Abstract
Introduction
Neoplasms in the parotid gland are often nonmalignant tumors, where about 60% to 80% of cases are considered benign. Although pleomorphic adenomas make up the majority of parotid gland neoplasms, Warthin’s tumors are the second most common benign tumors of the parotid gland, making up 15% of all epithelial tumors in the gland.1-3 Warthin’s tumors are histologically described as concentrated lymphoid stroma with architectural arrangements of both papillary and cystic structures.2,4 These neoplasms typically reside in the superficial lobe and inferior pole of the parotid gland, where roughly 15% of Warthin’s tumors display multifocal patterns within a single gland, while bilateral occurrences are noted in approximately 10% of cases.4,5
It is important to note that the incidence of Warthin’s tumors has been increasing in recent years, which can be attributed to improved diagnostic tools and imaging sensitivity. 6 Sometimes, Warthin’s tumors can be found incidentally and mimic cancer metastatic. Instances of parotid incidentalomas identified through fluorodeoxyglucose positron emission tomography (FDG-PET/CT) have been documented, potentially leading to misdiagnosis of malignancy.7,8 Nevertheless, Warthin’s tumors also exhibit significant FDG uptake, and their diagnosis can be confirmed through fine needle aspiration cytology (FNAC) or core needle biopsy (CNB).8,9 Both FNAC and CNB are accurate methods for diagnosing benign salivary tumors before patients undergo invasive procedures, a critical consideration for those with comorbidities who are unable to undergo general anesthesia or interventional operations.8,9 One study highlighted the higher sensitivity and specificity in CNB compared to FNAC for diagnosing benign masses in the salivary glands. 9
Although the exact etiology of these neoplasms is unknown, Warthin’s tumors typically present in middle-aged or elderly male patients who have a history of smoking.5,10 The rise in cases of Warthin’s tumors in older age groups may be attributed to an increase in smoking and body mass index (BMI) among patients.6,11 A study proposed that patients diagnosed with Warthin’s tumors exhibit higher BMI compared to individuals with other benign masses in the parotid gland. 11
Common methods in treating Warthin’s tumors include superficial or partial superficial parotidectomies. Other techniques include extracapsular or capsular dissection, microwave ablation, ultrasound-guided ethanol sclerotherapy, and radiofrequency ablation (RFA).5,12,13 Extensive surgeries involved in removing Warthin’s tumors have been associated with various adverse effects, including hematomas, seromas, facial nerve palsy, infection, Frey’s syndrome, and salivary fistula.13,14 One study examining postoperative complications following parotid gland study in 132 patients found that the rate of immediate facial paralysis was 53.8%, 43.8%, 41.5%, and 6.3% for extended parotidectomy, superficial parotidectomy, partial superficial parotidectomy, and extracapsular dissection, respectively. 15
The decision to pursue surgery is generally cosmetic. There are some patients who are averse to surgery or may have a medical history whereby general anesthesia risks outweigh the benefits of an elective, relatively cosmetic surgery. To mitigate these complications, Warthin’s tumors can be safely observed. Nonsurgical considerations, such as RFA, could also be considered. RFA is used as an alternative to thyroid surgery in a subset of benign thyroid cystic disease. Minimally invasive techniques may be similarly suitable for addressing Warthin’s tumors due to their gradual development, characterized by an annual growth rate of 8%, and a minimal risk of malignant transformation, which is less than 0.3%.13,16-18 RFA employs the application of heat generated by radiofrequency waves to directly eliminate or coagulate specific tissues and nerves.19-21 Coagulative necrosis in tissues occurs when temperatures exceed 50°C, with uncontrolled destruction observed at temperatures above 100°C. Consequently, the objective of RFA is to sustain a temperature range between 50°C and 100°C to ensure the accurate destruction of targeted tissues while avoiding coagulation in surrounding areas.22,23 RFA has been widely used in otolaryngology to treat benign thyroid nodules, thyroid cancers, lymphatic malformations, and tonsillar hypertrophy.20,24-26 The aim of this systematic review is to investigate the effectiveness and safety of RFA in the treatment of patients with Warthin’s tumors, while also evaluating any complications and postoperative outcomes associated with the procedure.
Methods
Study Design
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) review framework was used by 2 reviewers to identify articles for this systematic review. 27 This guideline was used in 2 medical databases, PubMed (MEDLINE) and Excerpta Medica (EMBASE). Five screening stages, which included our exclusion and inclusion criteria, were used in accordance with the PRISMA guideline. To identify articles on PubMed and EMBASE, the comprehensive search term “Warthin Tumor Treatment” was employed, which produced a total of 1299 articles. The PubMed database produced 747 articles with years ranging from 1955 to 2023 and the EMBASE database produced 552 articles with years ranging from 1973 to 2023. Figure 1 demonstrates the 5 steps associated with article identification in this systematic review. Our inclusion criteria included articles that assessed the use of RFA on the parotid gland. Our exclusion criteria encompassed various factors, such as articles not written in English, those primarily centered on non-RFA treatments, and those that did not investigate Warthin’s tumors.
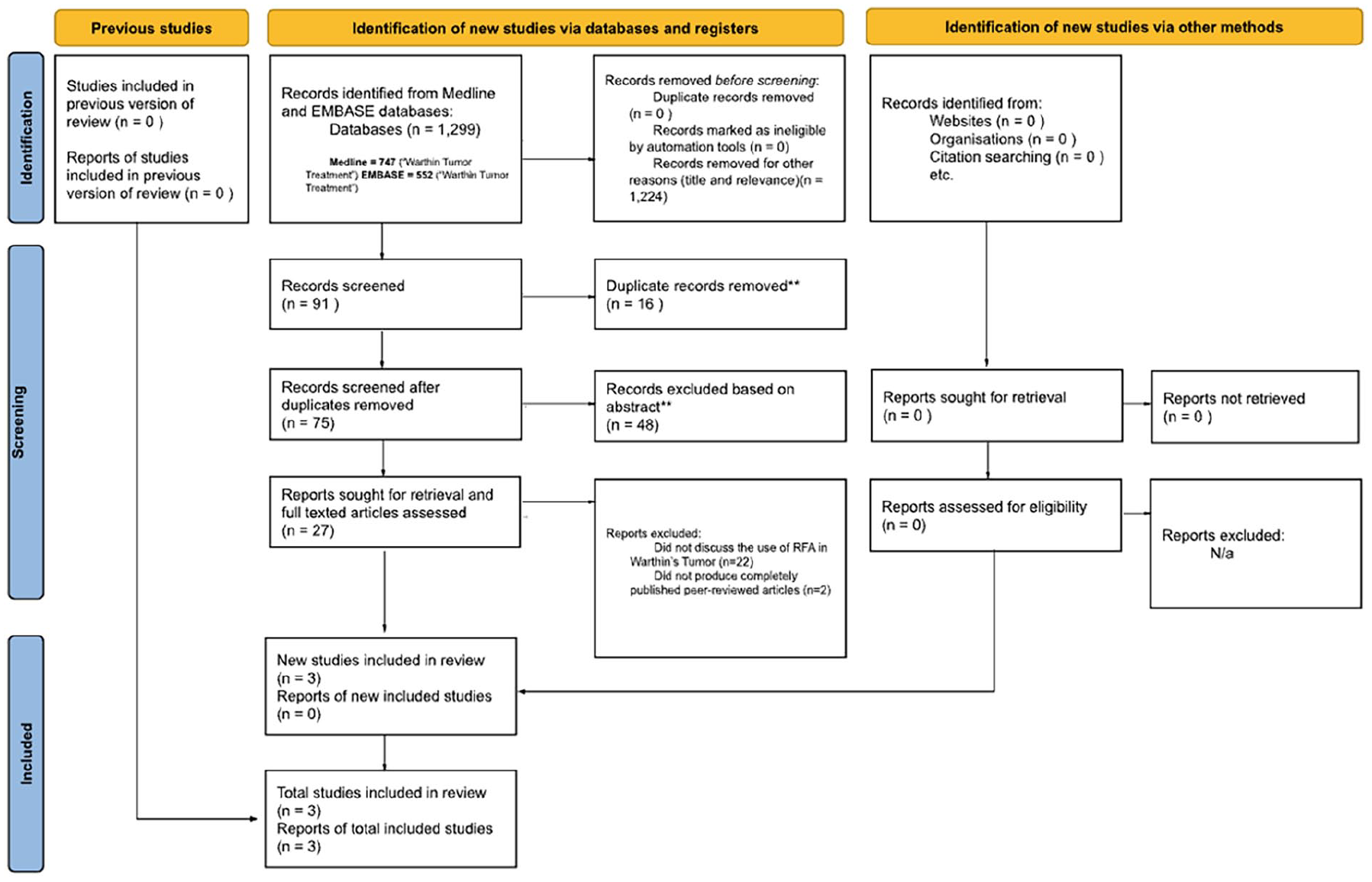
Study flow chart.
Study Selection
Step 1 of the 5 screening stages involved the use of the broad search terms to identify available articles throughout PubMed and EMBASE. Step 2 was the evaluation of titles. Step 3 was filtering the articles based on their abstracts. The remaining articles that were not excluded during Step 2 and Step 3 of the screening entered the fourth stage, which was examining the articles based on their full-length articles. Figure 1 highlights these screening stages. This systematic approach produced a total of 3 qualitative articles that fit our investigation on the treatment of Warthin’s tumors with RFA. Table 1 displays the average tumor size pre-RFA, average tumor size post-RFA, tumor size percent reduction, keen findings, cosmetic score, and more. The fifth step in Figure 1 shows articles excluded from this systematic review, which included those that did not address the applications of RFA in Warthin’s tumors, were not written in English, and were incomplete or lacking peer-reviewed status. The article screening and selection in this systematic review was independently filtered by 2 reviewers with the supervision of a senior reviewer.
RFA in Patients With Parotid Warthin’s Tumor.
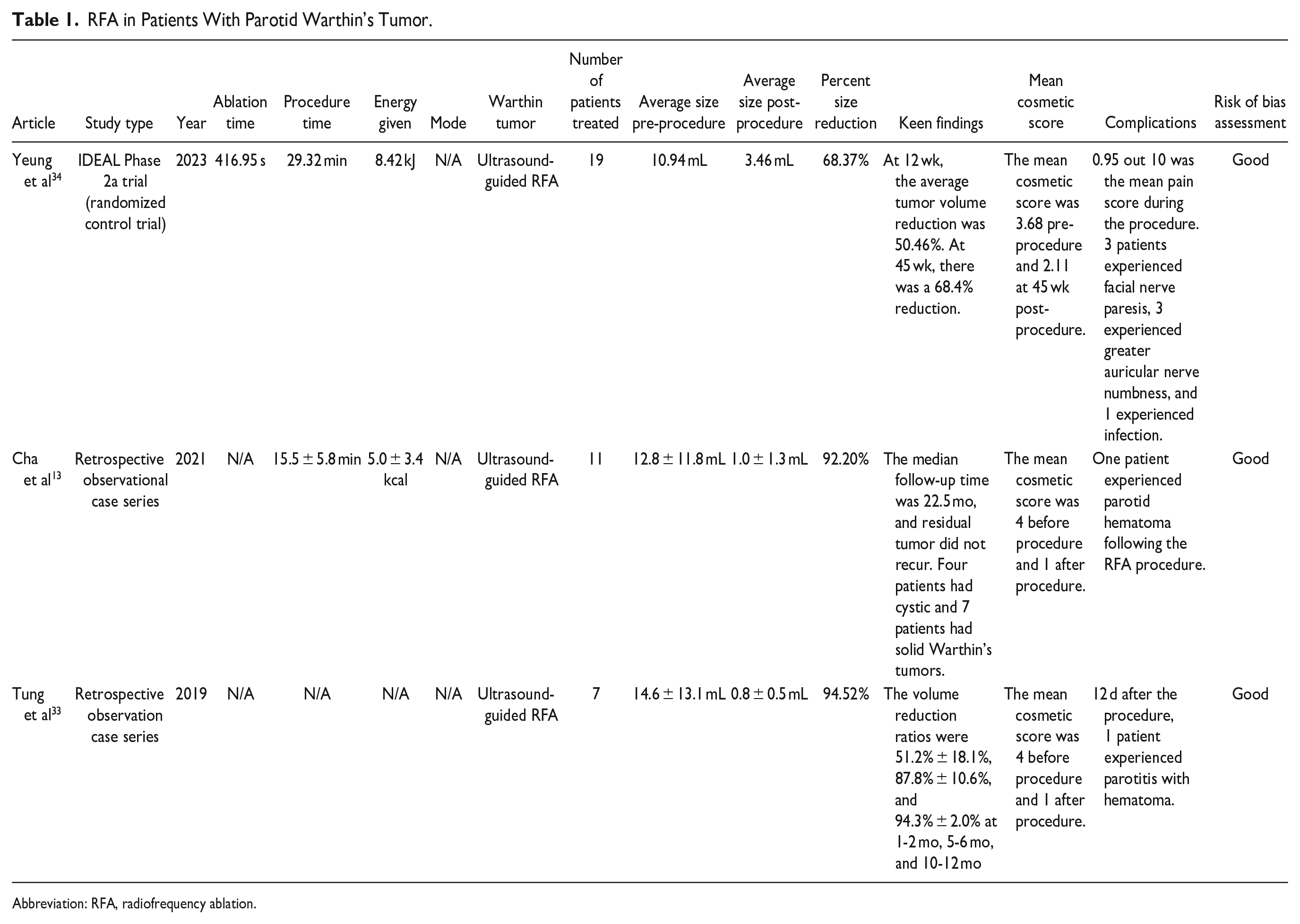
Abbreviation: RFA, radiofrequency ablation.
The investigators of this systematic review examined and aggregated the results from 3 studies that assessed the efficacy of RFA in treating Warthin’s tumors of the parotid gland. These 3 publications included 1 IDEAL Phase 2a clinical trial and 2 retrospective observational case series.
Quality Assessment and Data Abstraction
The National Institute of Health (NIH) Study Quality Assessment tool was referred by authors of this systematic review to rate the risk of bias for pre-post studies and case series. 28 Following this tool’s guidelines, pre-post studies were graded using 12 questions, where a score of 9 to 12 was considered good quality, 5 to 8 was considered fair quality, and 0 to 4 was considered poor quality. For case series, a 9-question assessment tool was used, where a score of 7 to 9 was considered good quality, 5 to 6 was considered fair quality, and 0 to 4 was considered poor quality. 28 The risk of bias and quality of these 3 studies are displayed in Tables 2 and 3.
Risk of Bias Assessment for Clinical Trials.
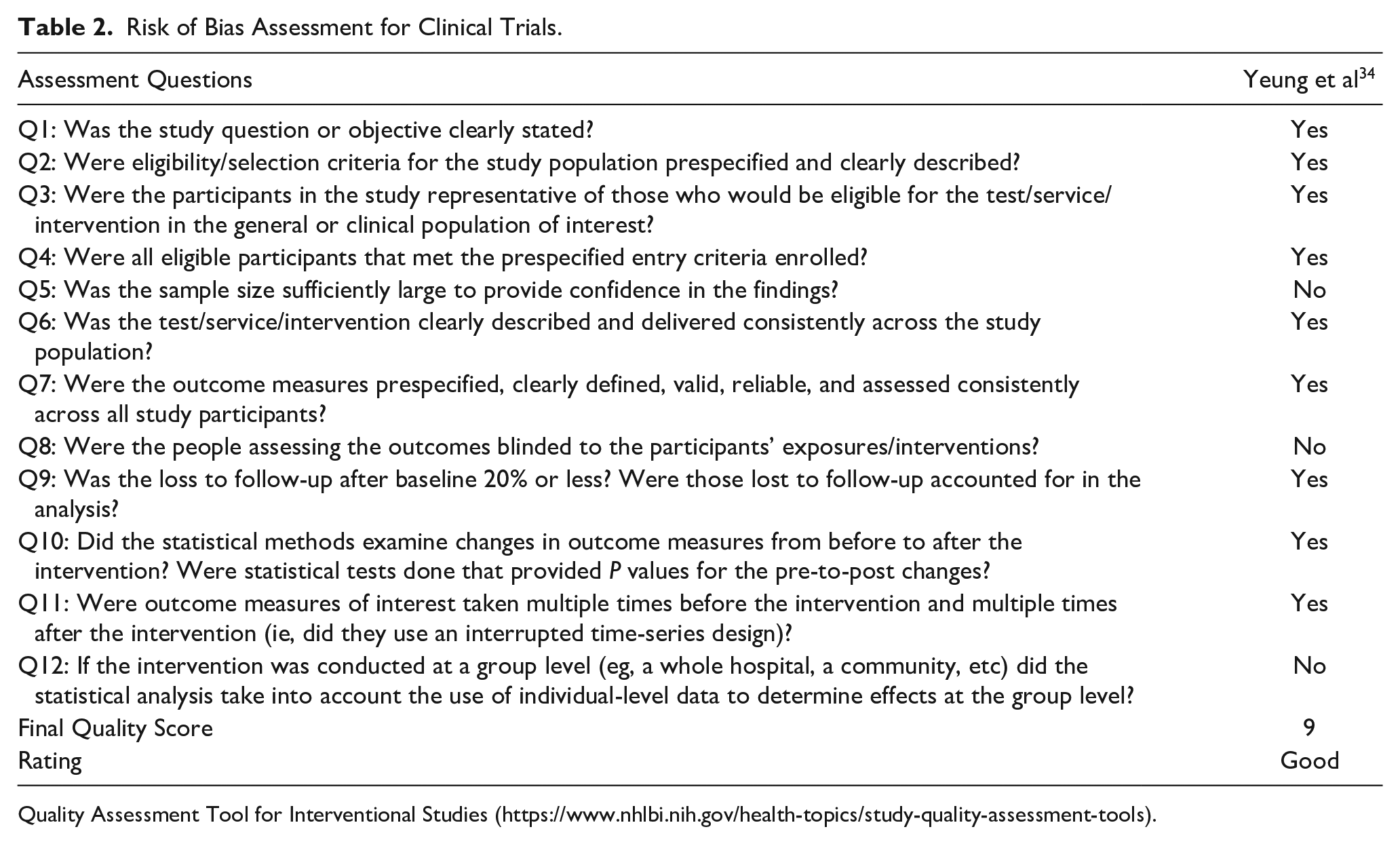
Quality Assessment Tool for Interventional Studies (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools).
Risk of Bias Assessment of Case Series.

Quality Assessment Tool for Clinical Case Series (https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools).
Definitions
RFA can be applied in a continuous or pulsatile manner. 29 In continuous RFA (CRF), high energy waves are kept at a constant rate to maintain a temperature of 60°C to 80°C necessary for tissue necrosis. 30 The temperature used in pulsed RFA is usually kept under 42°C, where thermocoagulation is typically done in multiple sessions lasting for 20 ms in intervals of 500 ms.30,31
The cosmetic score was also mentioned in the 3 studies included in this systematic review. This evaluation scale was based on the cosmetic evaluation tool used in the treatment of RFA in thyroid nodules.32,33 The scale used across the 3 articles in this systematic review rated the facial improvement, ranging from 1 to 4. A score of 1 indicated the absence of a palpable parotid tumor, 2 indicated the presence of a palpable tumor without a visible mass, 3 indicated a tumor that protruded during teeth clench, and 4 indicated a distinctly observable mass.13,32-34
Results
After the application of the PRISMA screening, 3 articles were included in this systematic review, with 1 being an IDEAL Phase 2a clinical trial and 2 being retrospective observational studies. These individual studies assessed the Warthin tumor size reduction and cosmetic scores in patients following RFA, as well as any complications associated with the procedures.
Study Screening
Our comprehensive search terms initially produced 91 articles with relevant titles. The reviewers then removed 16 duplicates. The abstracts of the remaining 75 articles were evaluated, which led to the removal of 48 publications. The final screening step involved assessing the full-length articles of the remaining 27 articles. After evaluation, only 3 studies were included in this systematic review. Throughout the screening process of titles, abstracts, and full-length articles, articles were excluded if they did not address RFA in the treatment of Warthin’s tumors, were not written in English, or were not fully published as peer-reviewed articles.
Study Quality
The NIH study quality assessment tools were used to examine the risk of bias in the 3 individual articles included in this systematic review. 28 The controlled intervention studies tool 28 was used to rate the risk of bias and quality for Yeung et al. 34 The case series tool was used to rate the risk of bias and quality for Cha et al 13 and Tung et al. 33 Two reviewers evaluated the 3 articles and determined that all of them were classified as “good quality” according to the NIH risk of bias guideline.
Study Characteristics
The 3 studies included in this systematic review examined the use of RFA in treating 37 patients with Warthin’s tumors. The average age of these patients was 62.5 years, and all of the patients were male. Most of the patients in these studies had a history of smoking as well. At approximately 12 months, the average reduction in tumor size was 85.03% across the 3 studies, with the reduction ranging from 68.37% to 94.52%. Complications were experienced by some of the patients across the 3 studies as displayed in Table 1. Out of the 37 patients observed in this systematic review, 3 experienced transient facial nerve paresis, 3 experienced transient greater auricular nerve numbness, 2 experienced parotid hematoma, and 1 experienced infection.13,33,34 The 3 patients with transient facial nerve paresis and 3 patients had the tumors identified within the superficial lobe of the parotid gland, with its central point situated specifically in the parotid tail. 34 This yields a complication rate of 24.3% across the 3 studies assessing 37 patients who received RFA for treatment of Warthin’s tumors.13,33,34
Clinical trial
Yeung et al conducted an IDEAL Phase 2a trial clinical trial that examined 20 patients who had parotid Warthin’s tumor and were treated with RFA. 34 One patient discontinued participation in the study, and only 19 patients were included in their statistical results. At 45 weeks, the average mean volume of the Warthin’s tumors across all 19 patients was 3.46 mL, which was a 68.37% reduction compared to the initial pre-procedure volume of 7.48 mL. The mean cosmetic score was 3.68 before the RFA procedure and 2.11 after the intervention. 34
Retrospective observational case studies
Cha et al 13 examined 10 patients, with 1 individual having an extra procedure, and Tung et al 33 examined 7 patients who had parotid Warthin’s tumors and were treated with RFA. At approximately 12 months, the average size of the tumors in Cha et al 13 was 1.0 ± 1.3 mL, which was a 92.20% reduction compared to the average size of 12.8 ± 11.8 mL before the procedure. In Tung et al, 33 the volume of the tumor was 14.6 ± 13.1 mL before the procedure and 0.8 ± 0.5 mL after the procedure at 6 to 12 months, marking a reduction of 94.52%. The mean cosmetic scores for both studies began at a 4 before RFA and ended with a 1 after the operation.13,33
Discussion
In the past few decades, RFA has emerged as an alternative choice to surgery for treating various conditions. This shift is attributed to its suitability for high-risk patients, minimally invasive approach, and cheaper cost. 20 The application of electrosurgery in otolaryngology was first pioneered in the 1920s by Harvey Cushing and William T. Bovie, where the first application of RFA in reducing tongue volume occurred in 1999 and in ablating benign thyroid nodules in 2006.35-37 Outside of otolaryngology, RFA is widely used in treating chronic pain, lung nodules, hepatocellular carcinoma, osteoid osteoma, malignant brain metastases, and more.38-41 This minimally invasive technique works by generating high thermal heat through radiofrequency current, usually between 3 kHz and 300 GHz, that leads to precise tumor necrosis or nerve neurolysis. Providers who perform RFA typically address these masses from deepest to most superficial to prevent destruction of nearby tissues.42,43
Surgical excision has been the traditional method in treating Warthin’s tumor of the parotid gland. These options include total parotidectomy, lateral parotidectomy, partial lateral parotidectomy, extracapsular dissection, and capsular dissection. 5 However, these techniques also include potential intraoperative and postoperative complications. In a retrospective study involving 104 cases of subtotal parotidectomy and 4 cases of total parotidectomy, it was discovered that complications occurred in 62.3% of the surgeries. 1 The increasing use of minimally invasive techniques, such as RFA and microwave ablation, presents viable alternatives for minimizing postoperative complications or for patients who may be at risk for surgery. One case report examining the use of microwave ablation in treating parotid gland adenolymphoma reported an 82% tumor reduction at 3 months following the procedure and no detection of the tumor at 8 months. 44 Even though RFA has been shown in the current literature to be safe and effective for patients, there has not been many studies assessing its application in treating Warthin’s tumors of the parotid gland. In this systematic review, we examined publications that studied the use of RFA in treating Warthin’s tumors.
Yeung et al assessed the efficacy and safety of using ultrasound-guided RFA in treating 19 patients who had Warthin’s tumor of the parotid gland. 34 Before undergoing the RFA procedure, Warthin’s tumors in these patients were confirmed on 2 occasions via FNAC. The mean age of the patients was 68 years, and all the patients were male, with 12 individuals having the tumors on their left side and 7 individuals having the tumors on their right side. The average ablation time was 416.95 seconds, the average procedure time was 29.32 minutes, and the average energy of the RFA was 8.42 kj. The 10 patients were followed at 4, 12, 26, and 45 weeks, where compared to the baseline size of 10.94 mL, the mean tumor volume was 8.77 (19.83% reduction), 5.42 mL (50.46% reduction), 5.04 (53.93% reduction), and 3.46 (68.37% reduction) for these weeks, respectively. The authors of this study compared the patients treated with RFA with 19 other patients who had undergone parotidectomy as well. The cost of the RFA at the hospital was around $14,600 Hong Kong Dollar (HKD) compared to $58,470 HKD for parotidectomy, which was mainly attributed to the shorter procedure time and shortened length of in-patient overnight stay required for RFA compared to parotidectomy. In this study, individuals who underwent RFA experienced an average procedure duration of 29.3 minutes with no overnight stays. In contrast, those who underwent parotidectomy had an average procedure time of 190 minutes and an average of 3.4 overnight stays. It is important to note that there were some complications following the RFA procedure, with 3 patients experiencing facial nerve paresis, 3 experiencing greater auricular nerve numbness, and 1 experiencing infection. 34
Cha et al examined 10 patients with Warthin’s tumors who underwent 11 procedures because 1 patient had 2 neoplasms in their parotid gland. 13 CNBs guided by ultrasound were utilized for the diagnosis and confirmation of the disease. These patients were treated with RFA under ultrasound guidance. The average procedure time was 15.5 ± 5.8 minutes and the energy of the RFA was 20.9 ± 14.2 kj. The imaging methods used preoperatively in this study to identify the tumors included ultrasound, computed tomography, magnetic resonance, and positron electron tomography. All the patients included in this study were male who had a history of smoking cigarettes. Four of the tumors in this study were classified as cystic and the remaining 7 were classified as solid. The mean volume of the cystic tumor was 21.18 ± 15,18 mL, and at 1, 6, and 12 months, size of the cystic tumors decreased to 4.63 ± 5.00 mL (77.9 ± 12.0% reduction), 0.95 ± 1.04 mL (95.1 ± 2.7% reduction), and 0.81 ± 0.70 mL (97.5 ± 1.8% reduction), respectively. The mean volume of the solid tumor was 7.96 ± 6.62 mL, and at 1, 6, and 12 months, the size of the solid tumors decreased to 4.31 ± 3.70 mL (47.3 ± 12.2% reduction), 1.77 ± 2.11 mL (80.6 ± 8.8% reduction), and 0.98 ± 1.41 mL (90.1 ± 9.5% reduction). The authors found that cystic tumors had more response and volume reduction to RFA than solid tumors. Besides the occurrence of a parotid hematoma in 1 patient with a cystic tumors, there were no other complications such as facial nerve paresis or injury. 13
Seven patients who had refused surgical resection of their Warthin’s tumors were treated with ultrasound-guided RFA in Tung et al. 33 Their diagnoses were initially confirmed with CNBs under the guidance of ultrasound. All the patients included in this study were male and had a median age of 60.0 years. The tumor size before the procedure was 14.6 ± 13.1 mL. At 1 month, the residual tumor size was 5.3 ± 3.4 mL and at 6 to 12 months, the tumor size was 0.8 ± 0.5 mL. According to the authors, the percentage of volume reduction at 1 to 2 months, 5 to 6 months, and 10 to 12 months following the operations were 51.2% ± 18.1%, 87.8% ± 10.6%, and 94.3% ± 2.0%, respectively. Only one patient experienced complications following the RFA procedure, where his parotitis with hematoma was successfully treated with antibiotics. 33
Limitations
One constraint of our systematic review is that only 2 extensive databases were used to identify individual studies examining the use of RFA to treat Warthin’s tumors of the parotid gland. The databases used in this systematic review were PubMed and EMBASE, and while these databases offer comprehensive literature coverage, it is conceivable that studies not indexed in these databases may not have been incorporated into our systematic review. Our extensive search terms yielded a total of 1299 articles, and following a thorough assessment by 2 reviewers, 3 articles met our criteria.
The different settings of RFA were not thoroughly examined in this study, which serves as another limitation to this systematic review. The power, ablation time, procedure time, and energy of RFA was available in only 2 of the 3 studies, and they are available in Table 1. However, other important information, such as the frequency, temperature, number of cycles, and continuous or pulsatile mode were not compared in this study. The effectiveness of RFA in treating Warthin’s tumors may have been influenced by the various settings in which these studies were conducted.
Furthermore, the 3 studies analyzed in this systematic review lacked comprehensive follow-up observations extending significantly beyond the 12 month mark. It becomes challenging to discern any alterations in the volume reduction rate or cosmetic score beyond this specific time frame, as well as recurrence rates in patients with Warthin’s tumors receiving RFA. The current literature suggests that the recurrence rate of Warthin’s tumors is 7% to 12%. 45 The individual articles did not examine next steps if RFA proves ineffective, especially after the 12 month period, such as the need for additional RFA sessions or subsequent surgery to address recurring Warthin’s tumors. Also, it is worth noting that the duration of patient follow-up was relatively short, which restricts the ability to conclusively determine the efficacy of RFA as an alternative surgical approach. Furthermore, the potential for malignant transformation posttreatment with RFA remains uncertain, as it may only result in partial remission rather than complete eradication of Warthin’s tumor. Notably, after excision of the tumor, recurrence at the same site is uncommon, with new foci of Warthin’s tumor typically observed instead. 2
Future directions may include the addition of a meta-analysis to the data in this systematic review. Other areas of focus include the direct comparison of RFA with other treatment modalities, such as parotidectomy, microwave ablation, and sclerotherapy, in terms of effectiveness in tumor reduction, postoperative complications, and monetary costs. There is also limited literature on the assessment of RFA in treating patients with Warthin’s tumors. Only 3 articles, including 1 clinical trial and 2 case series, were available across PubMed and EMBASE. More studies, especially randomized control trials, are needed in the future to thoroughly examine the safety and efficacy of using RFA to destroy Warthin’s tumor.
Conclusion
In summary, RFA is a promising minimally invasive treatment for Warthin’s tumors of the parotid gland. This study showed significant tumor size reduction and improved cosmetic outcomes in patients after RFA therapy. RFA presents a favorable safety profile in comparison to traditional surgical interventions with notable cost advantages. Future direction should entail a comprehensive exploration of RFA settings, a potential meta-analysis, and more randomized control trials to validate its safety and efficacy in treating Warthin’s tumors.
Footnotes
Acknowledgements
None.
Data Availability
Data are public information and are available on request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Not applicable because this is a systematic review and does not involve live human participants.
