Abstract
Introduction
The highest amount of fat tissue on the face is located above the zygomatic bone. Having pronounced and prominent zygomatic bones can be considered a sign of youth. The malar fat pad shrinks and loses volume as we become older. Because of this decrease, the ground is subsiding here. The leftover adipose tissue also migrates downward, further debilitating the area.
The midface’s superficial fat compartments are a dynamic soft tissue layer that moves in tandem with the underlying muscles and the dermis. Superficial fat pads securely bind the skin to the muscles via connective tissue fibers that go upward to the dermis.1-5 Changes in the volume of the infraorbital fat compartment, the medial cheek fat compartment (MCF), the middle cheek fat compartment (MiCF), and the nasolabial fat compartment (NLF) are all factors in the aging of the midface. 5
A dermal filler is a substance that is injected or implanted into the dermis. Patients are cautioned from touching the treated regions for fear of causing the substance to move. A cold pack is recommended to decrease inflammation. 6
The ideal dermal filler would be affordable, risk-free, pain-free throughout the injection process, hypoallergenic, and long-lasting. It should be easy to inject, produce minimal discomfort, work immediately, need no recovery time from the patient, and have minimal potential for side effects. Due to factors such as an aging baby boomer population, rising demand for “lunchtime procedures,” and the desire for a more youthful appearance, the pharmaceutical market has responded by giving the cosmetic surgeon more options to satisfy the needs of the cosmetic patient. 6
Hyaluronic acid fillers are frequently used to treat V-deformities and loss of volume during midface rejuvenation. Collagen induction, a vital component of the extracellular matrix, has been viewed as a fundamental impact of the rejuvenation process. Histologic investigations have pinpointed the location of hyaluronic acid filler in the subcutaneous adipose tissue, even though it is generally referred to as a “dermal” soft filler. When possible, a deep injection will result in longer-lasting effects.7,8
Injecting dermal filler into the midface has become standard practice in cosmetic clinics worldwide as a nonsurgical approach to augmenting the malar region or reshaping the midface.5,8-10
This research aims to examine how a filler operation on the malar area affects the patient’s visual quality and quality of life (QoL).
Patients and Methods
This retrospective and multicentric study is conducted in the Otolaryngology Departments of Eskisehir Osmangazi University, Kırıkkale University, and Bilecik Şeyh Edebali University and Dr. Oğuzhan Oğuz Wellnose Clinic according to the rules outlined in the Declaration of Helsinki. Ethics committee approval was taken from T.R. Bilecik Şeyh Edebali University, Non-Invasive Clinical Research Ethics Committee (Date: 05.12.2023, Number: 8/8). There is no need to take informed consent because the data were evaluated retrospectively.
Subjects
A total of 72 patients who underwent a malar filler procedure between March 2022 and March 2023 were included in the study. The patients were selected from those who applied to the Otolaryngology Departments of Eskisehir Osmangazi University and Dr. Oğuzhan Oğuz Wellnose Clinic. The mean ages of the patients were 50.5 ± 8.6 (ranging from 38.0 to 66.0).
Each patient received a 2 ml injection of hyaluronic acid filler, with 1 ml administered on the right side and 1 ml on the left side.
Inclusion Criteria
Patients who underwent malar filler procedure.
At least 1 month follow-up evaluation of the patients.
Exclusion Criteria
The study did not include patients who did not come for 1 month follow-up.
Methods
1.
Change criteria were rated on a scale of 1 to 5 as follows:
1—No change
2—Slight change
3—Moderate change
4—Significant change
5—Very significant change
2.
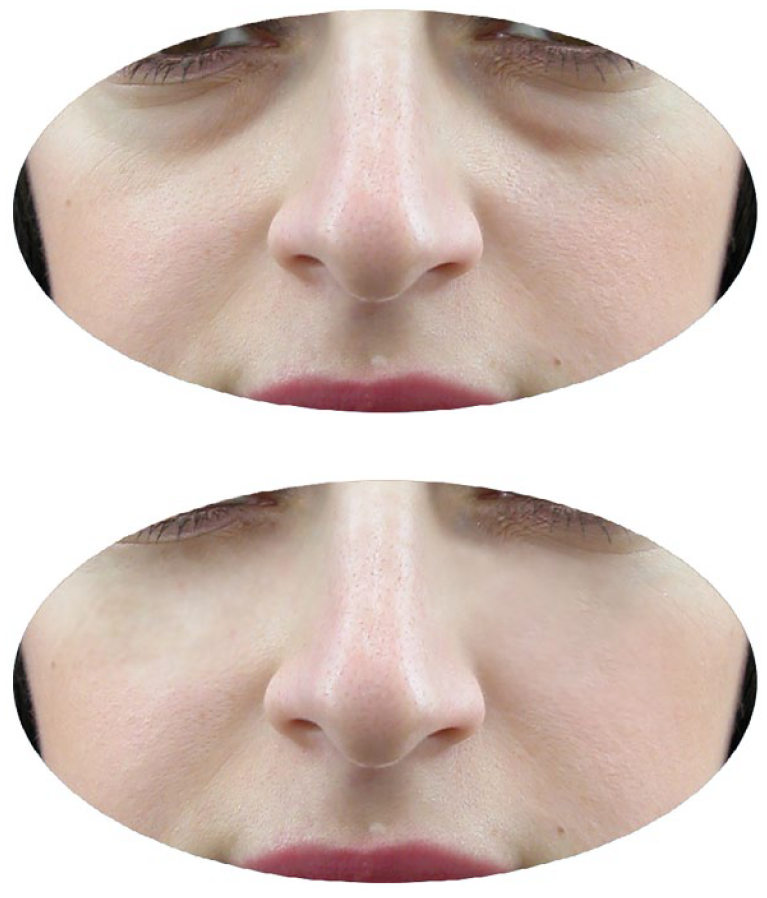
Case 1: Photos taken before the malar filler procedure and first month after the procedure.
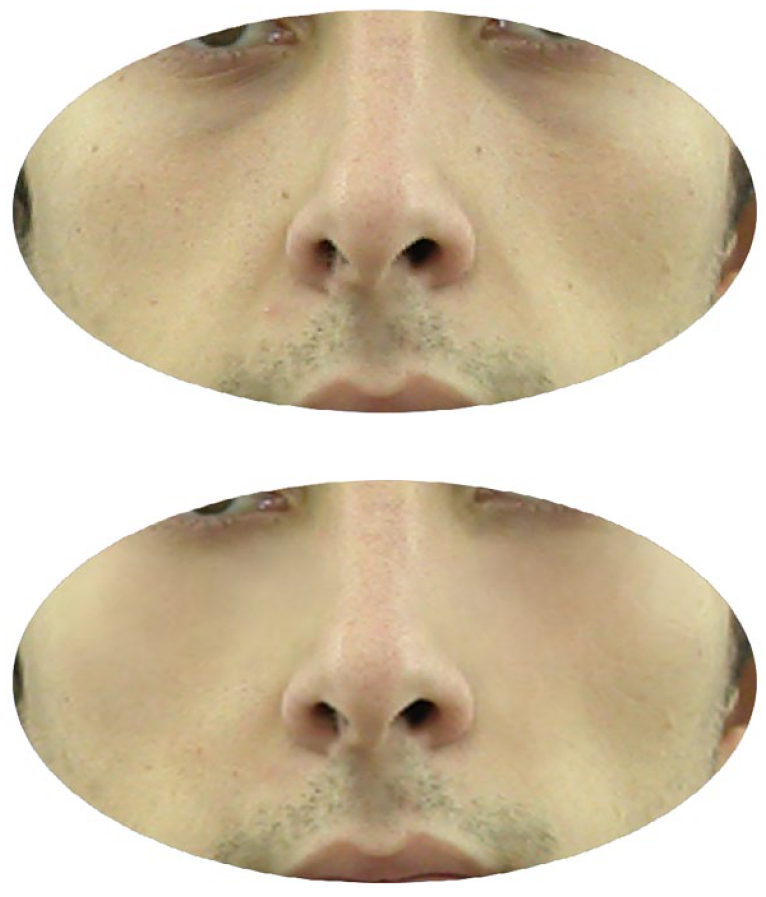
Case 2: Photos taken before the malar filler procedure and first month after the procedure.
Statistical Analysis
The data collected in this study were analyzed using the SPSS for Windows 16.0 software (SPSS, INC, an IBM Company). Descriptive statistics (mean, standard deviation, minimum, maximum) were applied and percentiles were defined for the genders.
Results
There were 27 (37.5%) males and 45 (62.5%) females.
Discussion
The MCF is located in the center of each cheek, directly above the deep medial cheek fat (DMCF)’s lateral section. Age-related MCF atrophy has been linked to an increase in the mid-cheek concavity depth and the mid-cheek groove’s emergence. 11 The medial cheek fold meets the medial cutaneous fold, the temporal cheek fat meets the MiCF laterally, and the superficial extension of the zygomatic cutaneous ligament meets the MiCF above. The MiCF’s volume decreases with age, exacerbating the MCF’s depression. 5
The NLF is the midface’s most medial superficial fat compartment. This rectangular fat pad is bordered on the inside by the medial cheek septum, the internal orbital rim, and the medial cheek fat pad, and on the outside by the nasal sidewall, the levator labii superior plaque nasi muscle, and the nasolabial septum. Nasolabial folds and nasojugal grooves become more apparent in older adults because the NLF widens and sinks inferiorly with age compared to surrounding fat compartments.5,12,13
Malar projection, broad cheeks, and a smooth convexity from the lower eyelid down to the nasolabial fold produce an ogee curve. These qualities are commonly considered aesthetically desirable in the midface. 1 This youthful midface shape often diminishes with age due to the generalized loss of volume that comes with age.5,14 A concavity may form at the intersection between the thin lower eyelid and the thicker cheek skin below. There may be an enlargement of the nasojugal groove and a flattening of the upper cheek. A sunken face with ample folds might be caused by alterations in muscular or ligamentous structures in addition to resorption of orbital and maxillary bones and atrophy or selective hypertrophy of the deep and superficial fat pads. 15
The skin’s primary glycosaminoglycan is hyaluronic acid. Injecting the skin with hyaluronic acid, which has a strong affinity for water, causes the skin to plump up, become more supple, and retain more moisture. These advantages include its role in cell proliferation, membrane receptor activity, and adhesion.6-8 Since volumizing hyaluronic acid fillers induce mechanical stress to not only fibroblasts but also adipocytes, and since deep injection itself causes minor trauma in the subcutaneous adipose tissue, we propose that the activation of adipose tissue-derived mesenchymal stem cells is responsible for the observed clinical effects. 7
In this study, we aimed to determine how malar area filler treatment affected the patient’s visual quality of life and QoL. Two milliliters of hyaluronic acid filler were injected into 72 individuals, with 1 ml placed on each side of the face. All patients had a “very significant change” in how they looked in the eyes of their doctors. Sixty-six patients (91.6%) were pleased with the operation, whereas 8 patients (8.4%) felt more could have been done and asked for more procedures.
Hyaluronic acid provides a viscoelastic network for collagen and elastin fibers to bond together and stabilize intercellular structures. Photoaging is characterized by the breakdown of these linkages, which leads to the accumulation of disordered collagen and elastin. Hyaluronic acid is a great dermal filler agent because of its many uses.16,17
To provide a more concentrated hyaluronic acid that is more resistant to chemical and physical degradation, the hyaluronic acid filler incorporates modified hyaluronic acid particles that have been cross-linked. Hyaluronic acid filler gradually loses concentration as water replaces it throughout deterioration and breakdown, but its volume remains constant. We call this process “isovolumetric degradation.” Depending on your geographic area, the precise type of filler you use, and the method of injection, the benefits of hyaluronic acid filler can last anywhere from 4 to 6 months. 18
Because malar projection and plump upper cheeks significantly contribute to a young appearance, midface rejuvenation is one of the most essential indications of hyaluronic acid dermal fillers. Over the past 2 decades, hyaluronic acid fillers have developed to fulfill various therapeutic demands, including increased projection power and the ability to accommodate natural face movement. Because of the promise of noninvasive treatment, instant results, and low downtime, they are becoming the treatment of choice for midface rejuvenation across all age groups. Injecting the midface may indirectly alleviate other cosmetic issues, such as infraorbital hollowing and nasolabial folds, due to the vital function played by the midface’s 5-layered structure. 5
The mechanism of action of hyaluronic acid when applied intraarticularly is identical. Synovial fluid and cartilage contain hyaluronic acid naturally; however, with osteoarthritis, both the concentration and the size of individual hyaluronic acid molecules diminish, reducing the viscosity of the synovial fluid. Half-lives for hyaluronic acid after injection into a joint range from 17 hours to 1.5 days. 19 Purified or synthetically produced hyaluronic acid formulations with a high molecular weight have a longer half-life. 18
Hyaluronic acid filler’s most frequent side effects include discomfort, bruising, redness, itching, and swelling. These adverse effects usually do not persist for more than a week. To lessen them, the patient should apply an ice pack to the injection site, keep their body upright, and stop taking any blood-thinning medications or supplements 1 week before the procedure. These include aspirin, nonsteroidal anti-inflammatory drugs (NSAIDs), vitamin E, fish oils, St. John’s wort, and Ginkgo biloba. 18
Rare but possible complications of injecting hyaluronic acid gel include infection, tissue necrosis, granulomatous foreign body, and herpes labialis activation. By using an aseptic technique and ensuring there is no current infection in the area, diseases caused by bacterial inoculation at the injection site can be avoided. Intra-arterial injection of hyaluronic acid filler can cause vascular occlusion, leading to tissue necrosis. This emphasizes the significance of knowing face anatomy and getting a blood aspiration test before injecting. Hyaluronidase, which breaks down hyaluronic acid gel particles, should be used immediately if this unwanted outcome is feared. While reactions to hyaluronic acid filler injections are uncommon, granulomatous foreign-body reactions have been reported on infrequent occasions. 18
It is recommended to rely on 2 anatomical lines, one from the medial canthus to the mandibular angle (the mid-cheek safety line) and the other from the middle of the zygomatic arch descending vertically (the lateral safety line), 5 as these follow the typical paths of critical neurovascular structures. After passing through the infraorbital foramen, the infraorbital artery splits into 2 or 3 major branches (palpebral, nasal, and labial branches). The 2 latter branches originate in the DMCF and go mainly under the mid-cheeks safety line, supplying the lateral nasal and superior labial areas. 20
One of the primary blood supplies to the cheeks is the transverse facial artery (TFA), along with the facial artery (FA). 21 Most people have a single TFA that emerges from the superficial temporal artery and divides into 2 branches, one that provides the upper part of the parotid gland and the parotid duct and the masseter, and another that supplies the deep part of the parotid gland and the parotid duct and the masseter. 22 It is essential to be careful while filling the superficial fat of the lateral cheek, as this is where the superior emerging branch emerges from the subcutaneous tissue. 23
The zygomaticofacial artery (ZFA) originates in the suborbicularis oculi fat (SOOF) from the lacrimal artery, emerging from the zygomaticofacial foramen. This foramen has only been documented a handful of times, but research has pinpointed its location between 12 and 15 mm below the lateral canthus and 5 and 9 mm to the side. A few patients lacked a ZFA and a foramen, according to a few publications.24-27 At or around the foramen, the ZFA divides into several smaller branches that can anastomose with the TFA. Underneath the Orbicularis oculi muscle (OOM), they connect with the zygomaticotemporal arteries to form a vascular system. However, these capillaries are theoretically too tiny to serve as an entry door for a possible hyaluronic acid embolus. 5
Conclusion
In our study, hyaluronic acid fillers 28 were administered to the malar area, and 91.6% of the patients expressed satisfaction with the operation. The doctors said the patients had “very significant change” in their appearance. Only 8.4% of people receiving therapy said it was not enough and asked for something more to be done. Patients’ requests for unnecessary operations should be noted and treated with caution since they may lead doctors in the wrong direction.
Footnotes
Acknowledgements
None.
Author Contributions
Oğuzhan Oğuz: Planning, designing, literature survey, data collection, active intellectual support. Nuray Bayar Muluk: Planning, designing, literature survey, statistical analysis, writing, active intellectual support, submission. Zeynel Öztürk: Planning, designing, literature survey, active intellectual support. Tarik Yagci: Planning, designing, literature survey, active intellectual support. Cemal Cingi: Planning, designing, literature survey, data collection, active intellectual support, English editing.
Availability of Data and Materials
All data for this study are presented in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Committee Approval
Ethics committee approval was taken from T.R. Bilecik Şeyh Edebali University, Non-Invasive Clinical Research Ethics Committee (Date: 05.12.2023, Number: 8/8).
Informed Consent
There is no need for informed consent because the data were evaluated retrospectively.
