Abstract
Introduction
Oral cavity is the most common site for head and neck mucosal cancers. 1 Traditionally, cancers within this site have been associated with smoking, tobacco use, as well as alcohol intake. Through the association with tobacco and alcohol use, socioeconomic deprivation negatively affects the development, treatment, and outcomes in the low socioeconomic status (SES) population.2-4 The impact of SES is so profound that low SES itself has been shown to have excess attributable risk for development of HNC even after controlling for alcohol and tobacco use.5,6
The impact of SES on oral cavity squamous cell carcinoma (OCSCC) incidence as well as survival has been previously reported.7-10 Multiple studies have shown that individual SES, neighborhood SES, marriage status, level of education, type of occupation, and income are associated with increased risk of developing HNC.7-10 Low individual and neighborhood SES also negatively affect overall and disease-specific survival independent of demographics, comorbidities, clinical stage, and treatment modality.11-15 The difference in survival is observed before the first 12 to 18 months following treatment and suggests that acute and subacute complications play a notable role in the observed disparity. 12
Despite the significant impact of SES on outcomes in HNC and OCSCC, there is a limited understanding as to how specifically the acute postoperative period differs in patients of low SES. Identification of such disparities will provide the foundation to improve care in this vulnerable and marginalized population. The Hamilton/Niagara region consists of a unique population given its mix of low SES with comparatively higher SES inhabitants. 16 We, therefore, sought to investigate the impact of SES on length of hospital stay (LOHS) and postoperative complications in OCSCC patients.
Materials and Methods
Study Design and Patient Cohort
This was a retrospective study intended to investigate the effect of SES on LOHS and postoperative complications in patients diagnosed with OCSCC. Consecutive patients diagnosed with OCSCC from 2010 to 2014 at a tertiary care hospital in Hamilton, Ontario, Canada, were included in this study. Inclusion criteria included age >18 years old, histopathological diagnosis of OCSCC, and primary surgical treatment with curative intent. Patients were excluded if they were undergoing palliative treatment or had previous head and neck oncologic surgery and/or radiotherapy. Incomplete datasets were also excluded from final statistical analysis. We received ethics approval from the Hamilton Integrated Research Ethics Board (HiREB: Approval no. 5298).
Medical chart review and data abstraction
For all included patients, we abstracted demographic, social, pathological, disease-specific, and treatment-specific data from medical charts. Further chart review was performed to determine LOHS, minor complication rates, major complication rates, and gastrostomy tube rates. 17 We used the seventh edition of the American Joint Committee on Cancer Staging to perform disease staging for all included patients. Complication rates were categorized using the Clavien-Dindo Grading System where minor complications were classified as Grade I or II (ie, conservative or medical intervention), and major complications were Grade III (ie, invasive intervention including interventional radiology, procedures, and/or operation) or higher (ie, requiring intensive care, end-organ injury, and/or death). 17 Postal codes were used to identify neighborhood-level socioeconomic variables via 2011 Canada Census Data. 18 Income quartiles were defined from groups of neighboring municipalities based on Canada Census definitions. 18
Statistical Analysis
Patients were stratified into income quartiles (Q1: lowest, Q2: second lowest, Q3: second highest, Q4: highest). Dependent variables and outcomes were analyzed in relation to SES. Descriptive statistics were used to compare patient demographics between income quartiles. Discrete variables were compared using a Chi-square test and continuous variables were compared using analysis of variance with a Bonferroni correction for multiple comparisons. Multivariable regression analysis was also used to identify a priori associations with LOHS and postoperative complications. An alpha level of <0.05 was considered statistically significant. All analyses were performed using SPSS 21.0 (SPSS Inc).
Results
In total, 174 patients met study inclusion criteria. Based on 2011 Canadian Census data income quartile definitions, we identified 74 (42.5%) patients in the lowest income quartile (Q1), 74 (42.5%) in the second lowest income quartile (Q2), 24 (13.8%) patients in the third income quartile (Q3), and 2 (1.1%) in the highest income quartile (Q4).
Patient Characteristics
All patient characteristics were defined in Table 1. There was a statistically significant difference in age between the groups (P = .041), much of this was a result of the higher relative age of patients in Q4. Analysis of Q1 to Q3 excluding Q4 found no differences in age (P = .120). Moreover, given the small sample size of the Q4 group, an overall analysis excluding Q4 was performed and found similar conclusions (Supplementary Data I).
Patient, Tumor, and Treatment Characteristics.
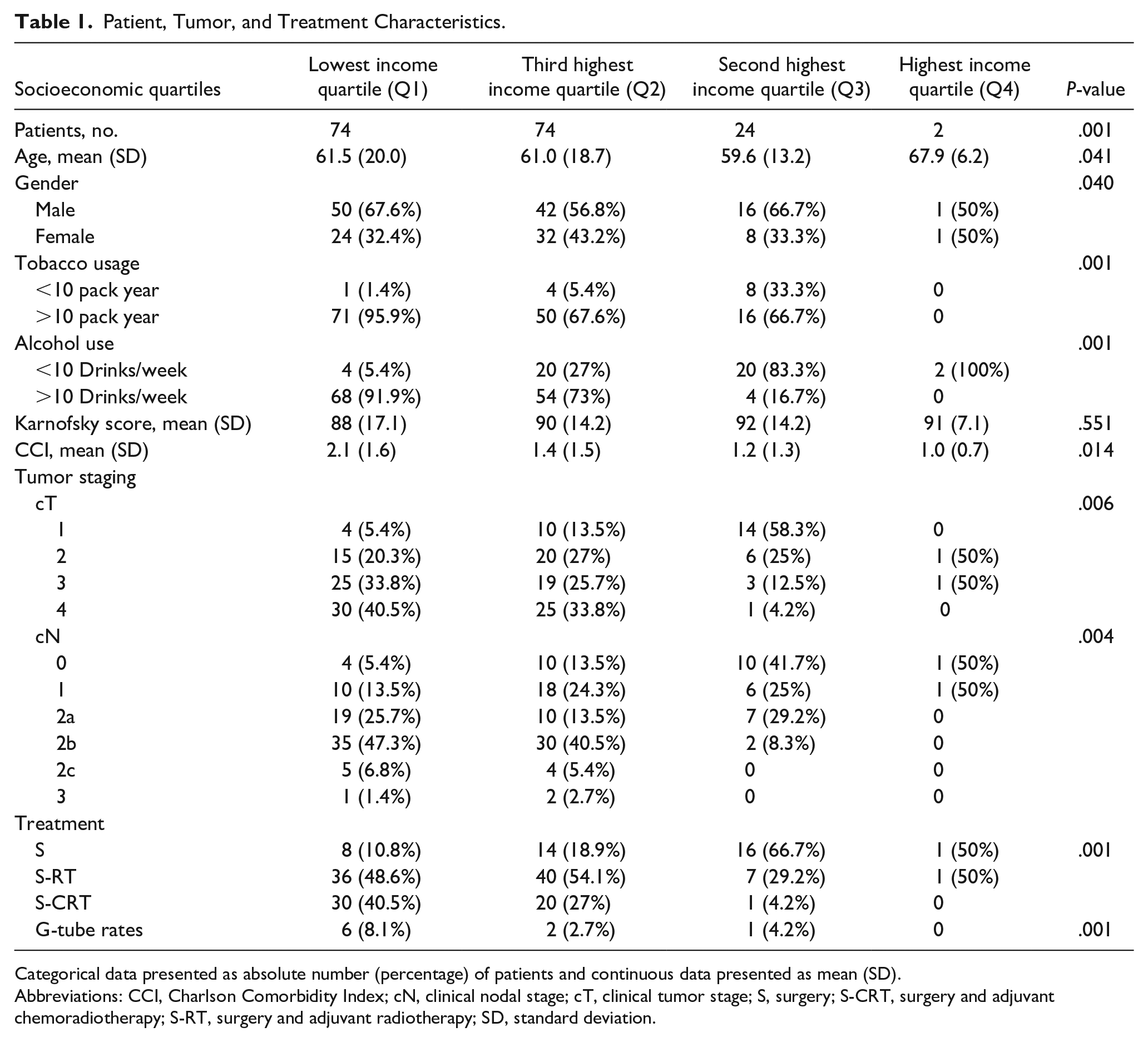
Categorical data presented as absolute number (percentage) of patients and continuous data presented as mean (SD).
Abbreviations: CCI, Charlson Comorbidity Index; cN, clinical nodal stage; cT, clinical tumor stage; S, surgery; S-CRT, surgery and adjuvant chemoradiotherapy; S-RT, surgery and adjuvant radiotherapy; SD, standard deviation.
Gender distributions varied between the groups (P = .040) with higher male predominance observed in Q1 (67.6% males) and Q3 (66.7% males), but not Q4, which had an equal 1:1 male:female ratio. There were increasing proportions of patients who reported greater than 10 pack year history (0%, 67%, 73.6%, and 95.9%, respectively) and greater than 10 alcoholic drinks (0%, 16.7%, 73.0%, and 95.9%, respectively) per week from Q4 to Q1 (P = .001 for both). Charlson Comorbidity Index (CCI) is a measure for prognostic comorbidity and predictive for 10 year survival; CCI progressively increased with higher degrees of neighborhood socioeconomic deprivation (P = .014). No statistically significant differences were observed in functional outcome between all groups as per Karnofsky score (P = .551).
Disease and Treatment Factors
Tumor and treatment modality are shown in Table 1. Statistically significant differences in local (P = .006) and regional (P = .004) disease at presentation were observed between the groups. In terms of clinical T stage, there was stepwise increase in proportion of T4 OCSCC from Q4 to Q1 (0%, 4.2%, 33.8%, 40.5%, respectively) and T3 OCSCC from Q3 to Q1 (12.5%, 25.7%, 33.8%, respectively), and reflective decrease in T1 OCSCC from Q3 to Q1 (58.3%, 13.5%, 5.4%, respectively). No trend was observed in the patients with T2 tumors. Breakdown of clinical N stage shows that there was a decrease in N0 (50%, 41.7%, 13.5%, 5.4%, respectively) and N1 (50%, 25%, 24.3%, 13.5%, respectively) disease with lower SES. Only the lowest 2 income quartiles (Q1 and Q2) had patients with N2c or N3 disease. N2b disease increased with lower SES (0%, 8.3%, 40.5%, 47.3%, respectively). Interestingly, no discernible pattern was seen in N2a disease and Q3 had the highest proportion at 29.2%. However, the overall trend suggests that lower SES is associated with higher clinical T and N stage. Reflective of more advanced disease, patients in Q1 and Q2 were less likely to be treated by primary surgery alone (10.8%, 18.9%, respectively) and were more likely to receive adjuvant radiation (48.6%, 54.1%, respectively) or chemotherapy (40.5%, 27%, respectively) (P = .001). G tube rates also increased with lower SES (0%, 4.2%, 2.7%, 8.1% from Q4 to Q1) (P = .001) reflective of more advanced disease and higher proportion of patients planned for adjuvant radiotherapy.
Socioeconomic Factors
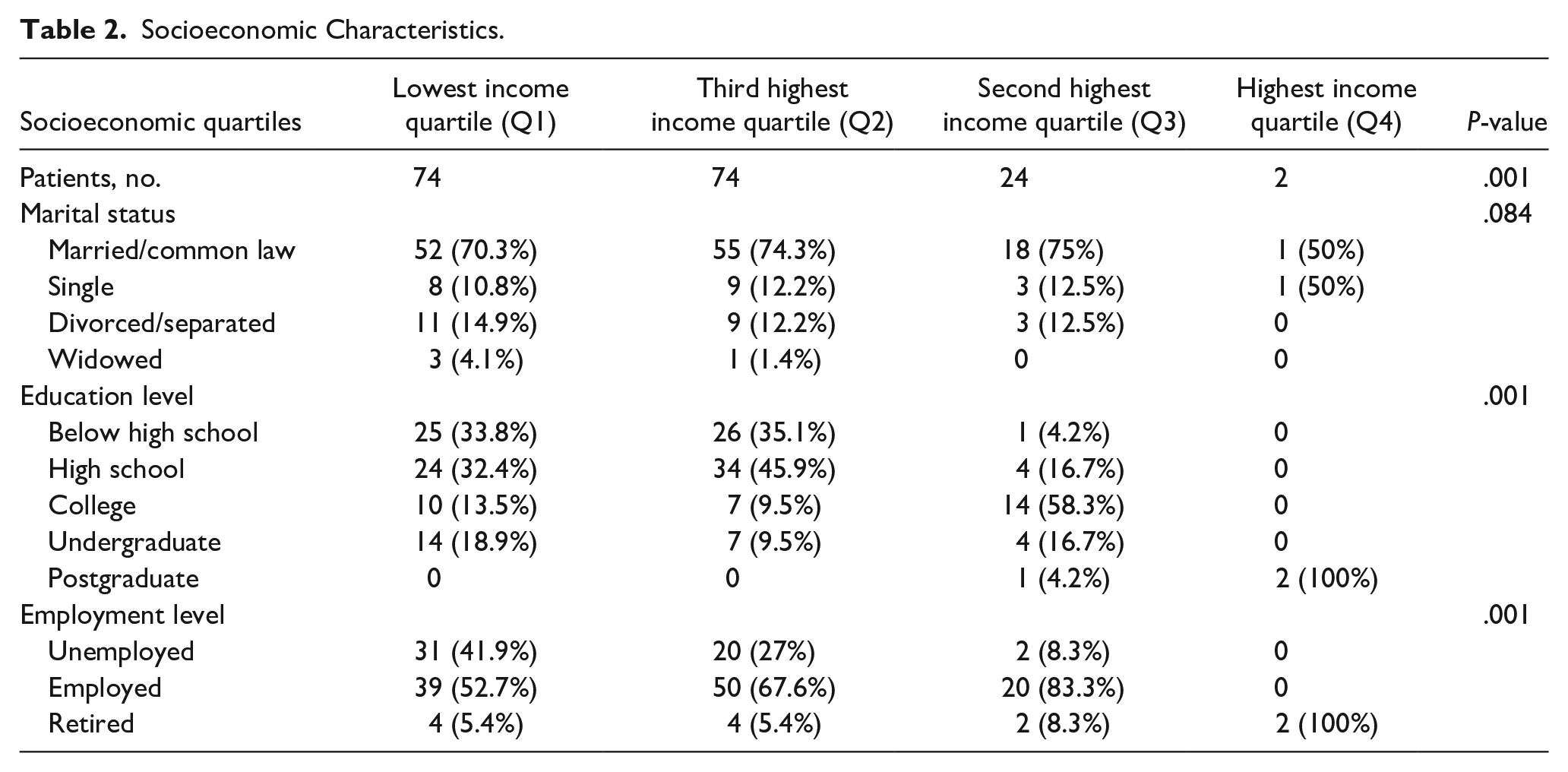
Socioeconomic factors are shown in Table 2. Patients in lower quartiles, Q1 and Q2, were less likely to have an education level beyond high school (P = .001). There was increasing levels of unemployment from Q4 to Q1 (0%, 8.3%, 27%, and 41.9%, respectively) (P = .001), although all patients in Q4 were retired. No statistically significant differences were observed between the groups in terms of marital status (P = .084) and within Q1 to Q3 approximately 70% of patients were married/common-law.
Socioeconomic Characteristics.
LOHS and Postoperative Complications
LOHS and complications are shown in Table 3. Mean LOHS had an exposure response relationship with socioeconomic depravation; from Q4 to Q1, LOHS increased from 7.5, 14.2, 18.4, to 19.4 days (P = .003). Similar trends were observed in rates of major and minor complication rates. Major complications were observed to be 0%, 4.2%, 2.7%, and 10.8% from Q4 to Q1, respectively (P = .007). Minor complications were observed to be 0%, 4.2%, 10.8%, and 18.9% from Q4 to Q1, respectively (P = .001). Major complications included free flap failure (n = 6), wound infection (n = 3), acute hematoma (n = 1), dehiscence with hardware exposure (n = 1), and wound dehiscence (n = 1): all of which required surgical intervention. Minor complications included atrial fibrillation (n = 3), hypotension (n = 1), first degree atrioventricular block (n = 1), pulmonary edema (n = 1), deep venous thrombosis (n = 2), Clostridium difficile (n = 1), wound dehiscence (n = 6), bleeding/hematoma (n = 2), and wound infection (n = 6) which required observation, nonurgent medical intervention, or nonoperative wound care. No medical complications required urgent or invasive interventions.
Postoperative Complications and Length of Hospital Stay.

Minor Complication (Grade I or II); Major Complication (Grade III or higher). 17
Abbreviation: SD, standard deviation.
A multivariable regression analysis (Table 4) identified lowest neighborhood level SES [β: 2.501 (95% confidence interval (CI) 0.200, 3.168); P = .014], tobacco usage [β 1.891 (95% CI 1.014, 2.427); P = .001], alcohol usage [β 1.373 (95% CI 0.021, 1.498); P = .004], clinical T stage [β 1.436 (95% CI 1.013, 2.178); P = .01], clinical N stage [β 1.248 (95% CI 1.076, 2.199); P = .012], CCI [β 1.043 (95% CI 0.851, 1.104); P = .001], and adjuvant therapy [β 2.876 (95% CI 0.044, 1.427); P = .0001] as predictors of complications. A multivariable regression analysis (Table 4) identified the same factors including lowest neighborhood level SES [β 2.028 (95% CI 1.062, 2.993); P = .0001], tobacco usage [β: 3.981 (95% CI 2.145, 5.432); P = .0001], alcohol usage [β 0.876 (95% CI 0.614, 1.087); P = .003], clinical tumor stage [β: 2.018 (95% CI 1.083, 2.998); P = .0001], clinical nodal stage [β: 1.053 (95% CI 0.650, 1.456); P = .0001], CCI [β 1.014 (95% CI 0.841, 1.542); P = .04], and adjuvant therapy [β: 4.788 (95% CI 1.063, 6.461); P = .0001] to be statistically significant predictors of LOHS.
Multivariable Regression Analysis of Complications and Length of Hospital Stay.
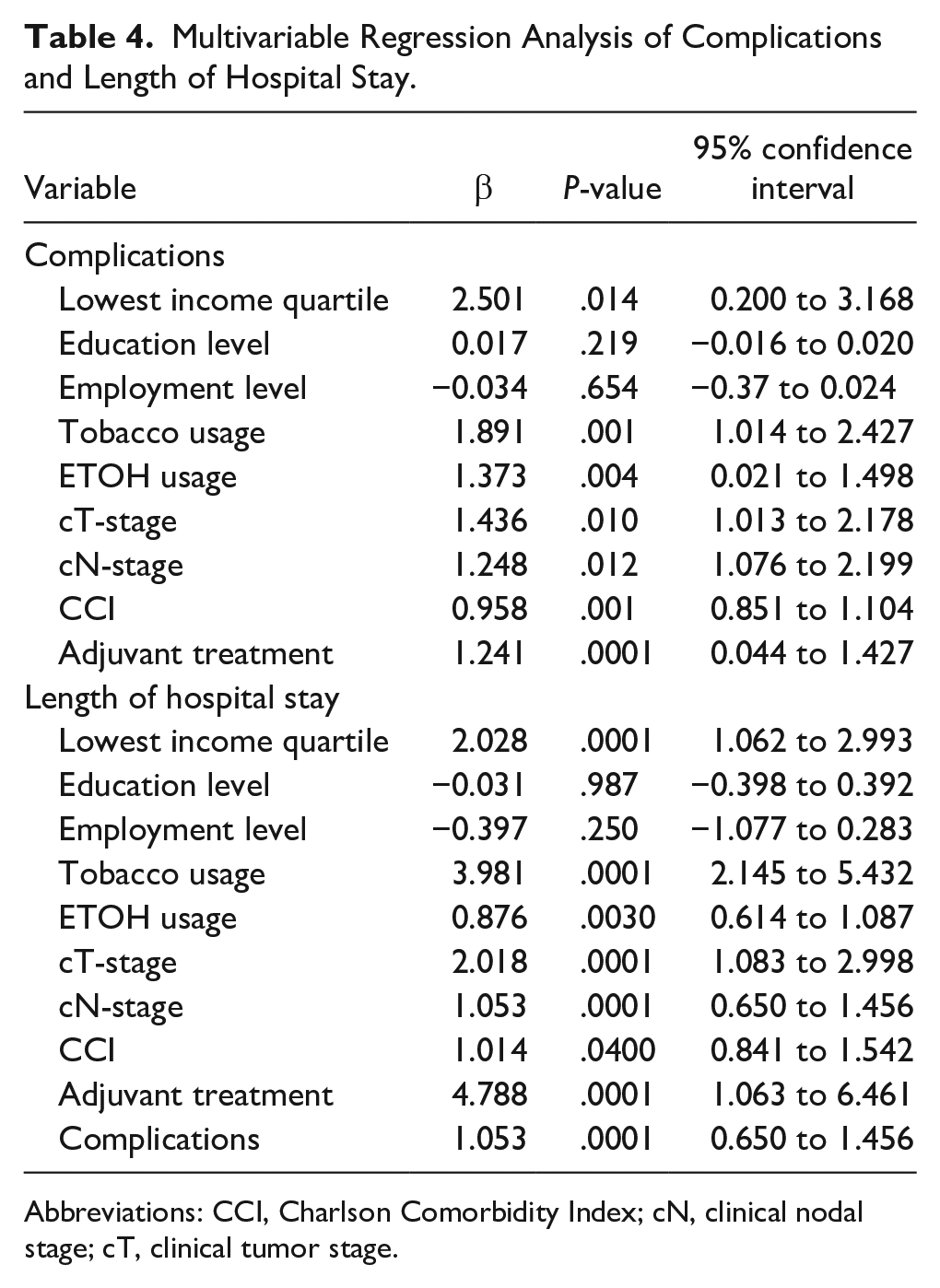
Abbreviations: CCI, Charlson Comorbidity Index; cN, clinical nodal stage; cT, clinical tumor stage.
Discussion
Socioeconomic deprivation is associated with decreased educational status.19,20 These factors are both associated with increased rates of tobacco and alcohol use, which can both lead to advanced stage cancer presentation.19,20 Previous studies have identified the detrimental effects of low SES on OCSCC incidence and survival outcomes, but the factors that lead to this decline in survival have yet to be delineated.7-15
We demonstrate that patients living in neighborhoods with low SES had higher proportions of tobacco use and alcohol use, in addition to reduced education levels and heightened unemployment rates. Similar to this study, Bonevski et al 21 found increased rates of tobacco and alcohol use in patients who lived in low SES areas and those with decreased education and employment. Moreover, we found that low SES patients had higher clinical tumor and nodal staging at time of diagnosis along with increased CCIs. Other studies have also observed an association between advanced stage OCSCC and socioeconomic deprivation, medical comorbidities, and less education.4,9,22 Patients with low SES are less likely to seek health services and cancer screening due to concerns with financial cost and misconceptions about health and disease screening.23,24 Hence, advanced stage disease appears to be a product of environmental, hereditary, lifestyle, and behavioral factors.
In Canada, low SES negatively affects access to primary care, thereby contributing to delays in diagnosis and possibly an increased rate of advanced disease on initial presentation. 25 We also found that the increased rates of advanced disease were reflected in the high need for adjuvant treatment and gastrostomy dependence. It should be noted, however, that the association between low SES and advance stage disease is controversial.4,26 This may be a result of heterogeneity in how SES is defined within the medical literature and geographical variation in the context of centrally based rather than distributed cancer care. 27
The current study results demonstrate that major and minor complication rates increased alongside decreasing SES while LOHS increased with social deprivation in OCSCC. Similar to previous studies, poor clinical outcomes in low SES patients are partially due to advanced stage disease presentation and have high rates of medical comorbidities and lifestyle risk factors.28-33 Interestingly, smoking and alcohol use have not been consistently identified in other multivariable analyses as independent predictors of postoperative complications and LOHS, and studies of these risk factors did not stratify patients by SES.28-31 Tobacco and alcohol use may have stronger impacts in low SES patients with OCSCC, but this notion will require validation with future studies.
Tobacco and alcohol use are well-established risk factors in HNC.32-36 Smoking and alcohol also increase the risk of advanced stage disease at presentation in OCSCC and can adversely affect postoperative healing.37-40 We found that OCSCC patients with low SES report more tobacco use prior to treatment when compared to higher SES patients. Although recommending preoperative smoking cessation is routine in HNC patients, Simmons et al 41 suggest that postoperative smoking relapse can be as high as 60% in patients who smoked 1 week prior to surgery. Smoking and alcohol interfere with wound healing and increase susceptibility to postoperative infection, and despite cessation, chronic tobacco exposure can continue to negatively affect wound healing.42,43 Previous studies, however, have found that controlling for smoking and alcohol use does not wholly account for the relationship between low SES and OCSCC.5,6
We found that low SES OCSCC patients had a high number of comorbidities as measured by CCI. Unsurprisingly, multiple studies have shown that increased rates of medical comorbidity lead to more postoperative complications in head and neck surgery.30,32-44 Moreover, studies of both HNC and general patient populations demonstrate that the high medical complexity in low SES patients may reflect the interfacing effects of lifestyle factors, alcohol and tobacco use, poor nutrition, and mental health disorders.45,46
Cramer et al 47 identified that HNC patients with advanced disease requiring moderate or major head and neck surgery, severe medical comorbidities, and postoperative complications were associated with a high likelihood for discharge to post-acute care facilities following treatment. Due to the logistics of organizing post-acute care resources, nonmedical delays may be encountered which can further delay care and hospital discharge. Furthermore, Lang et al 48 found that head and neck free flap patients often experience nonmedical delays due to staffing and resource challenges. In patients who develop postoperative complications, but are eventually discharged, additional outpatient resources are required (eg, wound packing, venous access catheters, and/or or tracheostomy and gastrostomy tube care). According to the Canadian Institute for Health Information, the median wait time for homecare services is 4 days in Ontario, and in patients who experience discharge delays, the median LOHS was extended by up to 9 days. 49 Despite the universal healthcare in Canada, our study suggests that OCSCC patients with low SES are exposed to risk factors that increase the likelihood of delayed discharge, thereby further straining institutional and system-wide healthcare resources.
Study limitations include the retrospective nature of data collection and small sample size in the Q4 group. However, statistical analysis excluding the Q4 group did not alter our results (Supplementary Data I). Despite the ecological fallacy which suggests that neighborhood SES may not adequately reflect individual SES, our results suggest that our study cohort’s education, employment, tobacco use, and disease stage at presentation is like other studies, thereby suggesting that in this study, neighborhood SES is a reasonable surrogate for overall patient SES. Perioperative optimization and intraoperative variables such as time under anesthesia, which may influence postoperative outcomes, were not controlled for in this study nor were sequelae of postoperative complications or LOHS. We also did not capture hospital readmission rates or details regarding nonmedical discharge delays or destination due to the lack of communication between electronic medical systems at community hospitals and community care networks. All these factors may contribute to quality of care in OCSCC and are fertile routes of investigation for future studies.
Conclusion
In summary, we found that low SES patients diagnosed with OCSCC demonstrate high tobacco and alcohol use and an increase in postoperative complications and LOHS. Behavioral lifestyle factors may negatively affect healing and immunity in the postoperative period following treatment for OCSCC. Low SES patients diagnosed with OCSCC are at a heightened risk for worse short-term postoperative outcomes and, therefore, may benefit from enhanced financial and nutritional support, in addition to alcohol and smoking cessation counseling to improve treatment outcomes and disease prognosis.
Supplemental Material
sj-docx-1-ear-10.1177_01455613241253146 – Supplemental material for Socioeconomic Status, Length of Stay, and Postoperative Complications in Oral Cavity Squamous Cell Carcinoma
Supplemental material, sj-docx-1-ear-10.1177_01455613241253146 for Socioeconomic Status, Length of Stay, and Postoperative Complications in Oral Cavity Squamous Cell Carcinoma by Michael Xie, Phillip Staibano, Michael K. Gupta, Nhu Tram Nguyen, Stuart D. Archibald, Bernard Stanley Jackson, James Edward Massey Young and Han Zhang in Ear, Nose & Throat Journal
Footnotes
Acknowledgements
Not applicable.
Authors’ Note
Presentation: This study was presented by Michael Xie at the AHNS Annual Meeting in Austin, Texas, on May 2, 2019. Submission: This material has never been published and is not currently under evaluation in any other peer-reviewed publication.
Author Contributions
M.X. was involved in the acquisition, analysis, and interpretation the data and was a major contributor in writing the manuscript. M.K.G. was involved in the conception and design, acquired and interpreted the data and was a major contributor in reviewing the manuscript. N.T.N. was involved in the analysis and interpretation of the data. S.D.A. was involved in conception and design, acquisition of data, and interpretation of data. B.S.J. was involved in conception and design, acquisition of data, and interpretation of data. J.E.M.Y. was involved in conception and design, acquisition of data, and interpretation of data. H.Z. was involved in the conception and design, acquired, analyzed, and interpreted the data and was a major contributor in writing the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. NB: The corresponding author has full access to the data in this study and takes responsibility for the integrity of the data and accuracy of the data analysis.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This study was approved by the Hamilton Integrated Research Ethics Board (HiREB). Consent for study participation was obtained from patients recruited to participate in the study. HiREB project number 5298.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
