Abstract
Keywords
Introduction
In the rapidly evolving field of oncology, head and neck cancer (HNC) remains a significant concern, ranking as the sixth most prevalent form of cancer globally. 1 Despite significant advancements in immunotherapy and technology, survival rates for late-stage diagnosis remain suboptimal. 2 A comprehensive approach that includes medical, surgical, and psychosocial interventions is increasingly acknowledged as integral to improving patient outcomes.
Recent studies have shed light on the challenges faced by HNC survivors, particularly the severity and prevalence of physical and psychological symptoms. These findings emphasize the importance of managing functional issues to enhance long-term quality of life (QoL). 3 In addition, there is a growing recognition of the need for treating physicians to be aware of common long-term sequelae that can be effectively managed to improve patient outcomes. 4
Despite these insights, gaps remain in our knowledge, especially concerning the differential impact of cancer localizations on symptom burden and QoL in HNC patients. In our subsequent research, we will build upon these findings by focusing specifically on this unexplored area. Given that our prior work indicated the importance of various influencing factors such as sociodemographic characteristics, therapeutic approaches, and oncological conditions, this study aims to extend this line of inquiry to examine how these factors interact with cancer localizations to shape symptom burden and QoL.
The forthcoming study aims to address the gaps in current knowledge by exploring the role of cancer localizations in influencing symptom burden in HNC patients. In addition, the study seeks to elucidate the relationships between QoL and different cancer localizations.
Conducting this research in a leading HNC center employing a multidisciplinary care approach will enable a more profound exploration of the specialized care needs of HNC patients. The insights gained from this study are intended to contribute to the development of more effective, personalized treatment strategies, thereby improving the QoL for individuals diagnosed with HNC.
Materials and Methods
This investigation serves as an extension of our previous studies. Both aimed to assess symptom burden and QoL in patients with HNC. The current study specifically targets the influence of cancer localization on symptom burden and QoL, employing data derived from the University of Washington Quality of Life Questionnaire (UW-QoL) Version 4.
Ethical clearance for this study was obtained from the Ethics Committee of the University of Erlangen (Approval No.: 486_18 B). The research protocol adhered to the tenets of the Declaration of Helsinki as well as the STROBE and CONSORT guidelines.5,6 Registration was duly completed with the German Registry for Clinical Studies under application No.: 00017122.
The initial patient pool comprised 503 individuals receiving care at the University Hospital Erlangen’s Department of Otolaryngology, Head & Neck Surgery, Germany, between July 7th and December 31st, 2019. Following the attainment of informed consent, patients were administered the UW-QoL questionnaire during their follow-up visit. However, to ensure a more homogenized study population and precise analysis, we refined our cohort from this initial pool. This refinement involved the exclusion of patients (n = 74) with diverse cancer types such as skin cancer, thyroid gland cancer, salivary gland cancer, and systemic cancer that manifests in the head and neck region, which could introduce significant variability in disease behavior and treatment response. In addition, patients who failed to return the questionnaire (n = 57), submitted incomplete responses (n = 57), or did not meet the inclusion criteria (n = 15) were also excluded. After these considerations, a total of 138 patients were deemed eligible for inclusion in this analysis, focusing on a more uniform group to enhance the relevance and applicability of our findings to HNC localizations.
Inclusion criteria stipulated that patients must be 18 years or older, diagnosed with HNC across any Union Internationale Contre le Cancer (UICC) stage (I-IV), possess complete medical records, and demonstrate adequate cognitive and linguistic capabilities to independently complete the questionnaire. Patients were also required to provide written informed consent after receiving detailed study information. The exclusion criteria encompassed those below the age of 18, those unable to complete the questionnaire unassisted, individuals suffering from mental incapacities, and those who personally opted not to participate in the study.
The UW-QoL questionnaire served as the primary instrument for the evaluation of symptom burden, featuring 12 domains: pain, appearance, activity, recreation, swallowing, chewing, speech, shoulder function, taste, saliva, mood, and anxiety. This validated tool employs a 0- to 100-point scale for each domain and two global questions concerning health-related and overall QoL, wherein higher scores denote reduced symptom burden.7,8
Comprehensive clinical and oncological variables such as the time-lapse from initial diagnosis to interview, regular medication regimen, Eastern Cooperative Oncology Group (ECOG) status, cancer localization, treatment modality, and cancer recurrence were retrieved from patients’ medical records.
Statistical Computation
Statistical analyses were conducted in R, using the package effect size. 9 Categorical variables are reported using absolute and relative frequencies (n/%). For inferential statistics on cancer localization, we used for nominal variables the χ2 test along with the Cramer’s V effect size. 10 In the event of a significant primary effect, we used pairwise comparisons (again with the χ2 test and phi) between individual groups. P-values were adjusted using the Benjamini-Hochberg correction. 11 For continuous variables, we report mean and standard deviation, in the case of normally distributed variables as descriptive measures or the median [25.;75 percentile] in the case of not normally distributed variables or ordinally scaled variables. To test for differences between cancer locations, we first conducted either a univariate analysis of variance or a Kruskal-Wallis test (in case of normally/not normally distributed continuous variables or ordinally scaled variables) and estimated the partial/rank η 2 effect size. 12 For significant main effects of cancer localization, we then calculated pairwise comparisons using the unpaired t-test/Wilcox test (with Benjamini-Hochberg adjusted P-values) and the Pearson/rank biserial correlation coefficient. 12 Effects with an (adjusted) P-value of ≤0.05 were defined as significant.
Results
Patient Collective
Between July 1, 2019 and December 31, 2019, a total of 503 patients participated in their scheduled follow-up visits at the University Hospital Erlangen’s Department of Otolaryngology, Head & Neck Surgery. Of this cohort, 446 individuals returned their questionnaires, resulting in an 88.7% response rate. In all, 219 patients, representing 49.1% of the returned questionnaires, were excluded due to incomplete responses. An additional 15 participants were also omitted as they did not meet the predefined inclusion criteria. Seventy-four patients were excluded because of their cancer types to ensure a more homogenized study population (refer to Figure 1).
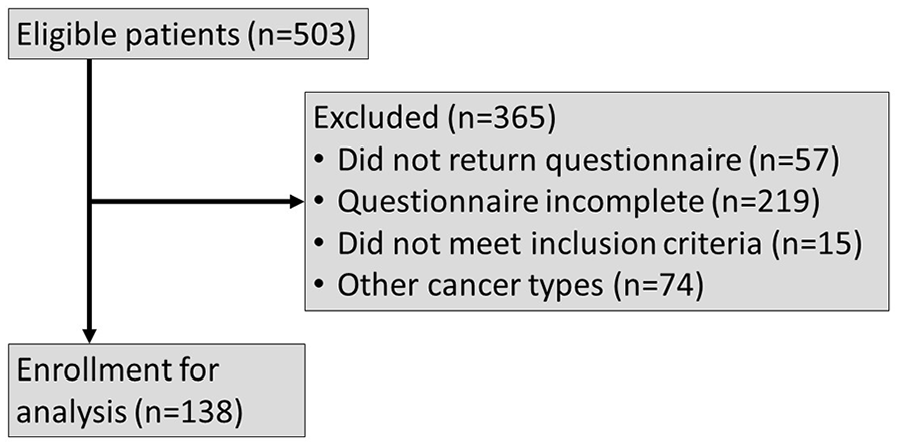
CONSORT flow chart for enrollment.
Patient Characteristics
Our study population included a total of 138 patients with various localizations of HNC, divided into six primary groups: oral cavity (N = 24), oropharyngeal (N = 48), hypopharyngeal (N = 11), laryngeal (N = 46), and cancer of unknown primary (CUP) (N = 9). For complete data, refer to Table 1.
Demographic data.
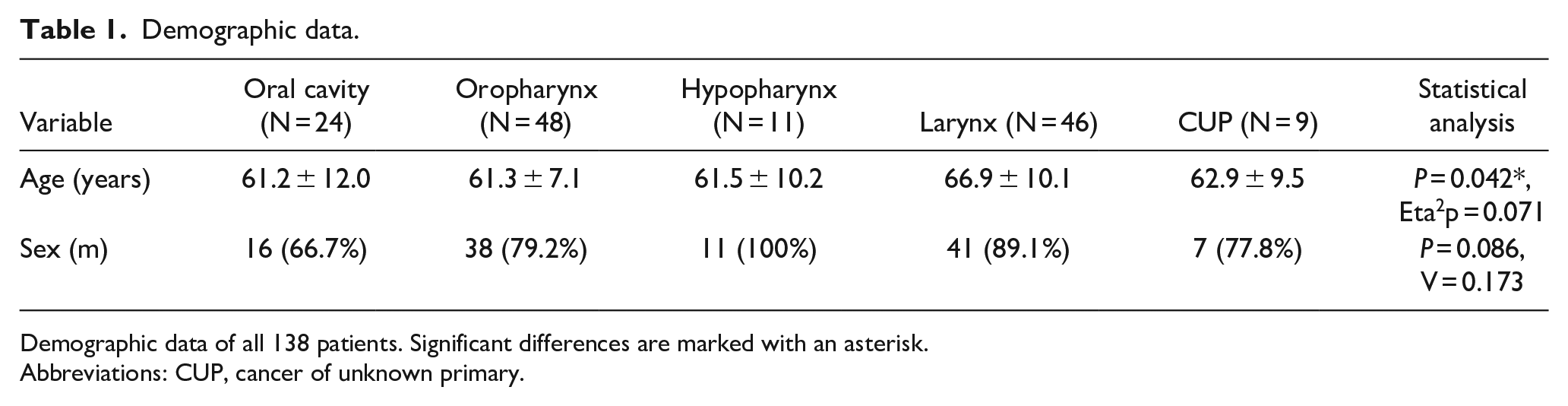
Demographic data of all 138 patients. Significant differences are marked with an asterisk.
Abbreviations: CUP, cancer of unknown primary.
A significant difference in mean age was observed across these groups (P = 0.042, Eta2p = 0.071). The ages varied, with oral cavity patients averaging 61.2 years (±12.0), oropharyngeal at 61.3 years (±7.1), hypopharyngeal at 61.5 years (±10.2), laryngeal at 66.9 years (±10.1), and CUP at 62.9 years (±9.5). Post hoc analyses, corrected for multiple comparisons, revealed a significant age difference, specifically a lower age in patients with oropharyngeal carcinomas compared to those with laryngeal carcinomas (P = 0.027, r = 0.642).
In terms of gender distribution, no notable differences were observed across these groups (P = 0.086, V = 0.173). The proportions were as follows: 66.7% males in the oral cavity group, 79.2% in the oropharyngeal, 100% in the hypopharyngeal, 89.1% in the laryngeal, and 77.8% in the CUP group. Adjusted post hoc comparisons for gender did not reveal any significant trends or differences.
Clinical Background
Our analysis of the clinical background of HNC patients revealed varied findings across different cancer localizations. For complete data, refer to Table 2.
Clinical data.
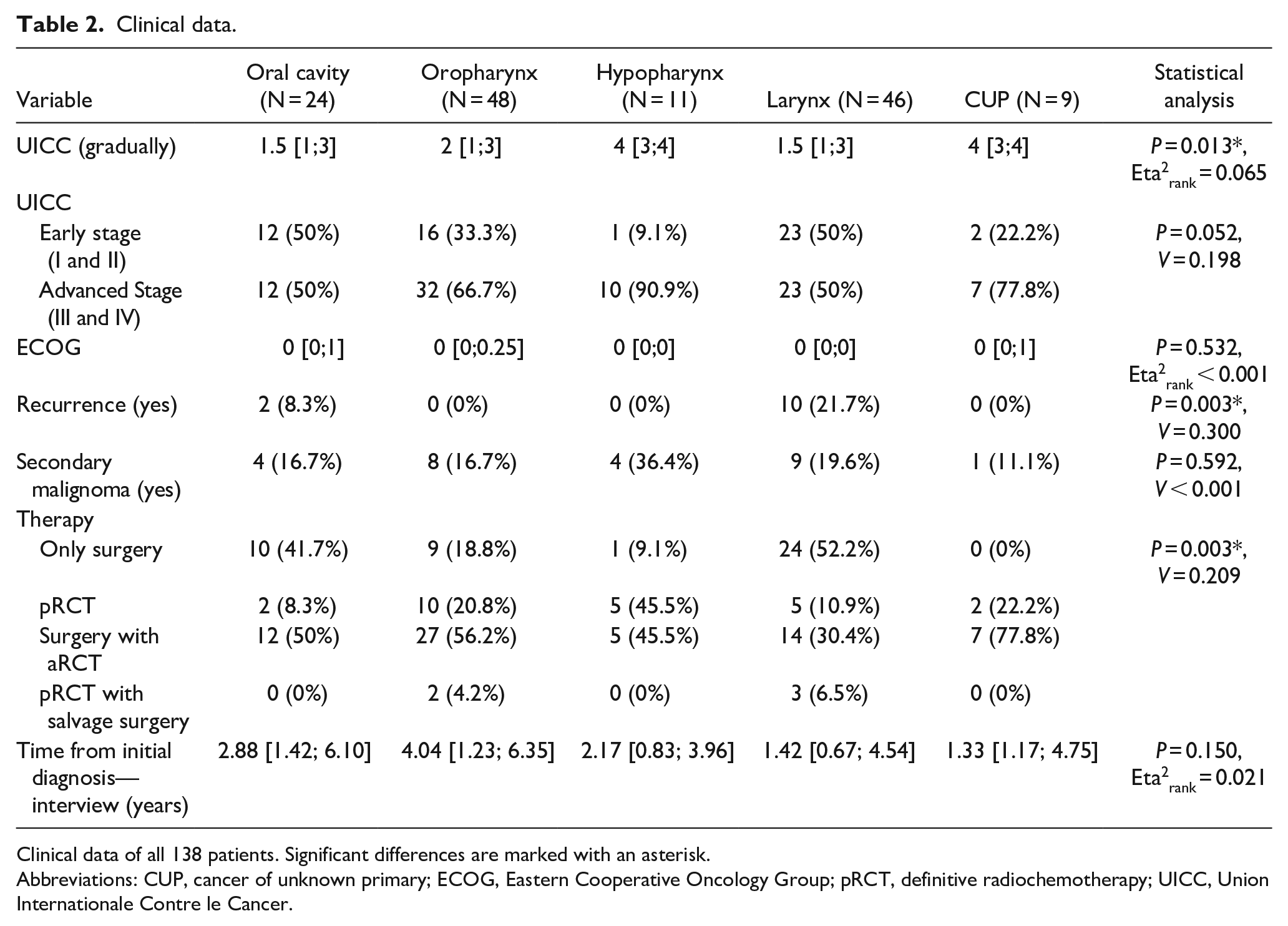
Clinical data of all 138 patients. Significant differences are marked with an asterisk.
Abbreviations: CUP, cancer of unknown primary; ECOG, Eastern Cooperative Oncology Group; pRCT, definitive radiochemotherapy; UICC, Union Internationale Contre le Cancer.
Significant differences were observed in the UICC stages across groups (P = 0.013, Eta2rank = 0.065). Specifically, patients with oral cavity, oropharyngeal, and laryngeal carcinomas had lower UICC stages compared to those with hypopharyngeal carcinomas. This was evident in pairwise comparisons: oral cavity versus hypopharyngeal (P = 0.042, r = 0.508), oropharyngeal versus hypopharyngeal (P = 0.038, r = 0.504), and laryngeal versus hypopharyngeal (P = 0.038, r = 0.520). A marginally significant effect was noted for grouped UICC stages (early vs. late; P = 0.052, V = 0.198), although no differences were found in post hoc tests. The ECOG performance status showed no significant differences (P = 0.532, Eta2rank < 0.001).
The rate of recurrences significantly differed (P = 0.003, V = 0.300), with oropharyngeal carcinoma patients exhibiting fewer recurrences compared to laryngeal carcinoma patients (P = 0.010, phi = 0.339). The occurrence of a secondary carcinoma did not show significant differences (P = 0.592, V < 0.001).
Significant differences were noted in treatment modality distributions (P = 0.003, V = 0.209). Laryngeal carcinoma patients more frequently underwent surgical intervention alone compared to oropharyngeal (P = 0.010, phi = 0.336), CUP (P = 0.020, phi = 0.368), and hypopharyngeal carcinoma patients (P = 0.033, phi = 0.318). In addition, CUP patients were less likely to undergo surgery alone compared to oral cavity carcinoma patients (P = 0.050, phi = 0.369).
Symptom Burden
A comprehensive assessment was conducted encompassing a wide array of symptoms: pain, appearance, activity, recovery, mood, anxiety, chewing, shoulder pain, and taste. For complete data, refer to Table 3.
Symptom burden.
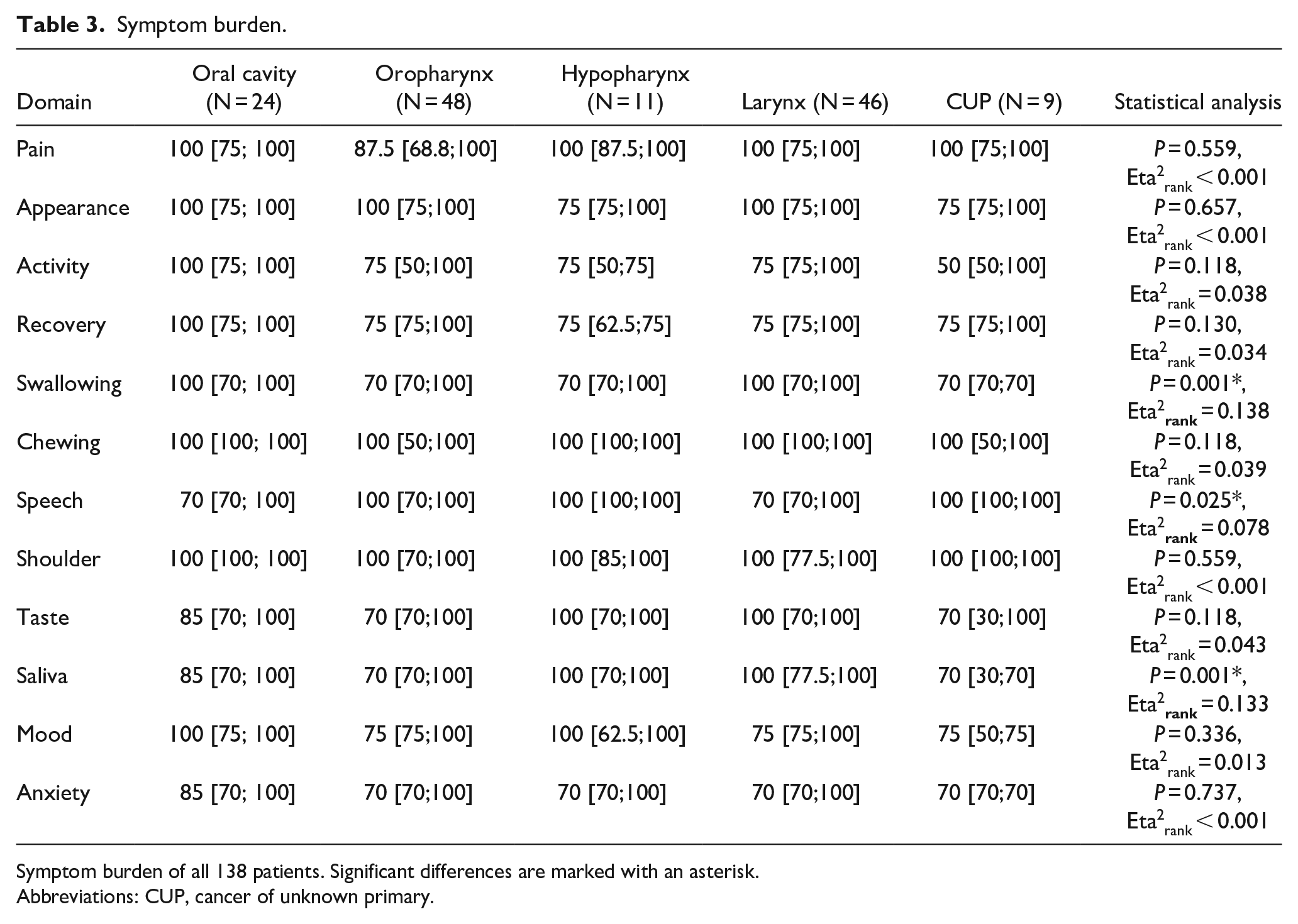
Symptom burden of all 138 patients. Significant differences are marked with an asterisk.
Abbreviations: CUP, cancer of unknown primary.
This evaluation revealed a consistent pattern across these symptoms, with no significant variations observed in their expression across different cancer localizations.
Uniformly high scores were noted across all groups in pain (P = 0.559, Eta2rank < 0.001). A majority of patients, regardless of cancer type, reported positive perceptions of their appearance (P = 0.657, Eta2rank < 0.001). A similar level of activity (P = 0.118, Eta2rank = 0.038) and recovery (P = 0.130, Eta2rank = 0.034) was observed in patients across all cancer types. The prevalence of mood (P = 0.336, Eta2rank = 0.013) and anxiety (P = 0.737, Eta2rank < 0.001) symptoms was comparably uniform across the groups. Furthermore, no significant differences were found for the symptoms of chewing (P = 0.118, Eta2rank = 0.039), shoulder (P = 0.559, Eta2rank < 0.001), and taste (P = 0.118, Eta2rank = 0.043).
While some symptoms showed consistency, other symptoms such as swallowing (P = 0.001, Eta2rank = 0.138), speech (P = 0.025, Eta2rank = 0.078), and salivation (P = 0.001, Eta2rank = 0.133) exhibited notable differences among the groups.
For swallowing distinct disparities emerged, especially between patients with oral cavity and laryngeal carcinomas versus those with oropharyngeal carcinomas. Oral cavity (100 [70; 100]) and laryngeal (100 [70;100]) groups reported significantly higher swallowing scores than oropharyngeal (70 [70;100]), particularly oral cavity versus oropharyngeal (P = 0.022, r = 0.362) and laryngeal versus oropharyngeal (P < 0.001, r = 0.448). In addition, laryngeal patients showed higher scores in comparison to CUP (70 [70;70]) patients (P = 0.022, r = 0.507).
Analysis of speech-related symptoms showed that patients with oral cavity (70 [70; 100]) and laryngeal (70 [70;100]) carcinomas had significantly lower scores compared to those with CUP (100 [100;100]). Marginal differences were also noted between oral cavity (P = 0.067, r = 0.402), and laryngeal (P = 0.067, r = 0.397) groups versus hypopharyngeal (100 [100;100]) carcinomas, and oropharyngeal (100 [70;100]) versus CUP (P = 0.067, r = 0.417).
Patients with laryngeal carcinoma reported notably higher salivation scores (100 [77.5;100]) compared to those with oropharyngeal (70 [70;100]) and CUP (70 [30;70]) carcinomas. Statistically, these differences were significant: laryngeal versus oropharyngeal (P = 0.001, r = 0.394) and laryngeal versus CUP (P = 0.001, r = 0.725).
Furthermore, marginal differences were observed in salivation scores when comparing oral cavity (85 [70; 100]) and hypopharyngeal (100 [70;100]) carcinomas with CUP. Specifically, oral cavity carcinoma patients tended to have higher salivation scores than those with CUP (P = 0.092, r = 0.426), but lower scores compared to Laryngeal carcinoma patients (P = 0.078, r = 0.283). In addition, hypopharyngeal carcinoma patients showed marginally higher salivation scores compared to CUP (P = 0.092, r = 0.525).
Quality of Life
A thorough evaluation of QoL was conducted. For detailed data, please see Table 4.
Quality of life.
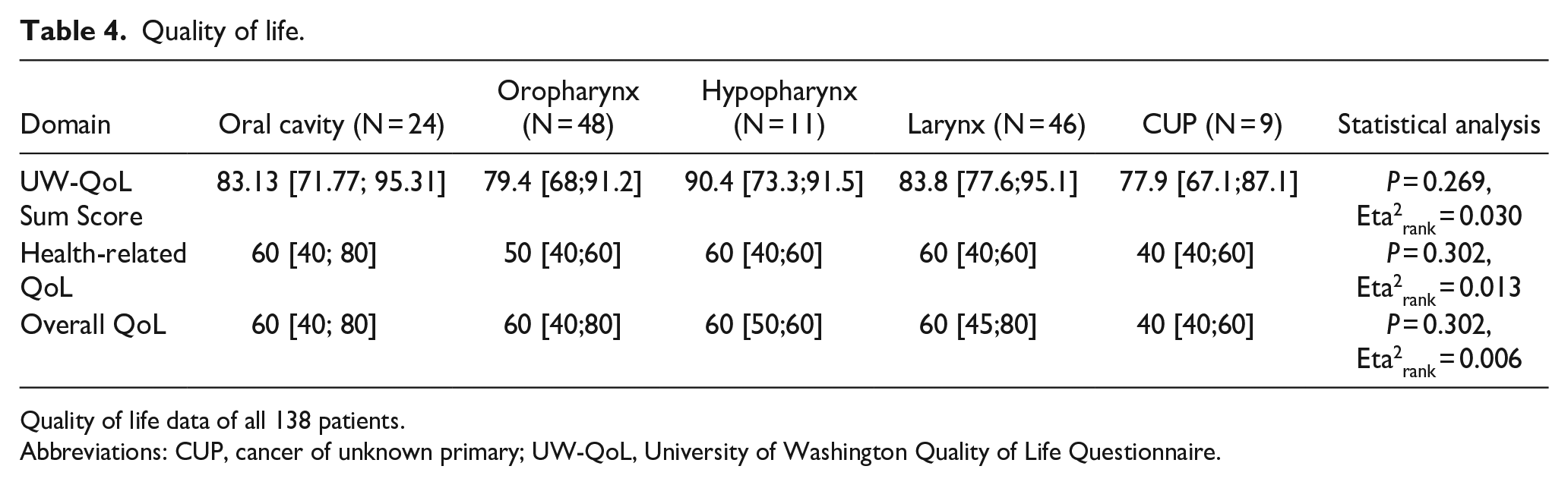
Quality of life data of all 138 patients.
Abbreviations: CUP, cancer of unknown primary; UW-QoL, University of Washington Quality of Life Questionnaire.
UW-QoL sum scores displayed a range across cancer localizations, with oral cavity at 83.13 [71.77;95.31], oropharyngeal at 79.4 [68;91.2], hypopharyngeal at 90.4 [73.3;91.5], laryngeal at 83.8 [77.6;95.1], and CUP at 77.9 [67.1;87.1]. However, statistical analysis revealed no significant differences among these groups (P = 0.269, Eta2rank = 0.030).
When examining the overall QoL scores, a consistent pattern was observed, with scores generally centering around the 60s mark for most groups, except for the CUP group, which scored slightly lower. Specifically, the scores were oral cavity: 60 [40;80], oropharyngeal: 60 [40;80], hypopharyngeal: 60 [50;60], laryngeal: 60 [45;80], and CUP: 40 [40;60]. The differences in these scores did not reach statistical significance (P = 0.302, Eta2rank = 0.006).
Similarly, in terms of health-related QoL, the scores ranged without significant variation among the different cancer localizations. The scores were consistent with the overall QoL scores, indicating a uniformity in health-related QoL across the cancer types (P = 0.302, Eta2rank = 0.013).
The lack of significant differences across various cancer sites underscores the uniform impact of HNC on QoL, regardless of cancer localization. For complete data, refer to Table 4.
Discussion
In this investigation conducted at the University Hospital Erlangen’s Department of Otolaryngology, we delve into the complex interrelationships between various HNC localizations and their consequent post-treatment symptoms and QoL. Our exploration reveals significant disparities in functionalities such as swallowing, speech, and salivation, particularly highlighting the struggles of patients with oral, oropharyngeal, and CUP syndrome. Despite these divergent symptomatologies, the QoL manifests as a consistent theme, seemingly unaffected by cancer localization.
Our study’s findings on age-related differences across various cancer localizations align with existing literature. The average age for oropharyngeal cancers in our cohort was 61.3 ± 7.1 years, closely mirroring the literature’s average of 64 years. 13
The younger onset age of oropharyngeal carcinoma compared to laryngeal (66.9 years ± 10.1, P = 0.042, Eta2p = 0.071) carcinoma could be attributed to the high prevalence of Human Pappiloma Virus infection in oropharyngeal cases, which is associated with younger patients and distinct lifestyle factors. 14 By contrast, laryngeal carcinoma, often linked to long-term tobacco and alcohol use, typically manifests in older individuals, with most patients diagnosed over the age of 65. 15
In our study, we observed significant differences in recurrence rates between oropharyngeal and laryngeal carcinoma patients, highlighting the importance of considering primary tumor sites in recurrence risk assessment.
These findings align with previous research, including a cohort study from Oslo, Norway, which identified primary tumor site, age, and tumor stage as key recurrence risk factors. Younger patients and those with supraglottic cancer faced higher recurrence risks, especially within the first 3 years post-treatment, emphasizing the need for vigilant follow-up in these groups. 16
Similarly, a retrospective study on oral cavity and oropharyngeal carcinomas underscored the impact of treatment approaches on recurrence rates. Surgery, followed by postoperative radiotherapy, was associated with the lowest locoregional recurrence rates, making it a recommended choice for initial clinical-stage cases. 17
In our post-treatment analysis, significant disparities in swallowing function were observed across different cancer localizations. Oral cavity cancer patients showed less compromised swallowing scores compared to oropharyngeal and laryngeal cancer patients. Swallowing dysfunction is a prevalent complication experienced by patients undergoing treatment for oral cancer, specifically due to interventions such as surgery, radiotherapy, and chemotherapy. 18 The mechanism of swallowing is extensively compromised due to impairments such as reduced mobility of the residual structures and sensory damage, induced by these treatment modalities. 19 Age, the precise location, and size of the tumor, alongside the chosen treatment protocol, are pivotal determinants influencing swallowing. 20
But for oropharyngeal cancer particularly medical interventions with extension of the cancer into the tongue base have been emphasized as pivotal contributors to declining swallowing function, substantially impacting the patient’s QoL.21–23
Patients with hypopharyngeal and laryngeal cancers displayed better swallowing outcomes, correlating with studies focusing on surgery-oriented comprehensive treatment in hypopharyngeal cancer, which illustrated satisfactory swallowing outcomes in 73.8% of patients. However, pharyngo-cutaneous fistula and local-regional recurrence were identified as independent factors impacting swallowing function. 24 A separate study emphasized the repercussions of partial laryngectomy on swallowing function and associated QoL in patients with laryngeal carcinoma. Despite the gradual improvement in swallowing function and related QoL post-surgery, full restoration remained elusive for some patients for up to 48 weeks, highlighting the need for enhanced postoperative care. 25 Lastly, another study illuminated the widespread occurrence of dysphagia following total laryngectomy and pharyngolaryngectomy, a finding that aligns with our results but does not severely impact the patients’ QoL. 26
Patients with CUP syndrome also experienced compromised swallowing function. In CUP, the primary cancer site is typically unidentified, leading to extensive radiation therapy that covers the oral cavity, pharynx, and larynx. This comprehensive radiation therapy can result in severe xerostomia, significantly impacting swallowing function. These impairments tend to peak at 3 months post-treatment and persist despite subsequent improvements. 27
In light of evaluating post-treatment speech outcomes, our research disclosed pronounced disparities in speech capabilities, contingent upon the specific sites of cancer localization. Notably, patients afflicted with oral cavity carcinomas manifested substantially diminished speech scores, a contrast to patients with CUP syndrome. This differentiation in speech outcomes may be attributed to surgical procedures typically necessitated for oral cavity cancers, often involving intricate reconstructive methods such as the use of free flaps, potentially impacting residual speech functionalities.
Drawing upon insights from a study by Suarez-Cunqueiro et al, it is elucidated that a substantial proportion of patients, 63.8% reported speech problems and 75.4% faced swallowing complications post-radical surgery for oral and oropharyngeal cancer. The association of speech and swallowing impairments was significantly correlated with various factors including sex, cancer location, pTNM stages, stage of the cancer, the modality of treatment, and the type of reconstruction employed. Specifically, patients who underwent combined radiotherapy and surgical tumor resection, those with advanced-stage tumors (III-IV), or those with tumors situated on the floor of the mouth were observed to be at a heightened risk of enduring severe speech and swallowing impairments. 28
Patients with hypopharyngeal and laryngeal carcinomas often exhibit compromised speech due to the necessity of complex procedures like partial or complete laryngectomy or even laryngopharyngectomy, resulting in permanent tracheostomy. These interventions, by removing the vocal cords, substantially alter speech quality, contrasting sharply with the experiences of those with cancers not directly involved in speech production.
This is consistent with a study focused on the repercussions of chemo-radiotherapy in patients with locally advanced laryngeal and hypopharyngeal cancers, demonstrating that even subclinical voice disorders post-treatment are common and can impact QoL. 29 In essence, both intricate surgical interventions and chemo-radiotherapy significantly impact speech in patients with hypopharyngeal and laryngeal carcinomas, underscoring the multifaceted nature of post-treatment experiences in such cases.
The findings of the study reveal a certain uniformity in the QoL outcomes across different cancer localizations post-treatment, as no significant differences were noted in the sum score of the University of Washington Quality of Life (UW QoL) questionnaire, the health-related QoL, and overall QoL after corrections for multiple comparisons were applied. These uniform outcomes across various cancer localizations suggest that the location of the tumor may not be a decisive factor in impacting the QoL, at least in the contexts measured.
In addition, while the study did not find significant differences in QoL scores across different cancer localizations, it is essential to acknowledge the specific challenges and symptom burdens associated with each cancer type, as discussed in previous sections, such as disparities in swallowing, speech, and saliva scores. Recognizing and addressing these specificities is crucial for providing comprehensive care and support to patients, focusing on alleviating symptom burden and enhancing QoL in a more targeted and personalized manner.
Acknowledging the potential limitations of our study is crucial to fully comprehend its scope and implications. A significant limitation stems from our dependence on the UW-QoL questionnaire, which is subject to the biases and inconsistencies inherent to self-reported data, potentially impacting the reliability and accuracy of our findings. Our stringent inclusion criteria, requiring participants to have adequate cognitive and linguistic abilities to complete the questionnaire independently, could limit the generalizability of our findings. This excludes a segment of the HNC patient population and potentially introduces selection bias, impacting the representativeness and external validity of our study. The considerable exclusion of participants due to incomplete responses or failure to meet inclusion criteria may also skew results and limit the comprehensive understanding of symptom burden across a broader spectrum of HNC patients. The reliance on clinical and oncological variables from existing medical records may omit significant factors impacting patient symptom burden and QoL, affecting the reliability of drawn associations between cancer localization and symptom burden. Another limitation stems from the broad categorization of oral cavity cancer, which encompasses tumors in the oral mucosa, gums, palate, tongue, and floor of the mouth. These sub-areas can impact symptom burden and QoL differently, potentially distorting our results. However, more precise insights into the influence of tumor localization within these smaller subunits of the oral cavity would require a larger population of patients with oral cavity carcinomas. In addition, the type of treatment performed on patients significantly influences symptom burden and QoL in HNC patients. Our results indicate that different treatments lead to varied outcomes across cancer groups. The statistical approach, despite meticulous application, has inherent limitations, and the assumptions made during statistical tests and the reliance on P-values might influence result interpretation, potentially overlooking critical nuances in the data. Lastly, focusing primarily on cancer localization without exploring a wide range of contributing factors might offer a somewhat unilateral perspective, restraining the depth of the study’s conclusions.
While our study noted uniform QoL outcomes, individualized management strategies are imperative, emphasizing specific supportive care corresponding to unique symptom burdens and challenges of each cancer type, ensuring optimal post-treatment QoL. Essentially, this nuanced, tailored approach to diagnosis, treatment, and management can significantly optimize overall patient outcomes in various HNCs, making care strategies more responsive and adaptive to individual patient needs and challenges.
Given the nuances in QoL outcomes across different HNC localizations identified in this study, future research should prioritize developing a deeper understanding of how cancer localization and its corresponding treatment impact specific aspects of QoL. It is critical that upcoming studies focus on creating refined, location-specific interventions and management strategies to mitigate symptom burden and substantially improve the overall QoL for patients, assessing long-term effects and diverse patient populations to ensure comprehensive insights.
Conclusion
Our study into various HNC localizations unveiled crucial insights, emphasizing the substantial disparities in post-treatment symptoms and QoL while showing uniformity in overall QoL scores. Specific symptom burdens such as swallowing, speech, and salivation exhibited significant variability, revealing the nuanced impacts of cancer localization and treatment focal points on patients’ post-treatment experiences. The revealed disparities underscore the imperative need for more individualized management and treatment strategies, focused on alleviating the unique symptom burdens associated with each HNC localization to optimize post-treatment QoL. Future research endeavors should delve deeper into the intricate relationships between cancer localizations and their specific impacts on diverse aspects of QoL, paving the way for the development of refined, location-specific interventions and management strategies, tailored to the unique needs and challenges of diverse HNC patient populations.
Footnotes
Authors’ Note
This article was created using the STROBE and CONSORT guidelines.
Author Contributions
M.A. conception and design, acquisition of data, analysis, and interpretation of data, wrote the main manuscript; A.R. acquisition of data, analysis and interpretation of data; M.G. conception and design, analysis and interpretation of data; R.R. drafting and critical revision of the article; J.G. drafting and critical revision of the article; M.S. drafting and critical revision of the article; S.M. drafting and critical revision of the article; M.K. critical revision of the article, final approval of the version to be published; H.I. critical revision of the article, final approval of the version to be published; F.P. critical revision of the article, final approval of the version to be published; T.W. critical revision of the article, final approval of the version to be published; M.A. conception and design, acquisition of data, analysis and interpretation of data, drafting and critical revision of the article, final approval of the version to be published; A.G. conception and design, acquisition of data, analysis and interpretation of data, drafting and critical revision of the article, final approval of the version to be published; M.A. and A.G. have contributed equally to the manuscript and share the last authorship; All authors have read and approved the manuscript.
Availability of Data and Materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request because the data cannot be anonymized.
Consent for Publication
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The Ethics Committee of the University of Erlangen-Nürnberg has approved the presented work (approval number: 486_18 B). The need for informed consent was waived off by the Ethics Committee of the Friedrich-Alexander-Universität (FAU), Erlangen-Nürnberg because this was a retrospective study. This study was carried out according to the Declaration of Helsinki (in accordance with the relevant guidelines and regulations).
Statement
During the preparation of this manuscript, the authors utilized the services of ChatGPT-4 and the ![]() translation tool for translating texts into English and checking grammar and spelling. Following the use of these tools, the authors thoroughly reviewed and edited the content as necessary. The authors take full responsibility for the content of the publication, ensuring its accuracy and coherence after the application of these language assistance tools.
translation tool for translating texts into English and checking grammar and spelling. Following the use of these tools, the authors thoroughly reviewed and edited the content as necessary. The authors take full responsibility for the content of the publication, ensuring its accuracy and coherence after the application of these language assistance tools.
