Abstract
Keywords
Introduction
Sudden sensorineural hearing loss (SSNHL), which is defined as an idiopathic hearing loss of greater than 30 dB in at least 3 contiguous frequencies within 72 hours, was first described by De Kleyn in 1944. 1 The pathophysiology of SSNHL is unknown. Several possible causes include viral infection, autoimmunity, and vascular injury. Various major treatments have been used clinically, including systemic corticosteroids, thrombolytics, vasodilators, hypertensive oxygen, and antiviral therapy. 2 In addition to oral and intravenous corticosteroid systemic use, intratympanic steroid injection (ISI) is also recommended as a treatment for patients with SSNHL. 3 Wu et al performed a double-blind, randomized, controlled trial comparing ISI with intratympanic saline injection in the rescue treatment of SSNHL, and found that the auditory outcome of the ISI group was significantly better than that of the intratympanic saline injection group, thus effectively demonstrating the effectiveness of ISI in the treatment of SSNHL. 4
Treatment of SSNHL with intratympanic corticosteroids was used primarily in the following 3 situations: as initial therapy with systemic steroids,5,6 as rescue therapy after failure of initial systemic steroid therapy,7,8 and as initial or rescue therapy alone in populations in which systemic steroids were contraindicated or not suitable for steroid use.9,10 There is no standardized method for intratympanic steroids for the treatment of SSNHL in the literature. There is no consensus on how to deliver steroids to the middle ear. In addition to the usual intratympanic injection method. Some scholars have introduced the method of injection into the middle ear via the ventilation tube or grommet placement and steroid eardrop application,11-14 microcatheter treatment,15,16 and the MicroWick™ device.17-20 Here, we describe a modification of the intratympanic injection method in light of the aforementioned method of administration, performed by continuous perfusion with a micropump inserted through the ventilation tube, compared with conventional intermittent intratympanic injection, and analyze the efficacy of 2 steroid-based regimens for SSNHL.
Patients and Methods
Patients
The study was approved by the Ethics Committees of the University of Hong Kong—Shenzhen Hospital, University (No. hkuszh2023092). From November 2018 to October 2022, we enrolled 60 subjects who were randomly divided into the continuously transtympanic steroid perfusion (TSP) group and the intermittent ISI group of 30 patients each. Subjects met both inclusion and exclusion criteria. The following inclusion criteria were used: (1) no hearing improvement after 1 week of initial systemic steroid therapy; (2) no initial systemic steroid therapy for more than 14 days after the onset of the disease, missing the optimal time for treatment; (3) hearing loss before rescue therapy, at least 3 continuous-frequency mean pure-tone audiometry (PTA) greater than or equal to 50 dB; (4) normal or near-normal hearing in the opposite ear (0.25, 0.5, 1, 2, and 4 kHz PTA <30 dB); (5) retreatment with salvage therapy less than 1 month after the first episode; A-type tympanogram; and (6) above 18 years in age. Cochlear structural abnormalities or retrocochlear lesions, such as vestibular schwannomas, were excluded from imaging. Examination of the ear excluded any history of hearing loss, Meniere’s disease, chronic inflammatory or suppurative ear disease, cholesteatoma, otosclerosis, previously had any type of ear surgery, and pregnancy.
Study Design
ISI groups
The intratympanic (IT) dose of 5 mg (1.0 ml of 5 mg/ml solution) of dexamethasone sodium phosphate was administered by injection into the middle ear cavity through the external auditory canal via a 27 gauge needle passed through a small posterior inferior myringotomy under endoscope, injected every other day (5 times in total).
TSP group
A T-type ventilation tube (model: 1016011, 1.14 mm ID, 12 mm l; Medtronic Inc., USA) was inserted after posterior inferior myringotomy under endoscope-guided. An epidural anesthetic catheter (model: 1.0 mm ID) was inserted through the ventilation tube, the catheter tip was inserted into the tympanic cavity as close as possible to the round window niche, the catheter ends attached to a syringe, continuous perfusion of a solution containing 1.0 ml of dexamethasone (5 mg/ml) was used a micropump twice daily for a week. Each time at the rate of 1 ml/hour for 30 minutes of continuous release, the middle ear cavity drug solution is injected at approximately 0.5 ml and the remaining drug solution is discarded. The ventilation tube and epidural anesthetic catheter were removed after 1 week (Figure 1).
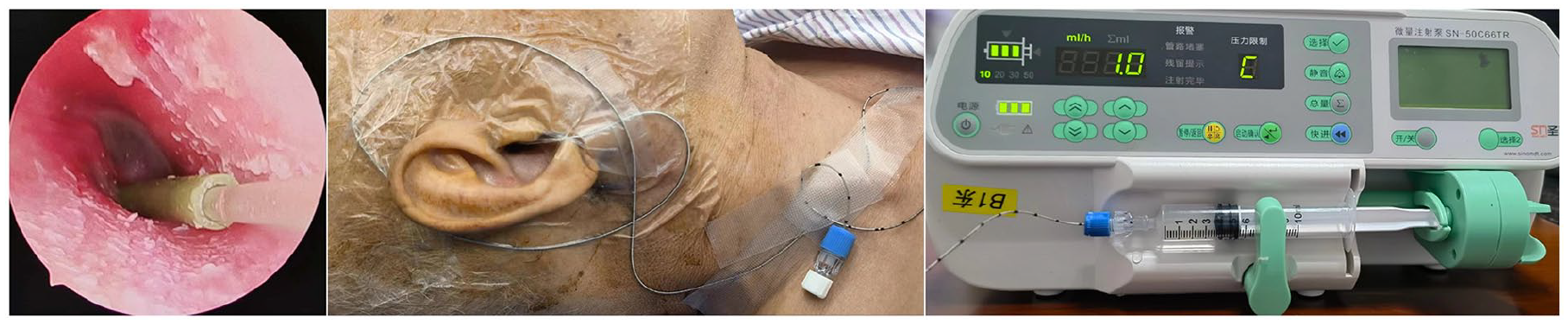
Schematic of TSP methods.
All patients who received intravenous methylprednisolone succinate 60 mg/day were administered simultaneously in both groups for 5 days, tapered to 40 mg/day for 2 days (Adolescent or weak female initiation of 40 mg/day tapered and as appropriate), and then 30 mg/day of oral prednisolone acetate for 3 days, tapered to 20 and 10 mg/day for 2 days. Simultaneous intravenous silver gland lobe extract was performed at a dose of 87.5 mg/day for 7 consecutive days. Oral mecobalamin tablets 0.5 mg, 3 times daily, betahistine hydrochloride 12 mg, if vertigo, 3 times daily, and oral aprazolam tablets 0.4 mg once per night in patients with sleep disturbance.
Outcome Analysis
All frequencies (250, 500, 1000, 2000, and 4000 Hz) and per frequency pure-tone average (PTA) was tested before the initial treatment and 2 weeks after treatment, respectively. The degree of hearing recovery after treatments was expressed according to Siegel’s criteria, as follows: complete recovery (more than 30 dB hearing gain and as final hearing better than 25 dB); partial recovery (more than 15 dB hearing gain and as final hearing between 25 and 45 dB); slight improvement (more than 15 dB hearing gain but with a final hearing poorer than 45 dB); and no improvement (<15 dB hearing gain and final hearing poorer than 75 dB).21,22
Statistical Analysis
Data were analyzed using the SPSS software (IBM SPSS Statistics 21.0, USA). PTA pre- and post-treatment were compared between the 2 groups. Differences in proportions and medians between the groups were analyzed using the χ2 test and 2-tailed Mann-Whitney U test. The average PTA and the degree of hearing recovery after treatments were analyzed using the paired t-test and the Chi-square test. The results are expressed as the means and standard deviations. The criterion for statistical significance was P < .05.
Results
Patients
A number of 60 patients met the criteria and were grouped (ISI group, n = 30; TSP group, n = 30). The demographics of the 2 groups were similar (Table 1).
Demographic Characteristics of the Patients.
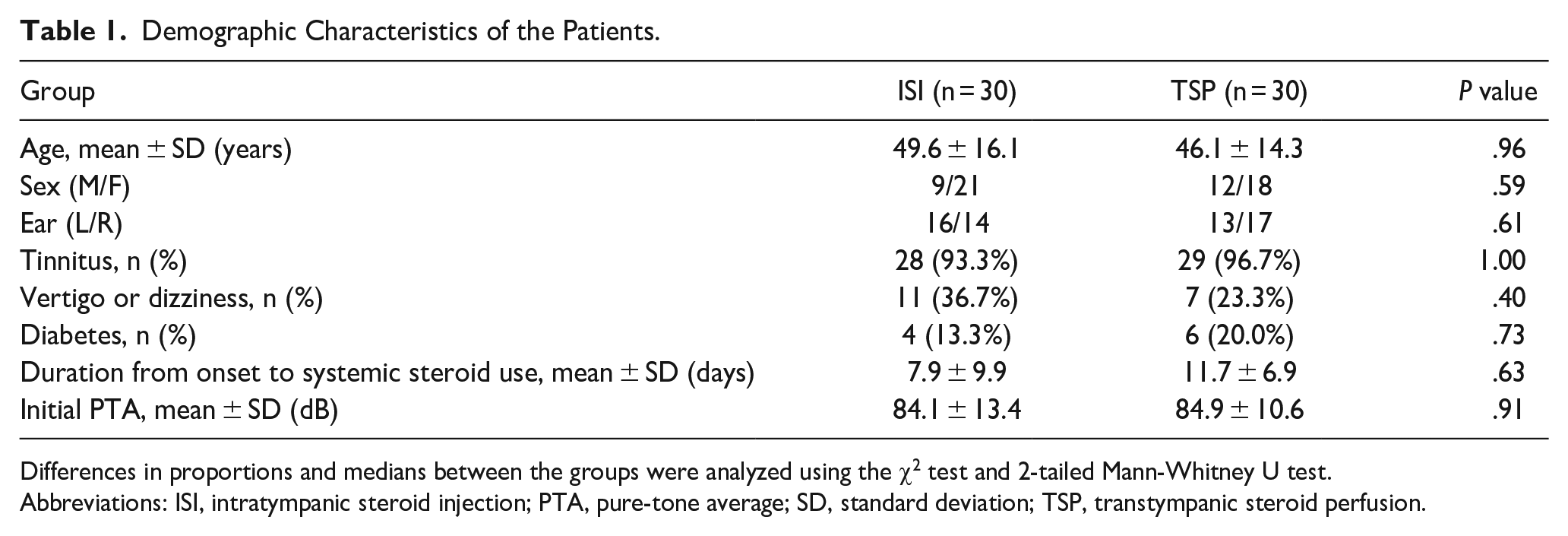
Differences in proportions and medians between the groups were analyzed using the χ2 test and 2-tailed Mann-Whitney U test.
Abbreviations: ISI, intratympanic steroid injection; PTA, pure-tone average; SD, standard deviation; TSP, transtympanic steroid perfusion.
Evaluation of Efficacy
The PTAs were 84.9 ± 10.6 dB in the TSP group and 84.1 ± 13.4 dB in the ISI group before treatment, the PTAs weres 60.3 ± 18.2 dB in the TSP group and 67.5 ± 22.6 dB in the ISI group after treatment. The threshold for hearing improvement was 24.6 ± 14.1 dB in the TSP group is better than that was 16.6 ± 14.9 dB in the ISI group (Figure 2). According to Siegel’s criteria, the response rate (hearing improvement of ≥15 dB) was 70.0% in the TSP group and 46.7% in the ISI group. There was a statistically significant difference in the degree of hearing improvement between the 2 groups (P < .05). In the analysis of hearing improvement at each frequency, we found that the low-frequency hearing had the greatest hearing improvement, with a maximal hearing improvement of 0.25 kHz in the TSP group, reaching 30.8 ± 3.3 dB (Figure 3).
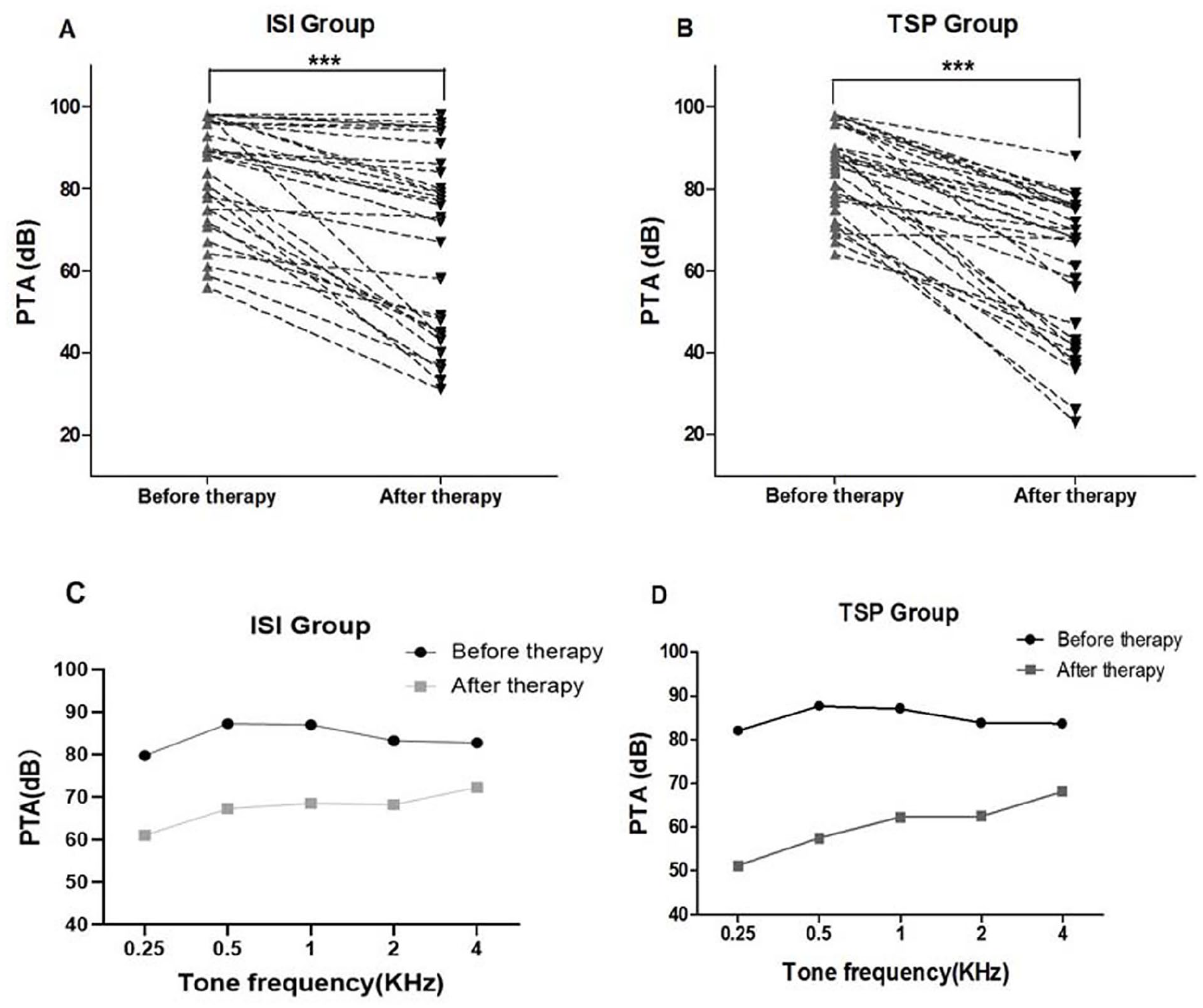
Comparison of the PTA improvements and hearing recovery in all frequencies after treatment in the ISI and TSP groups. Pre- and post-treatment PTA thresholds of patients in ISI group (A) and TSP group (B). PTA threshold of each frequency before and after treatment in ISI group (C) and in TSP group (D). ***The difference between the two is statistically significant (P < .05).
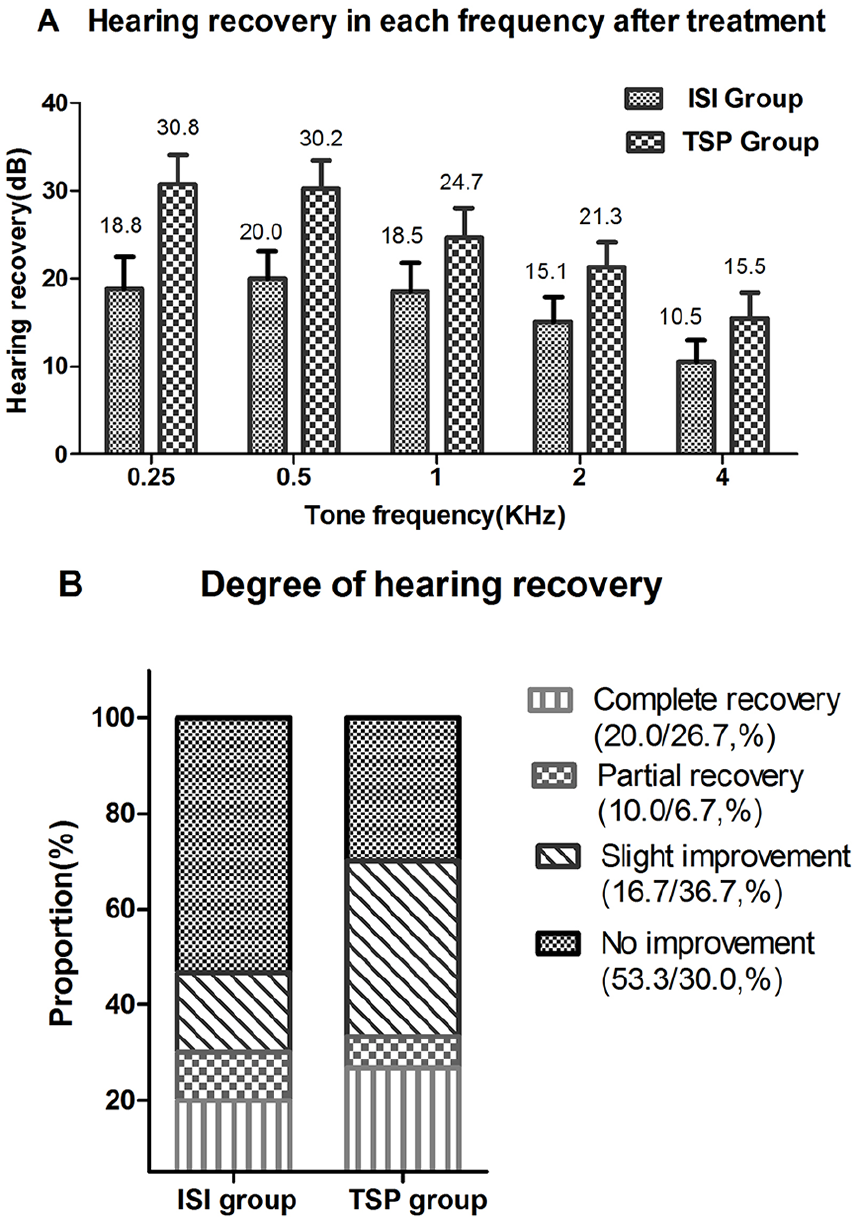
Comparison of the improvement of PTA of each frequency and the response rates in the ISI and TSP groups. (A) The improvement of PTA of each frequency by ISI therapy and by TSP therapy. (B) Comparison of the degree of hearing recovery in PTA between ISI and TSP groups.
Side Effects
There was no patient who showed severe vertigo and pain during the 2 groups. Four (13.3%) patients in the ISI group complained of transient dizziness, which was resolved spontaneously after rest; 6 (20%) patients in the TSP group showed ear fullness during the infusion. No significant signs of otitis media occurred during treatment in both groups. One (3.3%) patient had small perforation at discharge in the ISI group, 2 (6.7%) patients had transient perforation occurred in the TSP group after removal of the ventilation tube, which healed spontaneously after 3 months.
Discussion
ISI for SSNHL is a relatively popular and effective method, it was first reported in 1986 by Sakata et al. for the treatment of Ménière’s disease, 23 and in 1996 Silverstein et al. described intratympanic injection for the treatment of SSNHL. 24 Although systemic steroid therapy is recommended as the first-line treatment for SSNHL in most countries, Jin et al concluded that oral steroid therapy alone is decreasing and that patients receiving intratympanic injection in combination with systemic steroids is increasing. 25 Lee et al found that the combined treatment of systemic steroids with intratympanic injections was more effective than systemic steroids alone in treating SSNHL. Furthermore, repeated intratympanic injection steroids as salvage therapy in refractory SSNHL that fail to respond to systemic steroid therapy was effective. 26 Choi et al found that there was no difference in efficacy between intratympanic injection therapy and systemic steroid therapy in the initial treatment, but intratympanic injection therapy was effective in the salvage treatment of profound SSNHL. 22
Intratympanic injection therapy requires solving 2 questions: first, how to inject drugs into the middle tympanic cavity, and second, to prolong the duration of the drug’s stay around the round window membrane. Injecting drugs into the middle tympanic cavity is not the purpose of treatment in intratympanic therapy, but the ultimate purpose of treatment is to penetrate the inner ear through a round window membrane. Currently, intratympanic administration of drugs is commonly performed through myringotomy injection manually, but the greatest disadvantage of this method is the difficulty in controlling the rate of drug injection and the rapid loss of drug through the pharyngotympanic tube, which affects the effectiveness of this method. To compensate for these shortcomings, some researchers have attempted to extend the duration of medication stay around the round window membrane by means of intraoral drug instillation or injection, continuous microcatheter treatment, and MicroWick device, but these methods increase the risk of infection when medication stays outside the ear canal. In our study, the modified method of administration of the drug was introduced through the internal diameter of the tympanic ventilation duct into the anesthesia catheter, the end of the catheter was inserted into the tympanic cavity near the position of the round window, and the drug was injected directly into the tympanic cavity through the catheter without passing through the external meatus, thereby avoiding the potential risk of infection, thus reducing the risk of perforation of the tympanic membrane and infection.
Clinical reports of intratympanic administration have reported a 1.6% to 29.0% rate of perforation of the tympanic membrane.18,27 In this study, we used a grommet insertion to avoid the risk of pain and perforation caused by repeated myringotomy. Jin et al predicted that patients who received at least 5 repetitive myringotomy injections within 1 year had a 2.2-fold higher risk of needing tympanoplasty compared with patients who received only 1 injection. 25 It is difficult to grasp the volume and speed of conventional intratympanic injections, and excessively rapid injections of drugs are prone to drainage through the Eustachian tube and the myringotomy needle eye, and because of the pressure and limited volume of the tympanic cavity, they are unable to continuously inject large amounts of more drug, and the drug is unable to accumulate around the round window membrane; rapid infusion of drug into the tympanic cavity may form a level of fluid and gas on the surface of the round window membrane, which may affect the amount of drug that penetrates into the inner ear through the round window membrane. We replaced the micropumps to compensate for the deficiency of manual injections, allowing continuous, slow, and precise injection of the drug into the tympanic cavity, with enough drug accumulating around the round window membrane to increase the drug concentration in the internal ear lymph.
The basic principle of intratympanic therapy is the permeability of the round window membrane to the drug, which absorbs the drug into the inner ear and causes the concentration of the drug in the external lymphatic fluid to be greater than that in systemic therapy. Bird et al found that the concentration of steroids in the external lymphatic fluid after intratympanic injection was 127-fold higher than that after intravenous injection. 28 In a study by Dispenza et al, intracochlear drug release after intratympanic injection produced a significant gradient in drug concentration, with a trend toward a decreasing concentration of drug distributed from the basal portion of the cochlea to the apical portion of the cochlea, with the highest concentration at the basal portion of the cochlea and near the round window membrane and the lowest at the apical portion. 7 Fu et al observed that the fluorescence of spiral ganglion was significantly stronger than the apical coil after acoustic bubble injection of dexamethasone in animal experiments; in clinical studies, however, low-frequency hearing was significantly improved in patients treated with intratympanic injections, consistent with our findings. 6 But how can the highest drug concentration at the basal portion of the cochlea result in more pronounced low-frequency hearing improvement? The outcome appears paradoxical. However, in previous studies of these intratympanic injections, such results were due to neglect of the fact that the drug solution applied accumulated in the auditory vacuoles or tympanic cavities and completely soaked into the osseous ear sac. Mikulec et al used a continuous perfusion method to flood the entire auditory acoustic vacuole of guinea pigs and found significantly higher concentrations at the top of the cochlea. Thus, in addition to the round window membrane absorption pathway, there may be other entry of substances into perilymph through the bone of the otic capsule after intratympanic applications, 29 which explains our finding of better outcomes in low-frequency hearing restoration.
In order for the drug solution to become permanently filled and accumulate in the tympanic cavity, there is a need to choose a continuous medication regimen. Fu et al found in animal experiments that the peak concentration of fluorescence detected in the external lymph after a single injection of dexamethasone to the auditory bubble was maintained for only 30 minutes, followed by a gradual decrease in fluorescence. 6 Salt and Ma found that the drug is rapidly cleared from the external lymph after a single dose and that it is difficult to create a significant gradient in the drug concentration in the cochlea, and it often takes several hours to establish a stable gradient and to maintain this state, which can be achieved and maintained only by continuous drug administration. 30 Saijo and Kimura also showed that the pattern of intratympanic therapy did have a significant influence on the drug level in the external lymphatic fluid and continued intratympanic resulted in higher concentrations of drug in the external lymphatic fluid than a single administration. 31 In our study, we used the micropump continuous slow pumping drugs into the tympanic cavity instead of the method of manual injection, to overcome the adverse effects of rapid drainage after the injection of drug. We repeatedly injected 1 ml of fluid into the tympanic cavity twice daily to fill the tympanic cavity with drug, and the round window membrane and the promontory were constantly immersed in the drug. The drug continued infiltration into the internal ear producing lasting effects and improved the clinical treatment effect.
Summary
Standard systemic therapy for SSNHL still results in loss of hearing recovery in approximately one-third of patients. The results of this study suggest that TSP can achieve better hearing outcomes in initially ineffective patients with SSNHL and this approach can be used as a salvage therapy in patients with SSNHL. However, the efficacy of this approach, combined with systemic steroids, as initial treatment for SSNHL awaits further clinical observation. It is the key direction of future clinical research to continuously improve the method and way of administering drugs in the tympanic cavity, improve the permeability of drugs to the round window membrane, and explore the concentration gradient distribution and pharmacokinetics of drugs in the internal ear.
Supplemental Material
sj-xlsx-1-ear-10.1177_01455613241284154 – Supplemental material for Perfusion Steroid via Ventilation Tube as Salvage Treatments for Sudden Sensorineural Hearing Loss
Supplemental material, sj-xlsx-1-ear-10.1177_01455613241284154 for Perfusion Steroid via Ventilation Tube as Salvage Treatments for Sudden Sensorineural Hearing Loss by Yang Mingbao, Zhang Bei, Guan Yafeng, Liang Xiuni and Miao Beiping in Ear, Nose & Throat Journal
Supplemental Material
sj-xlsx-2-ear-10.1177_01455613241284154 – Supplemental material for Perfusion Steroid via Ventilation Tube as Salvage Treatments for Sudden Sensorineural Hearing Loss
Supplemental material, sj-xlsx-2-ear-10.1177_01455613241284154 for Perfusion Steroid via Ventilation Tube as Salvage Treatments for Sudden Sensorineural Hearing Loss by Yang Mingbao, Zhang Bei, Guan Yafeng, Liang Xiuni and Miao Beiping in Ear, Nose & Throat Journal
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received financial support from the Shenzhen Clinical Medical Research Center for Otolaryngology Diseases.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
