Abstract
Keywords
Introduction
Malignant peripheral nerve sheath tumor (MPNST) is one of the rare and most aggressive malignant lesions in the head and neck region. These are malignant tumors of peripheral nerve or nerve sheath cells excluding epineurium and vasculature.1,2 The incidence is 1 in 3000 globally, but it varies to region because of founder effect and mutation factors. 2 The majority of MPNSTs arise de novo or from malignant transformation of pre-existing neurofibromas, particularly in individuals with neurofibromatosis type 1 (NF1).
Progression from a recurrent sporadic neurofibroma to malignancy is an extremely rare event.
Patient Information and Time Line
We present a 26-year-old woman with a recurrent neurofibroma of the neck which transformed into MPNST after 3 excisions.
The patient had no cutaneous features or family history consistent with NF1.
She presented to the ENT outpatient department with complaints of multiple small swellings with skin ulcerations on the entire neck and mild pain for the past 6 years (Figure 1). She had a similar swelling on her left nape of the neck, for which she had an excision biopsy 3 years ago and was diagnosed as neurofibroma and repeat excision for the same after 1 year in an outside hospital.
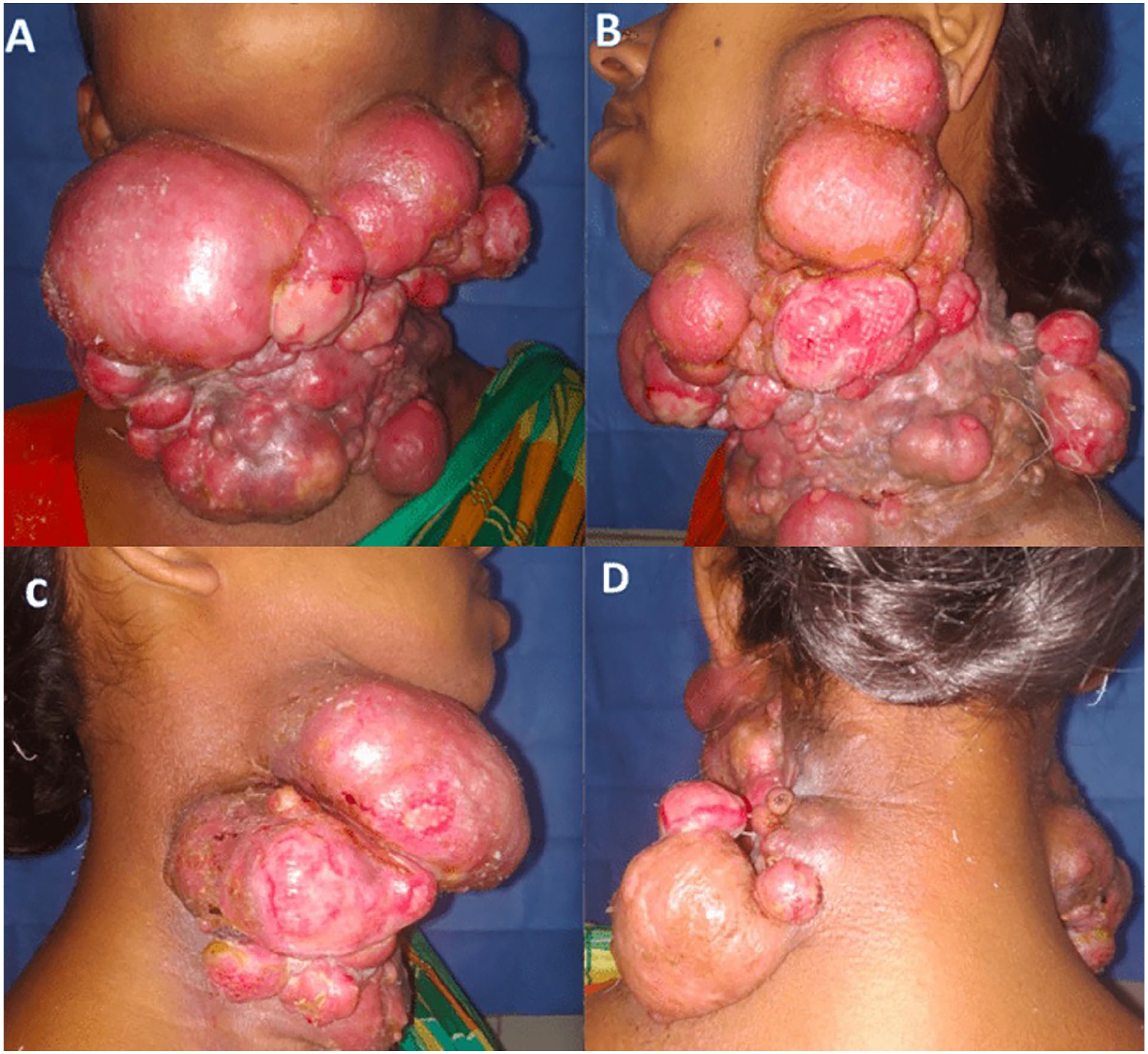
Picture showing multiple conglomerate swelling over the neck with skin ulcerations over the larger lesions which extend from the chin to the sternum in the midline (A) and from the posterior border of the right sternocleidomastoid muscle (C) to the anterior border of the trapezius muscle on the left side (B, D).
Clinical Findings
Physical examination showed multiple nodular swelling over the neck with skin ulceration on the larger lesions which extend from the chin to the sternum in the midline and from the posterior border of the right sternocleidomastoid muscle to the anterior border of the trapezius muscle on the left side (Figure 1).
Diagnostic Assessment
On radiological evaluation MRI revealed multiple well-defined lobulated lesions in the skin and subcutaneous plane on the left side of the neck and extending to midline and right which is iso tense on T1 and hyperintense on T2 with no involvement of soft tissues with mild diffusion restriction (Figure 2). Biopsy showed to be MPNST which on excision showed tumor showing cells with moderate nuclear pleomorphism with elongated nuclei with coarse chromatin, brisk mitotic activity can be identified. Immunohistochemistry (IHC) for S100 showed focal nuclear positivity (Figure 3).
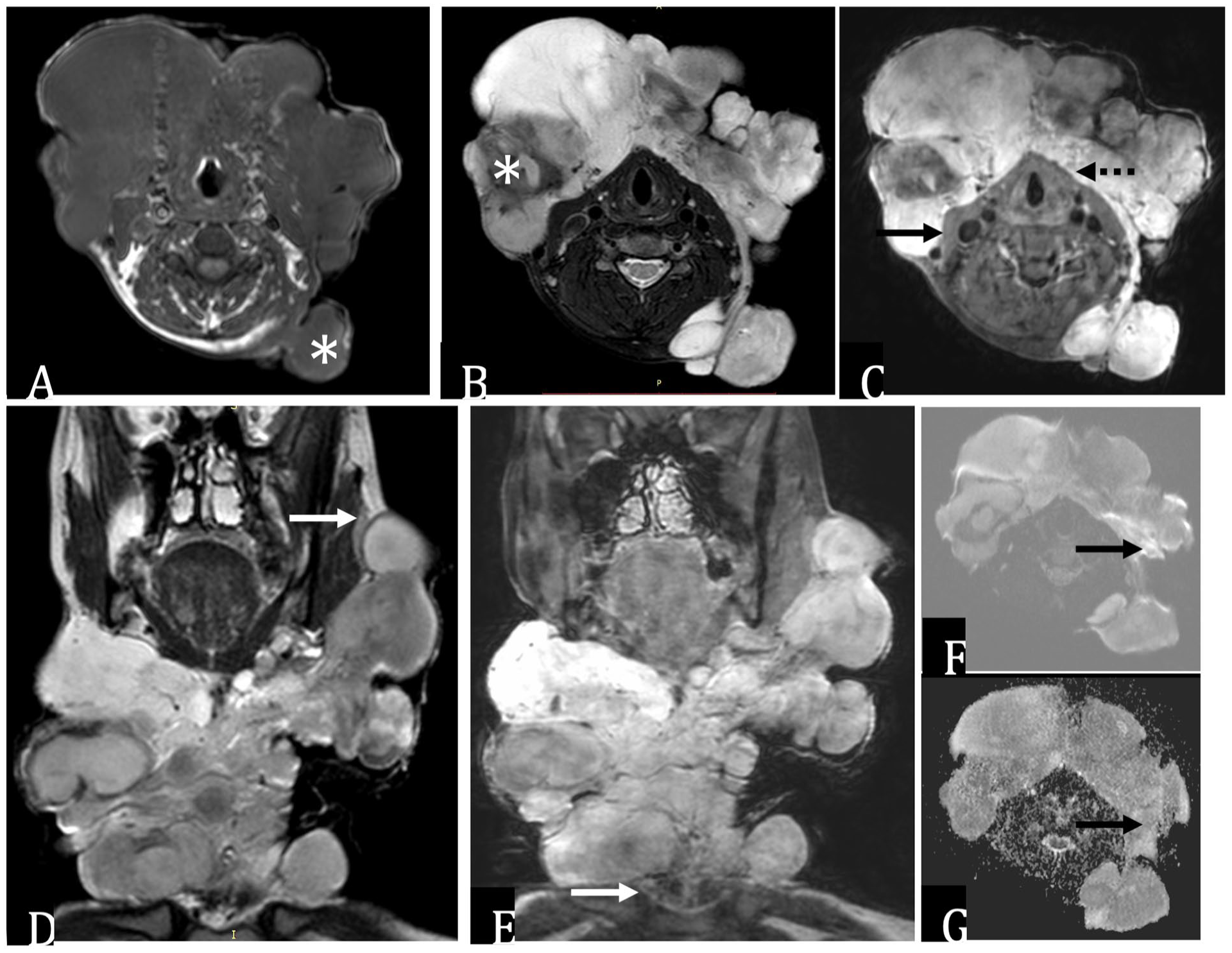
Contrast-enhanced MRI neck. (A) Axial T1WI, (B) T2WI, (C) post-gadolinium T1WI, (D) coronal T2WI, (E) coronal post-gadolinium T1WI, (F) axial high b value DWI, and (G) ADC map reveal a large lobulated mass lesion in the subcutaneous plane of neck, predominantly anteriorly with a lobulated posterior extension [* in (A)]. The mass is superiorly extending up to the level of the ramus of the mandible [arrow in (D)] with no evidence of intraorbital or intracranial extension. It is reaching up to the thoracic inlet [arrow in (E)] with no mediastinal extension. The lesion is superficial to the sternocleidomastoid muscle [arrow in (C)] and strap muscles [dotted arrow in (C)] with loss of fat planes but no deeper extension. The lobulated components are isointense on T1W and intermediate to hyperintense on T2W with few areas of T2 hypointensity [* in (B)] and intense, heterogenous post-contrast enhancement. Few areas show mild diffusion restriction [arrows in (F) and (G)]. Findings are suggestive of a large, plexiform neurofibroma in the subcutaneous space of the neck with a broad area of contact with the underlying muscles.
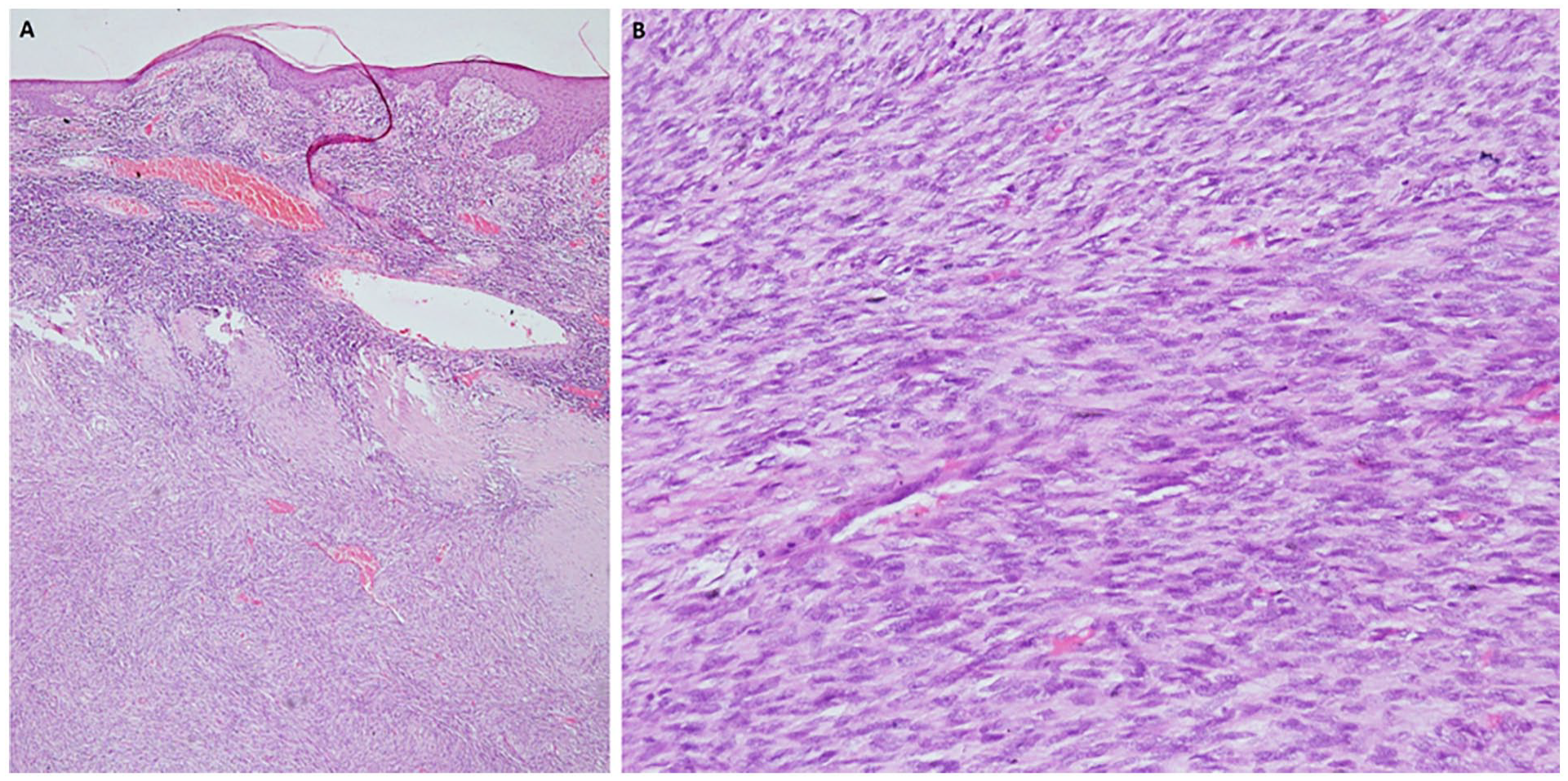
(A) Low-power histophotomicrograph of the tumor showing thinned out epidermis. There is a spindle cell tumor in the dermis and subcutis with cells arranged in fascicles which are interlacing at places. Focally alternating hyper and hypocellular zones are noted (H&E 40×). (B) High-power histophotomicrograph of the tumor showing cells with moderate nuclear pleomorphism with elongated nuclei with coarse chromatin, brisk mitotic activity can be identified (H&E 200×). IHC for S100 showed focal nuclear positivity (image not included).
Therapeutic Intervention
She underwent complete excision of the tumor which was seen in subcutaneous tissue and platysma with patchy infiltration of platysma and sternocleidomastoid muscles in some places. The tumor was seen infiltrating the underlying anterior scalene and middle scalene muscles and overlying skin (Figure 4A).
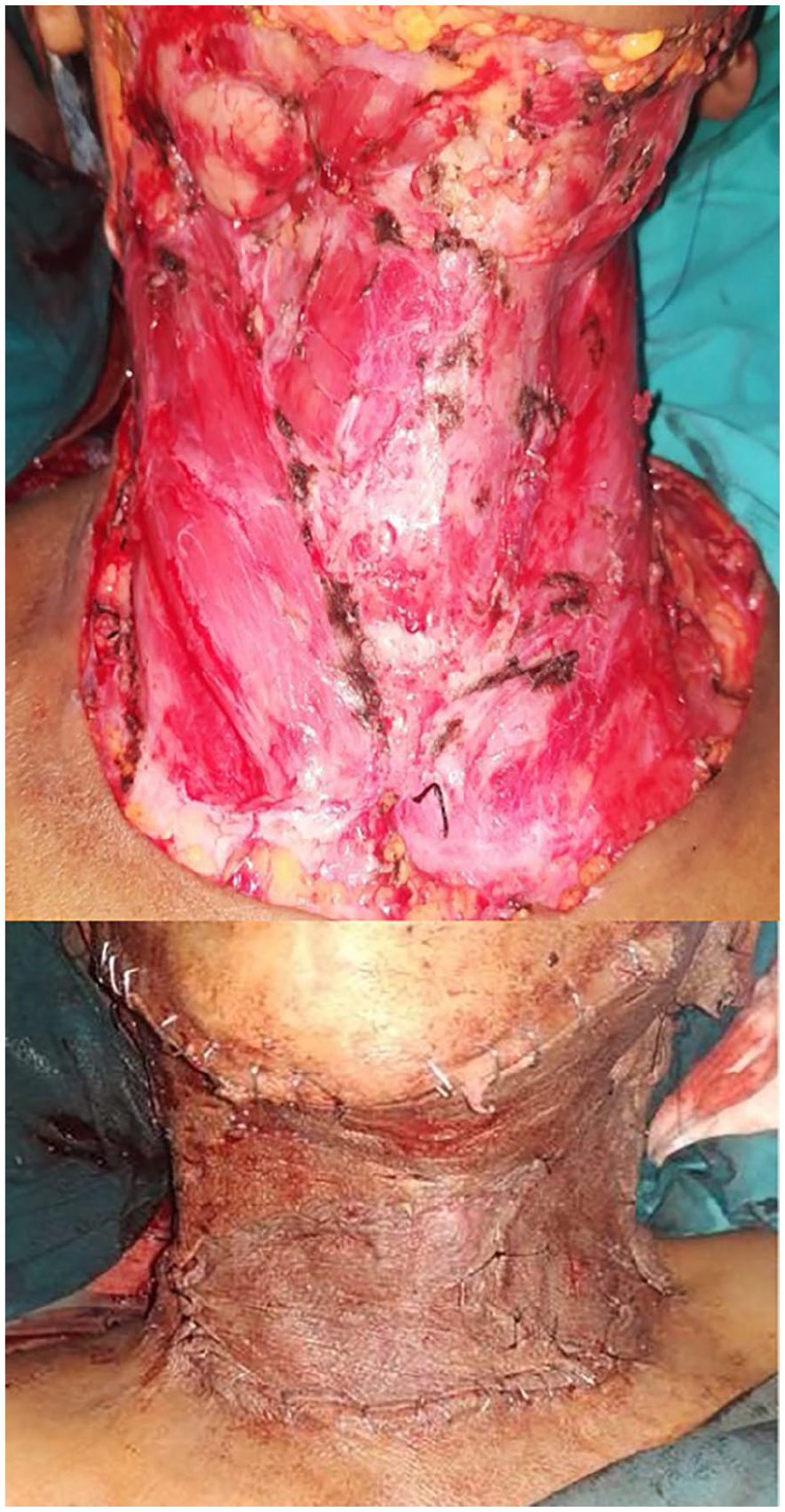
(A) Intraoperative picture showing the tumor bed after complete excision of the tumor (Top image). (B) After reconstruction of the defect with split-thickness skin grafting (Bottom image).
Split-thickness skin graft from the thigh was used to cover the defect (Figure 4B).
The postoperative histopathology showed MPNST with variable cellularity with scant cytoplasm and hyperchromatic nucleoli (Figure 3). The tumor cells are immunopositive for S100 with mitotic count 10 to 15/10 HP. The deep dissection margin was close to about 0.1 cm and was seen infiltrating into the muscles at a few regions along with the involvement of dermis. The patient received postoperative chemoradiotherapy in view of aggressive pathology and positive margins and tolerated well. She received 60 Grays of radiotherapy divided into 30 fractions over a period of 6 weeks along with 2 cycles of chemotherapy with Doxyrubicin and Ifosfamide. She had a good response following it (Figure 5A and B).
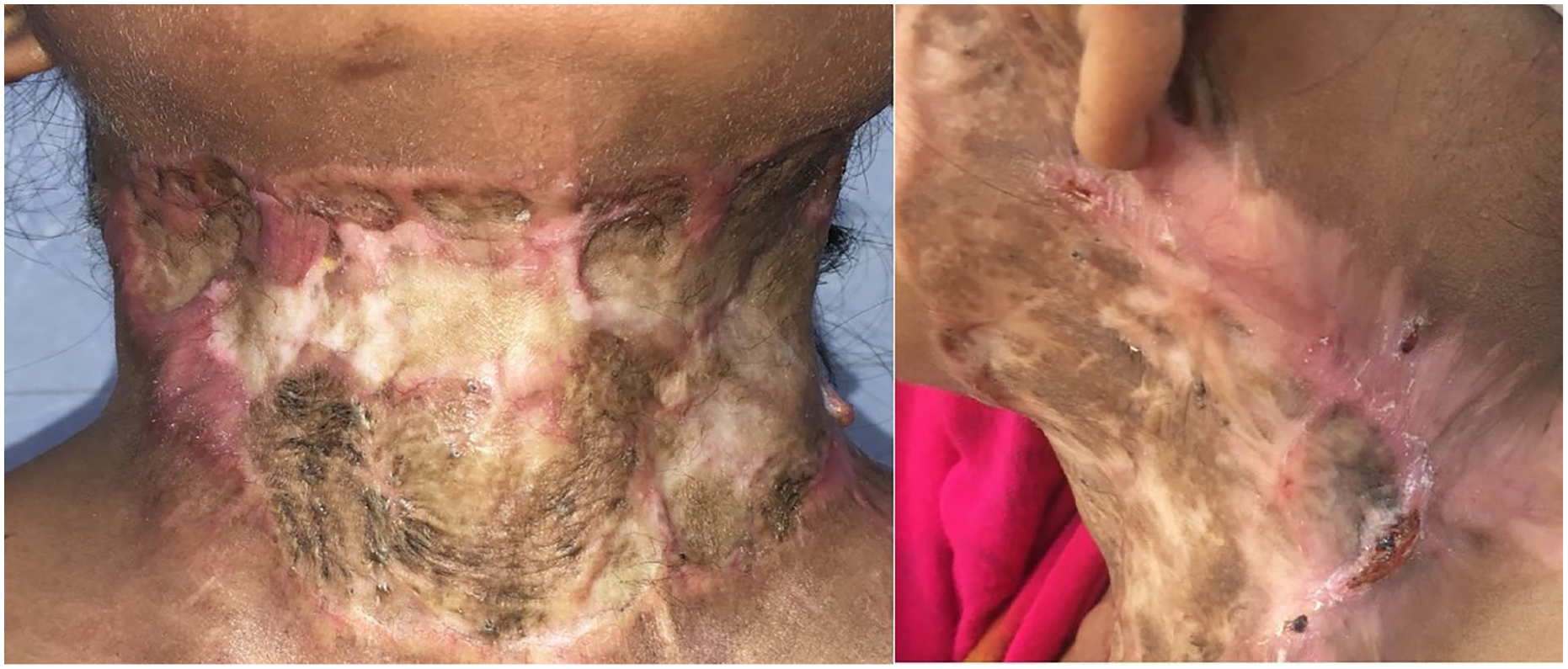
(A) Postoperative status after well-healed skin graft (anterio-posterior view). (B) Postoperative status of well-healed graft (lateral view) [5A- image in Left , 5B- image in right].
Outcome and Follow-Up
Postoperatively, the symptoms of compression improved, and on regular follow-up of the patient, she didn’t develop any recurrent swelling in the neck on 2 years follow-up which will be the longest survival for such a malignant lesion (Figure 5A and B).
Discussion
MPNSTs are rare and aggressive sarcomas that arise from Schwann cells of the peripheral nerves. They are often associated with NF1, which increases the risk of developing MPNSTs. Although MPNSTs account for only 5% to 10% of all soft tissue sarcomas, their occurrence in the head and neck is rare, representing less than 10% of all cases, with the neck being the most commonly affected site. These tumors are known for their rapid growth, aggressive behavior, and poor prognosis.
Genetic Background and Pathophysiology
NF1 is a well-known genetic predisposition for MPNST, with 8% to 13% of patients with NF1 developing into malignant tumors. The NF1 gene, located on chromosome 17q11.2, encodes for tumor suppressor protein neurofibromin, which plays a crucial role in regulating cell growth and differentiation through the RAS/MAPK pathway. Loss-of-function mutations in NF1 leads to unchecked RAS signaling, promoting oncogenesis. MPNSTs can also arise sporadically or secondary to prior radiation therapy.1,2
MPNSTs which originate from Schwann cells are histologically characterized by spindle-shaped cells and demonstrate markers of neural differentiation, including S-100 protein, which helps in differentiating them from the other soft tissue sarcomas. Genetic mutations in TP53, CDKN2A, and loss of SMARCB1 have also been noted in the pathogenesis of these tumors.3,4 Studies have also shown that MPNSTs have a higher degree of genomic instability compared to benign nerve sheath tumors. 5
Diagnosis
The diagnosis of MPNST is based on combined clinical, radiological, and histopathological assessments. Clinical features like rapid growth, pain, neurological deficits, and ulceration in a previously stable neurofibroma should always raise suspicion for malignant transformation. In our case, the patient’s history of recurrent neck swellings with pain and bleeding was seen pointing toward a malignant nature.
For imaging MRI is the modality of choice, showing tumors with low signal intensity on T1-weighted imaging and high signal intensity on T2-weighted imaging, often with signs of adjacent tissue invasion. PET-CT is also useful for detecting distant metastases. Histopathology reveals spindle cells with a fascicular arrangement and varying degrees of pleomorphism. IHC helps in diagnosis, with MPNSTs typically expressing S-100, SOX10, and Leu7, although these markers may be less pronounced in high-grade tumors.6-8
Treatment Options
The preferred treatment is surgical resection which is complete excision of tumor with clear margins. The positive margins are associated with a high rate of local recurrence. However, in the head and neck, attaining negative margins is always difficult due to underlying vital structures. In our case, complete excision was achieved and the defect was closed with split-thickness skin grafting.9,10
Several studies have shown the importance of surgery in managing MPNST. Widemann et al demonstrated that patients who underwent complete resection had significantly better overall survival rates compared to those with incomplete resections. 11 But even after radical surgery, local recurrence rates remain high, ranging from 40% to 60%.12,13
Early MPNST (stages 1 and 2) can be managed by single modality treatment preferably surgical, whereas late MPNST (stages 3 and 4) requires multimodality treatment; in our case, it was stage 3 with a size more than 5 cm with deep infiltration without distant metastases.
Adjuvant Radiotherapy
Radiotherapy is typically recommended for patients with high-risk features, which include tumors larger than 5 cm, high-grade histology, or positive surgical margins. Radiotherapy decreases the rate of local recurrence, especially in cases where achieving wide margins is difficult like head and neck regions as in our case. Study by Anghileri et al has shown that adjuvant radiotherapy significantly improves local control. 14
Chemotherapy
The role of chemotherapy in MPNST remains controversial. It is typically reserved for advanced or metastatic cases. Doxorubicin and ifosfamide are the most commonly used agents, but response rates are modest. A recent study by Ferrari et al reported a partial response rate of approximately 30% in patients treated with neoadjuvant chemotherapy for downstaging tumors prior to surgery. 15
Novel Therapies
Targeted therapies and immunotherapy are being explored in the treatment of MPNSTs. Preclinical studies on MEK inhibitors, given the involvement of the RAS/MAPK pathway in NF1-associated MPNSTs, have shown some benefits. Clinical trials involving drugs like selumetinib have demonstrated some efficacy, particularly in NF1-related tumors, but further research is required.16,17
Prognosis
The prognosis for MPNST, particularly in the head and neck, remains poor. Five-year survival rates range from 15% to 35%, significantly worser than for MPNSTs of the extremities. Factors contributing to this poorer prognosis include difficulty in achieving clear surgical margins and the proximity of vital structures, limiting radical surgical resection. Local recurrence occurs in up to 50% of cases, with distant metastases developing in approximately 33% of patients.18,19,20
In conclusion, while complete surgical resection remains the gold standard in MPNST management, multimodal treatment, including adjuvant radiotherapy and chemotherapy, is often required.
Footnotes
Acknowledgements
Dr Rajeev Kumar is the corresponding author. Dr Rajeev Kumar, Dr Ramaneeshwaran Murugesan, Dr Suresh Mani and Prof Rakesh Kumar were the treating physicians. Dr Ramaneeshwaran Murugesan designed the case report monitored by Dr Rajeev Kumar and Dr Prem Sagar with radiological data provided by Dr Ashu Seith Bhalla and Dr Smita Manchanda and pathological data by Dr Adarsh Wamanrao Barwad.
Data Availability
The required images have been uploaded along with the main document.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Our institution does not require ethical approval for reporting individual cases or case series.
Informed/Patient Consent
Written informed consent was obtained from the patient and her husband for patient information to be published in this article. Written informed consent was obtained from the patient and her husband for the publication of images and data included in this article.
Trial Registration and Date
Not applicable.
