Abstract
Angioleiomyoma (ALM) is a benign neoplasm marked by the presence of blood vessels and smooth muscle cells, commonly located within the subcutaneous or deep dermal layers. Its manifestation in the pterygopalatine fossa is infrequent, thereby posing difficulties in its diagnosis and distinction from other benign tumors within the same anatomical site. In the present case study, an ALM originating in the right pterygopalatine fossa of a 44-year-old female patient was investigated. The patient underwent surgical intervention for the vascular smooth muscle tumor, and there has been no recurrence noted during the postoperative monitoring period.
Introduction
Angioleiomyoma (ALM) is a rare neoplasm derived from vascular smooth muscle, primarily affecting the skin and subcutaneous tissues, commonly afflicting middle-aged individuals with a preference for extremities. Its presence within the nasal cavity is uncommon and may present diagnostic hurdles owing to vague clinical symptoms. In the present case study, a 44-year-old female patient with a hemangioma in the right pterygopalatine fossa underwent investigation to enhance the diagnostic and therapeutic approaches for this condition. As far as current knowledge, this patient is the first case of ALM in the right pterygopalatine fossa.
Case Presentation
A 44-year-old female patient was admitted to the Department of Otorhinolaryngology, Head and Neck Surgery of Jiangsu Provincial Su Bei People’s Hospital. The reason for hospitalization was the detection of a mass in the right pterygopalatine fossa during a physical examination 1 month prior. The patient did not report any symptoms such as nasal congestion, runny nose, reduced sense of smell, nosebleeds, headaches, dizziness, unpleasant nasal odor, visual disturbances, facial numbness, nausea, vomiting, or significant changes in hearing in her right ear. The patient’s vital signs were within normal limits. She exhibited good mood and normal nutritional status. Throughout the physical examination, the patient remained calm and cooperative. The neck was soft, and the trachea was centered. There were no deformities in the thorax, and the respiratory sounds of both lungs were symmetric, with no audible crackles. Abdominal examination revealed a flat and soft abdomen without signs of tenderness on compression or rebound pain. Although the external nose appeared nondeformed, a slight deviation of the septum with irregularities was noted. Bilateral chronic congestion and hypertrophy of the inferior turbinates were observed. No obvious signs of neoplastic growth were detected in the bilateral nasal passages, and there was no tenderness on palpation in the sinus region.
Sinus CT: The computed tomography (CT) scan of the sinuses revealed a soft tissue density shadow in the right pterygopalatine fossa, exhibiting inhomogeneous enhancement in the contrast-enhanced scan. This finding suggests the possibility of a neoplastic lesion. Please refer to Figure 1 for visual representation.
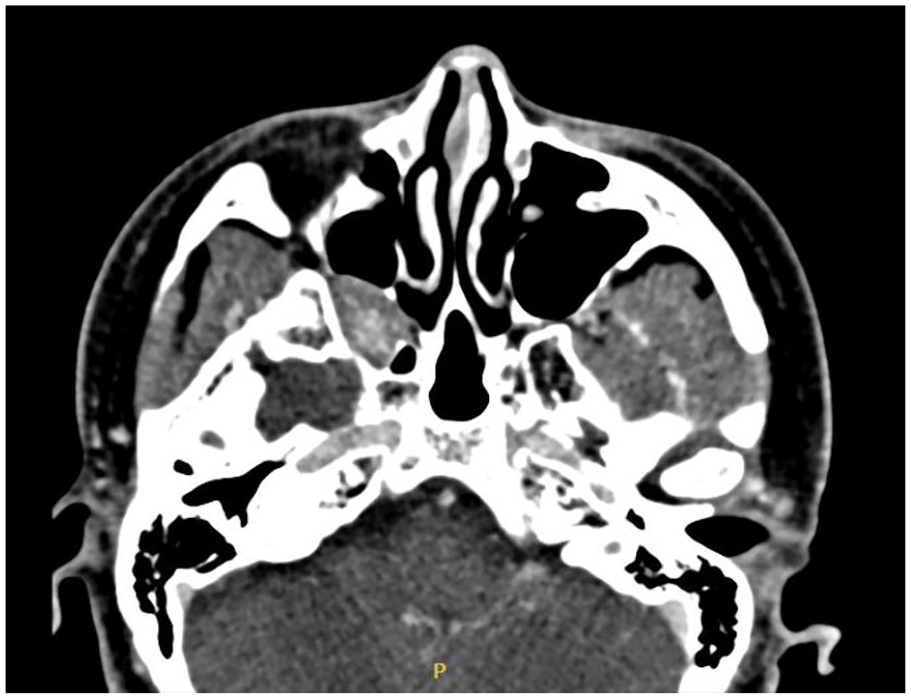
Sinus CT scan. CT, computed tomography.
Sinus MRI: The magnetic resonance imaging (MRI) scan revealed the presence of a mass located in the right pterygopalatine fossa, potentially indicative of a cavernous hemangioma, along with bilateral ethmoid sinusitis (Figure 2).
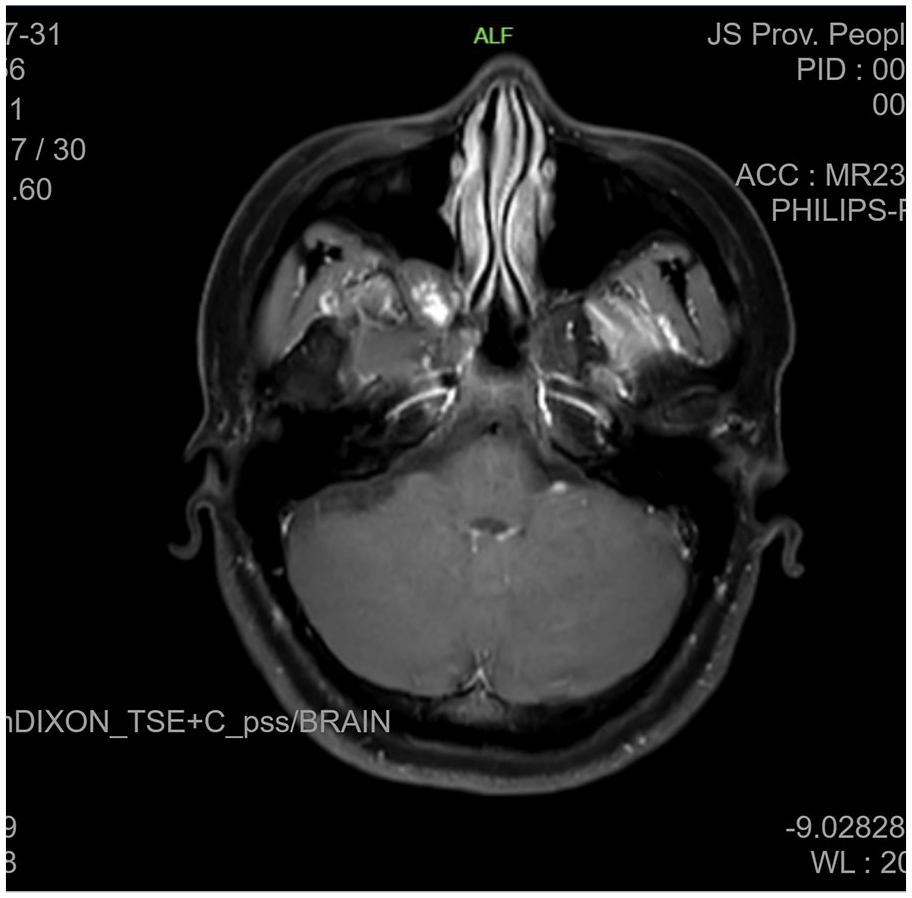
Sinus MRI scan. MRI, magnetic resonance imaging.
Laboratory tests performed after admission, encompassing routine blood tests, C-reactive protein assessment, tumor marker evaluation, and coagulation analysis, did not uncover any noteworthy abnormalities. The patient underwent endoscopic resection of lesions in the right pterygopalatine fossa, maxillary sinus, and ethmoid sinus, in addition to middle nasal turbinate mucosal flap repair (Figure 3).
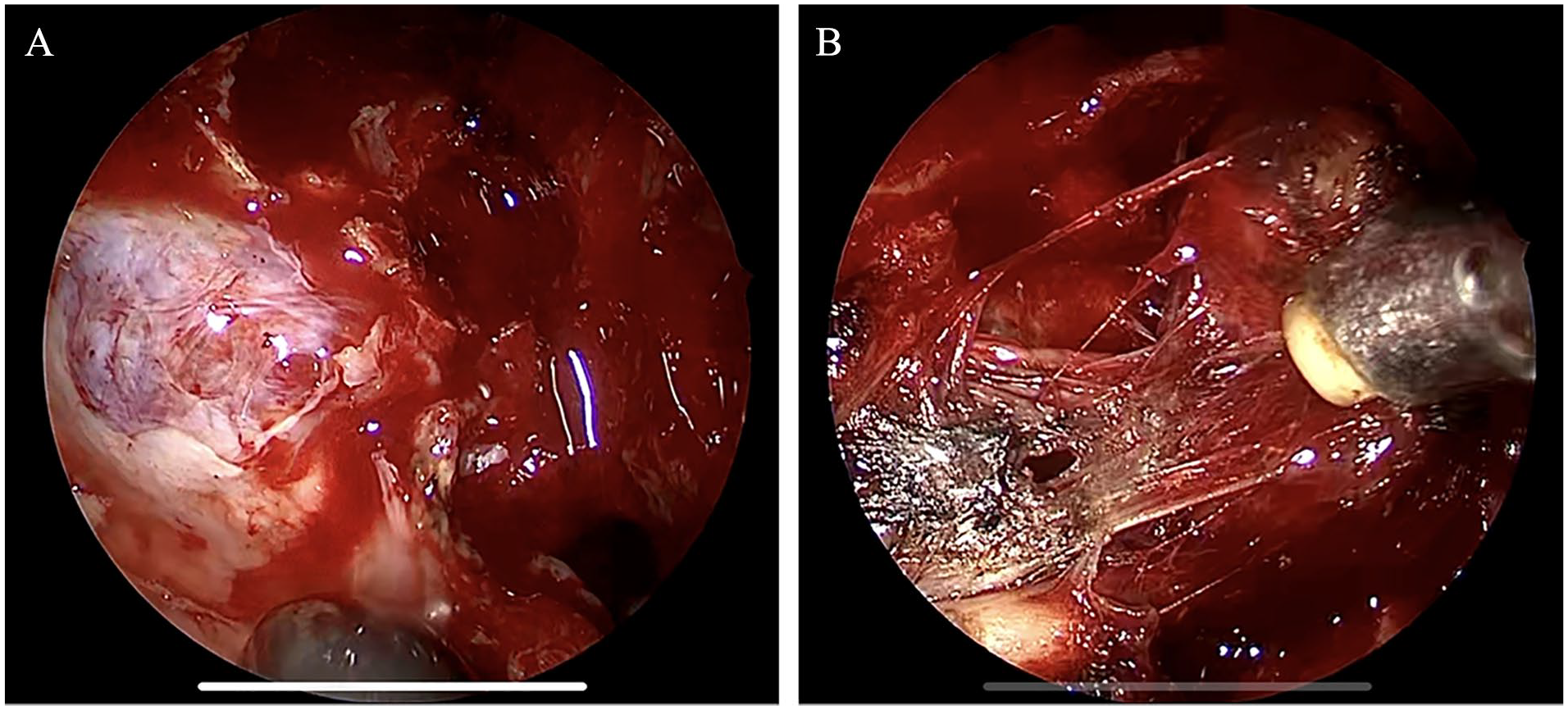
Intraoperative observation. (A) Open the posterior lateral wall of the maxillary sinus. (B) Disconnect the root of the tumor.
The postoperative pathology report indicated ALM (see Figure 4). Following the surgery, the patient reported experiencing mild nasal congestion without any episodes of nasal bleeding or fluid discharge. During the fourth-week postsurgery, a follow-up examination revealed an unobstructed nasal cavity with a small amount of dry scab present on the surface of the right pterygopalatine fossa. Notably, there has been no evidence of recurrence observed during the postoperative follow-up period.
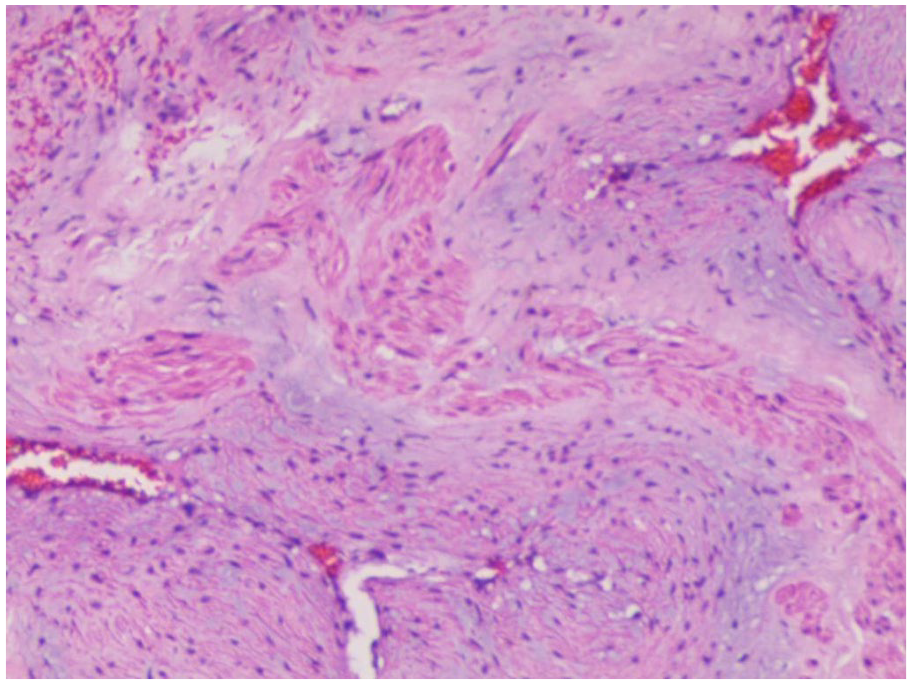
HE low magnification, visible vascular lumen, and smooth muscle interweaving, consistent with angioleiomyoma. HE, hematoxylin and eosin.
Discussion
ALM is a benign tumor frequently located in mesenchymal tissue, yet its specific tissue origin remains undetermined. 1 It may arise from undifferentiated ectopic embryonic mesenchymal tissue or smooth muscle cells within the vascular wall.2,3 The majority of cases, approximately 89%, manifest in the extremities, particularly in the deep dermis and subcutaneous layers of the calf. These tumors typically present as solitary lesions with a diameter of less than 2 cm. In contrast, occurrence of ALM in the nasal cavity is infrequent, constituting around 3% of cases 4 and approximately 1% of all benign nasal tumors. 5 Clinical symptoms of ALM are nonspecific, often leading to misdiagnosis. The etiology of ALM remains unknown, but potential factors include minor trauma, EB virus infection, and hormonal changes.6,7 In the present case, the patient developed a growth in the right lateral pterygopalatine fossa, an exceedingly rare location with an unidentified etiology.
Vascular leiomyoma is a rare phenomenon in the nasal region primarily attributed to the scarcity of smooth muscle in the nasal cavity, apart from within the vascular walls. The initial report of ALM in the nasal cavity dates back to 1966, documented by the Maesaka research group. 8 Since then, only a limited number of cases have been documented in the English literature, with the majority being case studies. Nasal ALM is typically observed more frequently in the turbinate, vestibule, and septum of the nose. Clinical symptoms typically include nonspecific symptoms such as nasal congestion, sinusitis, and epistaxis. The disease progression typically follows a protracted course. According to the 2022 World Health Organization Classification of Head and Neck Tumors (5th edition)9,10 and the 2020 World Health Organization Classification of Soft Tissue Tumors, 11 ALM is categorized as a benign vascular tumor within the soft tissue tumor classification. The tumor in ALM is distinguished by thick-walled blood vessels lined with a single layer of mild endothelium, surrounded by mature smooth muscle of variable thicknesses. Immunohistochemical staining displayed CD34 positivity in endothelial cells, smooth muscle actin (SMA) positivity, desmin positivity, and a lower Ki-67 positivity index in tumor cells. 12 ALM can be classified into 3 histological subtypes based on the composition of smooth muscle and blood vessels, namely the solid type, venous type, and sponge-like type. 13 The solid type is characterized by the presence of numerous narrow blood vessel lumens of varying sizes and thicknesses, intricately interwoven with surrounding smooth muscles within the tumor mass. The venous type presents as a smooth muscular nodule formed around a sizeable venous wall. In contrast, the sponge-like type comprises many dilated vascular lumens with fewer smooth muscle components. The author reports a patient with ALM exhibiting histopathological characteristics of a narrow, thick vascular cavity intertwined with smooth muscle. The leiomyoma was solid and located in the right pterygopalatine fossa, a relatively uncommon site for this condition.
ALM grows slowly and lacks specificity in radiography. CT scans of ALM often show apparent soft tissue masses, similar to hemangiomas. 14 On MRI scans, ALM commonly presents with uniform or slightly low signal intensity in T1-weighted imaging, low signal in diffusion-weighted imaging (DWI), and heterogeneous high and low signals, particularly at the edge ring, in T2-weighted imaging (T2WI). Significant enhancement is typically observed post-contrast administration. 15 Edo et al reported that angiogenic smooth muscle tumors exhibit the dark mesh sign in T2WI sequences, a typical manifestation of ALM meshwork. 16 In clinical practice, distinguishing ALM from conditions like schwannoma, cavernous hemangioma, and hemangiopericytoma can pose challenges. While imaging examinations lack distinctive features, preoperative CT and MRI scans play a crucial role in elucidating the location, size, extent, and adjacent relationships of the lesion. This information aids in selecting appropriate surgical strategies.
As the clinical specificity of ALM is not high, it must be distinguished from the following diseases: (1) Vascular smooth muscle lipoma: Mainly composed of mature adipose tissue, smooth muscle, and blood vessels. Positivity for HMB-45 is observed, while mature adipose tissue is rarely found in ALM. (2) Hemangioma: A typical vascular tumor lesion, usually asymptomatic and appearing red. CT scans reveal a distinct soft tissue mass with visible enhancement. Pathologically, the stroma does not contain smooth muscle bundles between its blood vessels. 17 (3) Perimuscular cell tumor: There is an oval- or spindle-shaped proliferation of perimuscular cells around thin-walled blood vessels, showing positive expression of SMA and negativity or focal positivity for desmin. (4) Nasopharyngeal fibrohemangioma: More common in the nasopharynx of adolescent males, consisting of many thin-walled blood vessels and a sparsely fibrous collagenous stroma. In some cases, the tumor body may contain fat components. (5) Smooth muscle sarcoma: Composed mainly of undifferentiated mesenchymal or fibroblast-like cells, along with myofibroblast-like cells. Tumor cells exhibit nuclear dysplasia and an increased mitotic rate. 18 (6) Neurogenic tumor: Pathological examination typically demonstrates positive expression of S100 protein.
Surgery is highly effective in eliminating ALM, often enabling complete resection of the mass along the periphery of the surrounding tissue. This approach typically results in a low recurrence rate post-resection and yields a favorable prognosis for patients. 19 If the tumor is sizable and imaging indicates a significant presence of blood vessels, it is advisable to consider comprehensive surgical treatment approaches coupled with vascular intervention and embolization prior to resection. This combined strategy helps mitigate the complexity and risks associated with surgery.
Conclusion
ALMs located in the pterygopalatine fossa are exceedingly uncommon. The primary focus in managing these tumors lies in their pathological diagnosis, with surgical intervention being deemed as a practical and preferred approach. ALM is characterized as a slow-growing benign tumor featuring a pseudocapsule and well-defined boundaries with surrounding tissues. Typically, ALM lesions do not invade surrounding structures and demonstrate a favorable prognosis following resection, although they may cause compression in adjacent tissues. Nevertheless, their rarity and vague clinical presentations render them easily overlooked in medical practice, potentially resulting in misdiagnoses or missed diagnoses. To mitigate this risk, it is advisable to thoroughly review the patient’s medical history, conduct imaging studies, and carefully consider differential diagnoses.
Footnotes
Acknowledgements
We express our gratitude to Bing Guan and Ma Wei for their invaluable review and guidance during the final revision process.
Author Contributions
The concept and layout of the study were proposed by Bing Guan. Data collection and analysis were performed by Haiyong Sun. The literature search was completed by Wei Ma. The core article was written by Haiyong Sun. Bing Guan performed editing and critical revisions. Bing Guan managed the study and provided funding. The final draft of the article was approved by all authors after review. All authors have read and approved the final article.
Availability of Data
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Jiangsu Province Natural Science Foundation (BK20201220).
Ethical Approval
The study was approved by the Ethics Committee of the Ethics Review Committee of the Yangzhou University Clinical Medical College (Approval number: 2020ky106) and was conducted in accordance with the Declaration of Helsinki.
Written Informed Consent for Publication
Written informed consent was obtained from the patient for the publication of this case report and any accompanying images.
