Abstract
Warthin's tumor is the second most common benign neoplasm of the parotid gland. Epithelial malignant transformation of Warthin's tumor, especially to squamous cell carcinoma, is a rare event. The authors present a case report of a 57-year-old male patient with a mass in the left parotid gland. This paper documents the appearance of squamous cell carcinoma within Warthin's tumor of the left parotid gland and reviews the other similar features of 12 cases.
Introduction
Warthin's tumor (WT) of the parotid gland, also known as papillary cystadenoma lymphomatosum, is the second most common benign neoplasm of the parotid gland, accounting for 5%–10% of all parotid tumors. It usually shows a slow-growing painless mass and occurs in male patients within the age range of fifth to seventh decades of life 1 . Histologically, Warthin's tumor is composed of oncocytic epithelial cells and lymphoid stroma. Malignant transformation of Warthin's tumor is quite common in the case of the lymphoid component, while epithelial malignant transformation is extremely rare (0.3%), 2 especially to squamous cell carcinoma (SCC). 3 In the present study, we report a case of SCC arising in Warthin's tumor of the left parotid gland and review the published literature to highlight SCC arising in parotid WTs. We also describe the features of computerized tomography (CT), ultrasonography (US), and histopathology of this tumor.
Case Report
Patient Information: A 57-year-old nonsmoking man presented with slow-growing painless mass in the left upper cervical region for six years. At the time of the first visit, the patient asked for help at West China Hospital of Sichuan University, and cervical enhanced CT showed a 2 cm encapsulated mass with a relatively clear margin in the left parotid region (Figure 1A & 1B). Clinically, a benign tumor was diagnosed and followed up regularly as instructed. However, eventually the patient was lost to follow-up as the mass did not cause any major discomfort. Finally, enlarging at a faster rate over a 6 month period, he sought medical advice and was hospitalized. The patient’s other past medical history was unremarkable, with no history of acquired immune deficiency syndrome or other immunosuppression. CT scan in 2016 (A, B) and 2020 (C, D, E, F, G, H) and US (I, J, K, L) of tumors of the left parotid gland. (A) A common CT scan showed a round mass (yellow arrows), measuring 2 cm, with a slightly high density. (B) Enhanced CT revealed a lesion with homogeneous enhancement and moderate enhancement of the tumor. (C) A common CT scan revealed a well-marginated heterogeneous lesion, measuring 3 cm in the axial images (yellow arrows) and approximately 6 cm in the coronal images. (D, E) Enhanced CT showed inhomogeneous image density, with lower density inside an oval shaped area. (F, G) Enhanced CT exhibited the vessel facing sign (dotted-line circle). (H) Axial contrast-enhanced CT shows an ill-defined mass located in the left parotid gland with heterogeneous enhancement, extensive intratumoral necrosis, and capsular invasion (solid-line circle). US showed a 55×26×37 mm well-defined mixture and vascularity echoic mass located in the left parotid mass. (I, K) Grayscale ultrasound demonstrated an oval-shaped heterogeneous mass (white thin arrows), with multiple hypoechoic areas (white thick arrows) in the transverse planes. (J, L) Color Doppler flow imaging showed peripheral vascularity.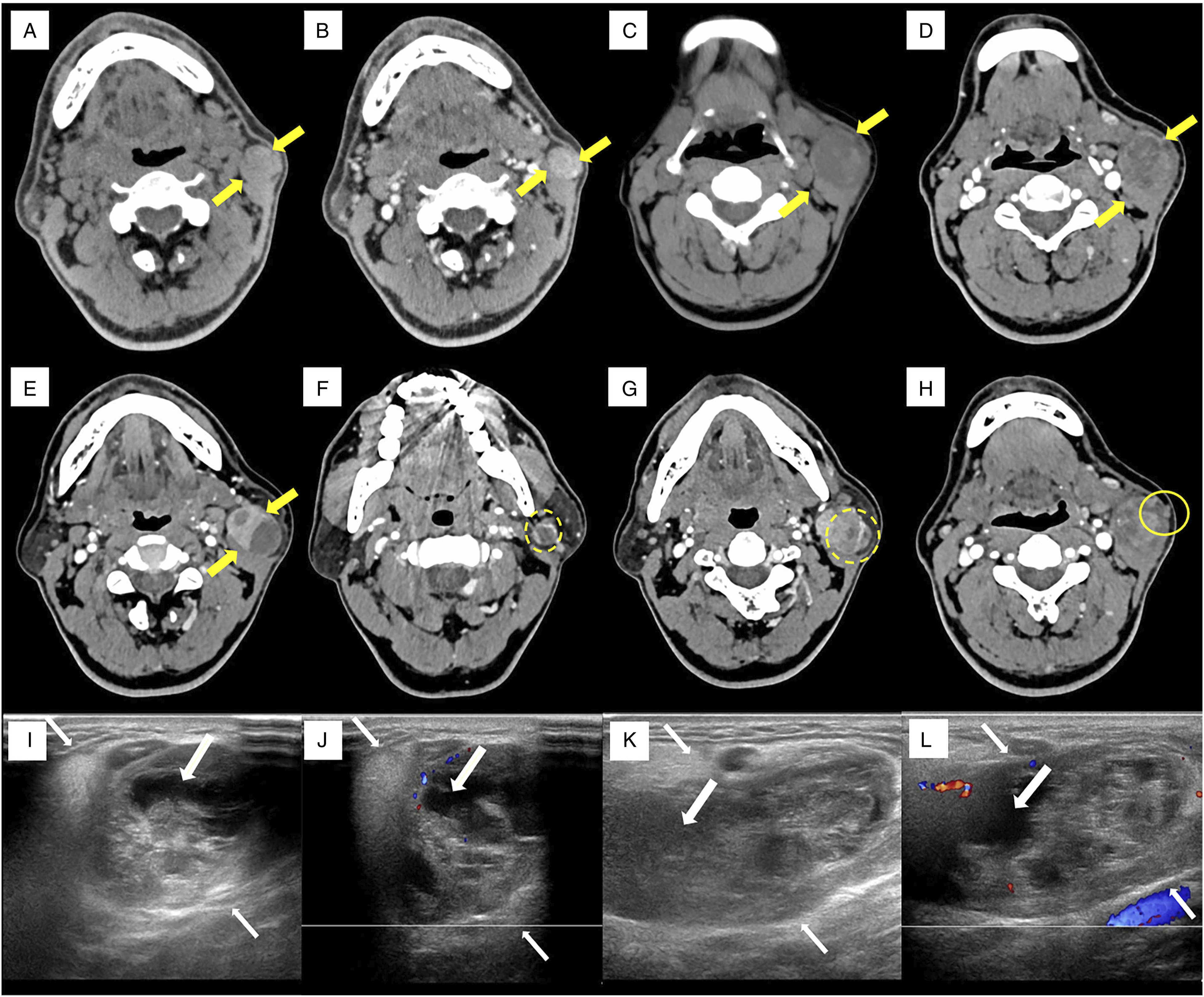
Clinical Finding: Physical examination revealed a 6×5 cm tender, well-defined, slightly hard and movable mass in the left upper cervical region. The skin over the swelling appeared normal with no evidence of scars, sinuses, discolorations, or punctum. The patient had no facial paralysis.
Diagnostic Assessment: The patient received CT and US examinations, revealing a 6 cm lesion (Figure 1). Complete blood count, inflammation indexes, biochemistry, CEA, CA15-5, CA19-9, CA-125, and CYFRA21-1 revealed no abnormal values.
Therapeutic Intervention: The patient underwent left partial parotidectomy under general anesthesia. The 5 ×3 × 4 cm mass was well encapsulated and cystic-solid, with grayish-brown viscous mucous fluid inside. Intraoperative freezing revealed SCC in Warthin's tumor (Figure 2). After dissociating the facial nerve trunk and its branches, the left parotid gland and lymph nodes in cervical I–III areas were resected. Postoperative biopsy indicated that the resection margins were found to be free of tumor and the cervical lymph nodes did not show metastasis. (A) Areas of typical Warthin's tumor. Warthin's tumor is composed of double-layered oncocytic epithelial cells and lymphoid stroma, (H&E, ×100). (B) Warthin's tumor was infiltrated by malignant squamous cells, (H&E, ×100). (C) An area of squamous cell carcinoma. Warthin's tumor underwent squamous metaplasia. (H&E, ×400).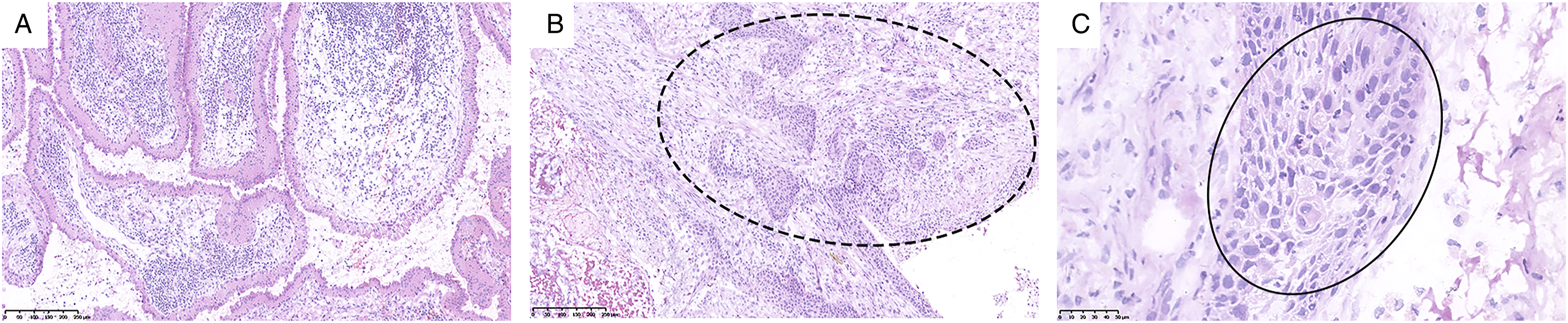
Outcome and Follow-up: On gross examination, the 5 ×3 × 4 cm mass was well encapsulated and cystic-solid, with grayish-brown viscous mucous fluid inside. Postoperative histopathology was consistent with the intraoperative frozen results, comprising WT and SCC. Postoperative positron tomography-CT (PET-CT) showed no other mass or lymphadenopathy. The patient remains well and without evidence of metastasis at this time, 19 months postoperatively.
Discussion
Summary of cases of squamous cell carcinoma arising in Warthin’s tumor.
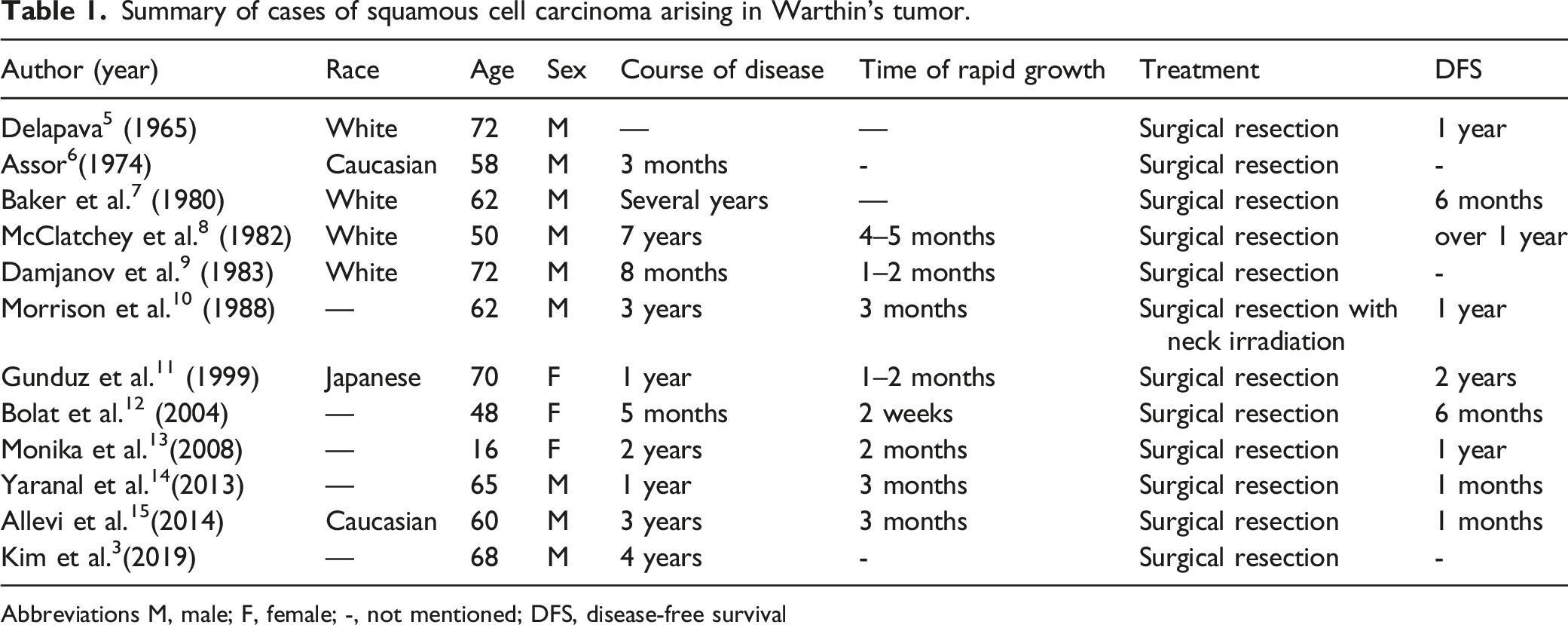
Abbreviations M, male; F, female; -, not mentioned; DFS, disease-free survival
Previous reports postulated that squamous carcinoma arises from a focus of squamous metaplasia.3,5-7,9,11,13-15 Significantly, Allevi et al. 15 in 2014 found that pathologists of WT squamous transition could have three stages of degeneration: WT, squamous metaplasia, and SCC, which are similar to the opinion of Bolat et al., 12 who reported that the diagnosis of malignant transformation of Warthin’s tumor is based on some standards. First, there was a preexisting benign Warthin’s tumor. Second, there are transitional zones from benign eosinophils to malignant epithelia; third, the peripheral lymphoid stroma is invaded. Finally, the metastasis of the lymphatic matrix component of primary extrasalivary tumors was excluded. In the present case, the hematoxylin and eosin (H&E) stained sections showed the typical characteristics of Warthin's tumor with malignant squamous epithelial cells (Figure 2A), consistent with the first and second points of the criteria. Malignant squamous epithelial cells were observed to infiltrate the lymphoid stroma (Figure 2B), satisfying the third condition. The patient had no history of other malignancies, and postoperative PET-CT showed no other mass or lymphadenopathy, excluding the metastasis of malignancy from other regions to the parotid gland. And the malignant squamous epithelial cells presented atypia with a high nucleocytoplasmic ratio and prominent nucleoli (Figure 2C). Therefore, SCC was thought to arise in preexisting WT. The etiology of malignant transformation in WT is not established. Damjanov et al. 9 in 1983 identified the process of the cylindrical epithelium of benign portions with abundant mitochondria into flat squamous cells with fewer organoids forming the areas of squamous metaplasia and carcinoma. These transitions were deemed to be due to infection or necrosis and ischemia, of which ischemia was thought to be the most likely etiology for squamous metaplasia. Clinically, 11 the tumor is usually an asymptomatic and slow-growing painless mass.3,5-10,12-15 Malignant transformation in WT is suggested by a recent history of rapid enlargement of a longstanding mass,8-11,13-15 and was also found in our patients. Furthermore, on the basis of immunohistochemistry, the presence of the keratin cytoskeleton of WT cells perhaps provided a clue to account for these cells undergoing squamous metaplasia.9,11,13 Consequently, if necessary, for a more accurate diagnosis, immunohistochemistry and electron microscopy should be carried out.
Concerning the preoperative assessment of WT, US, fine-needle aspiration cytology, and CT or magnetic resonance imaging are promising tools. In our case, the sonographic features exhibited a mass with a clear boundary without lymph node metastasis, and multiple cystic areas on grayscale images, consistent with the findings reported by Rong et al., 16 who agreed that cystic areas as characteristic characteristics of Warthin's tumors on US images. The results of US do not indicate signs of malignant transformation of the tumor. It is possible that the benign neoplasm was in the early stage of malignancy and that the surrounding tissue of WT was not infiltrated or was not obviously infiltrated. Thus, the conversion of WT into malignant carcinoma needs to be observed dynamically. Many studies have indicated that 2-phase CT showed a pattern of strong enhancement at early phase scanning with a decrease at delayed phase scanning of WT.3,17,18 However, the CT imaging features were atypical in our case. The vessel facing sign (VFS) has been reported to be a characteristic of WT, 19 found in our CT image (Figure 1F & 1G). In addition, the most malignant carcinomas have enhanced nodules on CT images inside the tumor 3 . Enhanced CT showed strong enhancement of the tumor with an intratumoral cystic or necrotic area and invasion of the capsule, revealing the possibility of malignancy (Figure 1D & 1E).
The differentiation between benign and malignant tumors is crucial to the choice of treatment strategy and the quality of life and prognosis of patients. Surgical resection remains the most common treatment modality. The present patient underwent left full parotidectomy with radical neck dissection. The long-term prognosis of these patients is not clear; but 6- to 12-month disease-free survival has been reported in the published literature.5-8,10-15 Our patient was disease-free at the 19-month follow-up.
Conclusion
In this report, we document the appearance of squamous cell carcinoma within WT of the parotid and review the other similar features of 12 cases. Although the development of malignancy arising from WT is a rare event, less than 1%, it was still found in some patients. Accurate preoperative assessment of WT, such as CT and US, is favorable in planning the operative approach. A rapid increase in tumor size clinically is due to malignant transformation, needing to be brought to the attention of surgeons.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
