Abstract
Introduction
Chronic rhinosinusitis (CRS) is an inflammatory lesion involving the nasal cavity and one or more sinus mucous membranes that is symptomatic for more than 12 weeks. 1 As an inflammatory disease of the sinus mucosa, CRS often coexists with rhinitis; therefore, it is also known as nasosinusitis. Based on nasal endoscopy, CRS can be classified into chronic rhinosinusitis with nasal polyps (CRSwNP) and chronic rhinosinusitis without nasal polyps (CRSsNP). 2 The case classification of CRS is complicated, and it is usually divided into neutrophilic infiltration, eosinophilic infiltration, lymphocyte infiltration, and mixed type, according to the infiltration of the inflammatory cells. 3 The main symptoms of CRS include pus, nasal congestion, facial pressure/pain, and altered/decreased sense of smell. 4 The pathogenesis of CRS is complex and involves several factors, including genetic, environmental, and anatomical factors.5,6 At present, drug therapy and surgical treatment are commonly used to treat CRS, and patients can obtain good control through single or combined treatment. 7 Therefore, secondary prevention, that is, early detection, early diagnosis, and early treatment, is of great significance for patients with CRS.
The global prevalence of CRS is approximately 5% to 10%, and there is an error in prevalence, given that various epidemiological studies have identified the disease differently.5,8 The prevalence of CRS increases gradually with age 9 and declines gradually after reaching a peak at 50 to 59 years of age. 10 Women are more likely to have CRSsNP, whereas men are more likely to have CRSwNP. 11 Similar to united airway diseases, 12 allergic rhinitis and asthma increase the risk of CRS, especially when combined. In patients, gastroesophageal reflux syndrome could promote the development of CRS through the effect of reflux fluid on the nasal mucosa or sinus mucosa.11,13 Autoimmune diseases and psychiatric disorders are associated with CRS. 11 Among the individual behavioral factors, smoking, especially active smoking, increases the risk of CRS. 14 In terms of occupational factors, the prevalence of CRS in blue-collar workers is higher than that in white-collar workers, and occupational exposure to dust, smoke, or toxic gases and cleaning-related work significantly increases the risk of CRS. 15 Socioeconomic factors, including social class and educational level, are not clearly associated with CRS. 16 Sulfur dioxide, nitrogen dioxide, carbon monoxide, and particulate matter in outdoor environments are associated with CRS, although only weakly, whereas those in indoor environments are not associated.11,17 However, in a multicenter cross-sectional study, researchers found that indoor environmental factors, such as winter warmth, summer air-conditioning time, frequency of exposure to humidity, and pet ownership, are strongly associated with CRS. 15
Existing epidemiological studies on CRS have focused more on environmental or genetic factors and less on other factors. Apart from researching about the pathophysiology of CRS, 18 it is also important to explore the association between clinical indicators and the disease. This study analyzed the association of routine blood parameters and biochemical markers with the disease in patients with CRS and healthy subjects from Ningbo, China. Considering that the development of predictive models for CRS is currently lacking, a disease risk prediction model was constructed and evaluated based on these results. This study aimed to develop and verify a risk prediction model for CRS based on Chinese population data to facilitate the early detection, diagnosis, and treatment of CRS.
Materials and Methods
Participants
The study included patients with CRS who were admitted to the First Affiliated Hospital of Ningbo University between January 2022 and September 2022. Healthy controls (HCs) were recruited from Ningbo No. 6 Hospital during the same period. We collated their clinical data and excluded those with more than 20% missing data. Finally, the clinical data of 256 patients with CRS and 440 HCs were included in the study, which met the criterion of events per variable of 10, with a sufficient sample size for analysis.
The following criteria were used to diagnose CRS: (1) The main symptoms of CRS are nasal congestion, mucus, or sticky purulent mucus, and the secondary symptoms are head/facial swelling and reduced or loss of sense of smell. The patient was diagnosed with CRS if 2 or more of the above-mentioned symptoms were present, one of which was the main symptom. (2) Through nasal endoscopy, the patient was found to have viscous or mucopurulent secretions in the middle nasal passage and olfactory fissures, nasal mucosa congestion, edema, and polyps. (3) The patient examined using a computed tomography scan showed a sinusostomal tract complex or inflammatory lesions of the sinus mucosa.
Inclusion criteria: Patients older than 18 years of age with CRS confirmed by 2 or more attending physicians in the Department of Otolaryngology and Head and Neck Surgery, according to the Chinese Guidelines for the Diagnosis and Treatment of CRS (2018). 19 Exclusion criteria: Patients with severe chronic diseases such as malignancy or severe systemic disease of the heart, brain, lung, kidney, or liver. This study was approved by the Human Ethics Committee of the First Affiliated Hospital of Ningbo University. All the participants provided written informed consent.
Clinical Data
Clinical data is divided into demographic data including age and sex and laboratory indicators, obtained through electronic health systems. All staff involved in the survey received standardized training. All blood tests were performed after admission and before medication. All the laboratory indicators were obtained from medical records, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transpeptidase (GGT), urea (Ur), creatinine (CREA), uric acid (UA), triglyceride (TG), total cholesterol (TC), white blood cell (WBC) count, red blood cell (RBC) count, hemoglobin (HGB), platelet (PLT) count, lymphocyte (LYM) count, neutrophil (NEU) count, eosinophil (EOS) count, basophil (BAS) count, monocyte (MON) count, lymphocyte percentage (LYM%), neutrophil percentage (NEU%), eosinophilic granulocyte percentage (EOS%), basophilic granulocyte percentage (BAS%), and monocyte percentage (MON%).
Statistical Analysis
Statistical analyses were performed using R version 4.2.1. In this study, continuous variables are shown as means ± standard deviations, and discrete variables are expressed as frequencies and percentages, N (%). In the univariate analysis, Student’s t test was used to compare measurement data, and the chi-square test was used to compare counting data. Independent risk factors were screened using logistic regression analysis and 10-fold cross-validation combined with the least absolute shrinkage and selection operator (LASSO). A CRS risk prediction model was established using logistic regression, which was represented by a nomogram. Finally, receiver operating characteristic (ROC) curve, calibration curve, and decision curve analysis (DCA) were used for evaluation. Variables with a variance inflation factor (VIF) greater than 10 were excluded from the model. All tests were two-sided with a significance level of .05; the significance level for entry was set at .05, and that for stay was set at .1.
Results
Characteristics of Participants
The clinical data from patients with CRS and HCs are shown in Table 1. The age of the 2 groups was 43.65 ± 15.47 and 40.47 ± 11.29 years old, and the proportion of men was 74.55% and 72.55%, respectively, with no statistical difference. The demographic data showed comparability between the 2 groups. Regarding biochemical indices, univariate analyses of AST, ALP, GGT, TG, and TC showed statistically significant differences; the levels of AST, ALP, GGT, and TC were higher in patients with CRS than those of HCs. There were significant differences in 6 routine blood indices, including HGB, LYM, MON, LYM%, NEU%, and MON%; of these, MON, NEU%, and MON% of patients with CRS were higher than those of HCs.
Univariate Analysis of CRS.
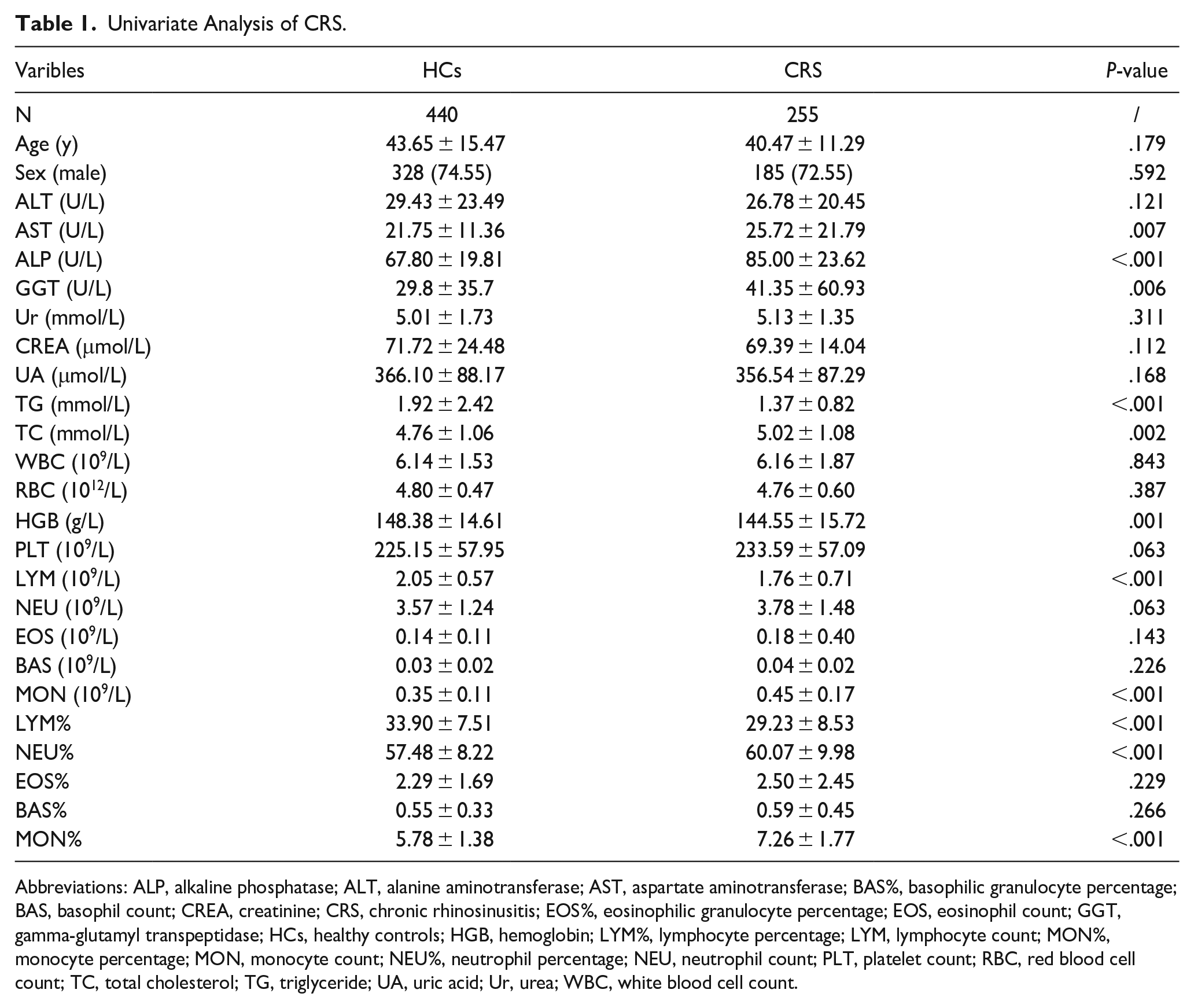
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BAS%, basophilic granulocyte percentage; BAS, basophil count; CREA, creatinine; CRS, chronic rhinosinusitis; EOS%, eosinophilic granulocyte percentage; EOS, eosinophil count; GGT, gamma-glutamyl transpeptidase; HCs, healthy controls; HGB, hemoglobin; LYM%, lymphocyte percentage; LYM, lymphocyte count; MON%, monocyte percentage; MON, monocyte count; NEU%, neutrophil percentage; NEU, neutrophil count; PLT, platelet count; RBC, red blood cell count; TC, total cholesterol; TG, triglyceride; UA, uric acid; Ur, urea; WBC, white blood cell count.
Independent Risk Factors
According to the results of the univariate analysis, variables with P < .05 were selected for further independent risk factor screening. The VIF for each variable was obtained using the VIF function in the car package in R. The VIF of LYM% was greater than 10, which indicated serious collinearity with other variables; therefore, it was not included in the screening of independent risk factors.
Logistic regression analysis revealed that ALP, TG, TC, HGB, LYM, MON, and MON% were independent factors influencing CRS. ALP, TC, MON, and MON% were risk factors, whereas TG, HGB, and LYM were protective factors against CRS (Table 2).
Multi-Factor Logistics Regression Analysis of Chronic Rhinosinusitis.
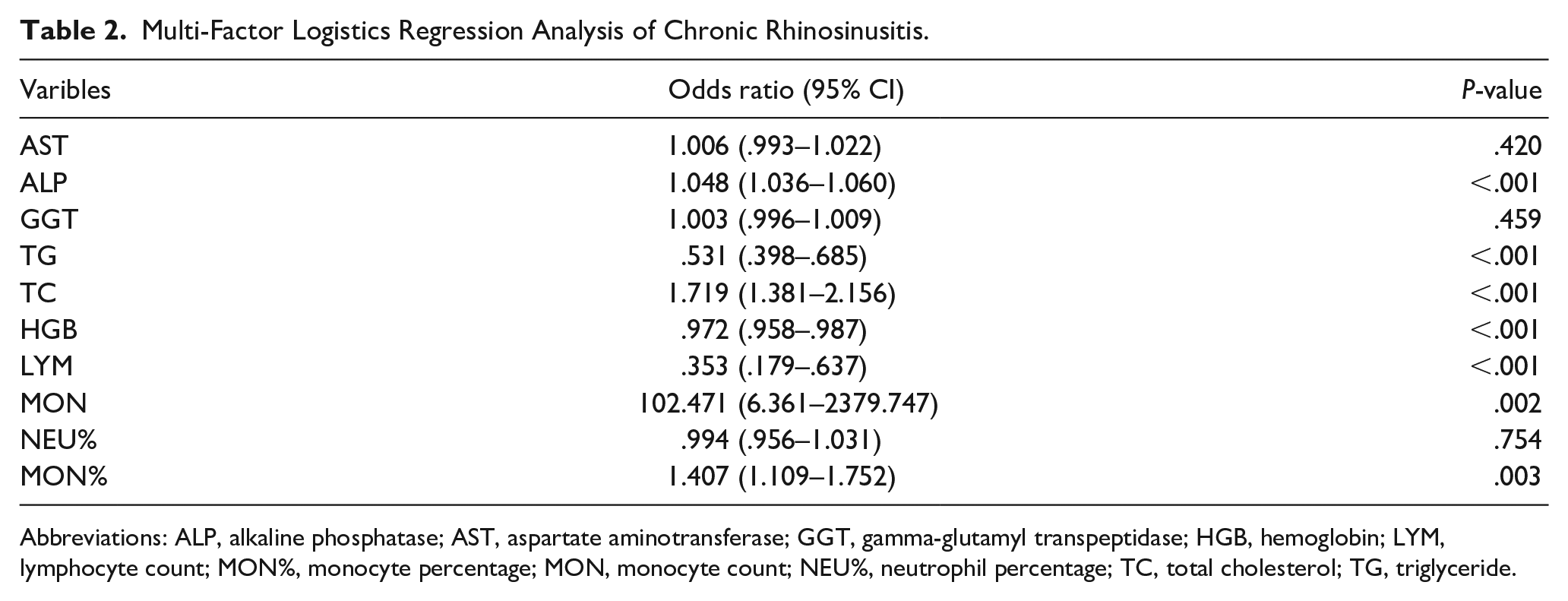
Abbreviations: ALP, alkaline phosphatase; AST, aspartate aminotransferase; GGT, gamma-glutamyl transpeptidase; HGB, hemoglobin; LYM, lymphocyte count; MON%, monocyte percentage; MON, monocyte count; NEU%, neutrophil percentage; TC, total cholesterol; TG, triglyceride.
Construction of Prediction Model
The training and test sets were selected through 10-fold cross-validation, and 13 candidate variables were screened from 23 variables using LASSO regression (Figure 1A-C). After building the model using the nomogram, 3 variables (points <20) were excluded. Finally, the prediction model was built according to the variables ALT, AST, ALP, CREA, TG, TC, RBC, HGB, LYM%, and MON% (Figure 1D).
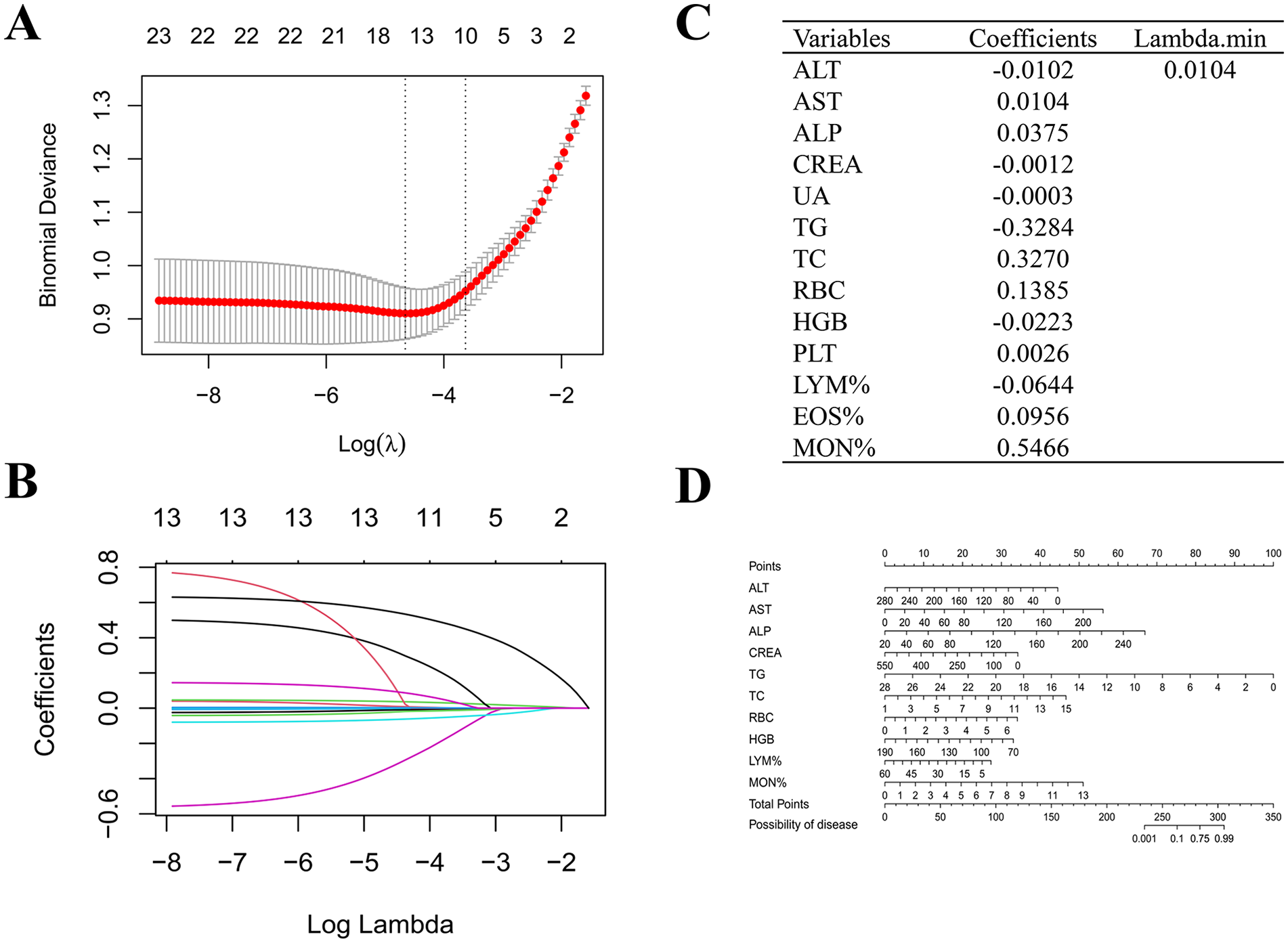
Construction of the prediction model. (A, B) Determination of the number of factors by the least absolute shrinkage and selectionator operator (LASSO) regression. (C) Factor coefficients and minimum λ values are obtained from LASSO regression. (D) The nomogram of the probability of chronic rhinosinusitis in healthy people. ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CREA, creatinine; EOS%, eosinophilic granulocyte percentage; HGB, hemoglobin; LYM%, lymphocyte percentage; MON%, monocyte percentage; PLT, platelet count; RBC, red blood cell count; TC, total cholesterol; TG, triglyceride; UA, uric acid.
Validation of Prediction Models
The ROC and area under the curve (AUC) were used to evaluate the discriminant ability of the prediction model. The sensitivity and specificity of the best cutoff value of the training set were 75.9% and 86.3%, respectively, and the corresponding AUC was 0.89 (Figure 2A). The sensitivity, specificity, and AUC of the optimal cutoff value for the test set were 69.6%, 97.8%, and 0.894, respectively (Figure 2B). Both AUC values were greater than 0.85, suggesting that the model had an excellent discriminative ability.
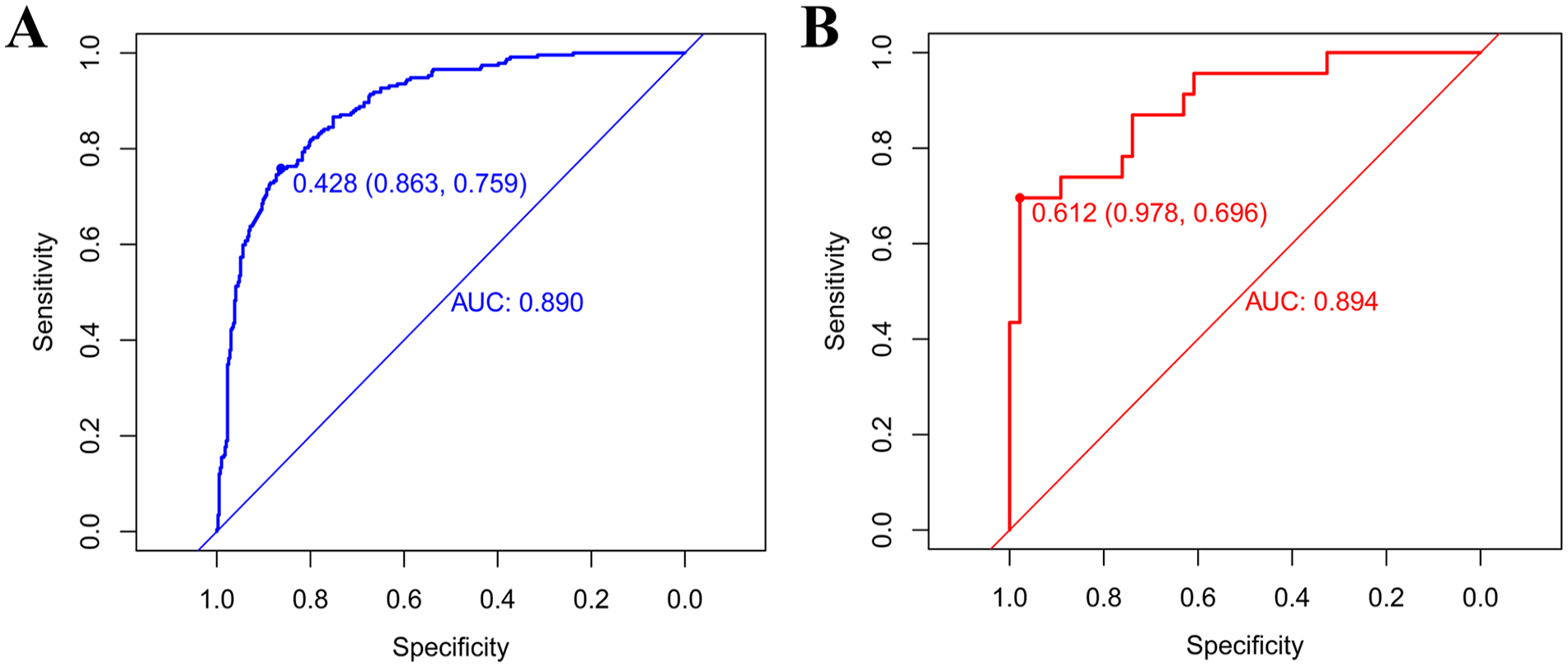
The ROC for the training and test sets. (A) Training set; (B) Test set. AUC, area under the curve; ROC, receiver operating characteristic.
The calibration curve is the fitting curve of the scatter graph formed by the actual and predicted probabilities, which essentially visualizes the results of the goodness-of-fit test of the model. The fitting curves of the training and test sets were distributed on both sides of the reference line and were close to the reference line, indicating that the model was well-fitted (Figure 3).
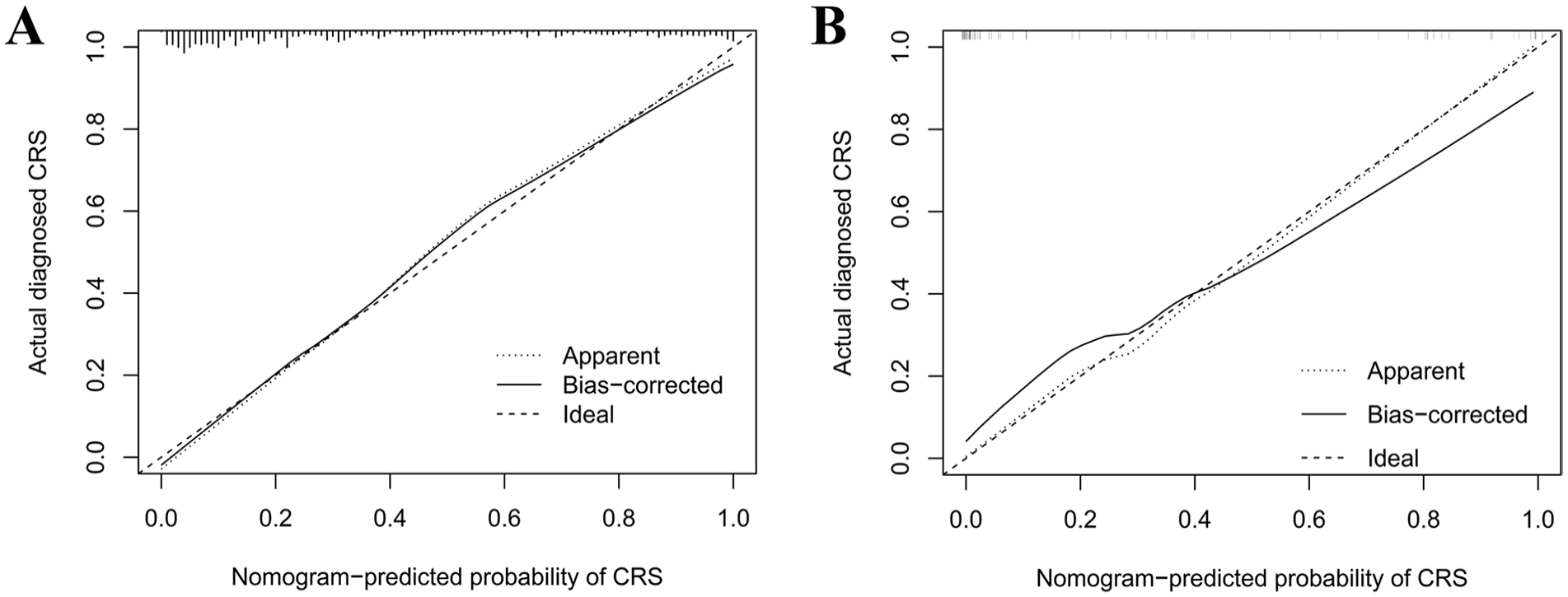
The calibration curve for the training and test sets. (A) Training set; (B) Test set.
DCA was used to calculate the relationship between the predictive model threshold probability and the clinical net benefit. In this study, the threshold probabilities of the training and test sets were 1% to 89% and 1% to 100%, respectively, indicating that the prediction model had a clinical net benefit within this range (Figure 4).
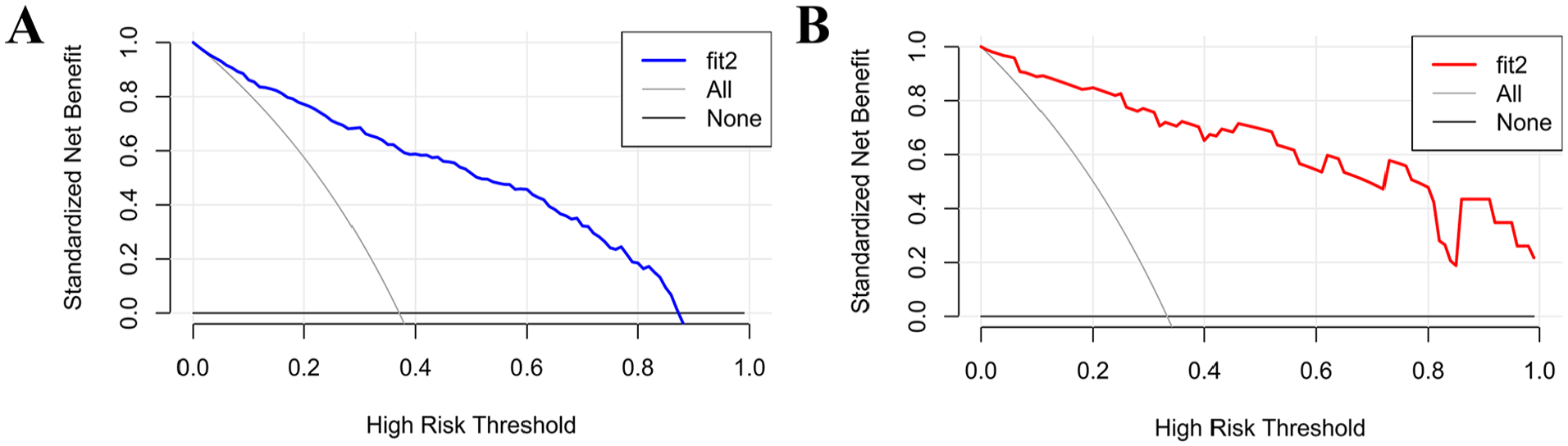
The decision curve analysis for the training and test sets. (A) Training set; (B) Test set.
Discussion
This case-control study was conducted in Ningbo, China, and included 255 patients with CRS and 440 HCs. Ten variables (ALT, AST, ALP, CREA, TG, TC, RBC, HGB, LYM%, and MON%) were selected to construct the CRS prediction model by logistic regression. The model was verified and evaluated in terms of discriminant ability, goodness-of-fit, and clinical benefit and was considered to be of great significance for the prediction and diagnosis of the disease.
The ratio of ALT to AST, which was included in the CRS prediction model in this study, can be used to indicate liver disease. This might indicate that patients with CRS have some liver damage, which has not been observed in other studies. Notably, glucocorticoids, macrolides, antibiotics, and other main drugs for the treatment of CRS have side effects, such as liver damage.7,19 Thus, the patients’ liver condition should be considered during CRS treatment. ALP is usually associated with hepatobiliary or bone remodeling, 20 and GGT is used as a test for liver homology 21 ; both of these did not show a difference in this study. Bone remodeling is common in patients with CRS and is dependent on the interactions of various cytokines, such as interleukin (IL)-13 and IL-17, which increases the disease complexity.22,23 The onset of CRS is related to the abnormal anatomical structure of the nose, 7 and patients with CRS also show pathophysiological changes in the sinus bones, 24 which supports the results of this study. Therefore, it is more likely that the association between ALP and CRS is related to bone remodeling rather than hepatogenic. While exploring biomarkers in urine samples from patients with CRS, Santarelli et al 25 adjusted the creatinine concentration as a baseline. However, the role and mechanism of action of creatinine in CRS remains unexplored. Studies have found that the prevalence of CRS in patients with metabolic syndrome is higher than that in patients without metabolic syndrome, 26 and that TG and TC are closely related to metabolic syndrome.27,28 In united airway diseases, serum TG and TC levels in patients with allergic rhinitis increase due to the action of cytokines such as IL-4, IL-10, and IL-17.29,30 Although there have been no direct studies investigating the role of TG and TC in CRS, their relationship has been well documented. In a case-control study, thalassemia was associated with an increased risk of CRS, and RBC and HGB levels were associated with changes in sinus structure. 31 RBCs crosstalk with immune cells to release damage-associated molecular patterns that enhance inflammation.32-34 This may explain why RBC count was an independent risk factor for CRS. The association between anemia and CRS remains unclear. One study on atopic diseases and anemia inferred that iron deficiency anemia may be secondary to food allergies and malnutrition caused by atopic diseases, whereas chronic inflammation may be secondary to anemia caused by chronic diseases. 35 Therefore, changes in hemoglobin, one of the main indicators of anemia, may be caused by chronic sinusitis rather than causing CRS. The immune response to CRS is mainly mediated by T helper cell type 2 and type 2 innate lymphoid cells (ILC2s). Numerous cytokines are released by T cells, ILC2s, mast cells, and eosinophils when pathogens invade and initiate an immune response. Cytokines also recruit monocytes and promote differentiation of M2-type macrophages, which play important roles in disease progression.1,36 Single-cell sequencing also revealed the recruitment of T cells and monocytes in patients with CRS. 37 The roles of lymphocytes and monocytes in CRS remain unclear; however, the absence of eosinophils in our study warrants further investigation.
It should be noted that the variables included in this study are common clinical indicators. These indicators can better show the overall changes of the human body, rather than having a very specific indication of CRS. These indicators are mainly used in the hope of using more convenient methods to provide some new help for the prevention and diagnosis of CRS. The clinical measures used to build the model in the study did not confirm a causal link between them and CRS. In other words, the study is more able to show the correlation between these indicators and CRS. In general, there is a certain relationship between the independent influencing factors obtained in the study and CRS.
Our study had several advantages. First, existing studies on independent risk factors for CRS have usually focused on genetic and environmental factors, whereas this study was the first to explore clinical indicators. Second, a prediction model was constructed and visualized using a nomogram, which is of great significance for the early diagnosis and prevention of diseases. The variables required for the construction of the model in this study were simple and easy to obtain and would certainly be useful in areas lacking medical resources. Finally, we evaluated the 3 dimensions of the model to ensure the validity and reliability of the results. Moreover, because this was a retrospective case-control study, bias caused by the study type could not be avoided. Therefore, we attempted to control the bias to a certain degree by including participants from 2 hospitals to control for Berkson’s bias and using objective indicators rather than subjective recall to control for recall bias. However, as this was a retrospective study, we could not verify weak causal associations. Ultimately, the sample size used in this study was sufficient. However, data from more regions and larger samples need to be collected for further analysis to enhance the accuracy and generalizability of the study.
Conclusion
In summary, this case-control study from Ningbo, China, identified 10 independent risk factors for CRS, from which a prediction model was constructed. The validation and evaluation of these models play an important role in the early diagnosis, prevention, and control of diseases.
Footnotes
Data Availability Statement
Data will be made available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical Health Science and Technology Project of Zhejiang Province (2023KY259, 2024KY1542, 2024KY341).
Ethical Statement
The protocols were approved by the Ethics Committee of The First Affiliated Hospital of Ningbo University (No. 2023-320).
