Abstract
Keywords
Introduction
Symptoms of chronic rhinitis include runny nose, congestion, itching, sneezing, and postnasal drip that leads to clearing of the throat and coughing. The severity of these symptoms can substantially impact a person’s quality of life.1-3
The NEUROMARK® System (Neurent Medical Ltd, Oranmore, Galway, Ireland) is an innovative multipoint, impedance-controlled, radiofrequency ablation device for the treatment of chronic rhinitis. The device uses 10 flexible leaflets to simultaneously reach proximal and distal treatment zones on the lateral wall of the nasal cavity. Low-power, bipolar radiofrequency is controlled through impedance thresholds and automatic cutoffs at each leaflet to create microlesions. The device design accomplishes concurrent neurolysis of nerves distributed both anterior and posterior to the middle turbinate attachment. The NEUROMARK console provides detailed feedback to the physician, confirming proper placement and treatment delivery data at each targeted treatment site.
A recent study demonstrated significantly improved symptoms through 3-month follow-up. 4 However, the sample size precluded subgroup analyses for the type of rhinitis and other potential influencing factors. The current study was undertaken to enroll enough participants for subgroup analyses and to examine the treatment effect on additional patient-reported outcomes (PROs) that have not been previously examined in this patient population. Additional symptoms we postulated might improve with treatment included ear symptoms, sleep, and nasal obstruction. We sought to study this prospectively by including the Eustachian Tube Dysfunction Questionnaire (ETDQ-7) and Nasal Obstruction Symptom Evaluation (NOSE) PROs. This is the first paper to include the ETDQ-7 in this patient population.
Patients and Methods
Study Design and Population
We conducted a prospective, single-arm, multicenter clinical study of impedance-controlled radiofrequency ablation of the posterior nasal nerve (PNN) in patients with chronic rhinitis. The study was approved by the Allendale IRB (Old Lyme, CT, USA). All participants signed the IRB-approved written informed consent before participating in the study. The study is registered at www.clinicaltrials.gov (NCT05591989).
Participants were adults (≥18 years) with at least 6 months of chronic rhinitis symptoms. The minimum total reflective Total Nasal Symptom Score (rTNSS) 5 for eligibility was 5 (out of 12), with moderate to severe rhinorrhea (rTNSS ≥ 2), and mild to severe nasal congestion (rTNSS ≥ 1). Participants were required to have an allergy test by a skin prick, intradermal testing, or an IgE immunoassay. Significant exclusion criteria included the presence of significant anatomical obstructions; previous interventions for rhinitis; mucosal erosion/ulceration, history of epistaxis or significant epistaxis event in the past 3 months; atrophic rhinitis; and previous head/neck irradiation. Participants taking anticholinergic sprays were instructed to discontinue use at least 14 days before the baseline visit and through the 6-month visit. Participants were also instructed not to change their baseline concomitant medications before the 6-month follow-up unless required to treat an adverse event.
All participants underwent bilateral PNN ablation using the NEUROMARK System under local anesthesia, according to the investigator’s usual practice, in the office setting.
Assessments
At baseline and at 1-, 3-, and 6-month follow-ups, participants completed the following assessments: rTNSS, postnasal drip and cough symptoms [scale of 0 (none) to 3 (severe)], and NOSE. The ETDQ-7 was completed at baseline and 6 months. The mini-Rhinoconjunctivitis Quality of Life Questionnaire (mini-RQLQ) was completed at baseline and at 3 and 6 months. Additionally, investigators completed the Clinical Global Impression of Improvement (CGI-I) for each participant at 3 and 6 months. Adverse events were noted at all visits.
Statistical Analysis
The primary efficacy endpoint was the change from baseline to 6 months in the total rTNSS. A 2-sided hypothesis using the Student’s t-test for paired differences was assessed on the primary efficacy endpoint. Using 90% power with a 2-sided alpha of .05, a sample size of 17 participants was determined to be adequate using a 1-sample Student’s t-test. The sample size was increased to a planned enrollment of 80 participants to allow for attrition and subgroup analyses.
Continuous data are summarized using descriptive statistics and include mean and SD or median and 95% CI, as appropriate. Changes from baseline were tested using paired t-tests or Wilcoxon signed-rank test. Categorical data are summarized using counts and percentages. An alpha level of .05 was considered statistically significant.
Minimal clinically important differences (MCID) were determined for the rTNSS and mini-RQLQ. A reduction of ≥1 point was used as the MCID for the rTNSS, 5 and a reduction of ≥0.4 points was used for the mini-RQLQ. 6 The rTNSS responder rate was defined as a reduction from baseline of ≥30% in the total score. 7 The NOSE score responder rate was defined as an improvement of ≥20% or 1 NOSE class over baseline.8,9
Two subgroup analyses were planned to compare outcomes for participants by rhinitis type (allergic vs nonallergic) and by baseline rTNSS (5-6 vs 7-9 vs 10-12).
The statistical analysis was performed by an independent statistician using Stata (version 17, StataCorp, LLC, College Station, TX), unless otherwise noted.
Results
Eighty participants were enrolled and treated at 11 clinical centers in the United States. Eighty participants completed the 1-month follow-up and 79 participants completed the 3- and 6-month follow-ups. One participant missed the 3-month visit and a second participant missed the 6-month visit. Both participants remain in the study.
Demographic and baseline medical history data are presented in Table 1. Average age was 59 years, the majority were female (64%), and 12.5% were non-White. A total of 34% of participants had allergic rhinitis and 60% had nonallergic rhinitis.
Demographics and Baseline Medical History.

Results are presented as mean ± SD, or % (n).
Of the 5 participants who had “unknown” allergic status, 2 did not complete allergy testing and 3 completed allergy testing but the results were indeterminant.
Some participants had more than 1 surgery.
History of medications administered to treat chronic rhinitis for durations ranging from 0.5 to 22 years, included corticosteroids (n = 40), antihistamines (n = 30), anticholinergics (n = 14), and decongestants (n = 12). All participants discontinued anticholinergic medications for 2 weeks prior to baseline and throughout the study period.
The primary endpoint was met with a statistically significant change from baseline to 6 months for the total rTNSS (−4.2, 95% CI, −4.8 to −3.6, P < .0001, Table 2). Responder rates at all 3 time frames were ≥75% and ≥90% of participants achieved the MCID. At 6 months, the responder rate was 77.2% (95% CI, 66.4% to 85.9%), and the MCID was 91.1% (95% CI, 82.6% to 96.4%). Additionally, there were statistically significant improvements in all 4 rTNSS subscores (congestion, rhinorrhea, sneezing, and itching) as well as postnasal drip and cough at all follow-up time points (P < .0001 for all, Figure 1).
Change from Baseline in Total rTNSS and Total NOSE Scores.
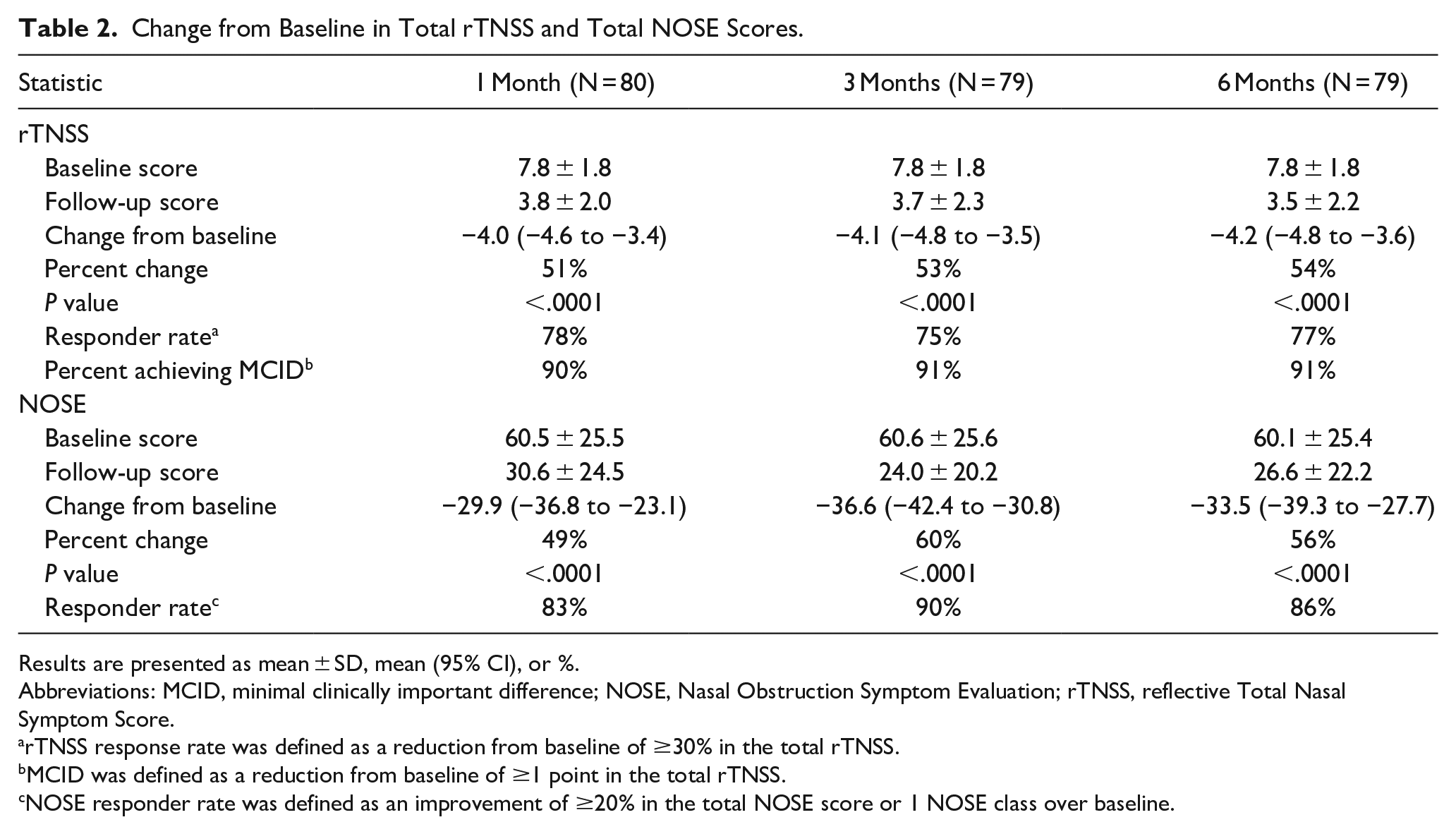
Results are presented as mean ± SD, mean (95% CI), or %.
Abbreviations: MCID, minimal clinically important difference; NOSE, Nasal Obstruction Symptom Evaluation; rTNSS, reflective Total Nasal Symptom Score.
rTNSS response rate was defined as a reduction from baseline of ≥30% in the total rTNSS.
MCID was defined as a reduction from baseline of ≥1 point in the total rTNSS.
NOSE responder rate was defined as an improvement of ≥20% in the total NOSE score or 1 NOSE class over baseline.
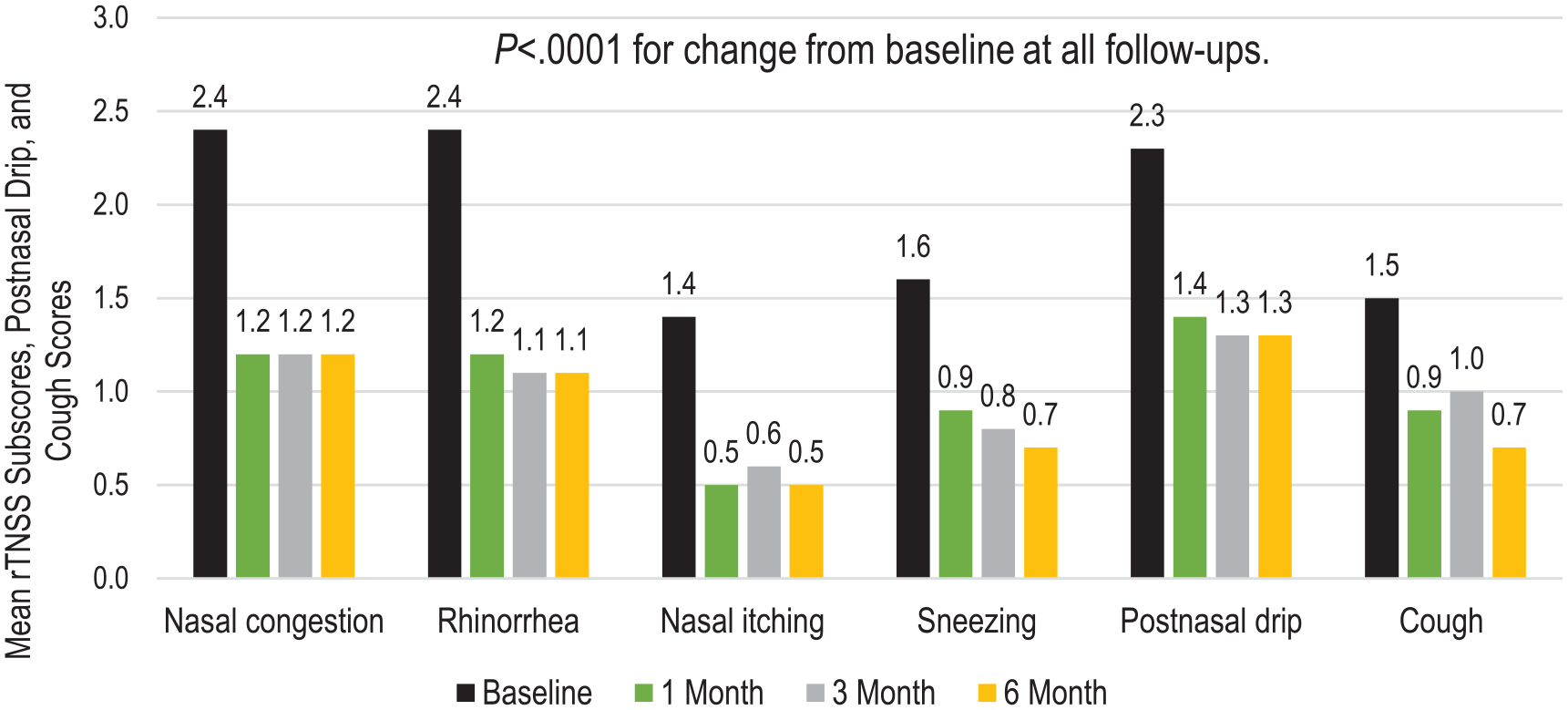
Change in mean rTNSS subscores, postnasal drip, and cough scores.
The ETDQ-7 overall score was significantly improved over baseline at 6 months (−1.2, 95% CI, −1.5 to −0.8, P < .0001, Figure 2). All 7 ETDQ-7 subscores (ear pressure, ear pain, clogged ears, ear problems with cold/sinusitis, crackling/popping, ringing in ears, and muffled hearing) showed significant improvement from baseline at 6 months (P < .0001 for all except P = .002 for ringing in ears). Ear problems with cold or sinusitis (45%), ear pressure (40%), clogged ears (37%), and crackling/popping (37%) subscores showed the greatest percent improvement from baseline.
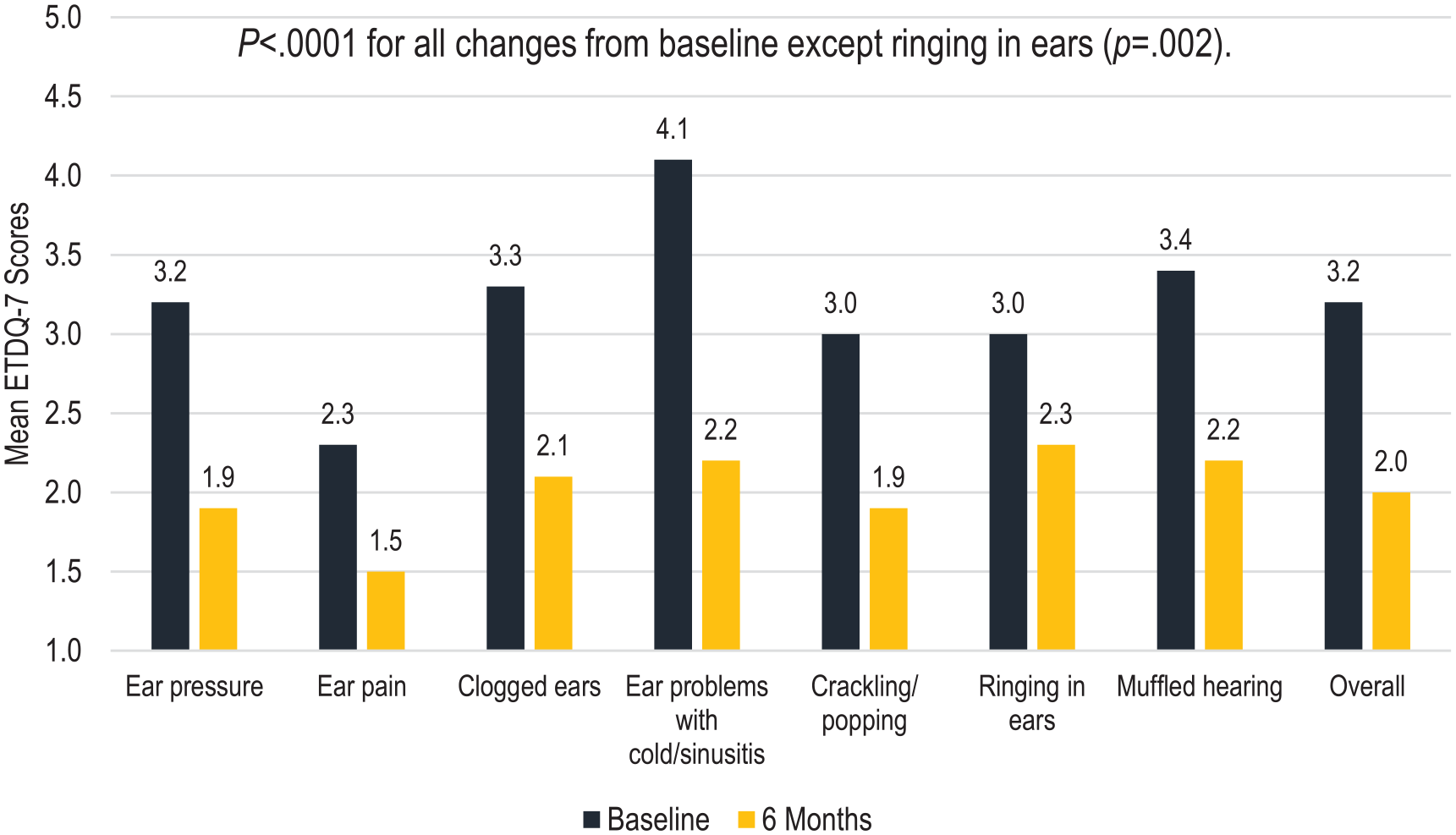
Change in mean ETDQ-7 scores.
Total NOSE scores were significantly improved at all time points (P < .0001) (Table 2). At baseline, the mean total NOSE score indicated severe symptoms (scores 55-75) with improvements at follow-ups to scores that demonstrated mild (5-25) or moderate (30-50) symptoms. Responder rates were 89.9% (95% CI, 81.0% to 95.5%) and 86.1% (95% CI, 76.5% to 92.8%) at 3 and 6 months, respectively. Significant improvement was demonstrated in all 5 NOSE subscores (P < .0001) (Figure 3). The largest percent improvement in NOSE subscores at 6 months was achieved for difficulty getting air through the nose during exercise (61% improvement), followed by trouble sleeping (58% improvement), and trouble breathing through the nose (57% improvement).
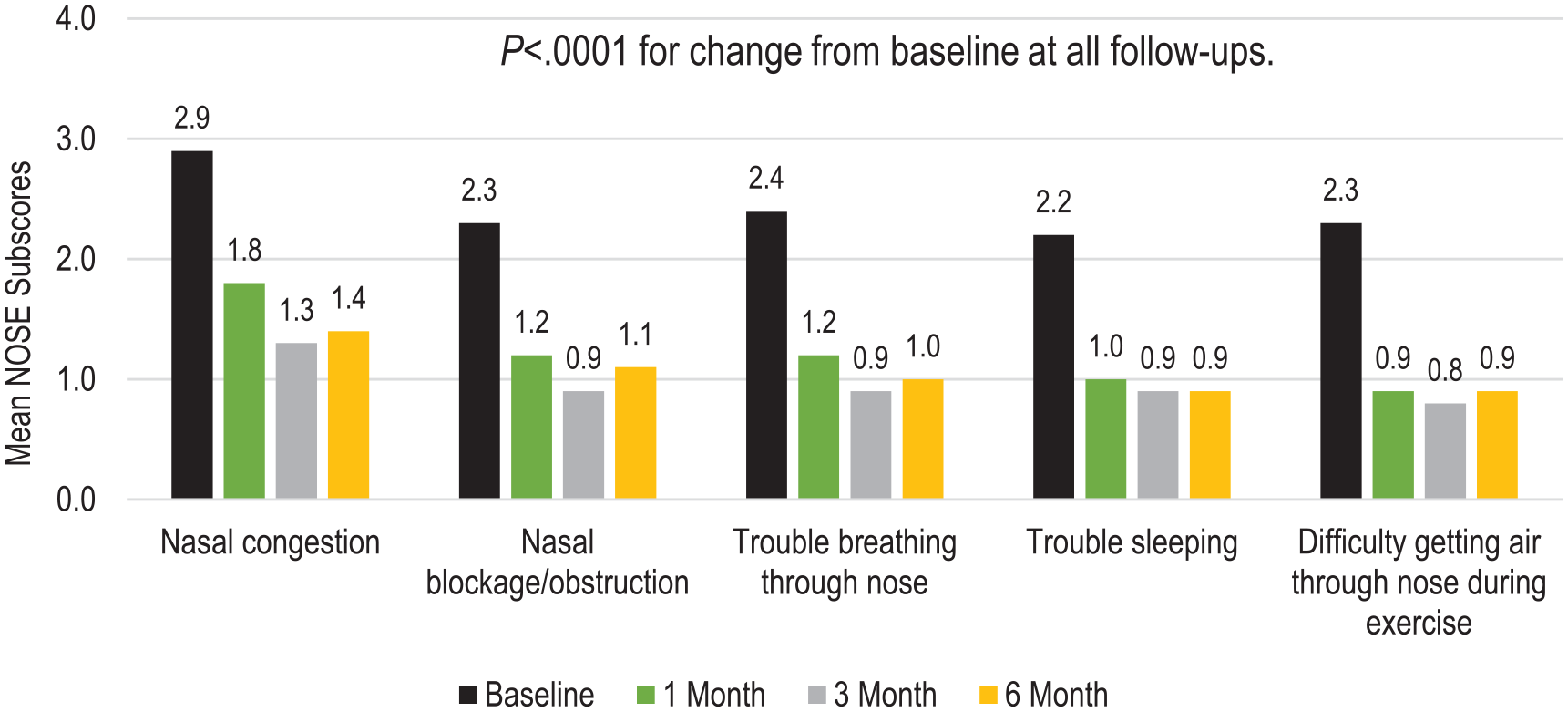
Change in mean NOSE subscores.
The overall mini-RQLQ showed statistically significant improvements from baseline to 3 and 6 months (−1.8 ± 1.3, 58% change, P < .0001 at both time points). The change from baseline to 6 months in mini-RQLQ overall and domain scores, which include activities (regular, recreational, sleep), practical problems, nose symptoms, eye symptoms, and other symptoms (tiredness/fatigue, feeling irritable), are shown in Figure 4. All domain scores demonstrated significant improvement (P < .0001 for all). At 3 and 6 months, 88.6% (95% CI, 79.5% to 94.7%) and 87.3% (95% CI, 78.0% to 93.8%) of participants achieved the mini-RQLQ MCID, respectively.
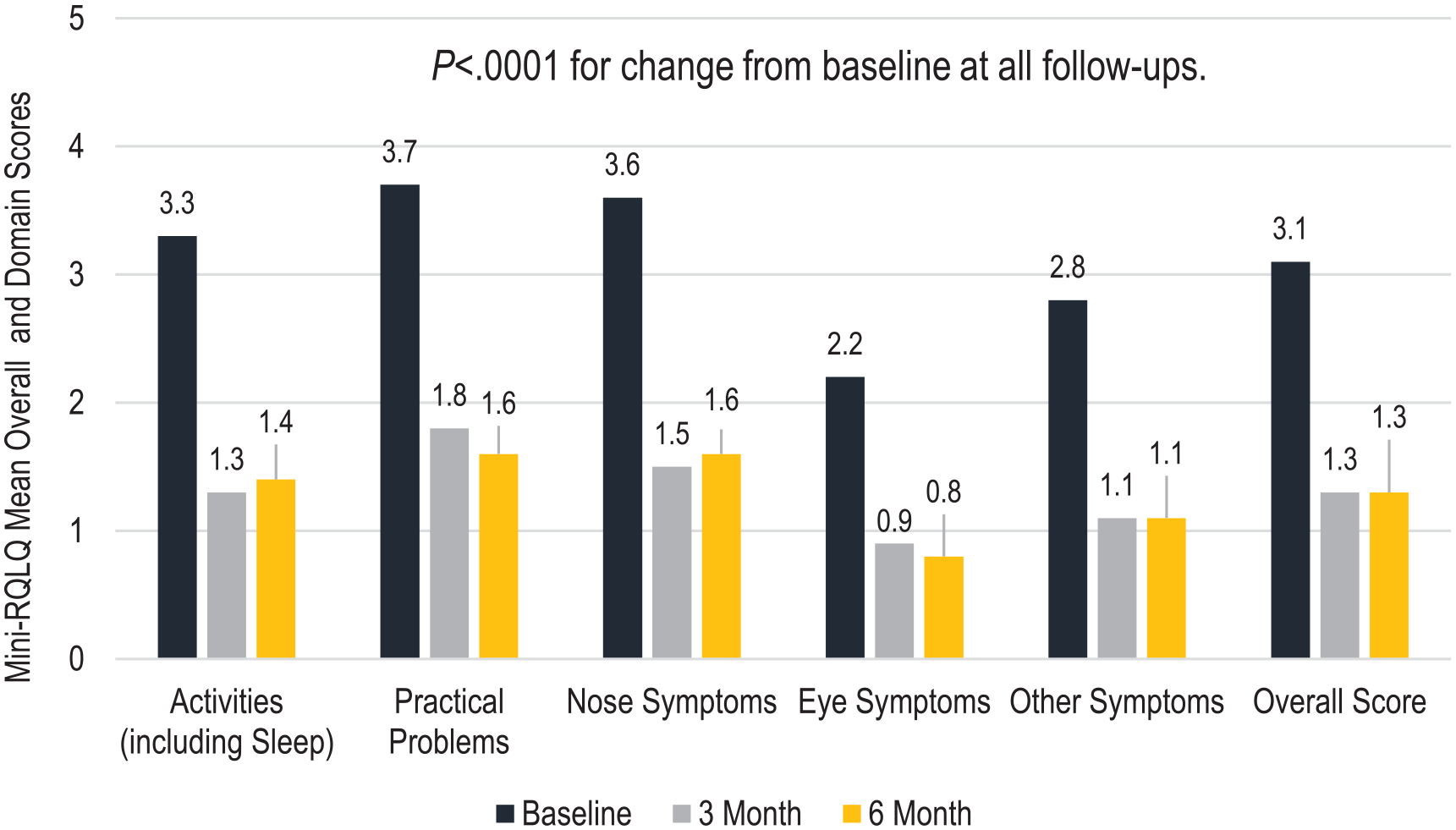
Change in mean mini-RQLQ scores.
Physician’s clinical judgment of the participants’ improvement, as measured by the CGI-I, reported 86.1% (95% CI, 76.5% to 92.8%) of participants showed improvement. Only 1 participant (1.3%) was judged to have symptoms that were worse than baseline at the 6-month visit. Greater than 60% of participants were assessed by the physician to have very much improved or much-improved symptoms over baseline at both 3 and 6 months.
Subgroup analysis by rhinitis type (allergic vs nonallergic) showed significant within-group improvements from baseline to 6 months for the total rTNSS, postnasal drip, cough, overall mini-RQLQ, total NOSE, and overall ETDQ-7 scores (P < .0001 for all assessments, except ETDQ-7 P < .001). There were no significant differences in any of the outcome measures between the allergic and nonallergic subgroups (Table 3).
Comparison of Change from Baseline to 6 Months in Symptoms by Rhinitis Type.
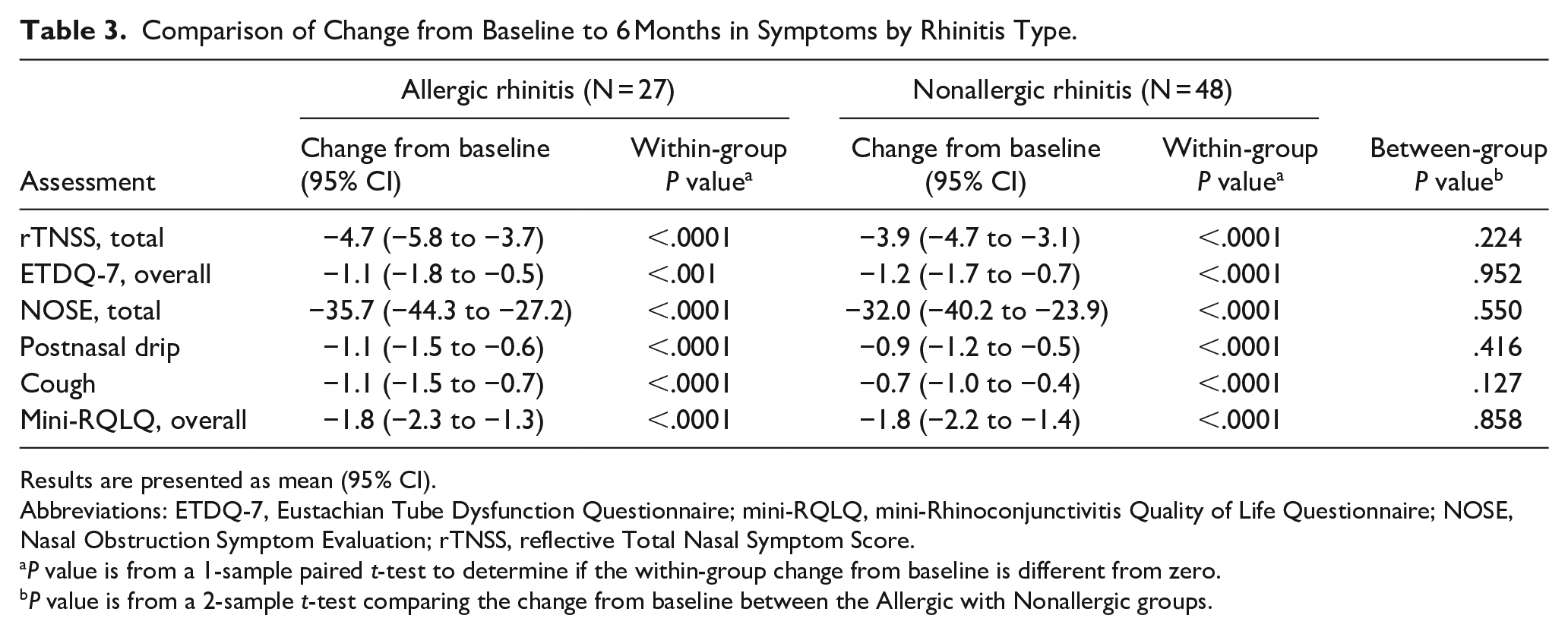
Results are presented as mean (95% CI).
Abbreviations: ETDQ-7, Eustachian Tube Dysfunction Questionnaire; mini-RQLQ, mini-Rhinoconjunctivitis Quality of Life Questionnaire; NOSE, Nasal Obstruction Symptom Evaluation; rTNSS, reflective Total Nasal Symptom Score.
P value is from a 1-sample paired t-test to determine if the within-group change from baseline is different from zero.
P value is from a 2-sample t-test comparing the change from baseline between the Allergic with Nonallergic groups.
Subgroup analysis by baseline total rTNSS (scores 5-6 vs 7-9 vs 10-12) showed significant within-group improvements from baseline to 6 months in all outcome measures for all 3 groups (P ≤ .035 for all). There were also significant differences between the groups for all outcomes except postnasal drip, with greater improvements associated with higher baseline rTNSS.
One participant experienced a serious adverse event (SAE) of epistaxis that required hospitalization. The event occurred 15 days after the procedure. The participant underwent surgical ligation of the sphenopalatine artery (SPA) with resolution of the event. Eight participants experienced 9 nonserious AEs that were considered related to the device or procedure. Events included sinus infection (3), facial pain/pressure (2), presyncope/syncope (2), nosebleed (1), and synechia (1). All the AEs were mild to moderate in severity and resolved.
Discussion
Here we report the results of a prospective, single-arm, multicenter study of a multipoint, impedance-controlled, radiofrequency ablation device. We report statistically significant improvements in the rTNSS and mini-RQLQ, which are consistent with the previous study of this device 4 as well as other radiofrequency 10 and cryotherapy devices. 11 Notably, all domains of the mini-RQLQ demonstrated improvements, including activities (regular, recreational, sleep) and eye symptoms. Separate assessments of postnasal drip and cough also indicated significant improvement, similar to other studies.4,12
We collected data on the ETDQ-7, NOSE, and CGI-I questionnaires, which are not commonly administered in this patient population. We observed statistically significant improvements in the total NOSE score after treatment with an overall shift from severe symptoms to mild/moderate symptoms reported. It is interesting that the NOSE subscores that improved the most were difficulty getting air through the nose during exercise and trouble sleeping. These are noteworthy quality-of-life findings that are important for clinician awareness.
This is the first study of chronic rhinitis patients to prospectively assess ear symptoms using the ETDQ-7, which is typically used to assess patients with Eustachian tube dysfunction (ETD). We wanted to assess ear symptoms in this patient population because we experienced feedback from some patients that their ear symptoms improved after being treated with PNN. The design of the device to treat a broad area with direct leaflet contact contributed to our desire to examine the impact of treatment on ear symptoms using the ETDQ-7. As this study did not require participants to have ETD, the baseline overall ETDQ-7 score (3.2) was slightly lower than that in the cohort of patients in studies examining the impact of interventions specifically aiming to treat ETD (baseline score 4.6). 13 However, our baseline ETDQ-7 values were similar to those observed in patients with chronic rhinosinusitis and ETD.14-16 We observed statistically significant improvement from baseline in the ETDQ-7 overall score and all subscores. The change in overall ETDQ-7 score is comparable to those reported after endoscopic sinus surgery for patients with chronic rhinosinusitis and ETD14-16 and after balloon dilation for treatment of ETD. 13 The pathophysiology of ETD in the absence of an obstructive lesion has long been debated but is thought to be related to functional obstruction and/or nasal cavity inflammation and nasopharyngeal inflammation. 17 In addition, current best evidence supports an association between allergic rhinitis and ETD; however, not all patients with ETD suffer from allergic rhinitis and vice versa.17,18 Our results showed both allergic rhinitis and nonallergic rhinitis groups had significant improvements in ETD symptoms. We believe that the current device effectively targets the region of inflammation (PNN region) responsible for hypersecretion and nasal congestion within the nasal cavity, 17 which subsequently contributes to the obstruction of the Eustachian tube. Recent literature suggests that radiofrequency ablation of the inferior turbinate, and critically the region posterior to it, significantly reduces the proportion of patients experiencing grossly impaired Eustachian tube function (P < .001). 19 The current device is engineered to precisely target this specific region of the posterior inferior turbinate and sphenopalatine region. Future research may be of interest to examine if radiofrequency ablation of the PNN, which disrupts overactive nasal nerves, has a role in impacting ear symptoms in patients with ETD.
Physician assessment of participants’ improvement, as measured by the CGI-I, agreed with the patient-reported assessments with physicians reporting that 86% of participants were improved at 6 months. We believe this supports the soundness of the PROs obtained in this study.
There was 1 SAE of epistaxis reported at 15 days posttreatment. Epistaxis, although low in incidence, is a known potential risk of PNN procedures. Epistaxis typically manifests itself within 2 weeks of surgery, but as late as 1 month of surgery. Reported operative interventions include SPA ligation, embolization, and cautery.20,21 There were no reports of “ice cream” headaches, which are a common effect after cryoablation therapy. 22 These results confirm the excellent safety profile noted in the previous study with this device. 4
This study was designed to evaluate the device with a sample size large enough to evaluate outcomes in subgroup populations. Most importantly, we wanted to evaluate the outcomes of patients with allergic rhinitis versus those with nonallergic rhinitis. For accuracy, we required all participants to be evaluated for allergies by a skin prick, intradermal testing, or an IgE immunoassay. We did not find any significant difference in outcomes by rhinitis type and both groups demonstrated significant improvement in all the outcomes. This finding is consistent with the findings of others.22,23
We found significant differences in outcomes based on the baseline rTNSS score. All baseline rTNSS subgroups showed significant improvement in all measures, but higher baseline scores were associated with greater improvement. This is an effect that has been previously reported for rTNSS in patients with chronic rhinitis.24,25
The major limitation of this study is the lack of a concurrent control; however, the improvement of each participant was based on their pretreatment assessments. Although we included ETDQ-7 as an assessment, our inclusion criteria did not require that participants have ETD, so the relevance of our findings in this cohort is speculative. Additionally, our study cohort was predominantly White with only 10% of our population being Black. This is an improvement over previous studies but more diverse racial enrollment in future trials would be valuable. Finally, longer-term follow-up on this cohort is ongoing.
Conclusion
Our results demonstrate the efficacy and safety of a multipoint, impedance-controlled, radiofrequency ablation device for the treatment of chronic rhinitis. There were significant improvements over baseline through 6 months in the rTNSS, ETDQ-7, NOSE, and mini-RQLQ. Results from physician-reported CGI-I assessment were consistent with the PROs. Both allergic and nonallergic subgroups showed significant improvement in all outcome measures with no significant differences between the subgroups. Participants with higher baseline rTNSS scores experienced more improvement than those with low baseline rTNSS scores.
Footnotes
Acknowledgements
The authors thank study investigators: Michael Armstrong, MD; Nicholas Beckmann, DO; Jeffery Bedrosian, MD; Brian Davis, MD; Marc Dubin, MD; Adil Fatakia, MD; Jarrod Keeler, MD; Kristopher Lay, MD; Matthew Leach, MD; Daniel Rontal, MD; Matthew Rontal, MD; Douglas Reh, MD; Michael Sillers, MD; and Daniel Smith, MD. The authors also thank Manya Harsch, MS, for assistance in the statistical analysis.
Author Contributions
G.E.D.: conception and design, data acquisition, revision, critical review, and final approval; R.A.O.: data acquisition, revision, critical review, and final approval; D.M.Y.: conception and design data acquisition, revision, critical review, and final approval; E.M.O.: drafting article, revision, critical review and final approval; A.G.D.S.: data acquisition, revision, critical review, and final approval.
Data Availability Statement
Research data are not shared.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: G.E.D., D.M.Y., and A.G.D.S. are medical advisor consultants. E.M.O. is a medical writing consultant to Neurent Medical Ltd. The other authors have no conflicts to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the research was supported by Neurent Medical Ltd. No financial support was provided for authorship and publication of this article.
Ethical Considerations
Ethical approval for this study was obtained from Allendale IRB (Old Lyme, CT, USA) on June 25, 2022.
Consent to Participate
All participants signed the IRB-approved written informed consent before participating in the study.
Consent for Publication
Not applicable.
