Abstract
Keywords
Introduction
Microtia is the second common congenial craniofacial deformity, primarily characterized by abnormal development of the external ear. It is often accompanied by varying degrees of craniofacial skeletal developmental abnormalities.1,2 The Nagata’s 2-stage and the expanded two-flap methods are most used for auricular reconstruction. The advantage of expanded two-flap method is that it can obtain sufficient and ideal thickness skin flap through tissue expansion, which can be used under different postauricular skin conditions. The expanded two-flap method for auricular reconstruction includes 3 stages. Cartilage scaffold exposure is one of the main complications after the second stage, which can lead to serious consequences such as cartilage absorption, infection, and necrosis.
The superficial temporal fascial flap is also called the temporoparietal fascial flap. It is located between the subcutaneous tissue of the temporal region and the deep temporal fascia. This fascial flap has a stable blood supply from the superficial temporal vessels, and the facial nerve and auriculotemporal nerve provide it with rich sensations. Moreover, it is flexible and can be deformed and turned at will. These anatomical advantages make it widely used in plastic and reconstructive surgery.3,4 In ear reconstruction using autologous rib cartilage, Nagata 5 and Park et al 6 have skillfully used superficial temporal fascia combined with skin graft to cover the framework. Reinisch and Lewin 7 have covered and wrapped the high-density polyethylene prosthesis stent with superficial temporal fascia for a large number of auricle reconstructions and achieved stable results. However, considering that cartilage exposure is not a rare complication. Brent 8 advocated not to use superficial temporal fascial flap as a routine material for auricular reconstruction, but to reserve it as a material that may be used for secondary repair. Using superficial temporal fascia flap combined with skin flap graft to cover the exposed or unsatisfactory cartilage framework can achieve good postoperative results.9,10
Patients and Methods
This study reviewed patients who underwent auricular reconstruction with the expanded two-flap method in the Department of Auricular Reconstruction at the Plastic Surgery Hospital, Chinese Academy of Medical Science and Peking Union Medical College (Beijing, China). The exclusion criteria were as follows: microtia as a part of syndromic congenital deformity except for hemifacial microsomia (HFM), previous trauma or surgery related to ear reconstruction, cartilage scaffold exposure due to trauma, mosquito bites, or infection. As a result, 853 patients (908 sides) were included in this study. Among these patients, 641 (75.1%) were males and 212 (24.9%) were females, wherein 493 (47.8%) with right microtia, 305 (35.8%) with left microtia, and 55 (64.4%) with bilateral microtia. The mean age was 10.4 ± 5.2 years. There were 32 cases of exposed ear cartilage, of which 23 were males and 9 were females; 14 cases were on the right side and 18 were on the left side. There was no statistically significant difference between the cartilage exposed group and the nonexposed group in gender (P = .662) and sides (P = .334; Table 1). The study was reviewed and approved by the hospital ethics committee (approval number: z201707).
Characteristics and Statistical Analysis of the 853 Microtia Patients (908 Ears) Treated With the Expanded Two-Flap Method.
In this study, all 32 cases of cartilage exposure occurred at the distal end of the postauricular fascia. It is mainly considered that the cut and reversed postauricular fascia has a single blood supply at this point, and the proliferation of the posterior auricular artery is not ideal, leading to ischemia and cartilage exposure. 11 To investigate whether HFM is a risk factor for cartilage scaffold exposure, we set up all patients with cartilage exposure as the case group and matched 1:1 sampling in the noncartilage exposure patients (821 cases) according to variables such as gender, age, and side, with the same number of samples matched as the control group (32 cases). A case-control study was performed to analyze the correlation between cartilage exostosis and HFM and the Orbit, Mandible, Ear, Nerve, and Soft tissue (OMENS) classification.
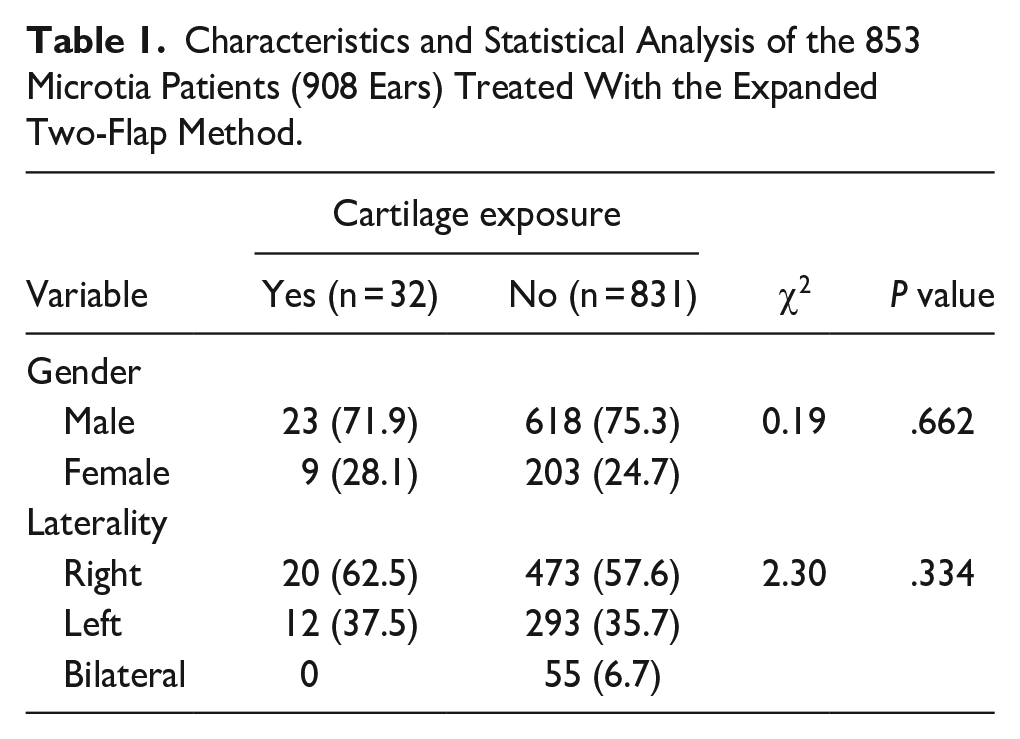
In the early stage, whitening could be seen in the local area, followed by scabbing. Within 3 months, the scab fell off and the cartilage exposed (Figure 1). All cases underwent surgical repair. Before the operation, Doppler ultrasound was used to explore the course of the superficial temporal artery to facilitate the intraoperative resection of the superficial temporal fascia flap. The surgical procedure included 3 steps. First, debridement was performed on the exposed cartilage scaffold to remove necrotic tissue on the surface, and soft tissue around the wound was excised to the active bleeding area. It also needed to be partially excised if the scaffold was necrotic. In the case of the amount of necrotic cartilage scaffold removed significantly affected the auricular morphology, partial residual ear cartilage should be harvested and spliced to the original framework with 5-0 absorbable stitches to repair the defect site to maintain the structure and stability of the auricle. Second, according to the location and size of the wound to decide the range of superficial temporal fascial flap. A curved incision was designed along the temporal superficial artery and the required superficial temporal fascial flap was carefully separated under the scalp fascia layer to forming an axial fascial flap with a vascular pedicle. Subsequently, it was flipped downward and folded over the exposure cartilage wound. Finally, a thin split-thickness scalp was grafted on the surface of the superficial temporal fascia. The graft area was covered with Vaseline gauze at first and then covered with general medical gauze roll with uniform stress. Finally, knotted and secured by 3-0 nonabsorbable stitches. After the operation, cephalosporin antibiotics were used preventively intravenously for 24 hours. The sutures were removed 10 days after the surgery.
(a) Localized whitening at the upper posterior margin of the helix; (b, c) exposure of cartilage scaffold.
All statistical analyses were performed using SPSS software, version 25.0 (IBM Corp, Armonk, NY). Unordered categorical variables were represented by percentages and were tested using the chi-square tests. Case-control studies were analyzed by logistic regression analysis and qualitative matched data were analyzed by Wilcoxon signed-rank test. A P value less than .5 was considered statistically significant.
Results
Sampling was conducted on a 1:1 match basis according to variables such as gender, age, and side, resulting in 29 precise matches and 3 fuzzy matches. All patients in 2 groups were elevated by the OMENS classification system proposed by Boston Children’s Hospital in 2011 and the MacQuillan method was used for the diagnose of HFM. A total of 30 patients were diagnosed as HFM, including 18 in the case group and 12 in the control group. The OR value was calculated to be 2.1 [95% confidence interval (0.788, 5.825)], which considering that HFM may be a risk factor for the complication of cartilage exposure, but its statistical significance needed further verification (Table 2). The differences in the severity of ocular deformity, facial nerve dysplasia, and soft tissue dysplasia between the 2 groups were statistically significant (Table 3).
Statistical Analysis of the Odds Radio Between HFM and Cartilage Exposure.

Abbreviations: CI, confidence interval; HFM, hemifacial microsomia.
OMENS Typing and Statistical Analysis of Patients in the Case and Control Groups.
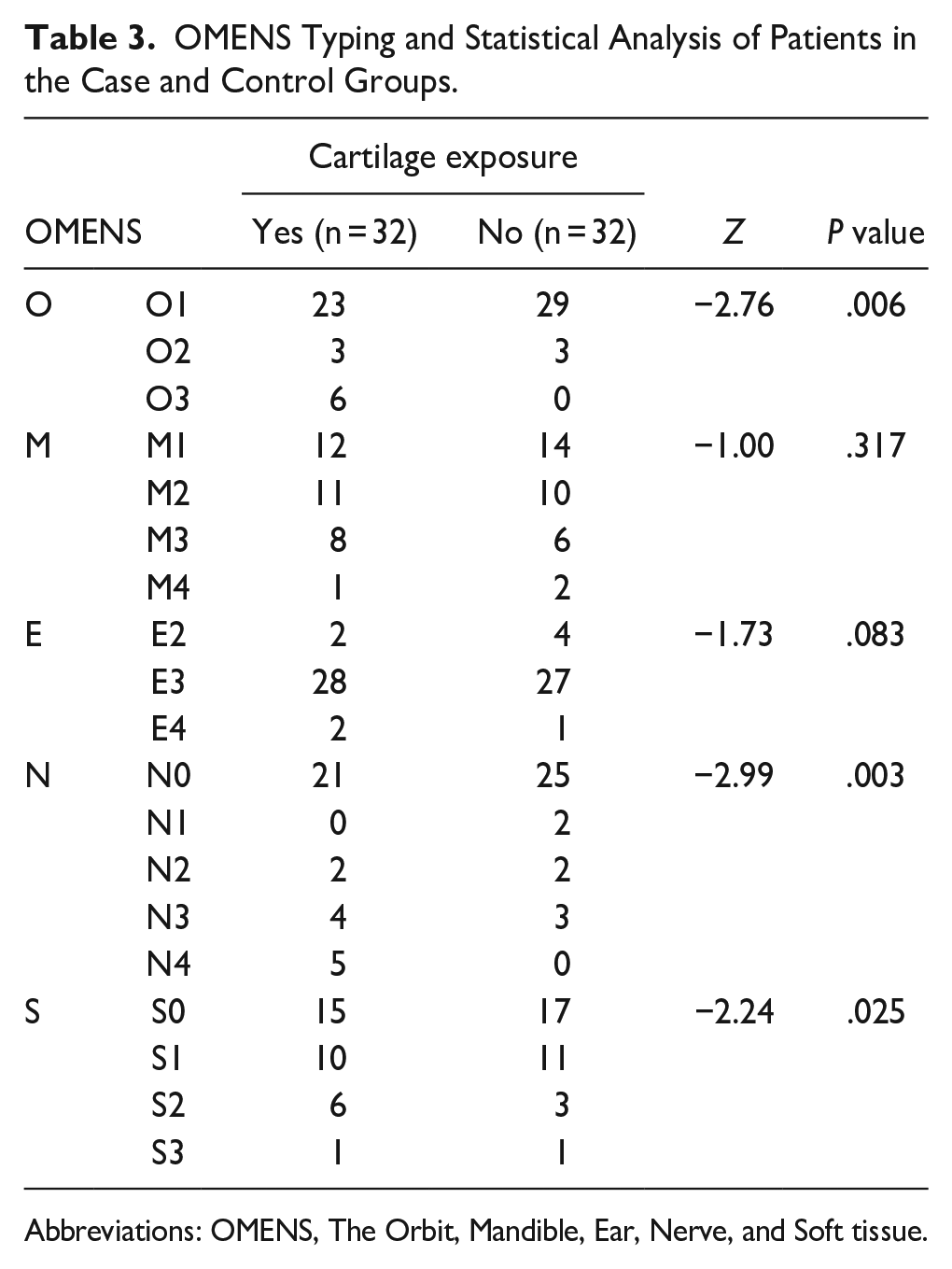
Abbreviations: OMENS, The Orbit, Mandible, Ear, Nerve, and Soft tissue.
Thirty-two patients in the case group were followed up at 1, 3, 6, and 12 months postoperatively. All wounds healed well, with good survival of the fascia and skin grafts, and no necrosis. Follow-up showed that the structure of reconstructed ear, such as the helix, antihelix, triangular fossa, and concha were in clear contours, with no swelling and no recurrence of cartilage exposure. All patients had satisfactory outcomes (Figures 2-4).
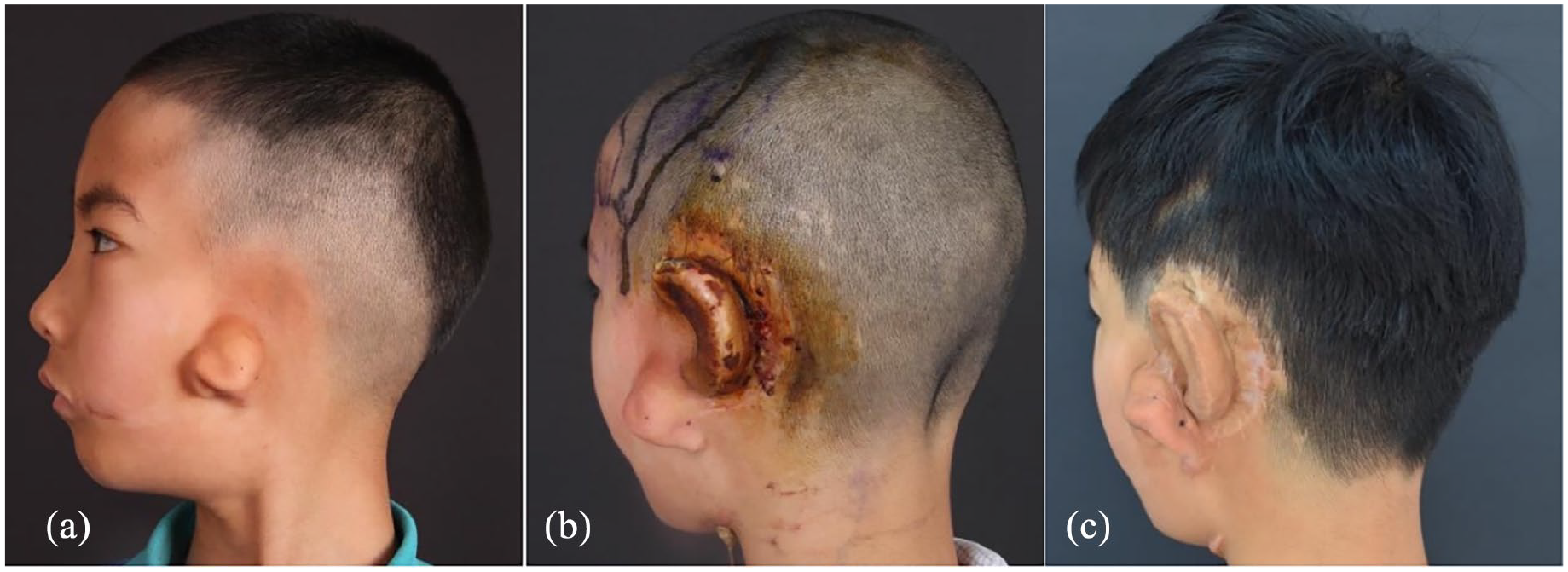
A 9-year-old hemifacial microsomia boy who underwent auricular reconstruction with the expanded two-flap method. (a) Left microtia before surgery; (b) cartilage scaffold exposed 2 months after the second-stage surgery; (c) 12 months after reparation with superficial temporal fascial flap and a free skin graft.
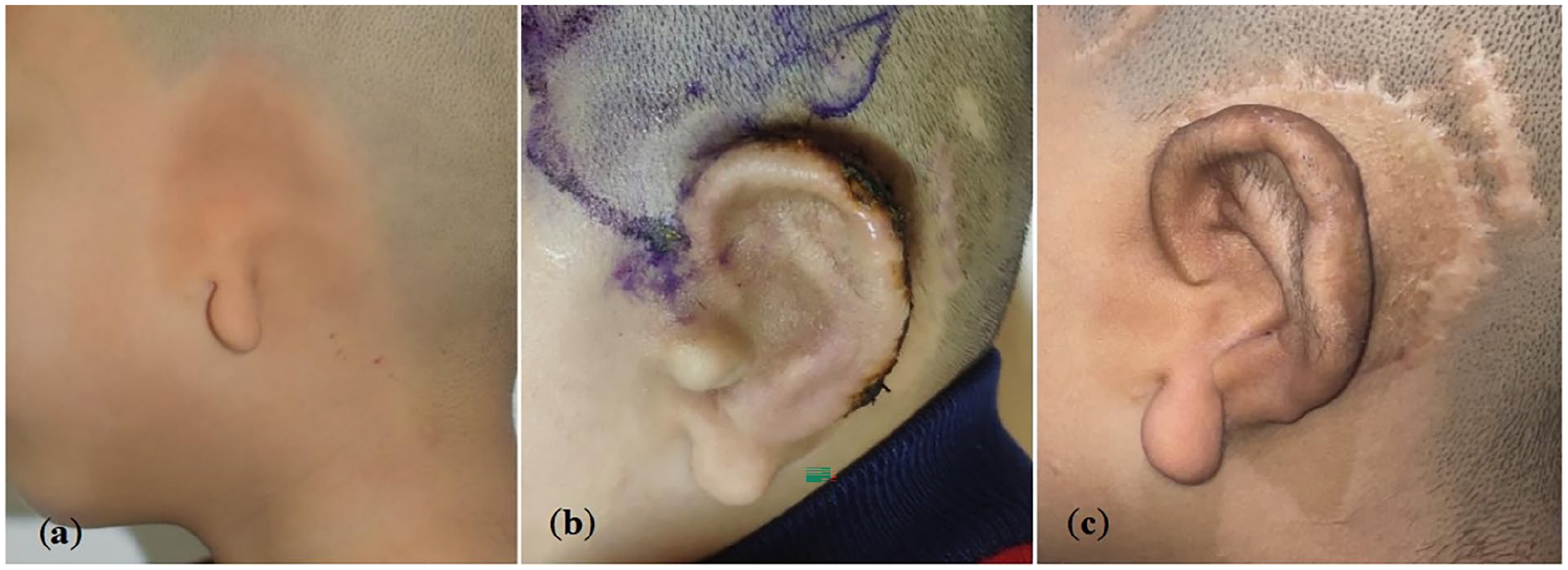
A 7-year-old hemifacial microsomia boy who underwent auricular reconstruction with the expanded two-flap method. (a) Left microtia before surgery; (b) cartilage scaffold exposed 2 months after the second-stage surgery; (c) 6 months after reparation with superficial temporal fascial flap and a free skin graft.
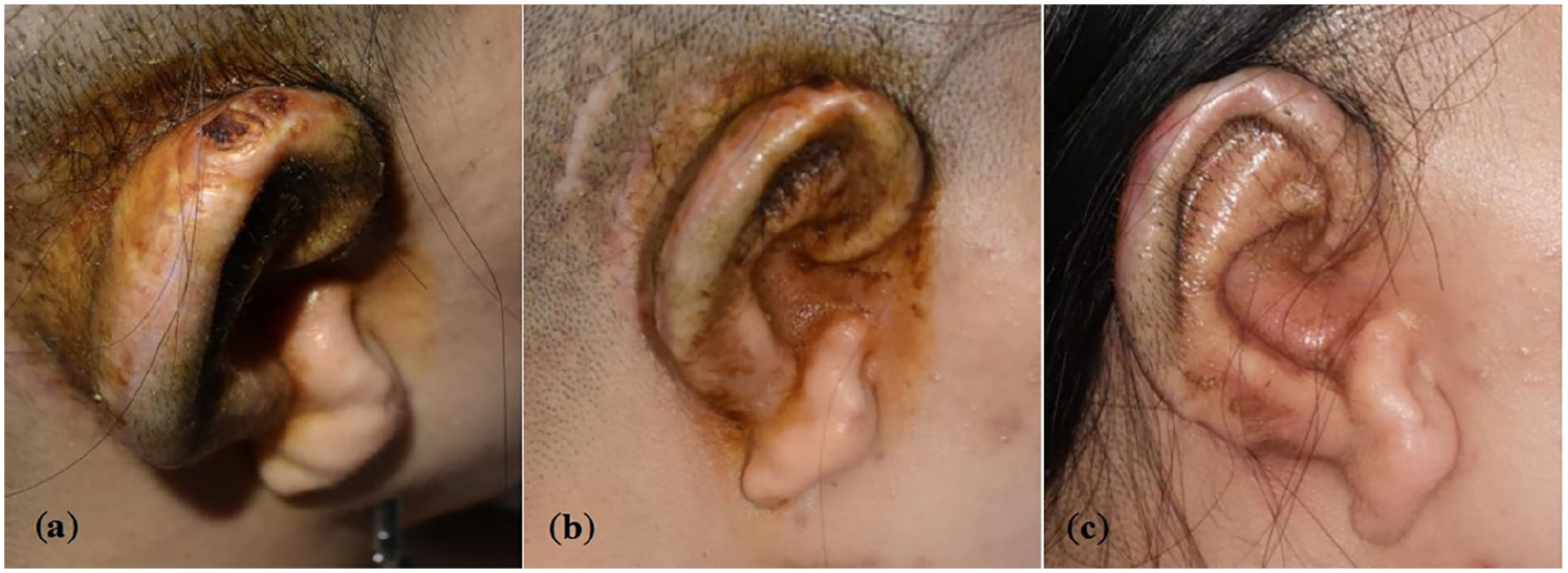
A 24-year-old hemifacial microsomia female who underwent auricular reconstruction with the expanded two-flap method. (a, b) Cartilage scaffold exposed 3 months after the second-stage surgery; (c) 12 months after reparation with superficial temporal fascial flap and a free skin graft.
Discussion
The expanded two-flap method is one of the most used surgical methods for auricular reconstruction. The procedure includes 3 stages. In the first stage, a 50 ml kidney-shaped expander is inserted into the subcutaneous layer to create a skin pocket in the mastoid region. The expander is gradually inflated to obtain an ideal thickness and range of skin flap. In the second stage, the expanded skin flap covers the anterior surface of the autologous cartilage auricular framework, while the posterior surface and the helical rim of the framework are covered by a combination of fascia transfer and skin grafting. In our study, postauricular fascial flap was used in the second stage, because it was richly supplied by multiple branches of the postauricular artery and a small number of superficial temporal artery branches (Yoshimura, 2000). 12 It was easy to reverse, which caused relatively small trauma to patients and the superficial temporal fascia was preserved.7,8 The third stage performed approximately 6 months after the second stage, generally involving lobule transposition, tragus reconstruction, and excision of ear remnants. Complications mainly occur after the first and second stages of surgery, among which cartilage exposure is one of the most serious complications. It can occur at the junction of the posterior superior margin of the helix, the connection between the reconstructed helix and the lobule flap, the tragus, and the antitragus. All exposed cartilage in our study were found at the junction of the expanded flap and the skin graft, suggesting there is a close correlation between cartilage scaffold exposure and blood supply at the distal of the postauricular fascia. HFM is the second most common craniofacial birth defect, which caused by dysgenesis of the first and second gill arches. 13 Most HFM patients have different degrees of external ear deformities and are companied with skin, soft tissue, and vessels dysplasia. They are characterized by thinner postauricular skin, less subcutaneous tissue, and local arteriovenous anatomical abnormalities.10,14-16 which can severely affect the blood supply of fascial flap. To investigate the correlation between HFM and cartilage scaffold exposure, we performed a case-control study on 2 groups by logistic regression analysis, considering that HFM may be a risk factor for cartilage scaffold exposure, but its statistical significance needed to be further verified. The OMENS classification evaluation was conducted on 2 groups of patients, and it was found that cartilage scaffold exposure was correlated with the severity of ocular malformation, facial nerve dysplasia, and soft tissue dysplasia. We speculated that these malformations are accompanied by different degrees of vascular dysplasia, resulting in a lack of blood supply to the fascia, which lead to cartilage framework exposure. Currently, there is no consensus on the surgical method for ear reconstruction in HFM patients. Chen et al 16 has used a single expanded flap method without fascial flap transfer and skin graft in 41 HFM patients’ ear reconstruction and achieved good results. Qian et al 10 has used an expanded two-flap method for ear reconstruction in 111 HFM patients, with only a 1.8% incidence of cartilage scaffold exposure. In conclusion, it suggests that we should elevate the severity of deformities in HFM patients carefully before auricular reconstruction surgery, including ocular deformity, facial nerve dysplasia, and soft tissue dysplasia. A thorough assessment of blood supply is necessary to reduce the incidence of cartilage exposure.
Once the cartilage is exposed, it often leads to infection, necrosis, and resorption, which directly affects the final outcome. Therefore, it needs to be dealt with promptly. For cartilage exposure caused by necrosis of the skin flap with a diameter of less than 2 to 5 mm, it can be promoted through conservative treatments such as regular dressing, the use of growth factor gel, or directly sutured without tension after debridement. Surgical repair is always required in larger and more severe exposure. The cartilage exposure occurs at the crus of helix, the concha, or the junction between the helix and the earlobe; local skin flap transfer is considered at first. Fascial flap combined with skin graft is needed in following situations: cartilage exposure occurs in the skin graft area or at the junction of skin graft. Postauricular fascial flap, mastoid fascial flap, occipital artery fascial flap, deep temporal fascial flap, and superficial temporal fascial flap had been reported for repair, but there was no consensus on the efficacy.9,17,18 Kim et al 9 concluded that surgeons should choose appropriate fascial flaps according to the location and size of the cartilage exposure. For example, the superficial temporal fascial flap or the deep temporal fascial flap can be selected for the exposure of the anterior surface or upper third of the scaffold. For the lower two-thirds or the posterior surface of the reconstructed ear can be repaired with a mastoid fascial flap. The occipital artery fascial flap is not preferred because it is relatively far from the reconstructed ear and the fascia is prone to poor blood circulation at the distal end after being folded over a long distance. However, it can be considered in cases where superficial temporal fascial flap has been used or destroyed in previous surgical, HFM with superficial temporal fascial misalignment or superficial temporal artery injury. 19 The superficial temporal fascia flap is rich in blood supply and is relatively close to the reconstructed ear. It is currently the most used material for covering the exposed scaffold.
The superficial temporal fascial flaps in children differ from that in adults, especially in children with HFM, the superficial temporal fascial flaps are thinner. Blood vessel diameters in children are also thinner, may be with varying degrees of vasospasm and anatomical variants. The blood supply pattern of the superficial temporal fascia also varies. In addition to the superficial temporal artery blood supply type, some studies have also reported of retroauricular artery blood supply type and the occipital artery blood supply type.6,20
Therefore, it is very important to use Doppler ultrasound to explore the vascular alignment preoperatively. It can directly explore the blood supply pattern of the superficial temporal fascial flap and guide the incision design and separation of the superficial temporal fascia. Several techniques can help increase the success rate of repair operation: (1) The skin incision should be made along the hair follicles to avoid affecting hair growth. (2) The range of separating under the scalp can be slightly larger, which helps ensure the integrity of the superficial temporal artery. (3) The length of the superficial temporal fascia flap can be slightly longer to reduce tension at the pedicle. (4) The fascia flap should be turned along the direction of the vessel during the entire process to avoid pedicle torsion. (5) The fascial flap covering the exposure cartilage can overlap with the surrounding soft tissue to a certain extent to reducing the risk of cartilage reexposure. In this study, all 32 cases of cartilage exposure were repaired with superficial temporal fascial flap and achieved satisfactory outcomes without recurrence of exposure.
Conclusions
HFM may be a risk factor for cartilage scaffold exposure, where the severity of ocular malformation, facial nerve dysplasia, and soft tissue dysplasia are correlated with this complication. Superficial temporal fascia flap combined with skin grafting is a good method for secondary repair after cartilage exposure.
Footnotes
Author Contributions
All authors did the review and editing. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
All data generated or analyzed during this research are included in this published article. Data supporting the findings of this study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China, grant number 81701930.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the hospital ethics committee (approval number z201707).
Statement of Informed Consent
All patients or their guardians provided written consent for the use of patients’ images.
