Abstract
Neuroendocrine neoplasms (NENs) represent a collection of highly varied tumors that originate from neuroendocrine cells. They are considered rare tumors that predominantly affect the lungs. Epithelial NENs can be categorized into neuroendocrine tumors (NETs) and neuroendocrine carcinomas. It is extremely rare for NET grade 1 (NET G1) to exist in the nasopharynx, these tumors are slow-growing and the onset of symptoms and identification of the tumor may take several years. The majority of cases occur in ages between 60 and 65 years. In this article, we present a case of a 26-year-old female who presented with recurrent epistaxis and nasal obstruction for one and a half years. Magnetic resonance imaging revealed a substantial nasopharyngeal mass. Subsequently, a biopsy was conducted, and the histopathological results indicated a NET G1. Our literature review revealed 5 cases of NET G1 in the nasopharynx, with our patient being the youngest among all published cases.
Introduction
Neuroendocrine neoplasms (NENs) comprise a diverse group of tumors that originate from neuroendocrine cells and predominantly affect the lungs. Their incidence is approximately 7 cases per 100,000 people in the United States. 1 Epithelial NENs can be categorized into well-differentiated NENs [neuroendocrine tumors (NETs)] and poorly differentiated NENs [neuroendocrine carcinomas (NECs)]. NETs grade 1 (NETs G1) were previously described as typical carcinoid tumors or well-differentiated NECs. 2 NET G1 is rare especially in the head and neck area, with the majority of cases occurring in individuals aged between 60 and 65 years. 3 Various names are used to refer to these neoplasms, making a comprehensive literature review challenging. Here, we present a case of NET G1 in the nasopharynx, in a 26-year-old female. This diagnosis is exceptionally rare, with only 5 cases of NET G1 in the nasopharynx in our literature review.3-7 This report aims to shed light on this rare entity and alerts the importance of not underestimating mild symptoms, as they may be indicative of serious diseases.
Case Presentation
A 26-year-old female patient with an unremarkable past medical and surgical history presented to our department with complaints of recurrent epistaxis and nasal obstruction over the last one and a half years. She also reported symptoms of nasal discharge, snoring, headaches, shortness of breath, and hoarseness of voice. On examination, a mass was observed in the nasopharynx.
Magnetic resonance imaging (MRI) of the neck revealed an enhancing soft tissue lesion centered at the right nasopharyngeal fossa, invading the skull base and the right temporal lobe with a pathological-looking enlarged ipsilateral cervical lymph node (Figure 1A-F).
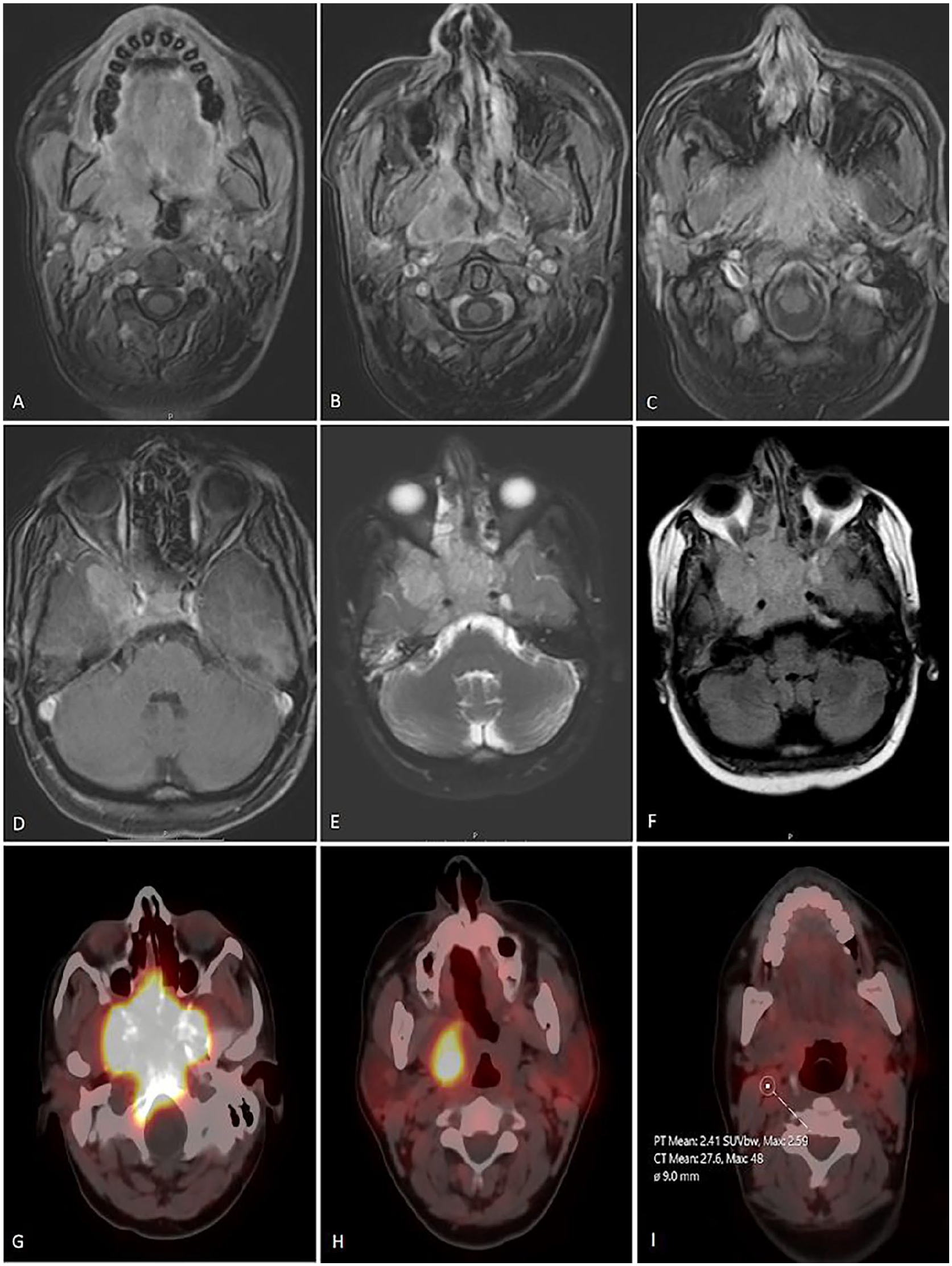
(A, B, C, and D) Axial T1-weighted post-contrast MR images of the nasopharynx showing an enhancing soft tissue mass centered at the right nasopharyngeal cavity and obliterating the right fossa of Rosenmüller. The tumor exhibits relatively homogeneous moderate contrast enhancement. The mass hyperintense in T2 and FLAIR images (E and F, respectively) enhance intensely after contrast. The mass is noted invading the clivus and the right temporal lobe superiorly (T4 tumor according to TNM staging). Multiple ipsilateral pathological lymph nodes are found (not demonstrated in the selected images). (G, H, and I) Gallium-DOTATATE PET CT scan demonstrating DOTATATE-positive large soft tissue mass lesion in upper nasopharynx invading the skull base. In addition, DOTATATE-positive right upper cervical parapharyngeal lymph node (level IIA). MR, magnetic resonance; PET, positron emission tomography; CT, computed tomography; FLAIR, Fluid-attenuated inversion recovery.
A biopsy was performed, and it showed that the tumor cells were composed of a single population of small, round to oval-shaped cells with inconspicuous nucleoli, scant cytoplasm, and a distinctive “salt and pepper” chromatin pattern. The tumor cells were arranged in cords (Figure 2A). The immunohistochemistry stains showed positive results for chromogranin A, pan-cytokeratin (pan-CK), synaptophysin, and neuron-specific enolase (NSE); however, the results were negative for TTF-1. The Ki-67 labeling index was less than 2%, and no mitotic activity was found. The histopathologic findings are consistent with NET G1 (Figure 2B-E).
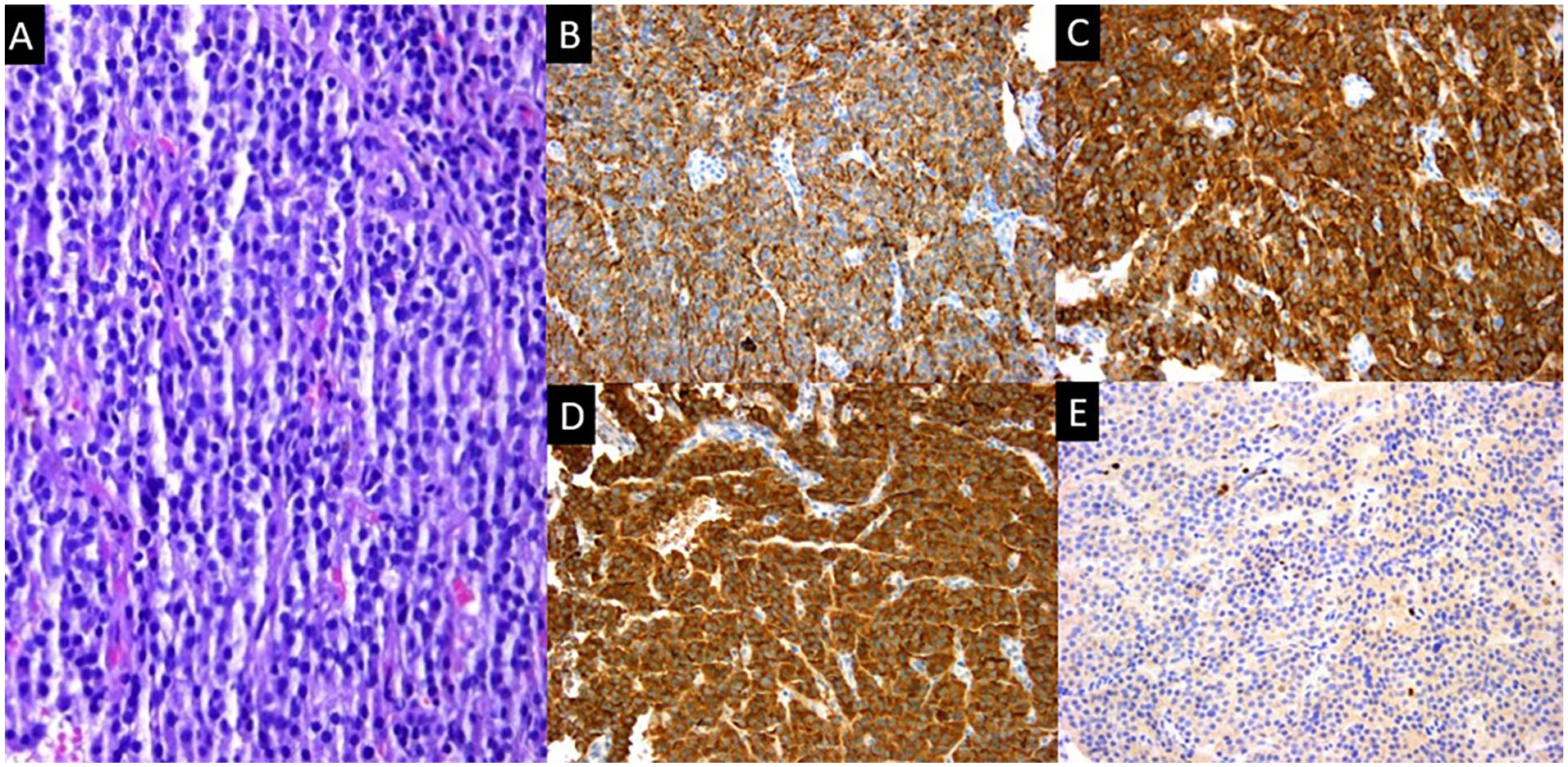
Hematoxylin and eosin (H&E) staining shows that the tumor cells are composed of a single population of small, round to oval-shaped cells with inconspicuous nucleoli, scant cytoplasm, and a distinctive “salt and pepper” chromatin pattern. These cells are arranged in cords (magnification, ×20) (A). The immunohistochemical studies demonstrate positive immunostaining of tumor cells for (B) pan-CK, (C) synaptophysin, and (D) chromogranin A. (E) The Ki-67 index is <10%. (H&E staining; magnification, ×20). pan-CK, pan-cytokeratin.
A Gallium-DOTATATE PET/CT (positron emission tomography/computed tomography) scan was done for staging; the nasopharyngeal mass and the ipsilateral cervical lymph nodes demonstrated positive high DOTATATE uptake with no evidence of distant metastatic hypermetabolic activity (Figure 1G-I).
The case was discussed in the head and neck multidisciplinary team, and since the mass was unresectable, the decision was made to treat the patient with palliative radiation and octreotide. The patient may benefit later from peptide receptor radionuclide therapy once the disease progresses on octreotide, with chemotherapy considered as subsequent therapy.
Discussion
NENs of the aerodigestive tract can be separated into pulmonary NENs and extrapulmonary NENs (EPNENs) based on where they are located. Aerodigestive NENs mostly affect the lungs, with EPNENs making up 9% of all cases. However, they are rare in the nasopharyngeal cavity—3% of sinonasal tumors and 0.1% of all cancer cases. 1 The 2022 World Health Organization (WHO) classification of epithelial NENs of the head and neck into 2 categories based on their morphologic differentiation status: well-differentiated NENs (NETs) which are divided into 3 grades based on proliferative features, and poorly differentiated NENs (NECs) which are divided into small and/or large cell subtypes. In the context of NET G1, characteristic histopathologic criteria include the absence of necrosis, a proliferation rate of less than 2 mitoses per 2 mm2, and a Ki67 index below 20%. 2 NET G1 of the head and neck are exceedingly rare, with the larynx being the most frequently affected area. 7 The aggressiveness of these NETs in the nasopharynx is not well understood. 5 There was no gender variation in its prevalence.1,8 The majority of the symptoms of NETs such as nasal congestion, runny nose, hyposmia, and headaches are compressive and are rarely accompanied by lymph nodes or distant metastases. 8 In addition, these tumors grow slowly, it could take several years for symptoms to manifest and for the tumor to be identified. 6
As far as we are aware, there have only been 5 cases of NET G1 in the nasopharynx documented in the literature (Table 1). The patient in our case was the youngest one among the previously published cases.
A Summary of the Characteristics of Worldwide 5 Cases and Our Case of Well-Differentiated Neuroendocrine Tumors in the Nasopharynx That Have Been Published Previously.
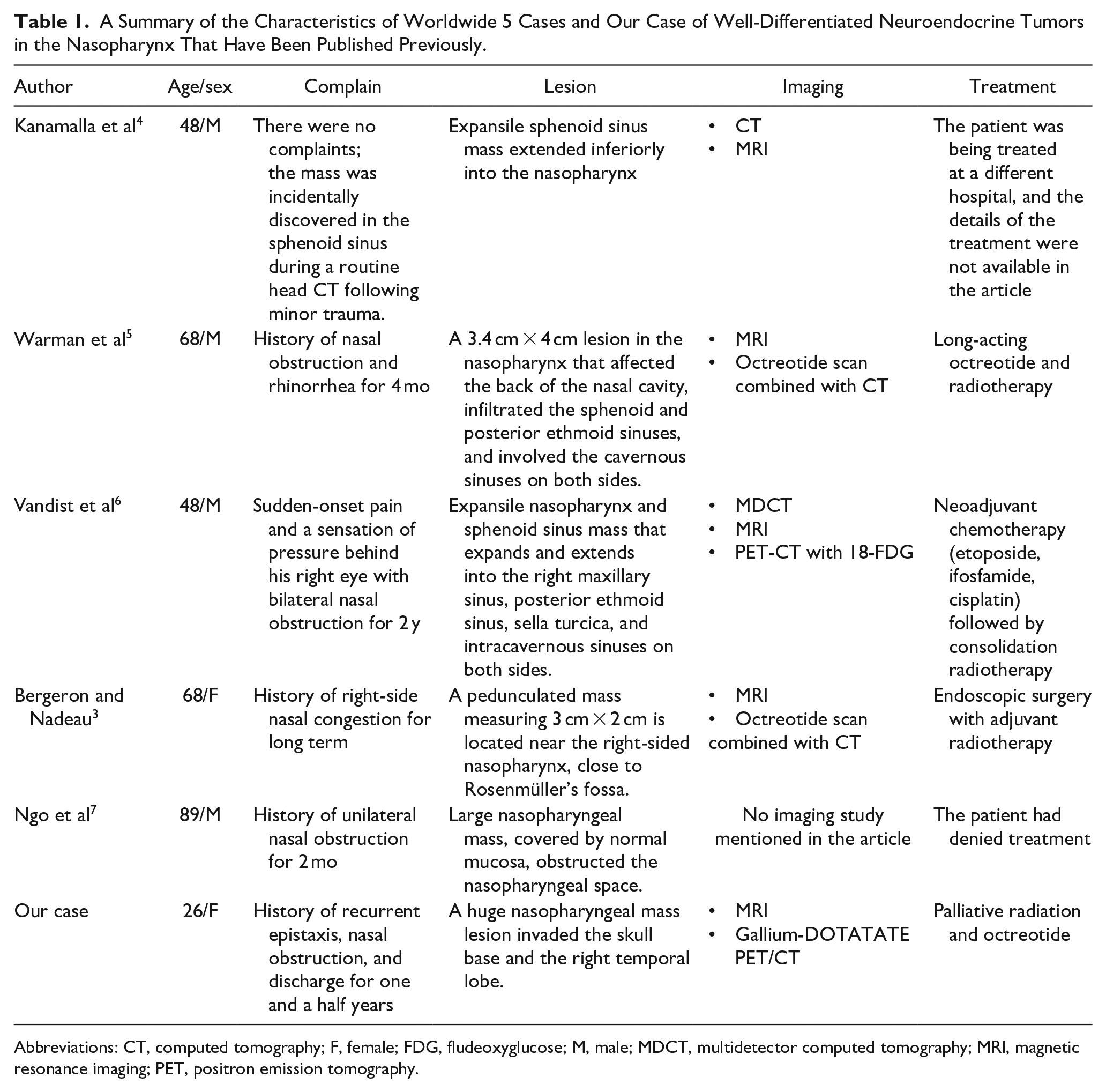
Abbreviations: CT, computed tomography; F, female; FDG, fludeoxyglucose; M, male; MDCT, multidetector computed tomography; MRI, magnetic resonance imaging; PET, positron emission tomography.
Both CT and MRI are used to precisely locate and measure tumors and monitor treatment response. 6 NETs were diagnosed through histopathological findings obtained from biopsy.7,8 When making a differential diagnosis of NETs, pathologists must consider both immunohistochemical and histopathological findings. 7 Previous studies have indicated that synaptophysin, NSE, CD56, chromogranin or neural cell adhesion molecule (NCAM), and AE1/AE3 are immunohistochemically positive markers for NETs.3,7,9 Somatostatin receptor expression in NETs can be shown by utilizing a radionuclide coupled to an analog of somatostatin, such as octreotide. The entire body can be scanned to detect metastatic disease, this method is very specific and sensitive for these tumors. 6
A standard strategy for the successful treatment of NET G1 in the nasopharynx has not yet been established due to the small number of cases. 7 Surgery is still the first line of treatment to resect the tumor completely.5,7 A somatostatin analog such as octreotide should be used as the first step in therapy for somatostatin receptor scintigraphy-positive tumors if curative surgery is not possible. 5 Utilizing an octreotide scan can be beneficial, as studies indicate that 20% to 25% of tumors exhibit a size reduction when treated with radioactive octreotide derivatives. 6 Previous cases of NET G1 have been treated with radiation and chemotherapy, but the effectiveness of those treatments is yet unknown. 7
Conclusion
NET G1 of the nasopharynx is extremely rare. The medical literature has only described 5 case reports before this case. This article tries to provide more information about this rare entity and should be considered in the differential diagnosis of a history of mild symptoms in the nasopharyngeal region because they could be signs of more serious illnesses. Further studies are required to develop optimal treatments aiming to improve patient prognosis for this rare disease.
Footnotes
Acknowledgements
The authors thank Polytechnic Medical Students’ Research Association—PMRA for their invaluable input and support throughout the research process.
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Statement
Ethical approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Patient Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
