Abstract
Keywords
Introduction
Acquired aural atresia and stenosis are uncommon diseases, and the annual occurrence rate is less than 0.6 cases/100,000 people. The main etiologies of acquired aural atresia and stenosis include trauma, inflammation, postoperative complications, and neoplasms.1 -4 The symptoms frequently include otorrhea and hearing loss, and surgical intervention is the curative standard for these diseases. The surgical aims are to prevent complications and restore function. Several surgical techniques have been introduced, and meatoplasty and canalplasty are the main methods used to obtain a widened external auditory canal (EAC).5 -8 In addition to surgical techniques, postoperative management is imperative to obtain good outcomes.9,10 However, the recurrence rate of acquired aural atresia and stenosis still ranges from 10.5% to 28.6%.2,9 This study included 4 patients with different etiologies of acquired aural atresia and stenosis. On the basis of these patients, we aimed to emphasize etiology management and prevention in clinical practice. In reference to the literature, we discuss surgical tactics and postoperative management to obtain positive outcomes. In particular, we suggest that maintaining EAC patency with a “Swiss roll” plastic sheet and an expansive sponge is highly important (Figure 1). Additionally, triamcinolone acetonide injections effectively prevent postoperative stenosis.

“Swiss roll.” (A) Intraoperatively, a plastic sheet was harvested from the pocket part of surgical film (AnBang, Xinxiang, China). (B) An expansive sponge (1 × 7 cm) produced by Medtronic of USA. (C) The plastic sheet and the expansive sponge were trimmed. Then, the plastic sheet was overlaid to the new external auditory canal and the trimmed-expansive sponges were filled into it via nasoscope. (D) A “Swiss roll” obtained from an expansive sponge with ear drops soaked and a plastic sheet.
Methods
Four patients with acquired aural atresia or stenosis who had received surgical treatment at the Xijing Ear, Nose, and Throat (ENT) department were selected according to their representative etiologies and postoperative management. Informed consent was obtained from these patients or their parents. A retrospective case review and literature review were conducted.
Patient 1
A 63-year-old woman had otorrhea and hearing loss in the right ear. She visited the local ENT department 6 years ago and received local treatment of antibiotic ear drops. The otorrhea improved after using antibiotic ear drops but it relapsed several times. One year later, she scheduled a consultation for progressive hearing loss with our department. Examination showed acquired aural atresia (Figure 2A-C) and surgical intervention was proposed. The operation was performed under general anesthesia, and an endaural incision was performed. After the removal of atresia tissue, a cholesteatoma lesion with bony erosion was observed. After the cholesteatoma was removed, the posterior skin had a partial defect but a normal tympanic membrane. Canalplasty and meatoplasty were performed and the skin flap from the cavum conchae was rotated to widen the EAC entrance and cover the denuded skin of the posterior bony erosion. The new EAC was supported by a plastic sheet (AnBang, Xinxiang, China) trimmed into a “Swiss roll” shape with a trimmed-expansive sponge (Medtronic, Jacksonville, USA) (Figure 1), and daily ototopical drops were applied. On postoperative day 21, the plastic sheet was removed, and the EAC was cleared. Expansive sponge replacement and clearance were performed every 3 weeks until 3 months after surgery. A well-epithelialized EAC and improved hearing function were achieved (Figure 2D-F).
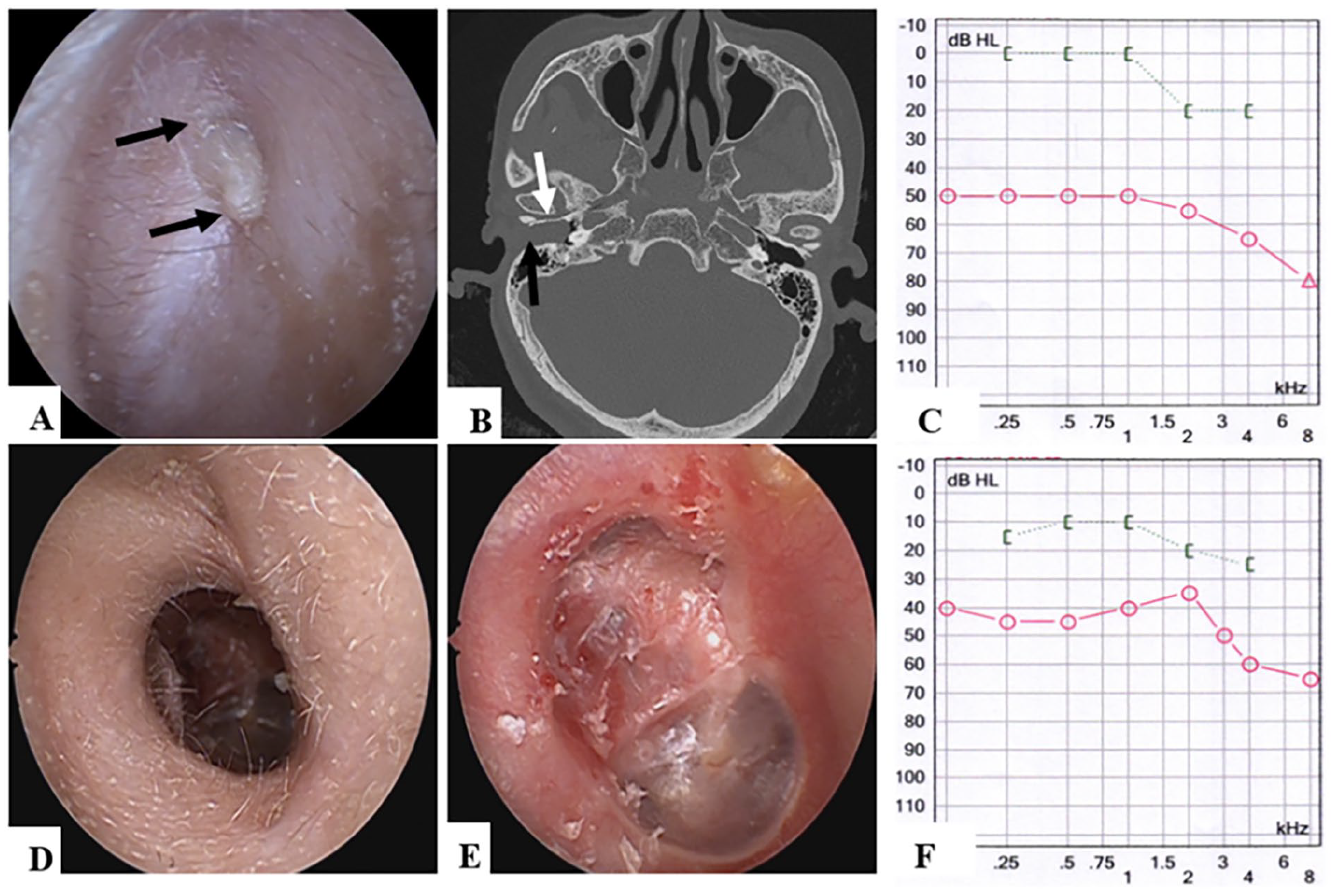
Patient 1 (right ear). (A-C) Preoperative examination results. (A) Atretic tissue (black arrow) was observed in otoscope; (B) Soft tissue (black arrow) and bony invasion (white arrow) showed in the CT; (C) Air-conduction level was significantly impaired. (D-F) 1-year postoperative outcomes. (D) A widened and well-epithelialized EAC obtained; (E) The retraction of tympanic membrane displayed in otoscope; (F) ABG significantly closed. CT, computed tomography; EAC, external auditory canal; ABG, air-bone gap.
Patient 2
A 50-year-old male with acquired aural stenosis of the left ear visited our ENT outpatient clinic. He had experienced maxillofacial trauma with bloody discharge and hearing loss in the left ear 5 years prior. The community hospital managed the maxillofacial trauma and cleared the bloody discharge without further treatment. Thereafter, the patient’s hearing function gradually decreased, and he visited the local ENT department 4 years ago. He was diagnosed with acquired aural stenosis, and a surgical operation was performed. However, restenosis occurred 6 months postoperatively, and the patient was referred to our department. A retroauricular incision, canalplasty, and meatoplasty were performed on the patient after the preoperative assessment (Figure 3A-C). The fibrotic plug was resected, and a postauricular free flap was harvested to cover the local skin defect (Figure 3D). Iodoform gauze was used to fill the new EAC, which was removed 3 weeks postsurgery. A trimmed-expansive sponge was subsequently applied to support the EAC, and daily ototopical drops were applied. A widened and stable EAC was obtained 4 months after surgery, and no reoccurrence occurred during long-term follow-up (Figure 3E and F).
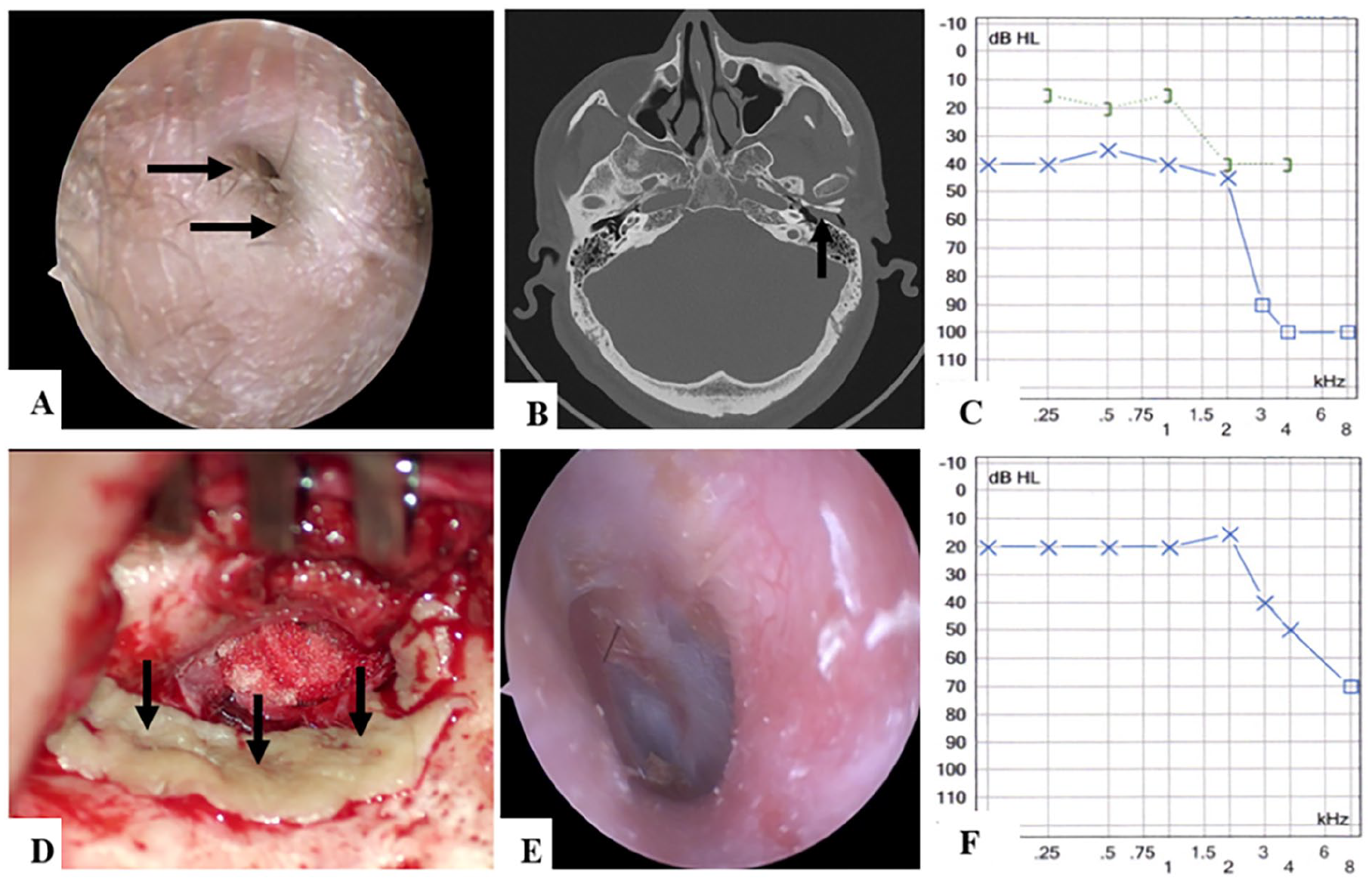
Patient 2 (left ear). (A-C) Preoperative examinations. (A) Severe stenosis (black arrow) in the opening of the EAC was observed in otoscope; (B) Soft tissue (black arrow) of the EAC showed in CT; (C) The ABG value ≥20 dB HL showed in pure tone audiometry. (D) Intraoperatively, the new widened EAC with coverage of postauricular free flap (black arrow) under microscope. (E, F) Results of 3-year postoperatively. (E) A well-epithelialized and stable EAC showed in otoscope; (F) The postoperative ABG closed. CT, computed tomography; EAC, external auditory canal; ABG, air-bone gap.
Patient 3
An 11-year-old boy underwent surgery for middle ear cholesteatoma in the right ear 2 years prior at a local hospital. One year later, he was diagnosed with acquired aural atresia in the local hospital. Surgery was recommended for this patient and he was referred to our department. Otoscopy and high-resolution computed tomography (HRCT) showed complete atresia in EAC (Figure 4A and B) and pure tone audiometry displayed conductive hearing loss (Figure 4C). The meatoplasty and canalplasty were performed. The whole fibrotic plug was removed and the EAC was widened. The cholesteatoma with involvement of mastoid air cells and partial destruction of the ossicular chain were detected. After cholesteatoma removal, a partial ossicular replacement prosthesis was implanted. A conchal flap combined with the temporalis muscle fascia was used for myringoplasty. Thin sectional skin was harvested from the temporal scalp and overlaid on the skin defect in the EAC. Routine postoperative care and follow-up were conducted. However, this patient developed hypertrophic scars 6 weeks after surgery, and cicatrix tissue formed in the cartilaginous segment of the EAC and retroauricular incision. Triamcinolone acetonide (at a concentration of 10 mg/mL) was injected into the local skin once every 2 weeks. It was circularly injected into the narrowest section via multiple injections. This injection was repeated 3 times, and subsequent EAC support with trimmed-expansive sponge was continued. The EAC was stable at 6 months after surgery, follow-up lasted 52 months, restenosis did not occur, and the hearing level remarkably improved (Figure 4D-F).
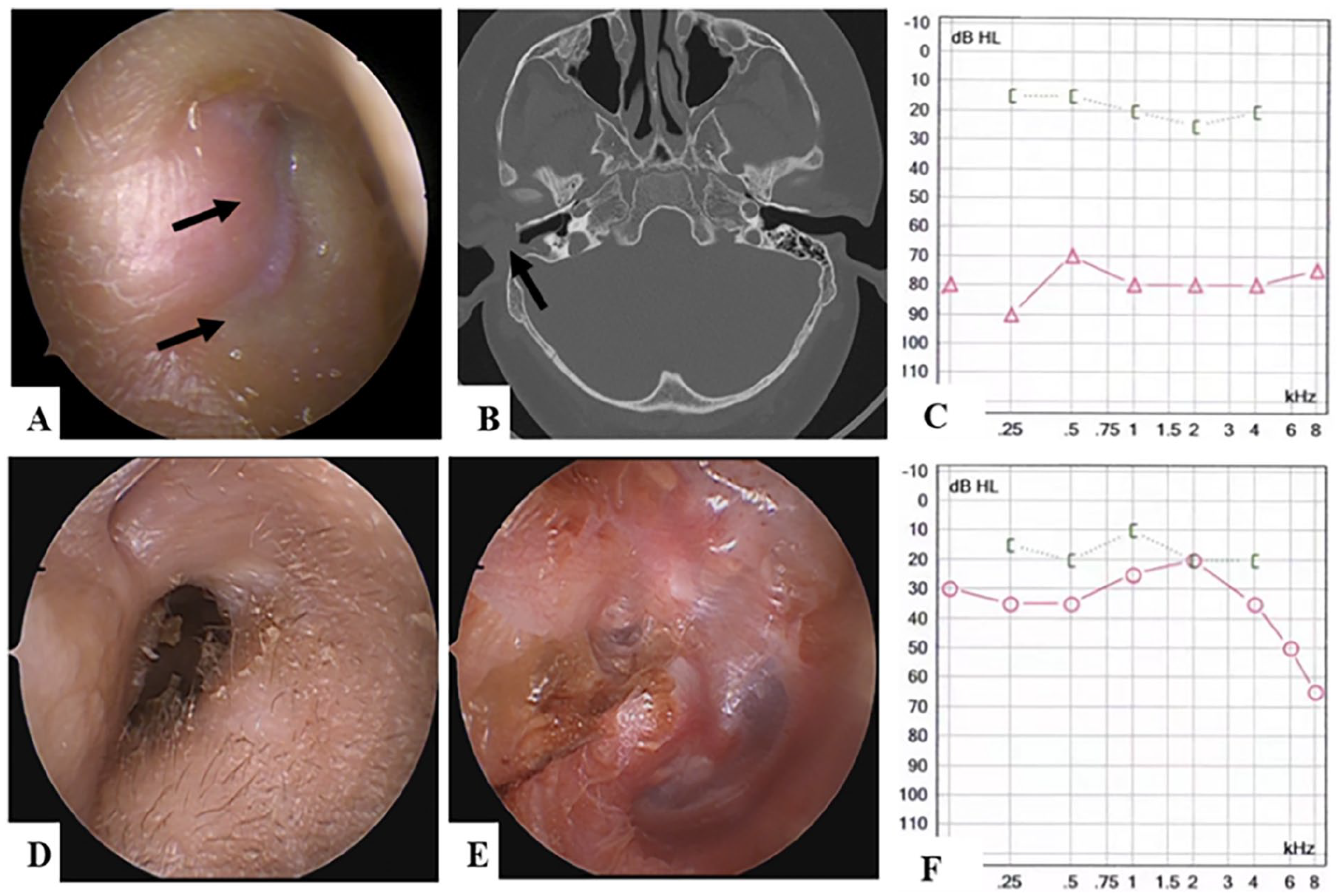
Patient 3 (right ear). (A-C) Preoperative examinations. (A) An atretic tissue (black arrow) was observed in otoscope; (B) Soft tissue blockage (black arrow) of the EAC was observed in CT; (C) ABG value >50 dB HL. (D-F) Outcomes of 1-year postoperatively. (D, E) Otoscopic manifestations, a stable-widened EAC, and intact tympanic membrane obtained; (F) Hearing level significantly improved with an ABG value of 12.5 dB HL. EAC, external auditory canal; CT, computed tomography; ABG, air-bone gap.
Patient 4
A 20-year-old man presented with hearing loss and otalgia in the left ear. He had a surgical history of cranial bones with fibrous dysplasia (FD). The relevant tests were carried out, and the patient was diagnosed with acquired aural stenosis caused by FD (Figure 5A-D). A retroauricular incision, canalplasty, and meatoplasty were performed. The bony EAC was drilled to 12 to 15 mm to completely observe the tympanic cavity (Figure 5E). There was no cholesteatoma in the EAC, and the eardrum was intact. A postauricular free flap was used to reconstruct the EAC in a tube (Figure 5F). The new EAC was supported with a plastic sheet and trimmed-expansive sponge as described above. Regular follow-up with EAC clearance and exchange of trimmed-expansive sponge was performed as previously mentioned. A widened and stable EAC was achieved at 6 months postsurgery, and the hearing level notably increased without decline for 2 years (Figure 5G-H).
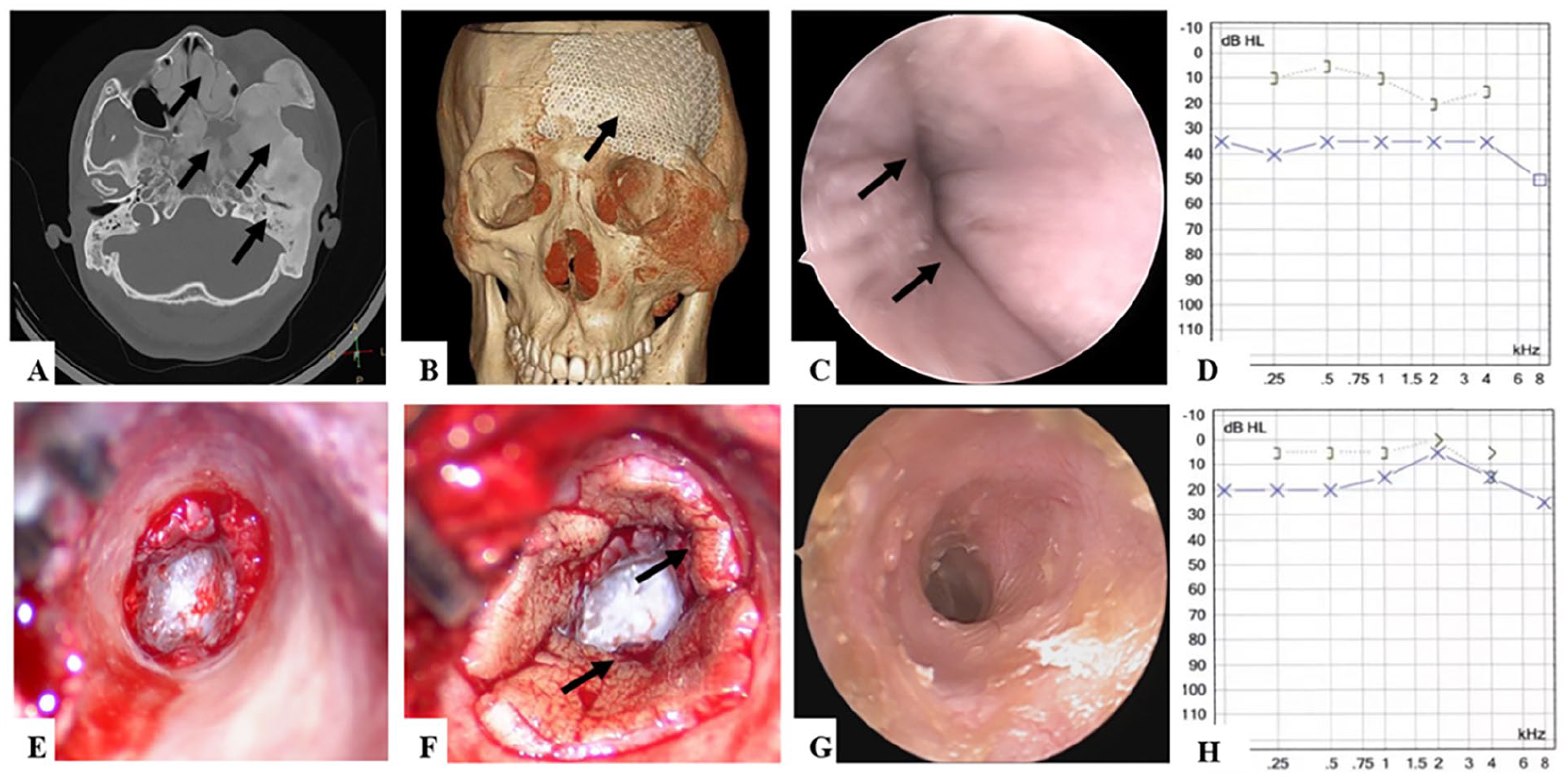
Patient 4 (left ear). (A-D) Preoperative examinations. (A) Patient 4 had the polyostotic type with multiple bones involvement (black arrow) showed in CT; (B) titanium mesh (black arrow) from previous surgery of cranial bones FD displayed in CT volume rendering; (C) Obvious stenosis (black arrow) of the EAC presented at otoscope; (D) ABG value was 22.5 dB HL. (E) Intraoperatively, the EAC diameter was widened to 12 to 15 mm. (F) A postauricular free flap was used to reconstruct EAC in a tube (black arrow). (G, H) A satisfactory result of 2 years postoperatively. (G) There was no re-stenosis occurrence in otoscope; (H) the ABG value decreased compared to preoperative level. CT, computed tomography; EAC, external auditory canal; ABG, air-bone gap.
Discussion
In this study, although patient selection was primarily based on different disease etiologies, 3 patients were male, aged 11 to 63 years, and 2 patients had right ear involvement. Both aural stenosis and aural atresia may trap epithelial cells and potentially carry a high risk of developing cholesteatoma. 11 In this study, cholesteatoma was found in Patient 1 and Patient 3. Surgical intervention is the optimal treatment modality, but the high recurrence rate still challenges ENT surgeons. Patient 2 had previously undergone surgery for acquired aural stenosis, which failed 6 months postoperatively. Surgical techniques for acquired aural stenosis and aural atresia are crucial2,9,10; however, in clinical practice, preventing recurrence and long-term postoperative management are also highly important.
Inflammation and trauma account for a large proportion of cases of acquired aural stenosis and aural atresia.10,12 Inflammation commonly derives from chronic otitis media and external otitis, which trigger an imbalance in the environment and damage the epithelial tissues of the EAC. Bonding and Tos displayed the gradual progress from the inflammatory stage to atresia formation in the following steps: epithelial damage, granulation formation, the formation of granulomatous fibrosis, and the development of a fibrous plug plate. 13 One study classified these pathological conditions into 2 stages: the wet stage and the dry stage. 14 Patient 1 initially presented with otorrhea and the medicine therapy could not result in a good outcome. Hence, recurrent inflammation or wet conditions may lead to subsequent disease progression-acquired aural stenosis or aural atresia. Surgical intervention is the standard treatment for the dry stage of hearing loss.
Patient 2 experienced a bloody left ear after maxillofacial trauma and did not receive reasonable treatment. Lin’s nationwide population-based study revealed that traumatic brain injury (TBI) significantly increased the likelihood of cholesteatoma and stenosis by 1.8- and 1.5-fold in contrast to patients without TBI. 9 Posttraumatic aural stenosis or atresia may stem from excessive inflammation, which occurs as a response to partial or circumferential loss of the epithelium. 12 Lin speculated that the potential mechanism of acquired cholesteatoma in relation to trauma was epithelial entrapment at the fracture line or tympanic membrane implantation into the middle ear and epithelial cells trapped in the EAC. 3 Zhang’s retrospective analysis demonstrated that the interval from trauma to cholesteatoma formation varied from 1 month to 30 years. 15 Hence, ENT specialists should not ignore early identification and treatment and should perform TBI follow-up. Early examinations involving otoscopy, CT, and audiological evaluation are conducive to identifying the effects of trauma. EAC support at an early stage also helps decrease the incidence of acquired aural stenosis or atresia. Owing to the association of cholesteatoma extension with disease course and stenotic severity, surgery is recommended for patients with aural atresia and aural stenosis with a diameter ≤1 mm once the general condition and ear condition are stable. 15
Patient 3 had previously undergone surgery for cholesteatoma in the EAC and postoperatively developed hypertrophic scars. Surgery associated with aural stenosis and aural atresia is due to circumferential loss of the epidermis and periosteum in the EAC without adequate coverage, and lateral blunting in overlay myringoplasty.12,16 Hence, avoidance of unnecessary damage to healthy skin is essential, preservation of diseased skin is sometimes necessary, and slightly residual cholesteatoma is allowed to prevent circumferential loss. Occasionally, as observed in Patient 3, keloid-like tissue in the incision leads to postoperative canal atresia or stenosis. 12 In addition to regular preoperative evaluations, obtaining a comprehensive medical history contributes to favorable outcomes. For patients who may develop hypertrophic scars or postoperative stenosis induced by a keloid-like tissue, local triamcinolone acetonide injections at an early stage or during surgery could effectively decrease the incidence of restenosis. 17 A combination of surgery followed by low-dose radiotherapy may also help to control potential recurrence. 18
Patient 4 presented with acquired aural stenosis caused by FD. Numerous neoplasms lead to acquired aural stenosis, and cutaneous malignancies are responsible for the majority of cases. 19 Aural stenosis caused by FD is uncommon because FD is a rare skeletal disorder, and temporal bone involvement accounts for nearly 10% of FD cases.20 -23 It progresses slowly and presents at any age, predominantly impacting individuals aged ≤30 years. 24 FD is classified into 3 categories depending on the distribution of lesions and related presentations: monostotic FD, polyostotic FD, and McCune–Albright syndrome.20,23 Patient 4 had the polyostotic type with the involvement of multiple bones, including the craniofacial bone, temporal bone, and sphenoid bone. Most studies recommend close clinical follow-up for asymptomatic patients because intervention is unnecessary for the majority of FD patients.21,23 Nevertheless, the incidences of aural stenosis and cholesteatoma of the temporal bone in patients with FD are 23% and 3% to 27%, respectively.21,23 Surgery is required in patients with obvious symptoms due to stenosis (as in Patient 4) or cholesteatoma. FD progresses slowly during adolescence, but its progression is relatively stable in adulthood. Hence, a higher recurrence risk should be considered in adolescent patients. In Patient 4 (an adult), a widened EAC was obtained and the patient did not develop restenosis from temporal bone FD.
Surgical outcomes are closely related to surgical techniques. Retroauricular and endaural incisions are universally adopted for acquired aural stenosis and atresia according to the surgeon’s preferences and related complications. We employed these 2 approaches in our daily practice. An endaural incision could further widen the opening of the EAC through the rotated flap from the cavum conchae. 6 A retroauricular incision could provide better visibility, and facilitate handling unpredictable conditions and skin transplantation from the postauricular region. Canaloplasty is indispensable for obtaining a widened EAC to effectively prevent recurrence. It is also convenient for intraoperative observation and operation. Generally, an EAC diameter of 12 to 15 mm with complete observation of the tympanum is optimal. Furthermore, meatoplasty is also essential for acquired aural stenosis and atresia. Acquired atresia frequently occurs in cartilaginous segments and bony‒cartilaginous junctions. 12 Simple excision of the fibrotic plug or atresia tissue contributes to treatment failure, and lining the denuded EAC with a skin flap could significantly improve treatment outcomes.12,16,25 Numerous grafting flaps have been described, including regional flaps, split skin grafts, and full-thickness skin transplantations.6,12,26,27 Skin grafts from the postauricular region and full-thickness skin grafts are frequently used in our practice because they are easily harvested via the retroauricular approach. However, these grafts lack ceruminous and sebaceous glands, leading to the absence of self-purification and anti-infection functions. Therefore, a study proposed that skin grafts transplanted from healthy EACs decrease the restenosis rate. 28
Postoperative management and follow-up are mandatory. Various materials have been used to pack EACs, including ribbon gauze, steel and silicone tubes, and some devices.6,29,30 In our clinical practice, iodoform sponges and particularly expansive sponges are employed to maintain EAC patency. Expansive sponges are low-cost, easy to trim, and provide pressure to the EAC via water or ototopical drops. However, iodoform sponges and expansive sponges pose a risk of adhering to the new EAC. A “Swiss roll” plastic sheet with an expansive sponge that fills the lumen could reduce the likelihood of adherence. Compared with silicone sheets, plastic sheets are relatively thin and malleable. Packing materials have been used in situ for 1 to 6 weeks in different studies.12,16 In our daily practice, the “Swiss roll” is removed 3 weeks postsurgery, and a new expansive sponge is placed. For infection prevention and observation, expansive sponge replacement and ear canal clearance were conducted once every 3 weeks from 3 to 6 months until a stable ear canal was achieved. The support period could be extended if necessary. However, excessive cleaning may injure the new skin of the ear canal and increase the probability of restenosis.
The risk of recurrence or restenosis in patients with acquired aural stenosis and aural atresia after canalplasty or meatoplasty remains greater than that in patients with other disorders.12,25 The mechanism remains unclear, but most studies have shown it may be due to the fibroproliferative inflammatory response and hyperplasia scarring. Mitomycin C has been intraoperatively used to reduce restenosis risk, and good results were obtained in 4 patients within 6 years. 31 However, there have been no further studies on a larger number of patients. The application of mitomycin C has several limitations: it cannot be repeated if it does not initially prevent recurrence, and a one-size-fits-all approach for patients may be unnecessary. Triamcinolone acetonide in local skin, which has a low recrudescence rate, is a feasible option. 32 A 73.3% (11/14) satisfaction rate was obtained from injection with support treatment after surgery for EAC stenosis, which was repeated 1 to 6 times at a frequency of 3 to 5 weeks. 17 Typically, a well-epithelialized and stable EAC is obtained 3 to 24 months postsurgery.31,33 However, the recurrence at 1 to 2 years is 21%, and patients with concomitant cholesteatoma may have a problematic treatment process. 16 Hence, regular long-term follow-up is conducive to early identification and intervention.
Conclusion
Prevention in clinical practice is conducive to preventing and decreasing the incidence of acquired aural stenosis and aural atresia. Surgical intervention is the gold standard for acquired aural stenosis and aural atresia. Surgical techniques, including widening the EAC via canalplasty or meatoplasty, native healthy skin preservation, and coverage with skin grafts, improve surgical success. Long-term follow-up is mandatory, and regular postoperative care consisting of EAC support and clearance is indispensable. Furthermore, local injections of triamcinolone acetonide are beneficial for managing early stage restenosis. However, this study was a case series, and a large-scale study is needed.
Footnotes
Acknowledgements
The authors thank these 4 patients for their consent.
Author Contributions
The study was designed by Y.H., and all of the operations were conducted by her. R.Y. and Y.Z. collected data and wrote this manuscript. R.L. manipulated the figures. C.Z., M.D., W.W., and X.L. participated in the patient follow-up. Some suggestions were provided by D.Z. and the revision work was conducted by him. All authors performed data interpretation and approved the manuscript.
Consent for Publication
Written informed consent was obtained from these patients or their parents.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81870719), the Foundation of Shaanxi Province (2023-YBSF-209). This project’s recipient is Yu Han.
Ethics Approval and Consent to Participate
This research was approved by the Medical Ethics Committee of the First Affiliated Hospital of the Air Force Medical University (KY20232313-C-1).
