Abstract
Introduction
Laryngomalacia is the most common congenital laryngeal malformation caused by partial or total supraglottic collapse into the airway during inspiration, resulting in upper airway obstruction symptoms. 1 The primary clinical manifestation is inspiratory laryngeal stridor, which accounts for about 58% to 74% of neonates and infants. 2 The pathogenesis of laryngomalacia remains unclear. However, its pathogenesis may involve structural abnormalities to the laryngeal cartilage, neuromuscular dysfunction, and gastroesophageal reflux that occur during the onset of the condition in children.3-5 In most children, laryngomalacia presents as mild to moderate condition and is mainly diagnosed 4 to 5 months after birth, and most symptoms disappear within 12 months. 6 About 10% of the laryngomalacia cases are severe, with flushing and high-pitched inspiratory stridor being the primary manifestation, noticed before 14 days of age at presentation. 7 Children with severe laryngomalacia exhibit several complications, such as severe dyspnea, apnea, hypoxia/hypercapnia, obstructive sleep apnea, pulmonary hypertension, secondary pectus excavatum, feeding difficulties, and growth dysplasia, 8 which require timely identification and treatment.
Posture adjustment and changes in feeding methods can lead to gradual improvement and recovery in children with mild to moderate laryngomalacia. Conversely, surgical treatment is preferred for severe cases of laryngomalacia. Supraglottoplasty is the primary surgical treatment of severe laryngomalacia, which improves the anatomical and morphological abnormalities of laryngeal cartilage at presentation. The success rate of the aforementioned surgical procedure is as high as 86.1% to 89.0%.9,10 However, some patients may still require repeat supraglottoplasty or tracheotomy after the initial operation, suggesting that severe laryngomalacia may be caused by other factors besides the abnormal anatomical structure of laryngeal cartilage at presentation. Therefore, several risk factors contribute to the occurrence of severe laryngomalacia in children. Edmondson et al 11 discovered a greater risk of laryngomalacia in premature infants. Kusak et al 12 also revealed that prematurity and medical comorbidities increased the risk of laryngomalacia. Dickson et al 13 found that the incidence of synchronous airway lesions in patients with severe laryngomalacia was significantly higher than that in patients with mild and moderate laryngomalacia. Bozkurt and Çelik 14 reported that vitamin D levels were reduced in infants with laryngomalacia compared to normal infants, suggesting that vitamin D deficiency may cause laryngomalacia. Therefore, we hypothesized that preterm birth, medical comorbidities, and vitamin D deficiency and other factors may be risk factors for laryngomalacia in children. Thus, our aim was to identify the clinical features and potential risk factors of severe laryngomalacia in children through a retrospective analysis and evaluation. Early diagnosis of severe laryngomalacia is important for timely intervention and treatment.
Materials and Methods
Study Design and Data Collection
All clinical data of children under 18 years diagnosed with laryngomalacia at Chongqing Medical University Children’s Hospital and Suzhou Municipal Hospital between January 2013 and January 2023 were retrospectively analyzed. The study protocol was approved by the Medical Ethics Committee of the Children’s Hospital of Chongqing Medical University (No. 2021251).The clinical data included the following: (1) basic information: gender, age at presentation, gestational age at birth, birth weight, delivery mode, feeding method, gravidity, parity, age of pregnancy, and calcium supplementation during pregnancy; (2) degree of laryngomalacia infestation (mild, moderate, severe); (3) medical comorbidities (cardiopulmonary disease, synchronous airway disease, neurological disease, congenital abnormality/syndrome, and gastroesophageal reflux). Children delivered before 37 weeks of gestation were classified as preterm infants. Breastfeeding for at least 6 months after birth was defined as exclusive breastfeeding. Low birth weight infants were those with less than 2500 g birth weight. Calcium supplementation during pregnancy, starting at 14 weeks, was defined as at least 600 mg of calcium per day and the calcium supplementation dose was adjusted according to the pregnancy cycle and diet. Patients were classified into no calcium supplementation or regular calcium supplementation.
Laryngomalacia is classified as mild, moderate, and severe according to the severity of symptoms and signs (see Table 1).15,16 Three otorhinolaryngologists participated in the assessment of patient grading, and the opinions of 2 or more physicians were taken. The 2 groups in this study (mild-moderate and severe) were divided according to the grading level of laryngomalacia.
Degree of Laryngomalacia.

Inspiratory 3-concave sign refers to the obvious depression of the superior sternocostalis fossa, the superior clavicular fossa, and the intercostal space during inspiration.
Statistical Analysis
Normally distributed data were analyzed using the Kolmogorov-Smirnov test (K-S test). Normally distributed data were expressed as mean ± standard deviation. Categorical and rank data were presented as frequency and percentage. Differences between 2 groups of nonnormally distributed data were analyzed using the Mann-Whitney U test. Differences between multiple groups of categorical data were analyzed using the chi-square test. Factors with statistically significant associations in the chi-square analysis were included in the logistic regression analysis. All statistical analyses were performed using SPSS 23.0 software (IBM). P < .05 was considered statistically significant.
Results
General Conditions and Comorbidities of Severe Laryngomalacia
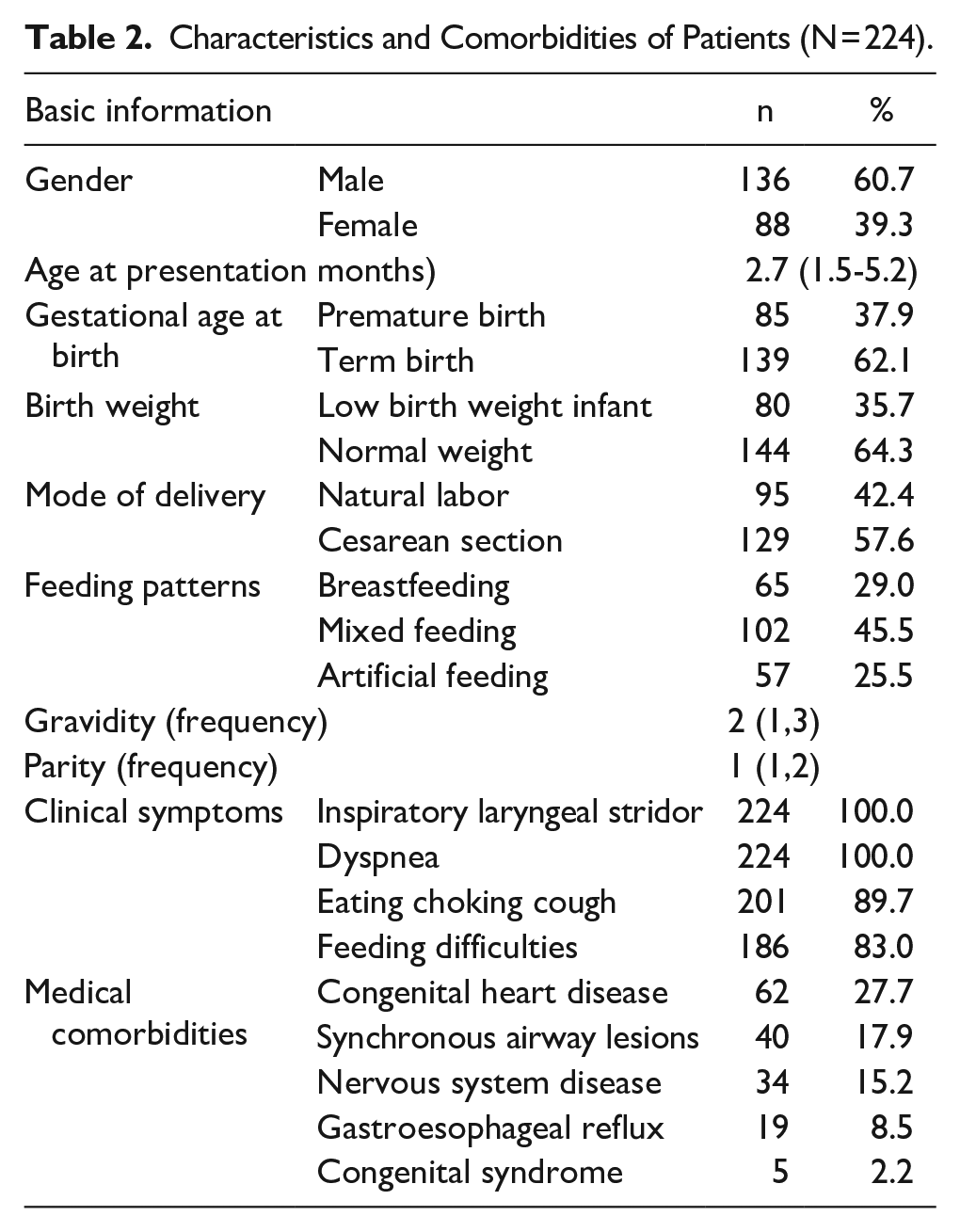
A total of 224 children with severe laryngomalacia were included in this study. The ratio of male to female patients was 1.55:1. The average age of the patients at presentation was 2.7 (1.5-5.2) months. Among them, 85 cases (37.9%) were premature, 80 cases (35.7%) had low birth weight, 95 cases (42.4%) were spontaneous delivery, and 65 cases (29.0%) were breastfeeding. All patients exhibited inspiratory laryngeal stridor and varying degrees of dyspnea. Comorbidities were classified based on the human body systems as follows: 62 (27.7%) congenital heart disease, 40 (17.9%) synchronous airway disease, 34 (15.2%) neurological disease, 19 (8.5%) gastroesophageal reflux, and 5 (2.2%) congenital syndrome disorders. Table 2 shows detailed information.
Characteristics and Comorbidities of Patients (N = 224).
Comparison of General Characteristics Between Mild-Moderate and Severe Laryngomalacia Patients
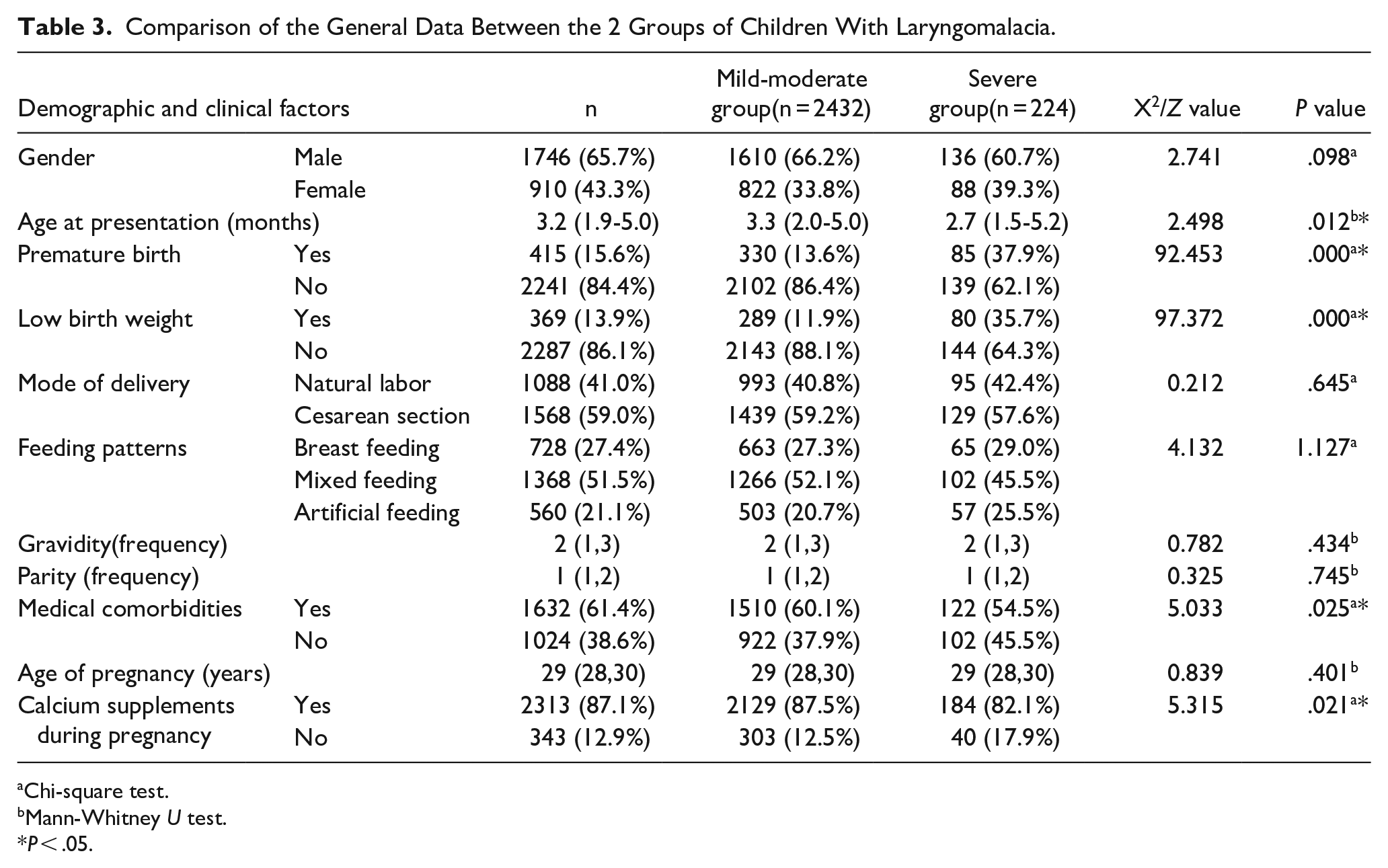
This study comprised 2656 children with laryngomalacia, with 2432 in the mild-moderate group and 224 in the severe group. There were significant differences among several variables, including age at presentation, premature delivery, low birth weight, medical comorbidities, and calcium supplementation during pregnancy between the 2 groups (P < .05). These parameters were considered to be potential risk factors for severe laryngomalacia. However, gender, delivery mode, feeding mode, gravidity, parity, and age of pregnancy were comparable between the 2 groups (P > .05; Table 3).
Comparison of the General Data Between the 2 Groups of Children With Laryngomalacia.
Chi-square test.
Mann-Whitney U test.
P < .05.
Multivariate Logistic Regression Analysis of the Risk Factors for Severe Laryngomalacia
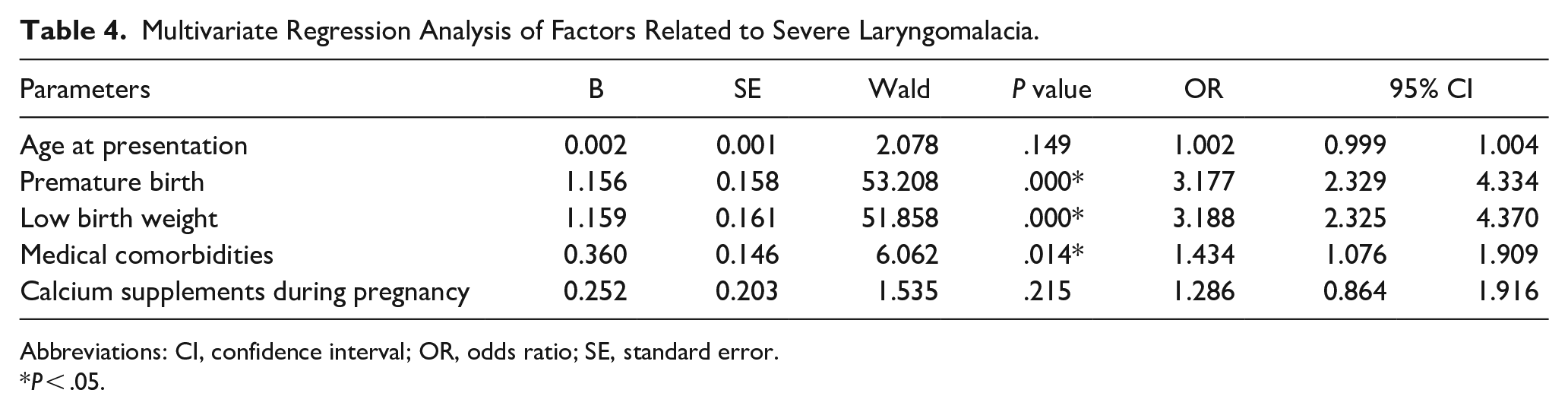
Factors strongly associated with severe laryngomalacia in the univariate analysis were included in the multivariate logistic regression analysis. The results showed that preterm birth [odds ratio (OR) = 3.177, 95% confidence interval (CI): 2.329-4.334], low birth weight (OR = 3.188, 95% CI: 2.325-4.370), and medical comorbidities (OR = 1.434, 95% CI: 1.076-1.909) were the independent risk factors for severe laryngomalacia in children (P < .05; Table 4).
Multivariate Regression Analysis of Factors Related to Severe Laryngomalacia.
Abbreviations: CI, confidence interval; OR, odds ratio; SE, standard error.
P < .05.
Discussion
Laryngomalacia is a congenital condition, most children with severe laryngomalacia gradually developed inspiratory laryngeal wheezing, dyspnea, and other symptoms shortly after birth. In this study, all the children exhibited inspiratory laryngeal wheezing and different degrees of dyspnea, which appeared early, with some present at birth. Moreover, the children had a median age of 2.7 months at presentation, consistent with other studies.12,17 This study revealed that children in the severe group were younger than those in the mild-moderate group (P < .05). Patients with severe laryngomalacia manifest with more aggressive clinical symptoms (laryngeal stridor, dyspnea, and other airway obstruction symptoms), and some of them appear at birth, so the age at presentation is earlier than mild-moderate laryngomalacia. In general, the symptoms of severe laryngomalacia manifest within 2 weeks of birth and usually resolve at around 2 years of age. 7 The present study comprised more males than females, consistent with a report by Toer et al. 18 Some studies have suggested that preterm birth and low birth weight are associated with laryngomalacia. 11 In this study, preterm birth and low birth weight infants accounted for 37.9% and 35.7%, respectively, with a higher incidence of both cases. Severe laryngomalacia is often accompanied by different types of medical comorbidities. The most common comorbidity in this study was congenital heart disease, accounting for 27.7%. In comparison, Yusuf and Utami 19 reported that 15.79% of children with severe laryngomalacia had congenital heart disease, which was significantly lower compared with that of our study cohort. Toer et al 18 found that the most common comorbidity was a neurological disease (35.1%), which was significantly higher compared with that of our study (15.2%). The incidence of synchronous airway lesions in this study was 17.9% which was lower compared with that reported by Glibbery et al. 20 The incidence of gastroesophageal reflux at presentation was 8.5%, significantly lower than in other studies. 21 This could be attributed to the fact that most of the children in this study were diagnosed with severe laryngomalacia at birth, and their condition was critical without relevant examination. Therefore, whether gastroesophageal reflux at presentation exists remains unclear, and its actual proportion may be higher.
The pathogenesis of laryngomalacia in children remains unclear. To date, few studies have investigated the risk factors associated with laryngomalacia. Preterm birth and low birth weight have been found to be strong predictors of laryngomalacia in children. 11 However, there are no reports of risk factors associated with severe laryngomalacia in children. We found that preterm birth was an independent risk factor for severe laryngomalacia. Moreover, severe laryngomalacia was 3.177 times higher in preterm infants than in the mild-moderate group. Some researchers have suggested that prematurity predisposes infants to atypical presentations of laryngomalacia, including apneic events with no observed stridor. 22 McCaffer et al 23 reported that preterm infant laryngomalacia is associated with higher secondary airway lesions and more severe dysphagia. Many studies have shown that preterm birth is an independent risk factor for poor prognosis in the surgical treatment of severe laryngomalacia.9,24 Most preterm infants were those with low birth weight, which was also an independent risk factor for severe laryngomalacia. The incidence of severe laryngomalacia was 3.188 times higher in children with low birth weights than in the mild-moderate group. Edmondson et al 11 also demonstrated that low birth weight is a strong predictor of laryngomalacia regardless of gender or race. Most children with severe laryngomalacia exhibit a lower weight gain rate, which could be attributed to increased respiratory effort, gastroesophageal or throat reflux during onset, and an uncoordinated aspiration-swallowing-breathing order. 12 Therefore, children with premature birth and low birth weight should be closely observed (because they are at high risk of developing severe laryngomalacia) to prevent delayed diagnosis and treatment.
Children with severe laryngomalacia are often present with medical comorbidities. In this study, medical comorbidities were independent risk factors for severe laryngomalacia. Medical comorbidities were 1.434 times higher in the severe group than in the mild-moderate group. Bhatta et al 17 reported that patients with severe laryngomalacia had synchronous airway abnormalities higher than that of the mild-moderate group. Thompson 6 also revealed that neurological, genetic, and congenital heart diseases are more prevalent in children with severe laryngomalacia. Laryngomalacia may be related to laryngeal cartilage at presentation plasia during embryonic development. 25 However, most medical comorbidities are congenital diseases such as congenital heart disease and synchronous airway lesions. These comorbidities are also related to the synchronous development of the embryo. Landry and Thompson 1 also demonstrated that underlying comorbidities aggravate the severity of laryngomalacia. Comorbidities are more prevalent in patients with severe laryngomalacia and may contribute to the development of systemic muscle hypotonia in children with neurological conditions, 26 the increase in respiratory function in children with congenital heart disease, 27 and the presence of anatomical airway obstruction in synchronous airway lesions. Many previous studies have revealed that medical comorbidities are also an independent risk factor for poor prognosis in the surgical treatment of severe laryngomalacia.12,28 Therefore, it is important to closely monitor children with medical complications who have been diagnosed with laryngomalacia because they are at a higher risk of developing severe laryngomalacia and at a higher risk of poor surgical outcomes.
During fetal growth and development, the mother provides the calcium needed for fetal growth and development. The calcium available to the newborn from the mother in the absence of exogenous calcium supplements during the pregnancy is only 25 to 30 g. Because this calcium level cannot meet the normal fetal bone development requirement, hypoplasia of the laryngeal cartilage may occur. In this study, the ratio of maternal calcium supplementation during pregnancy was significantly higher in the mild-moderate laryngomalacia than in the severe group. There are no studies on the relationship between maternal calcium supplementation during pregnancy and laryngomalacia. Vitamin D is known to promote calcium absorption, and there are some studies on the relationship between vitamin D deficiency and laryngomalacia. Hassan et al 29 suggested that laryngomalacia is an inflammatory disease secondary to maternal 25(OH)-vitamin D deficiency. Bozkurt et al 13 revealed that vitamin D levels were lower in infants with laryngomalacia compared to the healthy babies group, suggesting that vitamin D deficiency may be a factor in the etiology of laryngomalacia. However, the etiology of laryngomalacia remains to be elucidated. In this study, only the calcium supplementation during pregnancy was investigated, but calcium content in blood during pregnancy was not analyzed. In conclusion, appropriate and standardized calcium supplementation can improve the calcium level in pregnant mothers, increasing the density of fetal laryngeal cartilage at presentation and possibly reducing the occurrence of laryngomalacia.
This study had some limitations. Because this was a retrospective study, selection bias cannot be ruled out. Thus, further prospective studies are required to validate our findings. Moreover, the number of cases of severe laryngomalacia was relatively small. Therefore, larger sample sizes are required to validate our findings.
Conclusions
In conclusion, severe laryngomalacia in children is a multifactorial disease. Children with severe laryngomalacia exhibited persistent stridor at an earlier age at presentation. This study found that preterm birth, low birth weight, and medical comorbidities were independent risk factors for severe laryngomalacia. Persistent laryngeal stridor and dyspnea should be considered as risk factors for early identification and timely treatment of severe laryngomalacia.
Footnotes
Authors’ Note
Lu Chen collected data and wrote the manuscript, Yang Yang finished statistical analysis, Xinye Tang and Ling Ding checked statistical analysis and manuscript, and Ling Xiao proposed ideas and finished project administration.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
