Abstract
Introduction
Head and neck cancer (HNC) ranks 6th in Germany and 7th worldwide among all newly diagnosed malignancies. 1 Since early symptoms are often absent, about half of the patients already have an advanced tumor stage UICC III or IV (Union internationale contre le cancer) at diagnosis, and the 5-year survival is only 40%-50%, even in industrialized countries. 2
There are only very limited studies investigating the symptom burden and the quality of life (QoL) of patients with HNC at different tumor stages over a longer follow-up period. Available studies include mostly small numbers of cases or involve final-stage patients who are already in a palliative care unit.3–5
A useful tool for measuring the symptom burden of cancer patients is the standardized and validated MIDOS(2) (Minimal Documentation System) questionnaire. 6 It is the German version of the Edmonton Symptom Assessment Scale, which was developed in the early 1990s; it was validated, translated into German, and revised again in 2009. 7 The German Society for Palliative Medicine (DGP) recommends the use of the MIDOS(2) questionnaire for the self-reporting and follow-up of “symptom severity” and “best well-being”.6,7 The questionnaire is characterized not only by its high validity, but also by its brevity and practicability, and is especially recommended for patients with severe underlying disease. 8
The aim of this study is to evaluate the most common symptoms of HNC patients and their quality of life several years after initial diagnosis.
The relevance of the question is high due to the increasing incidence of HNC, an undersupply of this special patient population already described in the literature and growing awareness for good symptom-oriented therapy for improving QoL.
Patients and Methods
The present study was approved by the Ethics Committee of the University of Erlangen (No.: 486_18 B). It was conducted according to the Declaration of Helsinki and followed the Strengthening the Reporting of Observational Studies in Epidemiology (STROBES) guidelines. 9 The study was registered in the German Registry for Clinical Studies (DRKS) (application No.: 00017122) and adheres to Consolidated Standards of Reporting Trials (CONSORT) guidelines. 10
Between January 1, 2019 and December 31, 2019, 212 patients presenting for follow-up visits at the Department of Otolaryngology, Head & Neck Surgery of the University hospital Erlangen were included. The following inclusion criteria applied: HNC of all tumor stages (UICC I-IV), patients who had finished their cancer treatment, complete medical records, minimum age of 18 years, sufficient cognitive and language skills to independently answer the questionnaire, a completely and correctly filled in questionnaire, and written informed consent following detailed information about the study. Patients under current cancer treatment, under 18 years of age, and/or patients with cognitive impairments or language barriers who could not understand the content of the questionnaires, and/or with incomplete questionnaires and medical records were excluded from the study. During the survey period, each Wednesday a total of 30 questionnaires were prepared and handed out to patients presenting for follow-up examinations who verbally agreed to participate until all questionnaires had been distributed. The order in which the patients appeared for consultation and were consequently included was random (quasi-randomization).
All patients were asked to fill out the 10-item MIDOS(2) questionnaire. It surveys the ten most common symptoms that potentially affect patients during the course of their illness (pain, nausea, vomiting, shortness of breath, constipation, tiredness, loss of appetite, drowsiness, depression, and anxiety) on a four-point verbal categorical ranking scale with the options “none,” “mild,” “moderate” or “severe.” 11 The response options are assigned a numerical value in the database (0 = none, 1 = mild, 2 = moderate, and 3 = severe) to calculate a summed score (minimum 0, maximum 30). To analyze the absolute and relative frequency of symptoms, “none” was defined as “the patient reports no symptom (0),” while patients with “mild,” “moderate” or “severe” symptoms were defined as “the regarding symptom was shown (1).”
The information relates explicitly to the current day.
On two further scales, patients can name additional symptoms as well as their best well-being (“how you feel overall?”) using a five-point categorical ranking scale (“very bad,” “bad,” “average,” “good,” or “very good”). These answer options are represented by a numerical value (5 = very bad, 4 = bad, 3 = average, 2 = good, and 1 = very good) for statistical evaluation.
To elicit the patients’ QoL, we used the German version of the “University of Washington Quality of Life Questionnaire.” 12 Patients were asked to report their health-related and overall QoL during the past 7 days on a six-point scale (“outstanding” = 100, “very good” = 80, “good” = 60, “fair” = 40, “poor” = 20 and “very poor” = 0).
The survey of symptom burden (frequency and severity) of the ten most common symptoms in cancer patients as well as the severity sum score were defined as primary endpoints. Secondary endpoints included best well-being and QoL (health-related and overall).
In addition to the questionnaires, clinical and oncological characteristics (e.g., time from initial diagnosis to interview, regular medication intake, comorbidities, ECOG (Eastern Cooperative Oncology Group) status, tumor localization, treatment modality, and recurrence of cancer) were obtained from the patient’s medical records. Preexisting comorbidities were classified according to medical disciplines (cardiovascular, pulmonary, oncological, and neurological). Tumors were staged using the 8th edition of the tumor node metastasis classification and the UICC classification. 13 Treatment modalities were defined as follows: surgical treatment only, definitive chemoradiation, primary surgical treatment with adjuvant therapy (i.e., radiation or chemoradiation), and salvage surgery following failed initial cancer treatment of any kind.
Statistical Computation/Analysis
Categorical variables were expressed as absolute (n) and relative (%) frequencies. The continuous variables that were normally distributed were reported as mean (M) ± 1 standard deviation (SD) and those that were not normally distributed or ordinally scaled as median [25.; 75. percentile] and—for better comparison—additionally as mean ± 1 SD.
Independent t-tests were performed to detect significant differences between groups in continuous variables that were normally distributed. The Mann-Whitney U test was performed for ordinal or continuous variables that were not normally distributed. Nominal variables were tested for group differences using cross tables and the Chi 2 test. In the case of >2 × 2-dimensional cross tables, post hoc comparisons were performed using column-wise comparisons in SPSS (IBM, Armonk, New York, USA).
In the case of multiple comparisons, a Bonferroni correction was performed, and a new critical P value was defined. P-values that remained significant after Bonferroni correction were marked with an asterisk (*).
An effect size was calculated for each test: for the Mann-Whitney U test and the independent t-test r was reported; for 2 × 2 cross tables phi, in the case of >2 × 2-dimensional cross tables Cramer’s V.
A phi, Cramer’s V, and r of .1 can be interpreted as a small effect, .3 as a moderate effect, and .5 as a strong effect. Data analyses were performed with SPSS 28.0 (IBM Corp., Armonk, NY, USA) A P value of <.05 was considered statistically significant and was marked with an asterisk (*).
Results
Patient Collective
Between July 1, 2019 and December 31, 2019, 503 patients presented for their regular follow-up visit at the Department of Otolaryngology, Head & Neck Surgery of the University hospital Erlangen. Of these, 446 returned their questionnaire, which corresponds to a response rate of 88.7%. Two hundred nineteen patients (49.1%) had to be excluded from the study due to incomplete questionnaires. Another 15 patients were excluded because they did not meet the inclusion criteria (see Figure 1).
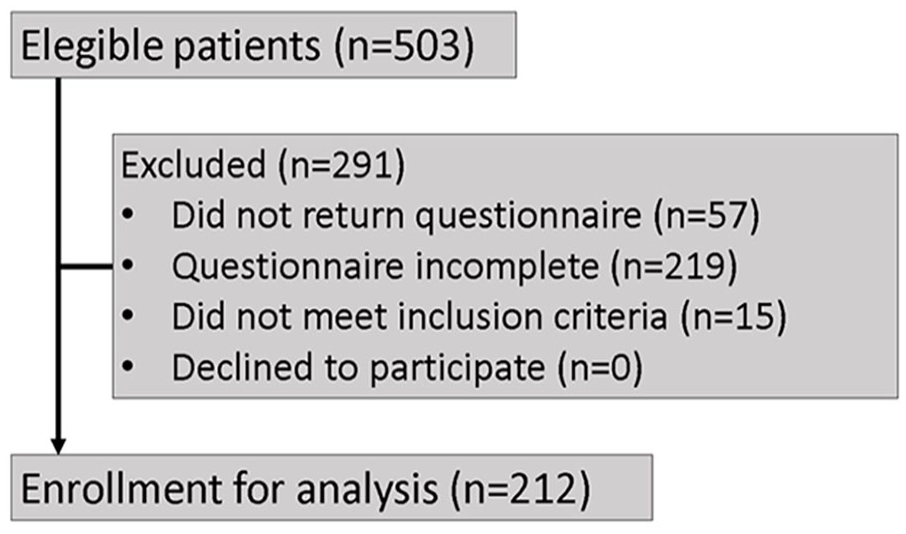
CONSORT flow diagram.
Out of the evaluated 212 patients 120 were diagnosed with tumor UICC stage I and II (early stages), and 92 with tumor UICC stage III and IV (advanced stages).
Patient Characteristics
22.5% of the patients with early stages were female, 62.84 ± 11.35 years old, and the average time between the initial diagnosis and the interview was 43.00 ± 40.95 months (Md: 31.00 [11.25; 73.25]). 25.0% of the patients with advanced stages were female, 62.67 ± 11.35 years old, and had a mean period of 47.40 ± 44.50 months (Md: 32.50 [11.75; 73.00]) between the initial diagnosis and the interview.
There was no significant difference regarding demographic variables, comorbidities, or ECOG status between the two groups (see Table 1 for statistical comparison).
Comparison of the Patient Population According to UICC Early and Advanced Stages.
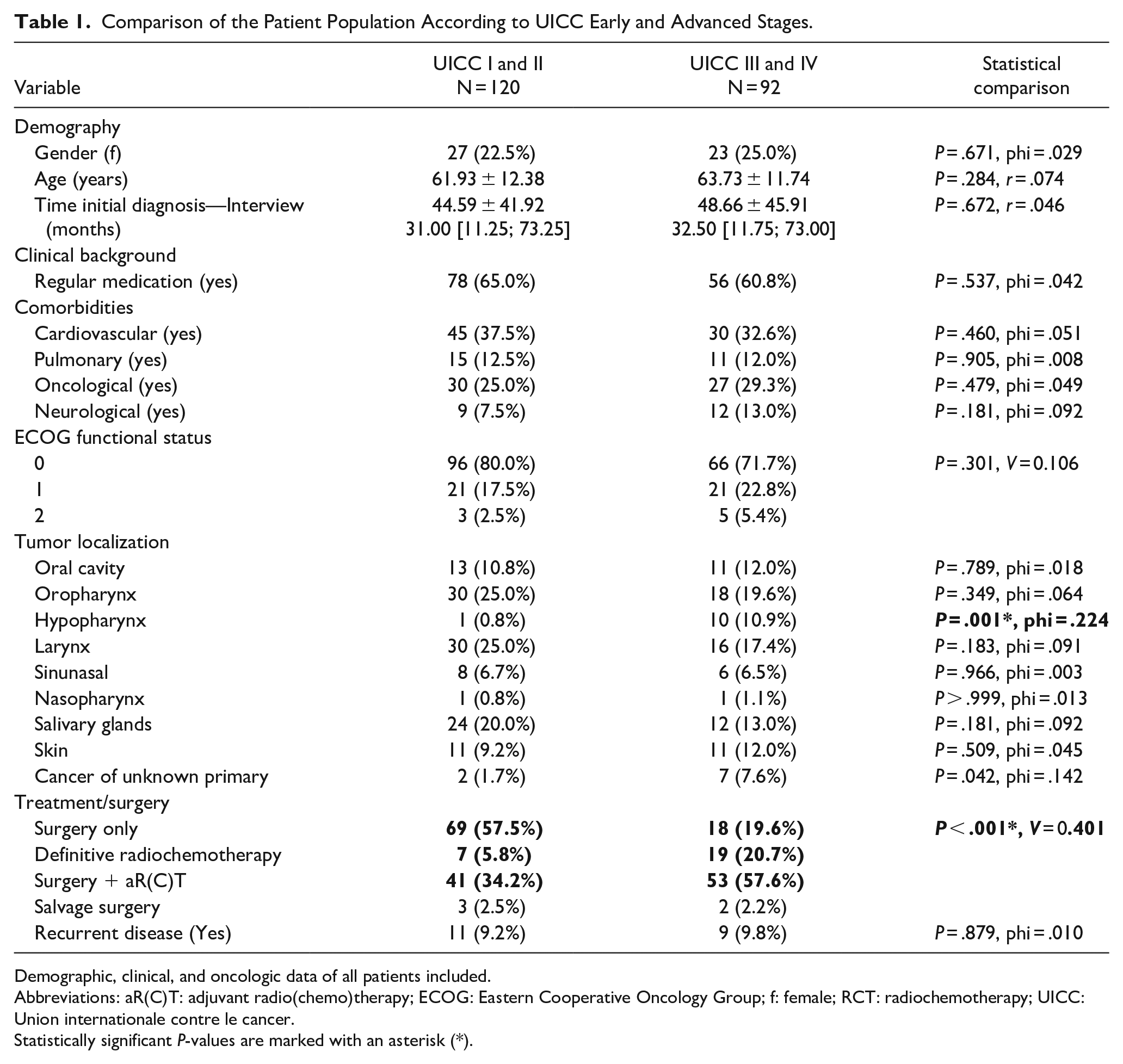
Demographic, clinical, and oncologic data of all patients included.
Abbreviations: aR(C)T: adjuvant radio(chemo)therapy; ECOG: Eastern Cooperative Oncology Group; f: female; RCT: radiochemotherapy; UICC: Union internationale contre le cancer.
Statistically significant P-values are marked with an asterisk (*).
The most frequent cancer localization was the oropharynx (25.0/19.6%), followed by larynx carcinomas (25.0/17.4%), salivary gland cancers (20.0/13.0%), and cancer of the oral cavity (10.8/12.0%). Less common tumor localizations were the nasopharynx (0.8/1.1%), cancer of unknown primary (CUP) (1.7/7.6%), and the hypopharynx (0.8/10.9%).
It should be noted that treatment modalities differed significantly between the groups depending on the stage (P < .001, V = 0.401): significantly more patients of the early-stage group received surgery only (57.5 vs 19.6%), while patients of the advanced-stage group more often received definitive radiochemotherapy (5.8 vs 20.7%) or a combination of surgery plus adjuvant radio(chemo)therapy (aR(C)T) (34.2 vs 57.6%). There was no difference regarding the relative frequency of salvage surgery (2.5 vs 2.2%).
Symptom Burden
The most frequently reported symptom was tiredness, reported by 47.5% of patients with early-stage disease and by 60.9% with advanced-stage disease (P = .053, phi = .133), followed by anxiety (35.8 vs 32.6%, P = .624, phi = .034), drowsiness (35.0 vs 44.6%, P = .157, phi = .097), and pain (30.0 vs 40.2%, P = .121, phi = .107). The least frequently reported symptoms were constipation (10.0 vs 10.9%, P = .837, phi = .014), vomiting (3.3 vs 5.4%, P = .452, phi = .052), and nausea (4.2 vs 9.8%, P = .103, phi = .112). A significant difference between groups was found solely for loss of appetite, with early-stage patients reporting diminished appetite significantly less frequently than patients with advanced UICC stages (15.0 vs 32.6%, P = .002, phi = .157) (see Table 2).
Frequency of reported symptoms according to UICC early and advanced stages.
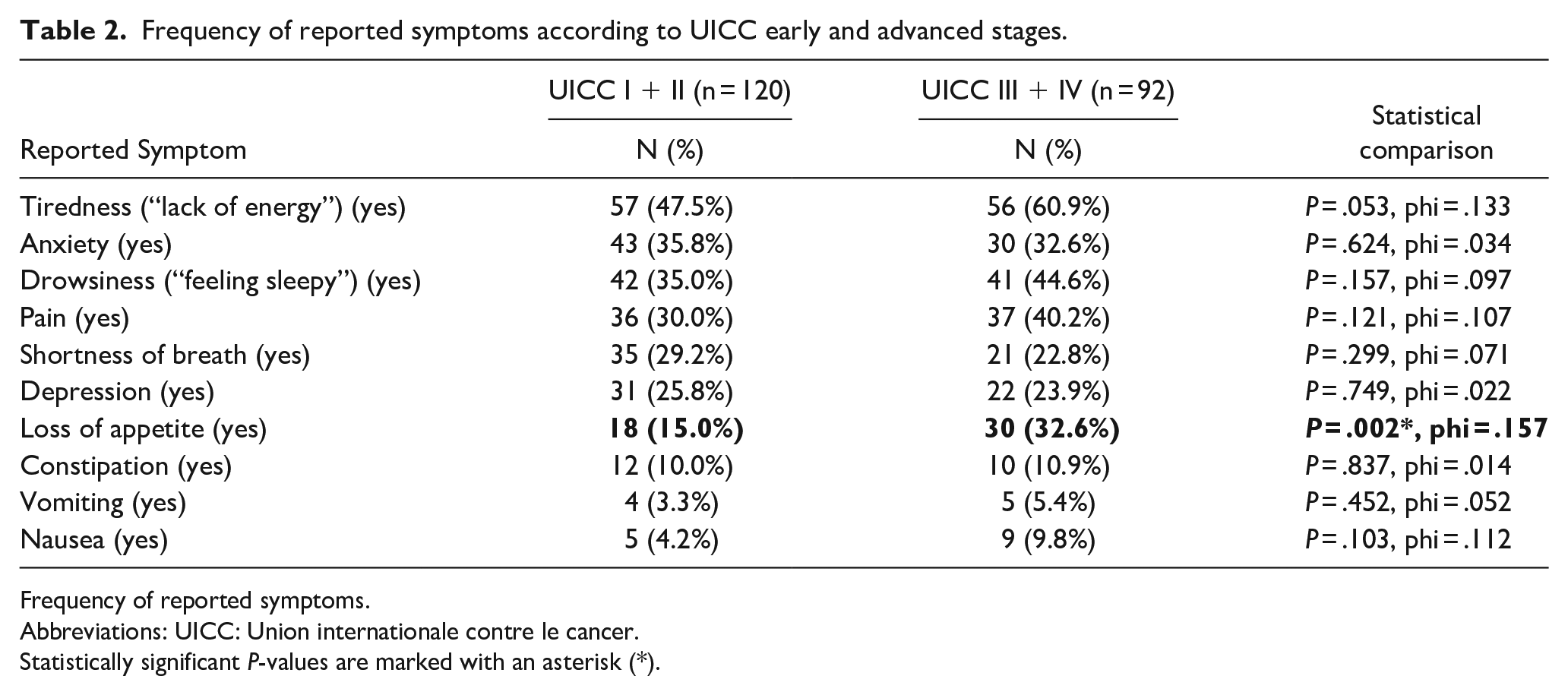
Frequency of reported symptoms.
Abbreviations: UICC: Union internationale contre le cancer.
Statistically significant P-values are marked with an asterisk (*).
The overall severity of symptoms was low in both groups and is illustrated by a severity sum score (min 0-max 30) of 3.04 ± 3.29 vs 3.58 ± 3.38 (P = .143, r = .100) (see Table 3). For almost every symptom, the median rating was 0 (=no perception, see Table S1 for further information on each item’s numerical distribution). Solely for tiredness, the median rating of the later-stage group was 1 [0;1] (= slight perception of fatigue), but no significant difference between both groups was found here (P = .073, r = .123).
Severity of reported symptoms according to UICC early and advanced stages.
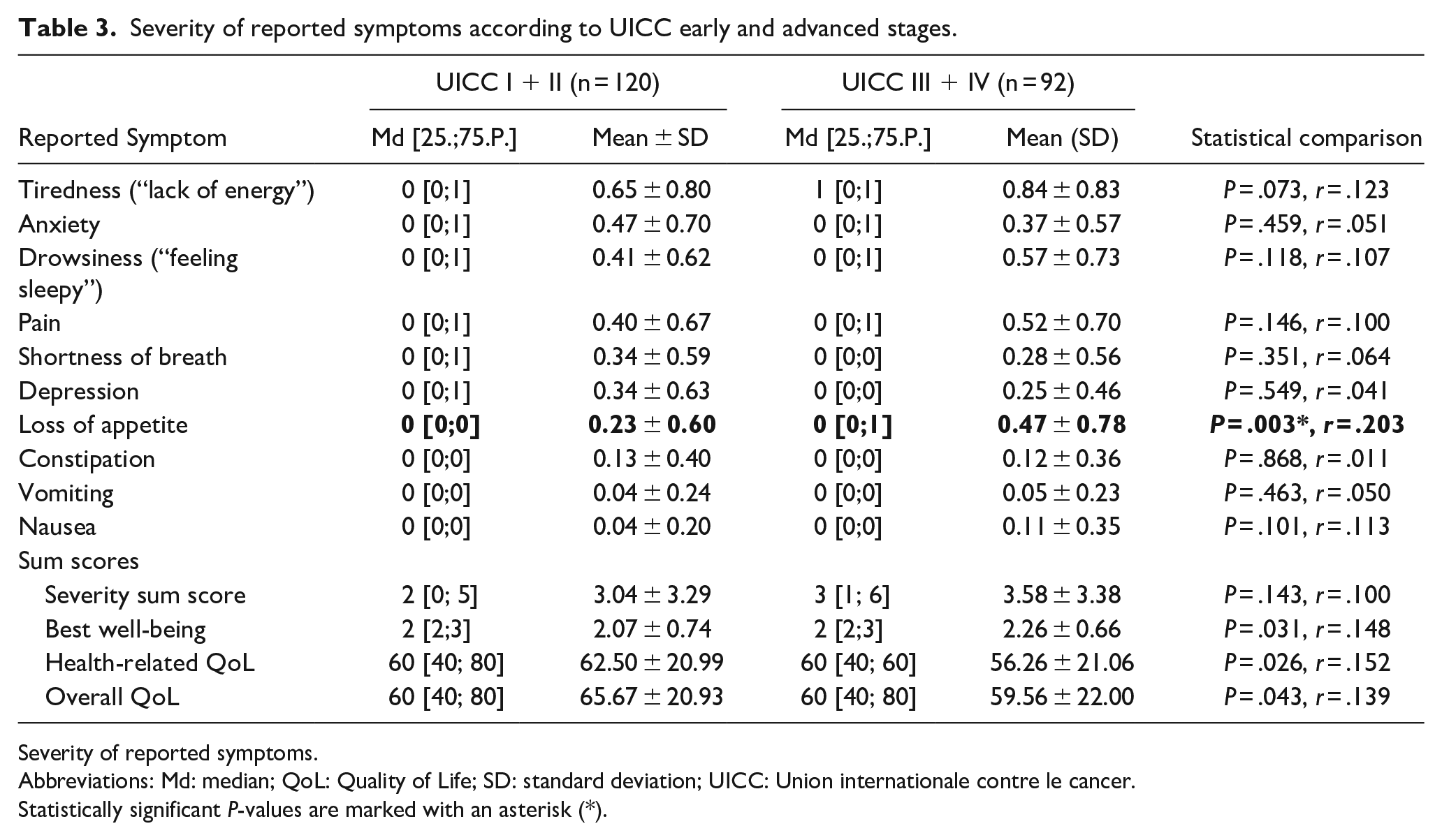
Severity of reported symptoms.
Abbreviations: Md: median; QoL: Quality of Life; SD: standard deviation; UICC: Union internationale contre le cancer.
Statistically significant P-values are marked with an asterisk (*).
The only significant difference between the two groups was detected regarding loss of appetite, which was significantly higher in the patients with advanced UICC stages (P = .003, r = .203), although both groups reported very low levels of loss of appetite (early stage: 0 [0;0], advanced stage: 0 [0;1]).
In general, after correction for multiple comparison, a trend toward a significantly lower symptom burden was found for “best well-being” (2.07 ± 0.74 vs 2.26 ± 0.66, P = .031, r = .148) for the early-stage group, which was accompanied by a tendency toward a higher health-related QoL (62.50 ± 20.99 vs 56.26 ± 21.06, P = .026, r = .152) and a higher overall QoL (65.67 ± 20.93 vs 59.56 ± 22.00, P = .043, r = .139).
Discussion
This study shows that long-term survivors of HNC complained most of tiredness, anxiety, drowsiness, and pain, regardless of the stage of the disease they were diagnosed with about four years previously, except for loss of appetite that was more frequently reported by patients treated for advanced-stage disease.
The severity of symptoms was encouragingly low for all evaluated cancer patients.
Consequently, all patients rated their health-related and general QoL as “good” on average, tending to have slightly better scores in the early-stage group.
In the “American Cancer Society Head and Neck Cancer Survivorship Care Guideline” “long-term effects” are defined as “medical problems that develop during active treatment and persist after the completion of treatment” and “late effects” are “medical problems that develop or become apparent months or years after treatment is completed.” 14
It is known from the literature that fatigue/tiredness/drowsiness, as in our study collective, are the most frequent and most burdensome symptoms of tumor patients that can persist for years after completion of therapy. 15 In a study involving 419 outpatient cancer patients of various entities, Vogelzang et al 14 reported that 78% of all patients complained of fatigue in the course of their disease, affecting their daily lives more than other symptoms. 16 In addition to diagnosing fatigue, it is important to identify possible underlying, treatable causes and encourage patients to be physically active.
Anxiety, along with depression, is also one of the common general psychological long-term and late effects in HNC patients reported in the literature. It is known that the pretreatment psychological status and caregiver support play significant roles in the magnitude of distress during and after treatment. 14 Therefore, the American Cancer Society Head and Neck Cancer Survivorship Care Guideline recommends “that primary care clinicians” “should assess HNC survivors for distress/depression and/or anxiety periodically (3 months posttreatment and at least annually).” Appropriate treatment options or referrals to specialists should be initiated and offered as needed. 14
“Pain” is “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage.” 17 It is known to have a severe impact on cancer patients’ QoL. A systemic review on pain prevalence and pain severity in patients with different cancers showed a pain prevalence of 39.3% after curative treatment in general 18 that is in line with our data. Pain in HNC patients can be caused by the tumor itself and/or different therapy modalities. Long-term and chronic pain in Head and Neck Cancer (HCN) patients primarily include shoulder pain, cervical dystonia (“painful dystonic spasms of the cervical muscles”), muscle spasms, and neuropathies. 14
There can be many reasons for loss of appetite. One of the reasons is an altered sense or loss of taste, called dysgeusia, mainly caused by head and neck radiotherapy. 19 Since radiation and chemoradiation were significantly more often performed in the advanced-stage group, this no doubt had an impact on the observed occurrence. Other possible causes of appetite loss may include chronic dysphagia or the occurrence of strictures. Both are known to be a common, challenging clinical problem. As in our study collective, chronic dysphagia is relatively rare in early-stage HNC, which is mainly treated with local radiation or single-modality surgery but estimatedly affects almost half of the patients with advanced-stage HNC treated with multimodal therapy. 20 The exact causes of reported appetite loss are beyond the scope of this study.
Nausea, vomiting, and constipation were among the least frequent and least intensely perceived symptoms in our patient population. This is most likely due to the fact that they are consequences of the acute treatment rather than of the long-term and late effects.
It is known from the literature that the health-related QoL in HNC patients is poor in the acute phase of disease and deteriorates with a combination of different treatment modalities. 21 Unfortunately, little is known about quality of life in long-term survivors. However, it appears that the QoL level is similar to that of the age-matched general population. The most prevalent problem that survivors faced was eating due to poor oropharyngeal functioning, psychological problems and substantial pain, which in some cases affected more than half of the patients. 22 Moreover, Mehanna et al 23 showed that the overall QoL at 10 years following diagnosis decreased significantly compared to pretreatment and years 1 and 2, while only poor pretreatment QoL could be identified as a risk factor. Our patients rated both their best well-being and their overall and health-related QoL as good on average. This is certainly related to the fact that we work at a large University Center with all diagnostic and therapeutic possibilities. This results in effective, long-term postoperative support for patients with correspondingly good results.
From our point of view, there are several ways to further improve the QoL of HNC patients. The top priority is certainly good symptom control. In recent years, it has become increasingly known that especially early integration of palliative medical co-care, in terms of good symptom control at an early stage, can significantly improve patients’ QoL and even prolong survival time.24–26 Therefore, palliative care should no longer be limited to end-of-life treatment, but should already be integrated into the early symptom-oriented treatment of tumor patients. 27
In addition, good communication between the primary care clinicians and the oncology team not only throughout diagnosis and treatment but also parallel to posttreatment care is essential for early problem identification and to ensure that care is evidence-based and well-coordinated.
Other influencing factors on QoL, such as financial and employment challenges or possible substance abuse, must also be kept in mind. Patients may be referred to social workers or mental health specialists in such cases.
Limitations of the study include the fact that only patients who were able to attend regular outpatient follow-up visits were included. This naturally leads to a selection bias of the study population, which is also reflected in the distribution of ECOG functional status. Moreover, it should be noted that the study was conducted at a hospital specializing in the treatment of head and neck tumor patients. It can be assumed that the results of symptom control are better than would be expected in less specialized hospitals. Patients with recurrent tumors, second primary cancers, and/or metastatic tumor stages were significantly underrepresented. 28 It must also be considered that the questionnaires were only distributed in a quasi-randomized manner and were completed by only half of all patients. The results must be interpreted accordingly. In actual fact, only the symptoms included in the MIDOS(2) questionnaire were studied. Thus, it is important to consider that other distressing symptoms, such as xerostomia, 29 trismus, 30 or persistent dysphagia, 20 which also affect many patients, may distinctly affect the QoL of HNC patients. It should be noted that only two patients took the opportunity to add additional symptoms under the item “other problem” of the questionnaire. One patient reported “distress,” another reported “difficulty concentrating.”
On the other hand, the results presented are based on a large patient cohort that represents the entire spectrum of HNC patients with a follow-up of several years. It is important to continue to investigate and improve factors potentially influencing symptom burden and QoL in HNC patients in the future.
Conclusion
This study shows that long-term survivors of HNC at a University Cancer Center report an encouragingly low overall symptom burden. Treating physicians should be aware of the most common long-term and late sequelae in HNC survivors after treatment that can be effectively treated to maintain or improve patients’ QoL. Nevertheless, further efforts are needed to develop personalized treatment options that further reduce long-term morbidity and improve patients’ QoL in the long term.
Supplemental Material
sj-docx-1-ear-10.1177_01455613241276776 – Supplemental material for Symptom Burden and Quality of Life in Long-Term Survivors with Head and Neck Cancer
Supplemental material, sj-docx-1-ear-10.1177_01455613241276776 for Symptom Burden and Quality of Life in Long-Term Survivors with Head and Neck Cancer by Magdalena Gostian, Moritz Allner, Atina Rak, Matthias Balk, Robin Rupp, Christoph Ostgathe, Konstantinos Mantsopoulos, Matti Sievert, Heinrich Iro, Markus Hecht and Antoniu-Oreste Gostian in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
Author Contributions
All authors meet the ICMJE authorship criteria.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication of the data collected.
Data Availability
The datasets generated during and/or analyzed during the current study are available from the Corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of the University of Erlangen-Nürnberg (04.09.2019 / approval number: 486_18 B).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
