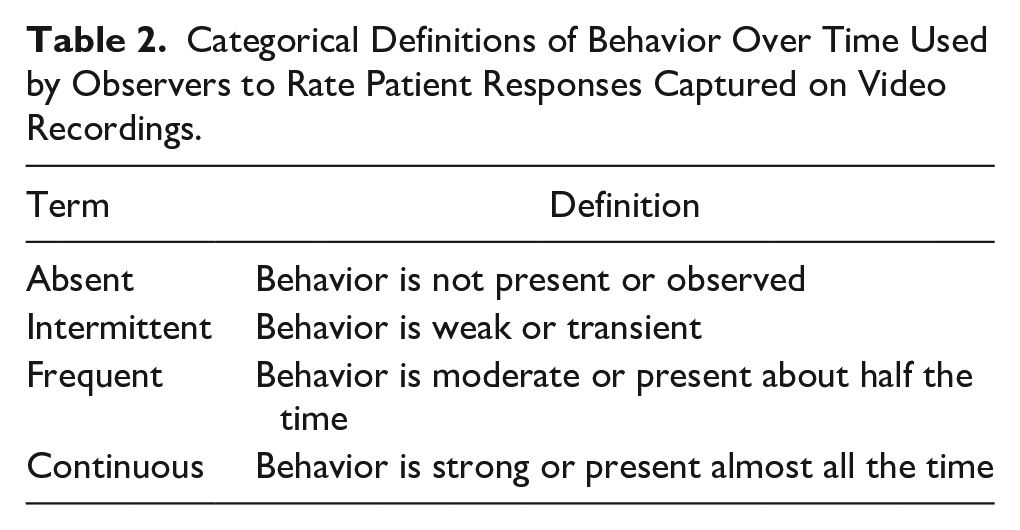
Definitions of the terms used to describe behavior over time (such as “intermittent” or “frequent,” as found in Table 2) were provided to the raters to standardize the use of these terms for elements in Table 1 where a frequency of the behavior is included. These definitions are consistent with those used in published scales with similar time components in their evaluation.17,23
Categorical Definitions of Behavior Over Time Used by Observers to Rate Patient Responses Captured on Video Recordings.
Subjective Assessments
Separate from the objective analysis of patient response, the clinician experts were asked to record their subjective assessment, in their opinion as a practicing clinician, if the child tolerated the procedure acceptably (yes/no). In addition, patient recovery was also assessed by the otolaryngologist and support staff. Recovery was defined as the time when the child was deemed calm and/or had no inappropriate crying (once back with parent, prior to leaving the clinic, or sometime after leaving the clinic).
Finally, a survey was distributed to parents/caregivers following the intervention at the same clinic visit to assess their overall satisfaction of the procedure. The survey consisted of 3 questions: (1) “As a parent, it is important to have an alternative to GA for my child’s ear tube procedure,” (2) “The reduced cost and increased efficiency associated with doing my child’s ear tube procedure in the office using the H-TTS is important to me,” and (3) “I would recommend doing ear tube procedures in office with the H-TTS to other parents.” Responses were recorded on a Likert scale: “strongly disagree,” “disagree,” “neutral,” “agree,” and “strongly agree.”
Results
Patient and Procedure Details
A total of 30 consecutive patients were enrolled at 5 otolaryngology sites across the United States from February to May 2020. The median age was 12 months (range 6-23 months), with females comprising 50% of the population. TTI was achieved bilaterally on all 30 patients with a single-pass success rate of 93% and a double-pass success rate of 100%. Phenol was successfully applied topically on the tympanic membrane in all 60 ears (100%). Median bilateral procedural time was 3.9 minutes (range 2.0-9.9 min).
Expert Video Review of Patient Responses
Each video was reviewed by each of 3 experts for a total of 90 reviews. The median patient response scores and 25th/75th percentile scores for each of the procedural phases are presented in Table 3; the P-values shown in the table for the latter 4 phases refer to comparisons to the baseline score of the patient entering the room.
Results of Expert Reviews of Procedure Videos Using Numerical Scale, Presenting Medians and 25th/75th Percentile Scores. P-Values Illustrate Significance of the Latter 4 Phases Compared to the Baseline Score of “Entering the Room.”
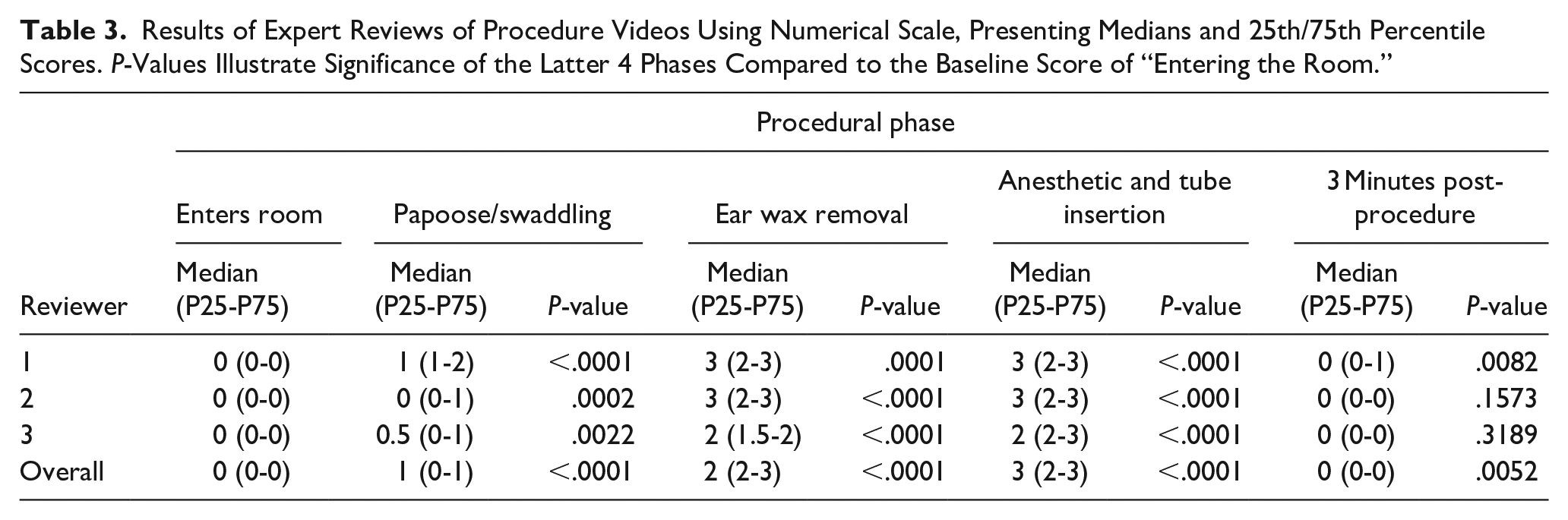
Based on overall median values, the reviewers rated the baseline response of entering the room as “no response” (score of 0). Patient response during the papoose/swaddling step rose to “mild” (median score of 1), increasing to “moderate” during the ear wax removal step (median score of 2), and further increasing to “significant” (median score of 3) during the anesthetic application/tube insertion phase. The response dropped back to “no response” (median score of 0) in the 3 minute post-procedure; however, the count of nonzero scores at that time was statistically significantly higher than at baseline.
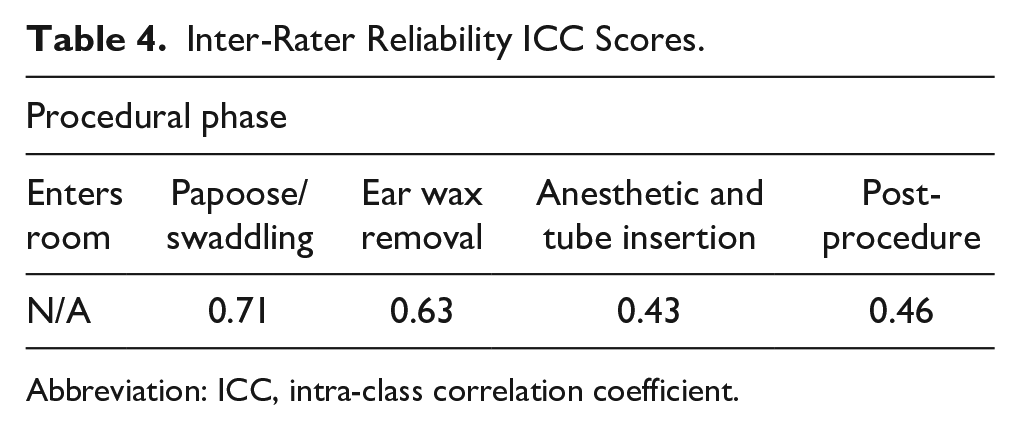
ICC used to represent inter-rater reliability range from a score of 0, representing no agreement between reviewers, and a score of 1, representing full agreement. The ICC scores for 4 of the phases are presented in Table 4; the score for the initial phase of the patient entering the room could not be calculated. The scores indicate a “good” to “very good” agreement between reviewers as defined by Regier et al. 24
Inter-Rater Reliability ICC Scores.
Abbreviation: ICC, intra-class correlation coefficient.
A summary of the response rates, stratified by procedural phase, is presented graphically in Figure 1. As described in the numerical analysis, the highest patient “no response” reviewer rates were reported for the baseline and the 3 minute post-procedure phases while the highest patient “significant response” reviewer rate was reported during topical anesthetic application and tube insertion.
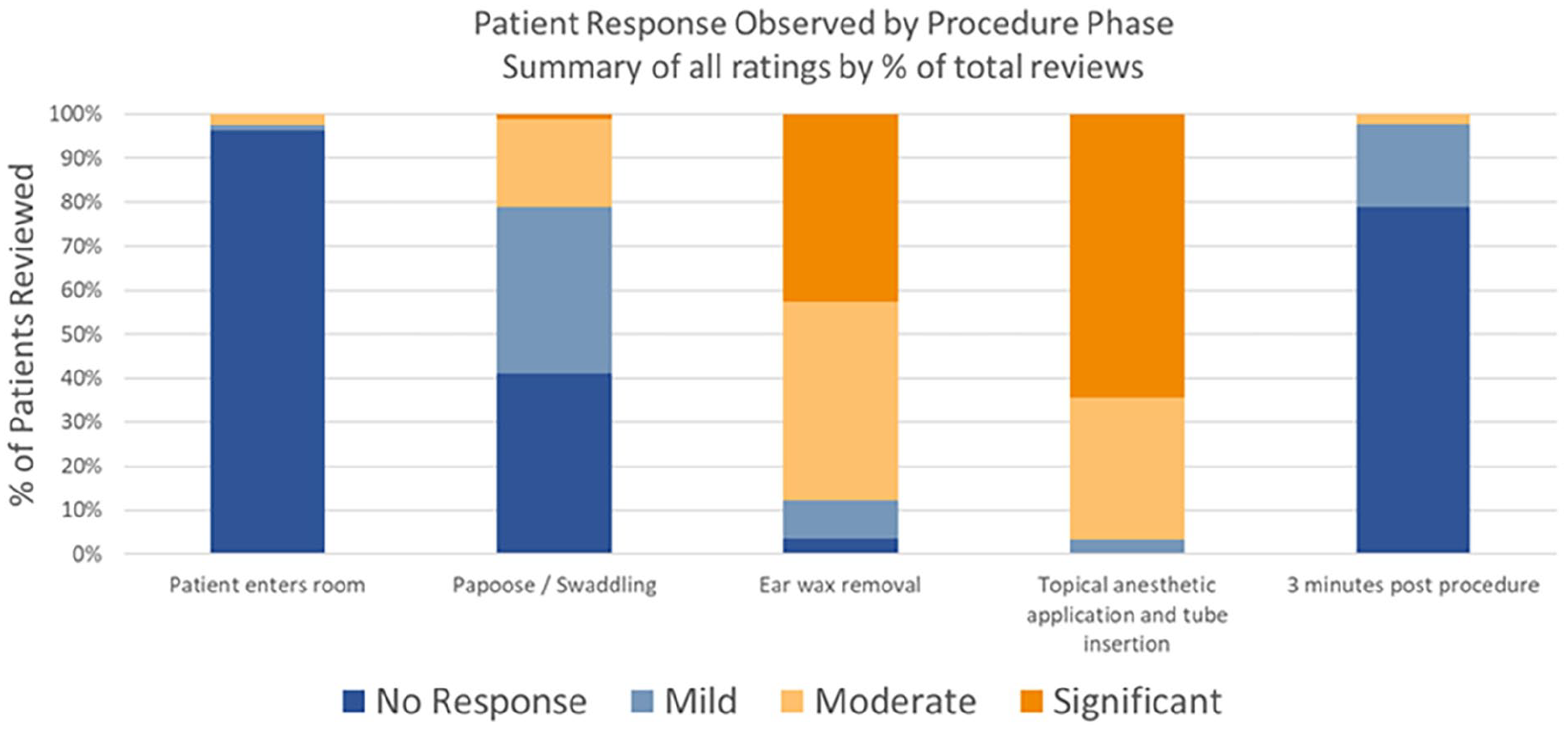
Summary of 30 individual patient responses during TTI at 5 procedural time points by 3 independent expert clinicians (n = 90). TTI, tympanostomy tube insertion.
Subjective Expert Assessment
Based on their opinion as a practicing clinician, all 3 reviewers rated that the entire cohort tolerated the procedure acceptably (100%).
Subjective Otolaryngology Staff Assessment of Child Recovery
Patient recovery, as assessed by the participating otolaryngologist and staff, occurred during the time immediately following the procedure to being back in their parent’s/caregiver’s arms in 27/30 patients (90%), with the remaining 3/30 (10%) patients requiring more time, but it occurred for all patients prior to leaving the clinic.
Subjective Parental Survey
According to the parental survey taken after the procedure, 93.3% of responders strongly agreed or agreed that an alternative to general anesthesia is necessary in TTI while 93.1% strongly agreed or agreed they would recommend the use of in-office TTI to other parents/caregivers (Figure 2). No caregivers recommended against the use of in-office TTI.
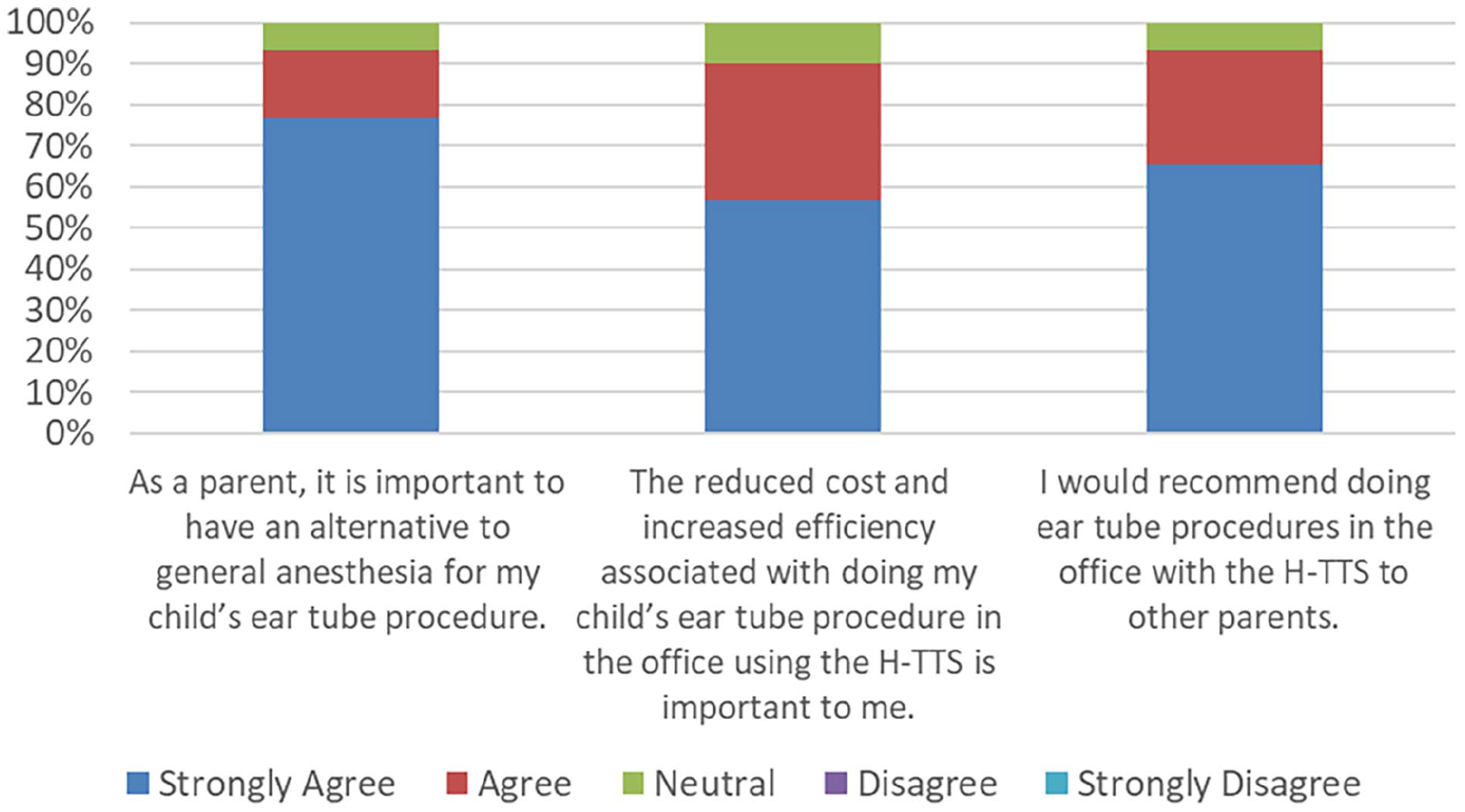
Parental response survey (n = 30).
Discussion
There is a clear focus on transitioning pediatric TTI to the office by the otolaryngologist community for many reasons including avoidance of general anesthesia risks and overall convenience.5,7,8,25 In 2017, the FDA recommended avoidance of prolonged exposure to general anesthesia (>3 hours) or repetitive exposures under age 3 years due to uncertain effects on neurodevelopment in children. 11 Furthermore, Warner et al. found the potential for decreases in fine motor coordination and processing speed associated with reading skills in children with multiple exposures to general anesthesia. 26 In-office TTI relies on local anesthesia to reduce the pain of the procedure, and the American Academy of Otolaryngology-Head and Neck Surgery issued a position statement in 2019 on office-based TTI for children that affirmed this option. 27
In this study, we analyzed tolerability outcomes in children under 2 years by using 4 assessment methods. The objective method focused on the child’s body language and vocal responses during the TTI procedure using a standardized assessment tool. Distress rates were generally low when patients entered the room and during papoosing/swaddling. The highest rates were encountered during anesthetic application and TTI. Recovery to baseline behavior was achieved relatively quickly as patients were returned to their caregiver, almost immediately after the insertion was completed. Results of the 3 subjective methods illustrated the in-office TTI procedure as being well-tolerated by the patients, with full patient recovery prior to leaving the clinic and with substantial support for the procedure by the patient caregivers.
In a study of 50 children with a mean age of 1.9 years undergoing bilateral TTI with general anesthesia, Fournier et al. reported a mean surgical time of 10.2 minutes with an average recovery time of 36.9 minutes in the postanesthesia care unit. 28 Caregivers were not allowed with their children during surgery. In comparison, the patients in the present study typically spent just 5 minutes not in their caregiver’s arms, including swaddling/papoosing and the TTI procedure itself, and required no postanesthesia recovery.
Child-centric assessment of procedure tolerability in young children has rarely been reported in the literature. Studies that did report on child tolerability outcomes mainly used subjective assessment methods, such as the visual analog scale and the Wong-Baker pain scale. However, these patient-reported assessment methods are not validated for the infant/toddler age range in our patient population29-31 as children younger than 4 years may not be able to provide meaningful reports of pain intensity. 32 Other studies have utilized the FLACC scale;3,33 however, the support for using this scale to assess procedural pain is limited.21,22
In contrast to the procedure used in the present study, Rosenfeld has described his experience with in-office TTI in children using acetaminophen premedication without local anesthesia and traditional myringotomy. He found no difference between TTI performed in-office using a papoose board for patient immobilization and an assistant for head stabilization versus in an operating room under general anesthesia, measured via caregiver reports of satisfaction of tube insertion or child’s recovery. 2 These results reflect the efficacy of in-office TTI seen in the present study even though procedural details may differ.
Cohen et al. 3 utilized behavioral support techniques, an investigational iontophoresis system to provide topical anesthesia and an automated tube delivery system for in-office TTI. Patients had to subjectively be able to follow simple verbal commands, sit still, and be able to cooperate during a routine ear exam and cleaning, since no papoose or swaddling was allowed. Research assistants assigned FLACC observational scores to patients 8 months to 17 years of age (mean = 7 years) from videos taken during the procedure. FLACC scores for patients up to 2 years of age mimicked those of the present study, with the highest distress seen with eardrum tap (considered as mild discomfort to moderate pain) 34 and tube delivery (moderate pain). Six patients (8.8%) with scores considered as moderate pain could not complete the procedure due to physical or behavioral issues. In contrast, all patients in the present study were able to complete the procedure without the need for behavioral support and without exclusion of patients, such as infants, who are unable to sit still during the procedure. In addition, specialized equipment was not required for anesthesia, only a manually-applied phenol solution, which has been previously reported as safe and effective in this study population. 35
A strength of this study is the demonstration, using objective evidence, of the acceptable pain tolerability of in-office TTI in a population where standard methods of pain assessment may not be capable of providing accurate results or are difficult to decipher. We employed a standardized, objective technique to assess the outcomes; however, this methodology is not validated. Additionally, our small sample size is another potentially-limiting factor in this study when considering generalizability to older children. Further investigation with larger patient sample sizes and a broader health care provider user base is required to fully assess the tolerability of tympanostomy tubes placed in children in the office.
Conclusion
This study found a concordance between the objective assessment and the 3 subjective assessments supporting the overall tolerability of in-office TTI. This procedure in young children was judged by independent video review to be universally well tolerated in young children. It is well perceived not only by clinician experts and health care providers but also by caregivers who would recommend office TTI to others for their children. Results from this study should provide reassurance to the otolaryngology specialist that in-office ear tube placement is a beneficial, well-tolerated option in young children and lends support for in-office TTI as an option for young children with middle ear disease.
Footnotes
Clinical Database Registry
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Franklin L. Rimell, MD is a part-time consultant for Preceptis Medical. Theodore Truitt, MD, has performed limited consulting work for Preceptis Medical. The remaining authors of this study (S.C., J.B., G.N., J.R., N.T, T.T) have no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preceptis Medical, Inc was the sponsor and provided the funding for this study.
