Abstract
For most cystic craniopharyngiomas, intracapsular debulking is a good strategy to get a large operation space and protect vital structures. However, this surgical strategy may lead to the residual and recurrence of the tumor capsule wall. Therefore, there is an urgent need for a new surgical strategy without residual capsule walls for the removal of cystic craniopharyngiomas. We reviewed a 45-year-old male with vision loss and visual field defects, whose head MRI revealed a suprasellar cystic lesion. The patient underwent extended endoscopic transsphenoidal surgery. The surgical strategy of total cystic wall decollement was adopted, which was that the lesion surrounded by the capsule was completely separated from the surrounding tissue without destroying the capsule and maintaining the tension of the capsule. The lesion was totally resected and pathological findings confirmed the diagnosis of craniopharyngioma. After the operation, both the visual acuity and pituitary function were significantly improved. In addition, he suffered from transient diabetes insipidus, which was subsequently relieved. During the 33-month follow-up, there was no tumor recurrence. Compared with the traditional surgical strategy of intracapsular debulking, the surgical strategy of total cystic wall decollement has the advantages of less residual tumor capsules, low tumor recurrence rates, etc. Therefore, for specific cystic craniopharyngiomas, the surgical strategy of total cystic wall decollement may be an effective surgical strategy to reduce tumor recurrence.
Keywords
Introduction
Craniopharyngiomas are rare congenital benign tumors originating from remnants of the pouch of Rathke. 1 The incidence rate of craniopharyngioma is only 0.13 cases per 100,000 people and presents a bimodal age distribution, that is, the peak age of incidence is mainly concentrated in 5 to 14 years old (children) and 50 to 74 years old (elderly). 2 Due to the space-occupying effect, the common symptoms of craniopharyngioma are visual impairment, hypopituitarism, and symptoms of increased intracranial pressure (headache and vomiting), etc. According to the tumor location, craniopharyngiomas can be divided into pure intrasellar, intrasellar, suprasellar, and pure suprasellar types. 3 While according to the tumor texture, craniopharyngiomas can be divided into cystic, solid, and mixed types. 4 Among these, cystic craniopharyngioma is characterized by cystic components in the tumor, which can become voluminous and may lead to obstructive hydrocephalus or compression of the optic nerve and optic chiasma. Generally, surgery is the first choice for the treatment of craniopharyngiomas. Because craniopharyngiomas are located in the skull base, endoscopic endonasal transsphenoidal surgery (EETS) is very suitable for resection of craniopharyngiomas and has become the standard surgical method. At present, the common surgical strategy for the removal of cystic craniopharyngiomas is intracapsular debulking, which is conducive to getting a large operation space and protecting important structures, such as the hypothalamus, pituitary gland, and optic nerve. However, this surgical strategy also has limitations, such as tumor residual and tumor recurrence. To break through these limitations, we proposed a surgical strategy of total cystic wall decollement, which is that the tumor surrounded by the capsule is completely separated from the surrounding tissue without destroying the capsule and maintaining the tension of the capsule. In this paper, we report a case of suprasellar cystic craniopharyngioma who underwent EETS with total cystic wall decollement.
Case Presentation
History and Examination
A 45-year-old male went to other hospitals for examination due to vision loss and visual field defect. Cranial MRI revealed a suprasellar cystic lesion, and then he was referred to our department. The preoperative clinical examination suggested 1-m blurred vision and temporal visual field defects in both eyes, while the hormone examination indicated pituitary dysfunction. Figure 1A and B are the preoperative sagittal and coronal T1-weighted MRI, respectively, presenting a suprasellar cystic lesion, marginal calcification, pituitary compression, and hypointense, which all suggest that the lesion is a suprasellar cystic craniopharyngioma.
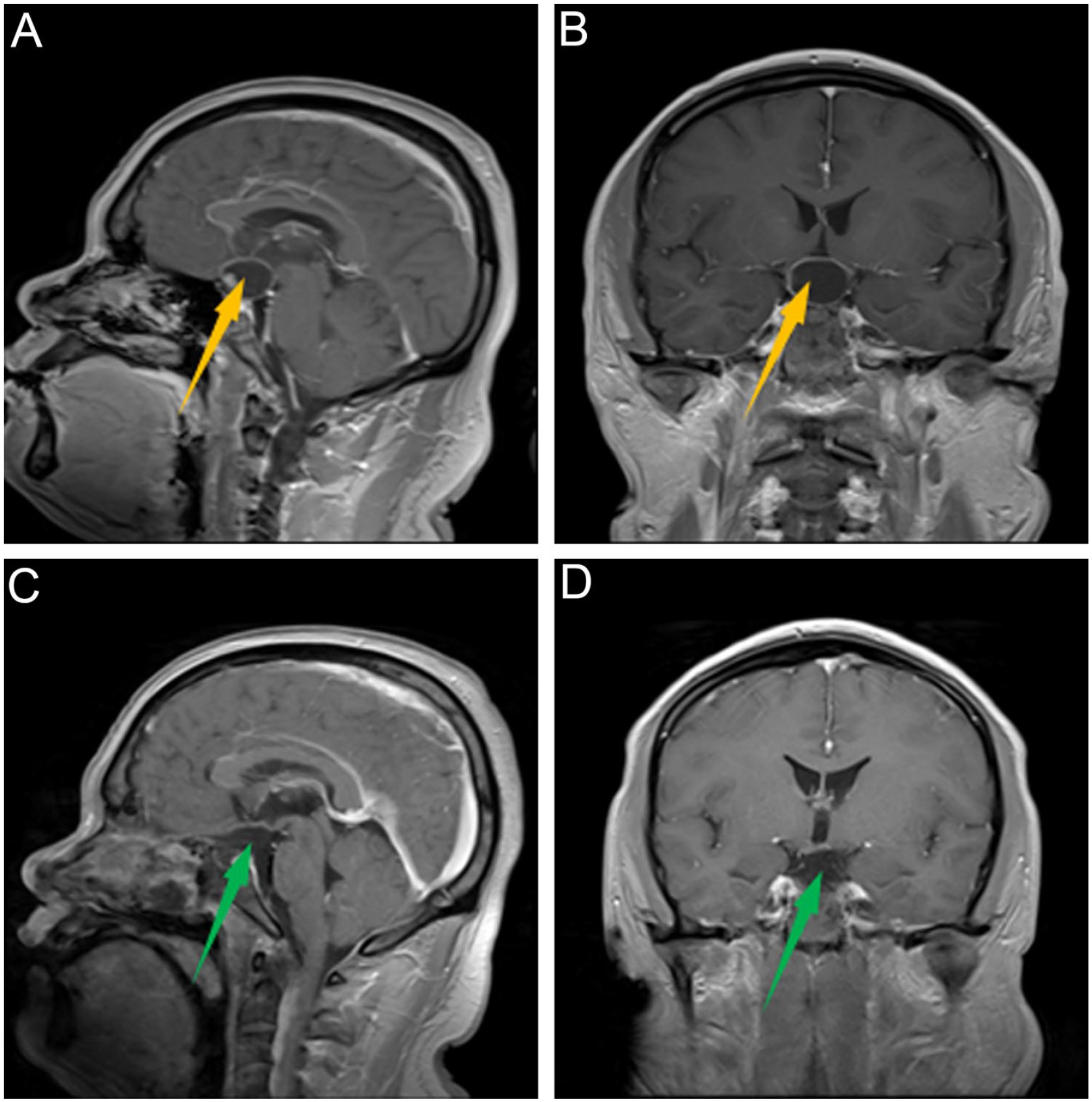
Preoperative and postoperative MRI. (A) preoperative sagittal MRI, (B) preoperative coronal MRI, (C) postoperative sagittal MRI, (D) postoperative coronal MRI. The yellow and green arrow directions are a preoperative tumor and vacant after tumor resection, respectively.
Operation
Under general anesthesia, the patient was placed in the supine position with the head tilted slightly to the right. First, the surface projection of the tumor was located under neuronavigation and the endoscopic was placed along the right nasal cavity. Under the endoscopic, the middle turbinate resection was used to reveal the opening of the right sphenoid sinus, and the dura mater and intercavernous sinus were exposed, as shown in Figure 2A. Then, the dura mater was opened, and the cavernous sinus was coagulated, the arachnoid above the pituitary stalk was dissected, and the cystic lesions containing straw yellow cystic fluid were displayed, as shown in Figure 2B. After the tumor was visible, the tumor and surrounding tissues were separated along the tumor capsule wall without destroying the tumor capsule wall and maintaining the integrity of the capsule wall. During the separation process, we found that the lateral capsule wall of the tumor adhered to the optic chiasma, as shown in Figure 2C, and the deep capsule wall of the tumor adhered to the bottom of the third ventricle, as shown in Figure 2D. The cystic wall of the tumor was slowly separated from the adhesive tissue. By slowly separating the tumor capsule wall and adhesive tissue, the tumor wrapped by the capsule wall was completely stripped off. Figure 2E shows that the tumor originated from the pituitary stalk. To achieve gross total resection (GTR), the tumor was removed from the pituitary stalk, as shown in Figure 2F. During the operation, we did not destroy the tumor capsule, maintained the tension of the tumor capsule, and completely stripped the capsule. Finally, the skull base was repaired with a vascular nasal septal mucosal flap, gelatin sponge, and artificial dura mater. In particular, it should be pointed out that in the process of peeling off the tumor capsule wall to cutting off the tumor origin, the capsule wall is always kept intact so that it has enough tension, which is more conducive to the separation of the capsule wall from the surrounding adhesive tissue with no residual tumor.
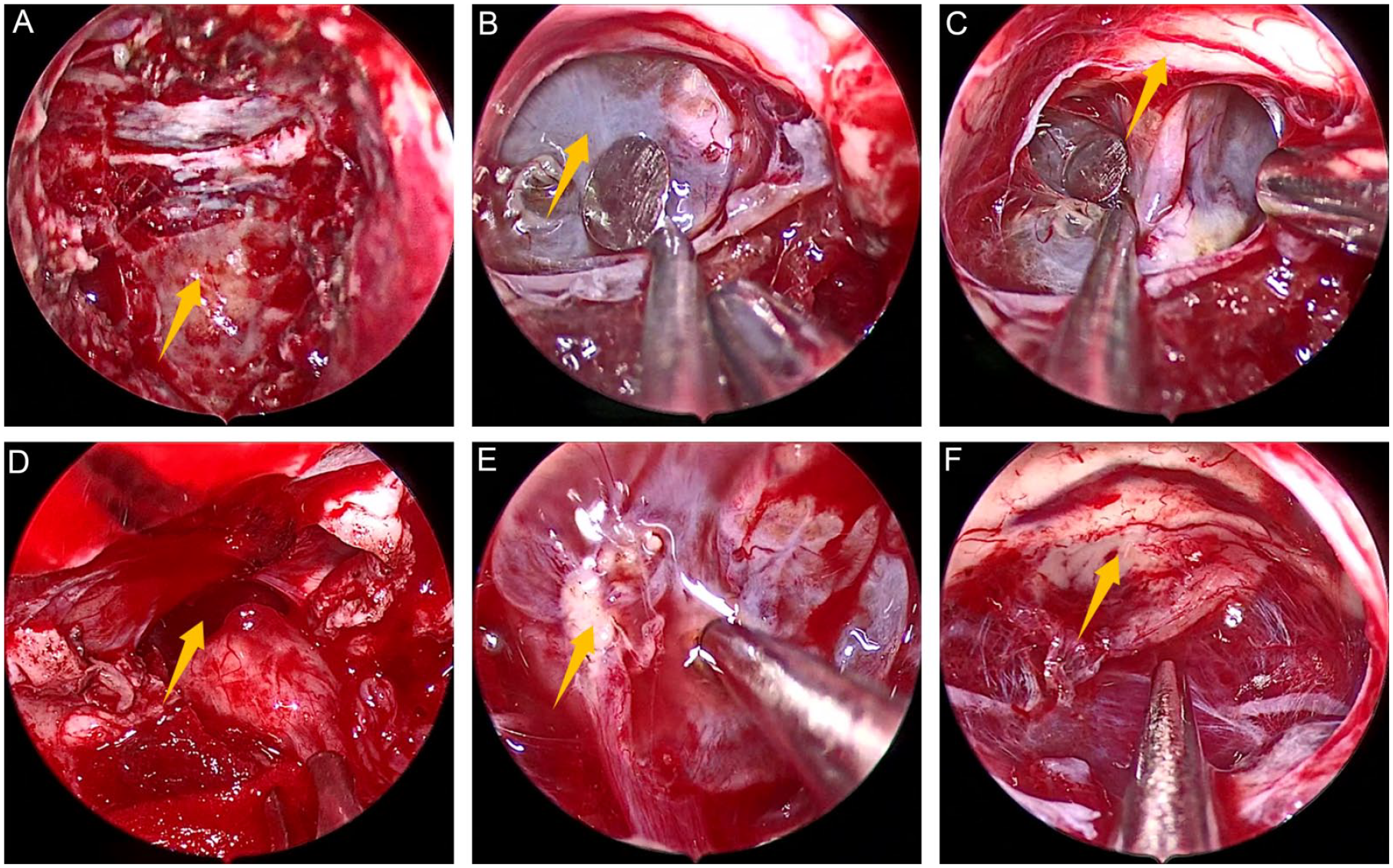
Intraoperative images. (A) exposure of dura mater, (B) exposure of tumor, (C) exposure of optic chiasma, (D) exposure of the bottom of the third ventricle (E) relationship between pituitary stalk and tumor, (F) complete resection of tumor. In (A-F), the directions of arrows are dura mater, tumor capsule, optic chiasma, the bottom of the third ventricle, pituitary stalk, and tumor cavity respectively.
Postoperative Course
After surgery, his vital signs were stable and pathological findings confirmed the diagnosis of craniopharyngioma. Clinical examination indicated visual disturbance improved, and hormone examination indicated mild hypopituitarism. On the third day after surgery, cranial MRI showed that there was no secondary intracranial hemorrhage and the tumor was grossly totally resected (Figure 1c and d). During hospitalization, he suffered transient diabetes insipidus, which was relieved at a 4-week follow-up. During the 33-month follow-up, there was no tumor recurrence.
Discussion
Craniopharyngioma is a rare benign tumor originating from remnants of the pouch of Rathke, which can lead to increased intracranial pressure, optic nerve compression, and pituitary stalk compression, thus presenting clinical symptoms such as headache, vomiting, visual impairment, pituitary dysfunction, etc. 5 Regardless of the type of craniopharyngioma, surgery is always the first choice. The location, size, and shape of the tumor are common factors in choosing the surgical approach, with the location of the tumor being particularly crucial. Kassam et al classified craniopharyngioma into 4 types based on the relationship between the tumor and the infundibulum. 6 Specifically, Type I, preinfundibular; Type II, transinfundibular; Type III, retroinfundibular; IIIa, retroinfundibular/third ventricular; IIIb, retroinfundibular/interpeduncular cistern and Type IV, purely third ventricular. The prechiasmatic-preinfundibular craniopharyngioma (Type I) is located in the suprasellar cistern and extends from the subchiasmatic to the prechiasmatic space, with the pituitary stalk located at its posterior edge. For this tumor, the pterional approach (PA) or the subfrontal approach (SA) can effectively remove it. 7 The subchiasmatic-transinfundibular craniopharyngioma (Type II) grows along the long axis of the infundibulum, widens the stalk, occupies the subchiastic space, and expands through tuber cinereum to invade or elevate the bottom of the third ventricle. The upper portion of the tumor is closely related to theanterior of hypothalamus. For this type of tumor, the PA and SA are equally applicable. 5 The retrochiasmatic-retroinfundibular craniopharyngioma (Type III) occupies the interpeduncular cistern and retroinfundibular space, often extending downward into the posterior fossa and occasionally extending upward into the third ventricle, which can cause the midbrain to move backward and the optic chiasm to move forward. The orbitozygomatic modification of the pterional approach craniotomy is recommended to improve the exposure of the contents of the interpeduncular cistern and enhance control of the neurovascular structure. 8 Although PA can treat the retrochiasmatic-retroinfundibular tumor craniopharyngioma, it requires manipulation of neurovascular structures to access the interpeduncular space and makes it difficult to visualize the hypothalamic surface. 9 Traditionally, the purely third ventricular craniopharyngioma (Type IV) is resected using an open microsurgical approach, including transendocortical approach and transventricular approach. 10 In fact, EETS is not only suitable for the treatment of skull base lesions but also has an excellent view, which makes it a standard method for the removal of the 3 types of craniopharyngiomas except for the pure third ventricle.11-13 For tumors of different sizes, there is still no clear standard for determining the surgical approach. 14 Generally, different approaches are adopted based on actual conditions and doctors’ preferences.
Table 1 shows the cases of cystic craniopharyngioma resected by surgery combined with intracapsular debulking of surgical strategy in recent years, including the number of patients, tumor location, surgical approach, and GTR rate, follow-up time, recurrence, postoperative complications, adjuvant therapy, etc.15-17,15-38 It can be seen from Table 1 that most of the cystic craniopharyngiomas were located in the sellar and suprasellar regions, and occasionally in the intraventricular, orbital cavity, and foramen magnum. The surgical approaches for craniopharyngioma are the endoscopic transorbital eyelid approach, endoscopic transcortical transventricular approach, endoscopic endonasal transsphenoidal approach, fronto-basal interhemispheric approach, sublabial-transseptal approach, microsurgery PA, microsurgery transsphenoidal approach and SA, etc. The GTR rate ranged from 35.2% to 100%, and the recurrence rate ranged from 0% to 82.0%.18,27 Im et al reported that GTR was achieved in all 6 patients, but 1 patient relapsed, which may be due to a small amount of residual cyst wall of the tumor not found by intraoperative endoscopic images and postoperative cranial MRI. 35 More residual cyst wall leads to a higher recurrence rate and reflects the fact that the surgical strategy of intracapsular debulking brings a higher risk of residual tumors and tumor recurrence. However, Zhu et al reported 8 patients (72.7%) who obtained GTR, but no patients relapsed, which may be due to the short follow-up period (18.5 months). 20 To prevent the recurrence caused by residual tumors, postoperative radiotherapy has also become a necessary adjuvant treatment and is widely used.19,23,32,33,36 Unfortunately, although postoperative adjuvant radiotherapy has been performed, there will still be tumor recurrence. Therefore, some patients must undergo reoperation or multiple operations,15,30 which brings huge pain and economic burden to patients. The main postoperative complications were cerebrospinal fluid leakage (CSFL), diabetes insipidus (DI), cerebral meningitis, hydrocephalus, hyposmia, intracranial infection, etc. Among them, CSFL and new onset DI were relatively high,20,39 which may be caused by surgery and the deficiency of the pituitary hormone. In addition, the occurrence of meningitis and intracranial infection may also be attributed to the release of cystic fluid. Considering the GTR, residual cyst wall, recurrence, and postoperative complications, intracapsular debulking is not a perfect surgical strategy. The main problem of intracapsular debulking is that once the tumor capsule is opened, the tension of the tumor capsule wall disappears, making it difficult for the tumor capsule to completely separate from the surrounding normal tissues, resulting in the residual and recurrence of the tumor capsule wall. Therefore, we propose a surgical strategy of total cyst wall decollement, that is, the tumor around the cyst wall is completely separated from the surrounding tissues, without destroying the cyst wall and maintaining the tension of the cyst wall, which is conducive to the separation of the tumor from the surrounding tissues.
Reported Cases of Cystic Craniopharyngioma Treated by Surgery.
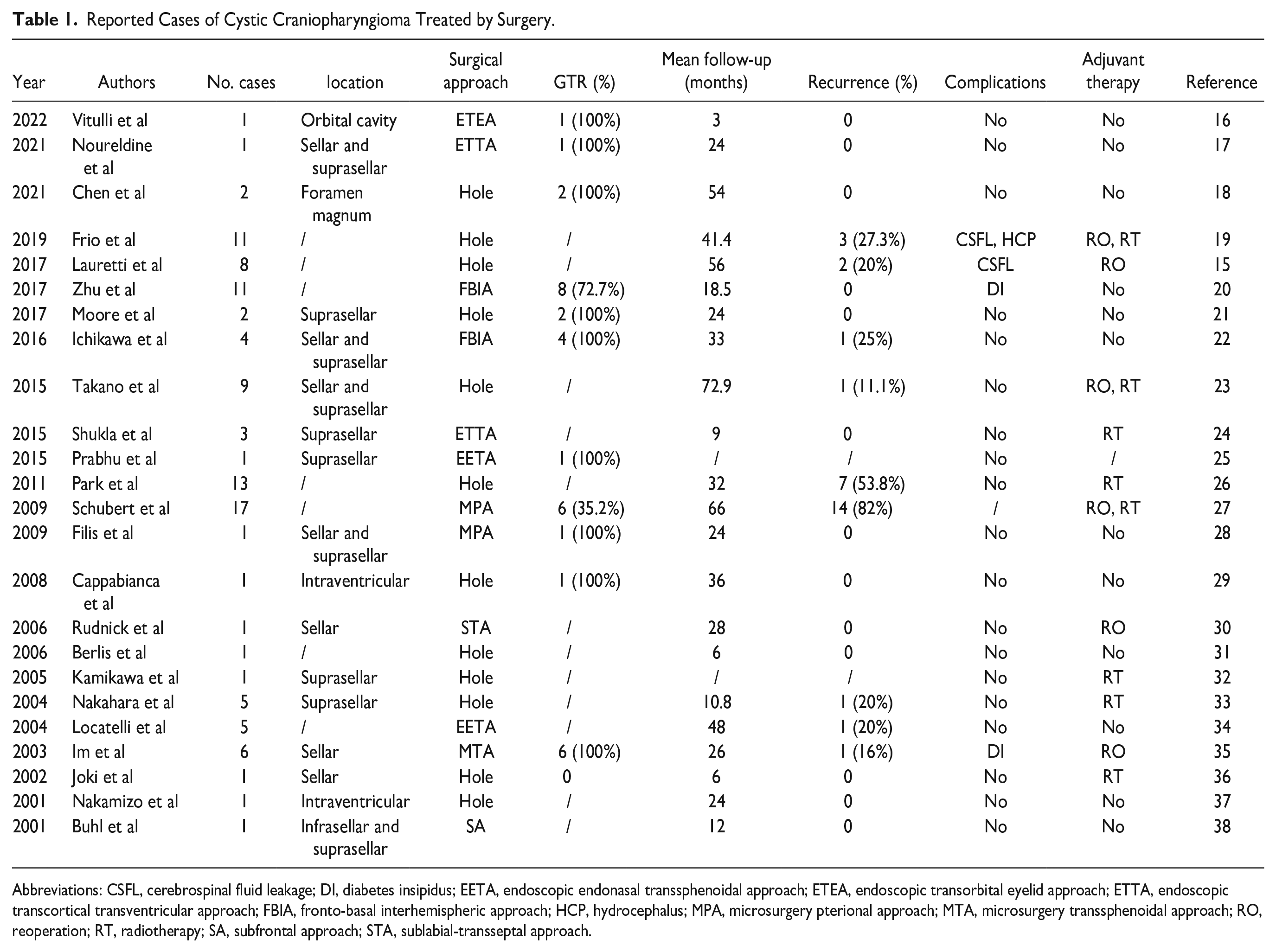
Abbreviations: CSFL, cerebrospinal fluid leakage; DI, diabetes insipidus; EETA, endoscopic endonasal transsphenoidal approach; ETEA, endoscopic transorbital eyelid approach; ETTA, endoscopic transcortical transventricular approach; FBIA, fronto-basal interhemispheric approach; HCP, hydrocephalus; MPA, microsurgery pterional approach; MTA, microsurgery transsphenoidal approach; RO, reoperation; RT, radiotherapy; SA, subfrontal approach; STA, sublabial-transseptal approach.
In this paper, a 45-year-old male with suprasellar cystic craniopharyngioma underwent EETS with total cyst wall decollement. During the operation, the tumor wrapped in the capsule wall is completely stripped off without destroying the tumor capsule and maintaining the tension of the tumor capsule. Finally, the tumor was cut from the pituitary stalk to achieve total tumor resection. After the operation, the visual acuity and pituitary function were greatly improved. There was no CSFL, meningitis, and other serious complications. Only transient DI appeared and was relieved later. There was no adjuvant radiotherapy after the operation. During the 33-month follow-up, there was no tumor recurrence. These results show that the EETS with total cyst wall decollement is a safe and effective surgical method for specific cystic craniopharyngiomas.
Conclusion
We retrospectively analyzed a case with suprasellar cystic craniopharyngioma treated by extended endoscopic transsphenoidal surgery. The tumor achieved GTR with no recurrence during follow-up. After the operation, both the visual acuity and pituitary function were significantly improved and there were no serious complications. A surgical strategy of total cyst wall decollement was adopted during the operation. Specifically, the tumor around the cyst wall is completely separated from the surrounding tissue without destroying the cyst wall and maintaining the tension of the cyst wall. Compared to the common surgical strategy of intracapsular debulking, which may lead to residual and recurrence of the tumor capsule wall, the surgical strategy of total cystic wall decollement has the advantage of obtaining a higher resection rate and lower recurrence rate and preservation of pituitary function.
Footnotes
Acknowledgements
None.
Author Contributions
J.L., J.W.W., P.W., N.W. contributed to the study conception and design. J.L. and J.W.W. were material preparation, data collection. J.L. and P.W. were analysis and interpretation of data. J.L. and N.W. were drafting the article and revising. All authors read and approved the final manuscript.
Consent
Written informed consent was obtained from the patient for publication and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This article was approved by the committee ethics of Chongqing General Hospital.
