Abstract
Introduction
Sudden-onset deafness constitutes a prevalent and urgent otologic emergency, frequently characterized by an abrupt and unexplained loss of hearing. Concurrently, affected individuals may exhibit clinical symptoms such as vertigo, tinnitus, and a sensation of ear blockage, 1 typically manifesting unilaterally and occasionally bilaterally. The escalating prevalence of type 2 diabetes mellitus has contributed to a rising incidence of comorbid cases involving sudden deafness. International studies indicate that approximately 16% to 20% of patients with sudden deafness are concomitantly diagnosed with type 2 diabetes mellitus, although the precise pathophysiology remains elusive. Some studies propose an association with blood circulatory disruptions, viral infections, and autoimmune mechanisms. Experimental models have confirmed the inducibility of sudden deafness in patients with diabetes, potentially attributed to the hypercoagulable state and microangiopathy characteristic of type 2 diabetes mellitus.
Presently, therapeutic interventions for sudden deafness in conjunction with type 2 diabetes mellitus primarily rely on vasodilatory agents, neurotrophic agents, and glucocorticoids. Hyperbaric oxygen therapy (HBOT) has emerged as a progressively employed modality in the management of sudden deafness, frequently serving as a complementary approach when conventional drug therapies prove ineffective. In this study, we aim to assess the efficacy of combining HBOT with standard pharmacological interventions in the treatment of sudden deafness coexisting with type 2 diabetes mellitus.
Materials and Methods
General Information
A cohort of 96 patients diagnosed with type 2 diabetes mellitus concomitant with sudden deafness at our hospital between October 2020 and September 2023 was selected for this study. The patients were randomly assigned to either a treatment group (n = 50) or a control group (n = 46) using a random number table method.
In the treatment group, there were 28 females and 22 males, aged between 22 and 68 years, with a mean age of 46.29 ± 6.92 years. Among them, 32 cases exhibited left ear lesions and 18 cases exhibited right ear lesions, with a duration ranging from 2 to 10 days and a mean duration of 6.36 ± 2.28 days. The degree of hearing impairment was mild (26-40 dB) in 28 cases, moderate (41-70 dB) in 20 cases, and severe (70-90 dB) in 2 cases.
In the control group, there were 25 females and 21 males, aged between 19 and 70 years, with a mean age of 39.81 ± 5.72 years. Among them, 26 cases exhibited left ear lesions, and 20 cases exhibited right ear lesions, with a duration ranging from 3 to 12 days and a mean duration of 7.11 ± 2.07 days. The degree of hearing impairment was mild in 25 cases, moderate in 19 cases, and severe in 2 cases.
Statistical analysis revealed no statistically significant differences in general information between the 2 groups (P > .05), establishing the comparability of the 2 groups for the purpose of this study.
Diagnostic Protocol
The diagnosis of type 2 diabetes adhered to the diagnostic criteria outlined in the Chinese Guidelines for the Prevention and Treatment of Type 2 Diabetes Mellitus (2017 edition). 2 Concurrently, the diagnostic criteria from the Guidelines for the Diagnosis and Treatment of Sudden Deafness (2015) 3 were consulted in accordance with Western medical standards.
Exclusion Criteria
Exclusion criteria for participant enrollment: (1) claustrophobia syndrome; (2) inner ear diseases; (3) individuals with an allergic constitution or multiple drug allergies; (4) severe cardiovascular, cerebrovascular, hepatic, renal dysfunction, and coagulation abnormalities; (5) malignant tumors and a history of psychiatric disorders; (6) patients presenting other contraindications such as active peptic ulcers, severe hypertension (blood pressure ≥ 180/100 mmHg), glaucoma, and those incapable of cooperating with treatment or participating in regular follow-up visits.
Treatment Approaches
The control group underwent conventional Western medicine therapy, primarily encompassing anticoagulation, neurotrophic interventions, microcirculation improvement, and glucocorticoid administration.
The treatment group received HBOT in addition to conventional treatment. The specific protocol involved placing patients inside a medical oxygen pressurized chamber. The air was pressurized for 20 minutes until the chamber reached a pressure of 0.20 MPa. After the pressure stabilized, patients wore a mask and inhaled pure oxygen for 30 minutes. Following a 10 minute rest interval, patients repeated the 30 minute oxygen intake via the mask. Subsequently, decompression occurred over a total period of 20 minutes. On decompression completion, patients exited the chamber. HBOT was administered once daily. This HBOT regimen comprised two 10 day courses with a gap of 1 day between the 2 courses. The entire treatment duration involved 20 days of oxygen inhalation.
Observation Indicators
Pretreatment and posttreatment assessments involved the extraction of 5 mL of fasting venous blood in both instances. Subsequently, the extracted blood samples were centrifuged for 10 minutes to obtain the supernatant. The supernatant was then preserved in a controlled environment at −30°C for the subsequent measurement of fasting plasma glucose (FPG), total cholesterol (TC), triglyceride (TG), and low-density lipoprotein cholesterol (LDL-C) levels in both the control group and the treatment group. Comparative analyses were then conducted to assess any discernible differences between the 2 groups.
The efficacy criteria for this study were established based on the extent of restoration of impaired hearing thresholds, referencing the Diagnostic and Therapeutic Guidelines for Sudden Deafness (2015). Recovery was defined as restoring hearing to normal or achieving the healthy ear function/pre-disease level. A positive effect was attributed to an improvement of more than 30 dB, while an improvement exceeding 15 dB was considered effective. Failure to meet any of these criteria was deemed ineffective.
The positive effect rate was calculated as (recovery + positive effect) divided by the total number of cases, multiplied by 100%. Similarly, the effective rate was determined by (recovery + positive effect + effective) divided by the total number of cases, multiplied by 100%.
Statistical Methods
The statistical analysis in this study employed SPSS 22.0 software (International Business Machines Corporation, Armonk, NY, USA) for data processing. Quantitative data are presented as mean ± standard deviation (x̅ ± s) and analyzed using the t test. Categorical data are presented as frequency/rate (%) and assessed through the chi-squared test. Statistical significance was determined when the P value fell below .05.
Results
Comparison of Biochemical Indicators
Following the intervention, levels of FPG, TC, TG, and LDL-C exhibited a decrease in both cohorts when compared to the pretreatment phase (P < .05). Notably, the reduction was more pronounced in the treatment group in comparison to the control group (P < .05; refer to Table 1 for specific data).
Comparison of Biochemical Indicators in Both Study Groups: Pre- and Posttreatment Assessment [mmol/L, (x̅ ± s)].
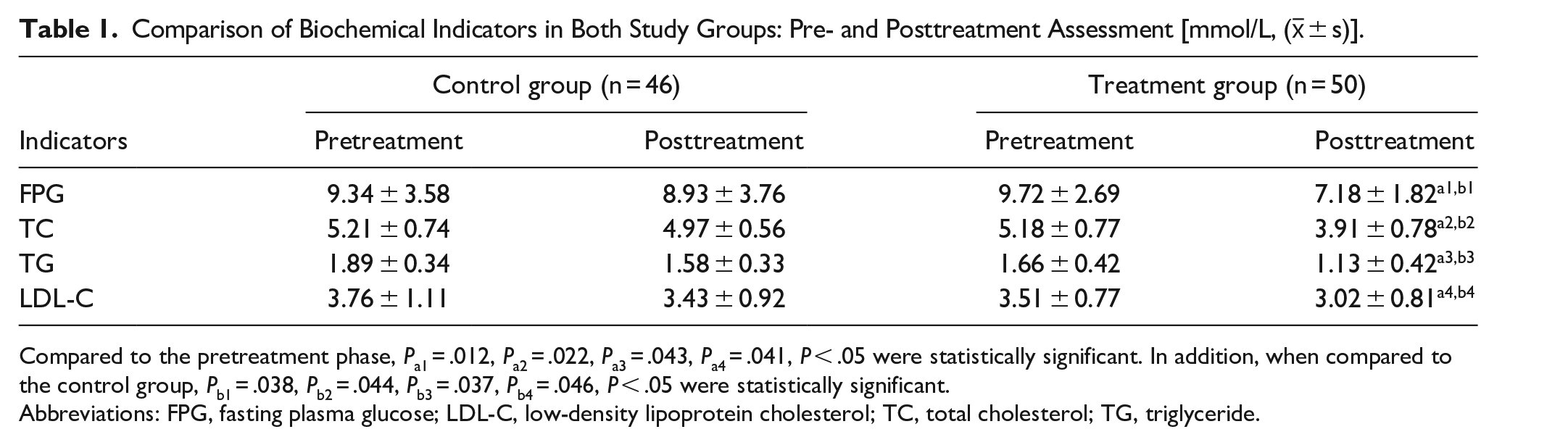
Compared to the pretreatment phase, Pa1 = .012, Pa2 = .022, Pa3 = .043, Pa4 = .041, P < .05 were statistically significant. In addition, when compared to the control group, Pb1 = .038, Pb2 = .044, Pb3 = .037, Pb4 = .046, P < .05 were statistically significant.
Abbreviations: FPG, fasting plasma glucose; LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglyceride.
Comparison of Clinical Efficacy Between the 2 Groups
The treatment group exhibited a total effective rate of 94.0%, while the control group demonstrated a rate of 89.1%. The disparity between the 2 groups was determined to be statistically significant (P < .05), as detailed in Table 2.
Evaluation of Biochemical Markers Pre- and Posttreatment in Both Study Groups [mmol/L, (x̅ ± s)].

Compared with the control group, P value = .012.
Comparison of Mean Hearing Thresholds Between Groups, As Well As Before and After Treatment
In assessing the initial hearing thresholds of both groups prior to treatment, no statistically significant difference was observed (P > .05). Subsequent to treatment, the average hearing thresholds in both groups exhibited a statistically significant reduction compared to their respective pretreatment levels (P < .05). Furthermore, the disparity in the average hearing thresholds between the treatment group and the control group reached statistical significance (P < .05), as outlined in Table 3.
Evaluation of Average Hearing Thresholds Pre- and Posttreatment, As Well As Between 2 Distinct Groups (x̅ ± s).
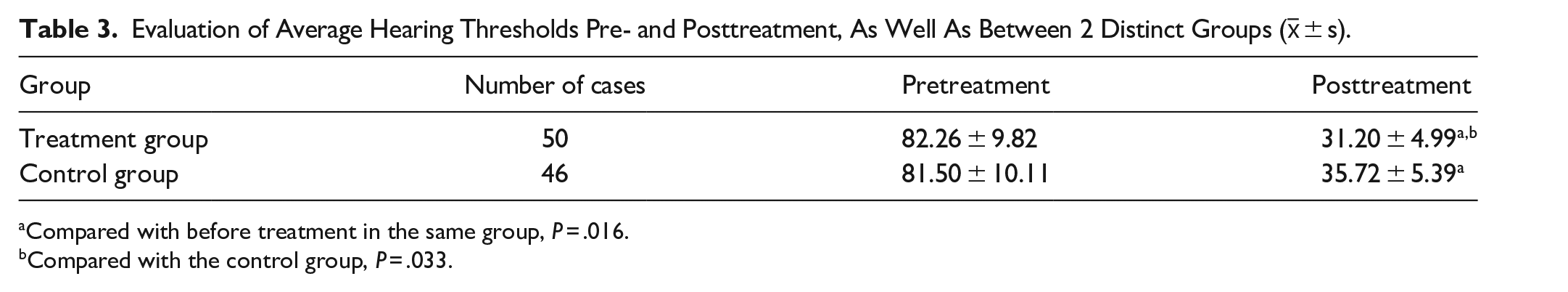
Compared with before treatment in the same group, P = .016.
Compared with the control group, P = .033.
Correlation Between Deafness Classification and Therapeutic Outcomes
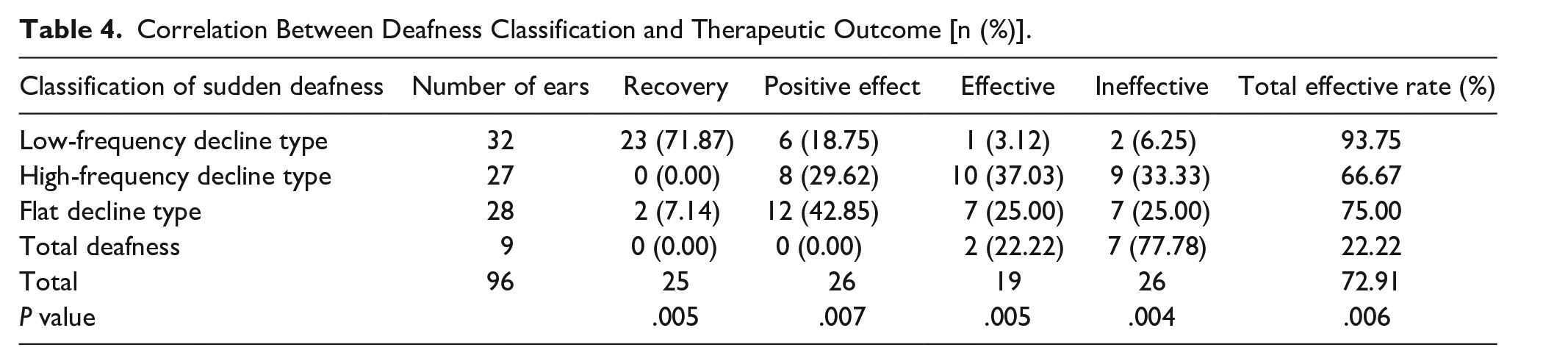
Among the classifications, the low-frequency decline type exhibits the most substantial total effective rate at 90.91%, whereas the total deafness type demonstrates the lowest total effective rate at 16.67%. The variances in both indicators among the groups exhibit statistical significance (P < .01), as outlined in Table 4.
Correlation Between Deafness Classification and Therapeutic Outcome [n (%)].
Discussion
Several studies have indicated an association between sudden deafness and circulatory disorders, viral infections, and autoimmunity. Notably, sudden deafness can be induced in patients with diabetes, as confirmed in animal models. This phenomenon may be attributed to the hypercoagulable state of blood and microangiopathy prevalent in patients with type 2 diabetes.
In recent years, HBOT has demonstrated notable clinical efficacy in the management of sudden deafness. Research suggests that hyperbaric oxygen may exert its effects through various mechanisms:
Rapidly increasing blood and tissue oxygen partial pressure may enhance the diffusion distance of oxygen in tissues, thereby improving inner ear circulation and alleviating hypoxia.
Reducing capillary permeability and mitigating tissue edema.
Accelerating the repair of hair cells and vestibular nerves.
Modulating the immune function of the organism and inhibiting the release of inflammatory factors.
Potentially increasing the sensitivity of pancreatic β cells to insulin, with positive implications for blood glucose and lipid reduction.
Possibly improving the state of vascular endothelium and ameliorating the hypercoagulable state of blood.
Early investigations into the mechanism of HBOT primarily concentrated on optimizing blood and oxygen delivery to the inner ear. Elevated pressure during HBOT results in increased oxygen tension in arteries and tissues, leading to enhanced oxygen-carrying capacity by hemoglobin. This, in turn, mitigates microcirculatory hypoxia in individuals experiencing sudden deafness.
As the trajectory of HBOT research advances, significant updates to the conceptual framework and mechanisms have emerged. In the context of both microthrombosis and vascular endothelial injury, HBOT extends the inhibitory phase of neutrophil activation, mobilizes stem cells derived from the vasculature, and enhances antioxidant capacity. These alterations establish a robust mechanism for safeguarding against ischemia-reperfusion.
In summary, effective HBOT demonstrates the potential to prevent early reductions in blood flow and local tissue ischemia in patients with sudden deafness. In addition, it exhibits a capacity to ameliorate the chronic inflammatory state. 4
Sudden deafness is frequently concomitant with varying degrees of capillary cell edema, resulting in a sustained reduction in the external lymphatic oxygen pressure within the body and an insufficient supply of oxygen to the cochlea. This in turn expedites the synthesis of inflammatory substances.5,6 HBOT has the capacity to swiftly augment oxygen delivery to the brain and the ischemic penumbra of the inner ear. This acceleration facilitates the removal of local hematomas, expedites the restoration of damaged neurons, and hastens the process of cellular regeneration. 7 As an adjunctive treatment, hyperbaric oxygen contributes to the enhancement of the partial pressure of oxygen and facilitates the restoration of hair cell function. 8
HBOT for patients experiencing sudden deafness facilitates local vasoconstriction, diminishes capillary permeability, and enhances microcirculation within the inner ear tissues. This process leads to a significant reduction in exudation, promoting the efficient elimination of edema. Consequently, it facilitates the restoration of local vascular perfusion and supply. Simultaneously, it effectively prevents edema and hypoxia, contributing to the overall recovery of the condition. 9
In addition, hyperbaric oxygenation markedly elevates the partial pressure of oxygen and oxygen content in the inner ear tissue. This augmentation promotes the supply of bio-oxidative energy and aids in the restoration of auditory function. 10 The vital role of HBOT in the recovery of auditory cells is evident through the increase in blood oxygen content, critically influencing the restoration of auditory cells. 9
Research has revealed that patients with sudden deafness often exhibit sluggish blood flow in the inner ear, rendering them susceptible to blood stasis and fat deposition. In such cases, blockages can lead to a hypoxic environment within the inner ear, exacerbating damage to the hair cells. Early intervention with medications to enhance microcirculation and nourish nerves proves effective in averting these complications and promoting overall recovery. 11
The findings of this study align with those of Chi et al. 12 In their randomized controlled trial, 60 patients experiencing sudden deafness were allocated randomly to either a control group or an intervention group receiving HBOT in addition to standard medication. The control group received medication alone. The outcomes revealed a superior improvement in hearing within the HBOT group compared to the control group, and no evident complications were observed. This suggests the potential efficacy of HBOT in the management of sudden deafness.
In a retrospective study, 13 HBOT was promptly initiated within 3 days for individuals with sudden deafness, serving as the exclusive therapeutic intervention without hormonal therapy. The results exhibited complete hearing recovery in 71.4% of patients, complete resolution of tinnitus in 32.3%, and a reduction in the severity of tinnitus (albeit to a lesser extent) in 45.2%.
The most recent Clinical Practice Guideline for Sudden Deafness (2019, USA) advocates the integration of HBOT and corticosteroid therapy as the primary treatment approach within a 2 week window following onset, and as a salvage treatment within 1 month of symptom manifestation. This guideline delves into the advantages of HBOT, outlining standardized treatment options for sudden deafness based on updated systematic review and meta-analysis reports. Notably, the guideline emphasizes the necessity for heightened awareness regarding the potential efficacy of HBOT in the context of sudden deafness. 14 We posit that there is a need for further investigation into the effects of HBOT, with the aim of progressively establishing a standardized treatment protocol to advance efforts in the management of sudden deafness.
Conclusion
The correlation between type 2 diabetes mellitus and sudden deafness is noteworthy, and HBOT demonstrates promising efficacy as a therapeutic intervention. HBOT has proven effective in addressing sudden deafness arising from diverse etiologies, displaying a favorable safety profile with minimal adverse reactions and complications. Its ease of acceptance by patients renders it a viable treatment option warranting wider endorsement. Particularly in instances where conventional treatments prove ineffective, the integration of HBOT into a comprehensive treatment approach is strongly recommended.
Footnotes
Acknowledgements
We are particularly grateful to all the people who have given us help on our article.
Author Contributions
Conceptualization:Hua-Qin Chen.
Data curation: Yao Peng, Yu Feng, Tian-li Jin. Data analysis: Hua-Qin Chen, Yao Peng, Yu Feng. Statistical analysis: Tian-li Jin, Yu Feng. Roles/Writing—original draft: Hua-Qin Chen. Writing—review and editing: Hua-Qin Chen.
Availability of Data and Materials
Date will be made available on request.
Consent for Publication
Consent for publication was obtained from every individual whose data are included in this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This study was conducted in accordance with the Declaration of Helsinki. This study was conducted with approval from the Ethics Committee of The First People’s Hospital of LanZhou City (2023A-12). A written informed consent was obtained from all participants.
