Abstract
Congenital granular cell epulis (CGCE) is a rare disease, particularly when it affects multiple oral and maxillofacial regions. We present the case of a female neonate with 4 asymptomatic masses attached to the maxillary and mandibular gingiva as well as the tongue. Due to the size of the masses causing feeding and respiratory difficulties, lesion removal was performed under general anesthesia. Histopathological analysis revealed tightly arranged polygonal cells with abundant granular eosinophilic cytoplasm. Immunohistochemically, the cells were negative for Smur100 (S-100), neuron-specific enolase (NSE), cluster of differentiation (CD) 68, and Sry-related HMG box (SOX)-10, and positive for alpha-1-antitrypsin (AAT) and vimentin. The diagnosis of multiple CGCEs was established through clinical examinations, histomorphometrical, and immunohistochemical analyses. In conclusion, CGCE should be considered in the differential diagnosis of intraoral lesions in neonates.
Introduction
Congenital granular cell epulis (CGCE) is a rare benign tumor located in the oral cavity, primarily on the maxillary or mandibular alveolar ridge. This condition was first described by Ernst Christian Neumann in 1871. Its incidence is estimated to be 6:1,000,000. 1 The most gingiva, particularly the maxillary anterior region (spanning from the maxillary central incisor to the maxillary canine), is the most common intraoral site for these tumors. Lesions are found 3 times more frequently in the maxilla than in the mandible.2,3 Although these lesions predominantly affect the maxilla and mandible, they can also appear on the ventral surface of the tongue and may be either sessile or pedunculated in nature. Clinically, lesions typically present as pink or reddish masses, with diameters ranging from a few millimeters to several centimeters. They may protrude from the neonatal oral cavity, potentially causing difficulty in mouth closure. 4 Diagnosis relies on clinical and pathological assessments. Approximately 10% of CGCE cases involve multiple lesions, termed multiple CGCEs. 2 Surgical excision is recommended for sizable or multiple lesions causing feeding and respiratory challenges. This report details the case of a female neonate diagnosed with multiple CGCEs undergoing surgical resection.
Case Presentation
A female neonate was admitted to the hospital due to the presence of 4 round, pedunculated masses in the oral cavity. Her medical and family histories were negative, and she experienced difficulty breastfeeding, although respiration and swallowing were normal. On thorough physical examination, 3 of the 4 masses were identified at the alveolar processes of the maxilla and mandible, while 1 was located ventrally on the tongue. The largest mass was 3.5 cm in diameter (Figure 1).
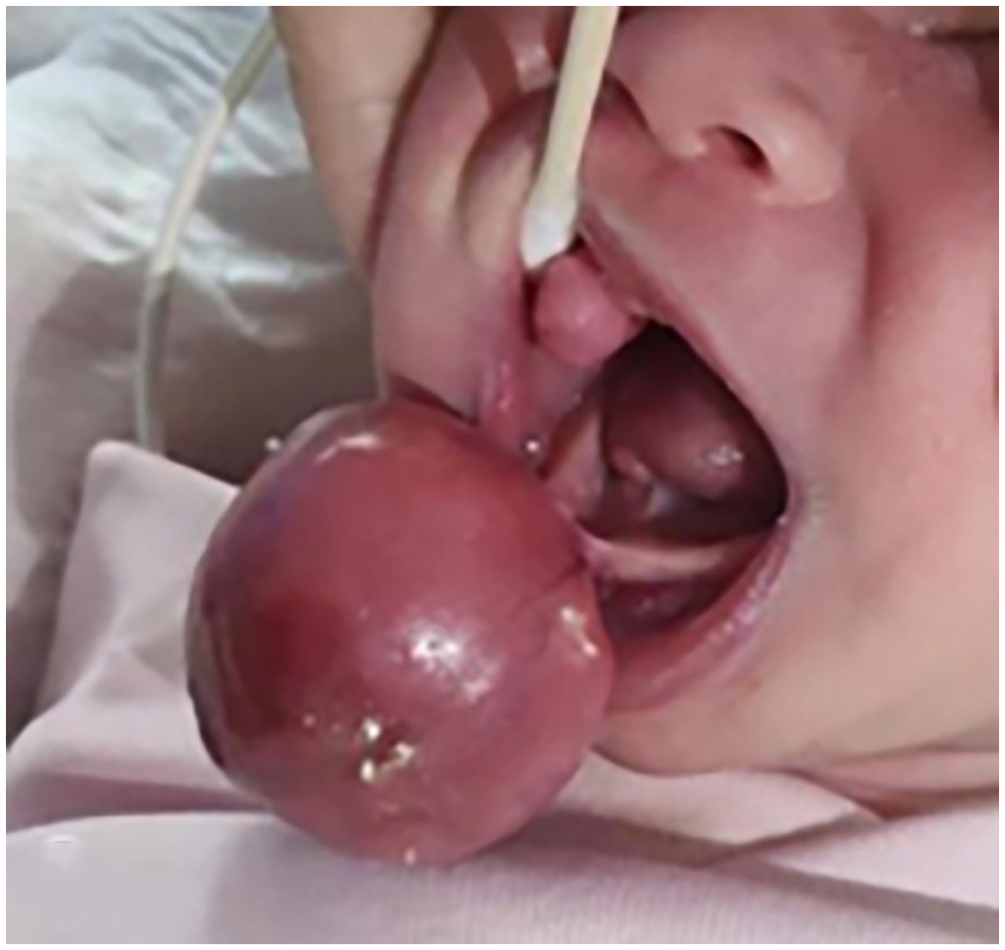
Preoperative aspect of the tumor mass localized at the anterior part of the alveolar bone and tongue.
On the second day of life, the lesions were surgically excised under general anesthesia. Histopathological examination with hematoxylin and eosin staining revealed densely arranged polygonal cells with abundant granular eosinophilic cytoplasm (Figure 2). Immunohistochemical analysis demonstrated that the cells were negative for Smur100 (S-100; Figure 3), neuron-specific enolase (NSE), cluster of differentiation (CD) 68, and Sry-related HMG box (SOX)-10, while being positive for alpha-1-antitrypsin (AAT) and vimentin. A diagnosis of CGCE was confirmed.
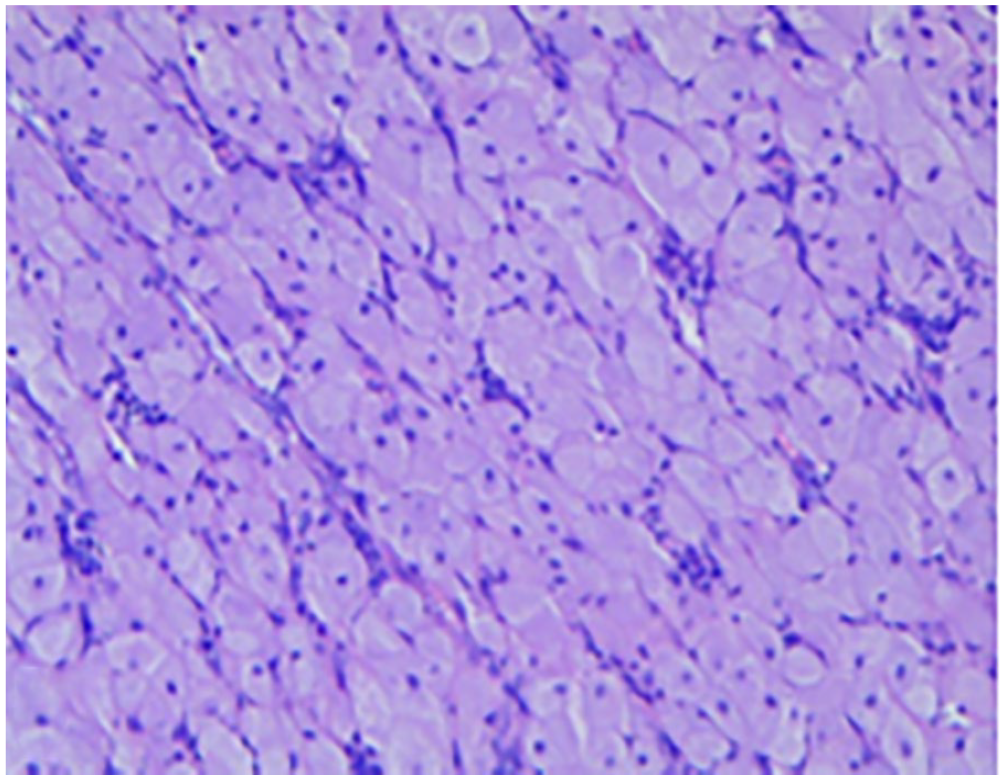
Tightly arranged polygonal cells with copious amounts of granular eosinophilic cytoplasm, no atypia nuclei were observed (H&E, ×10). H&E, hematoxylin and eosin.
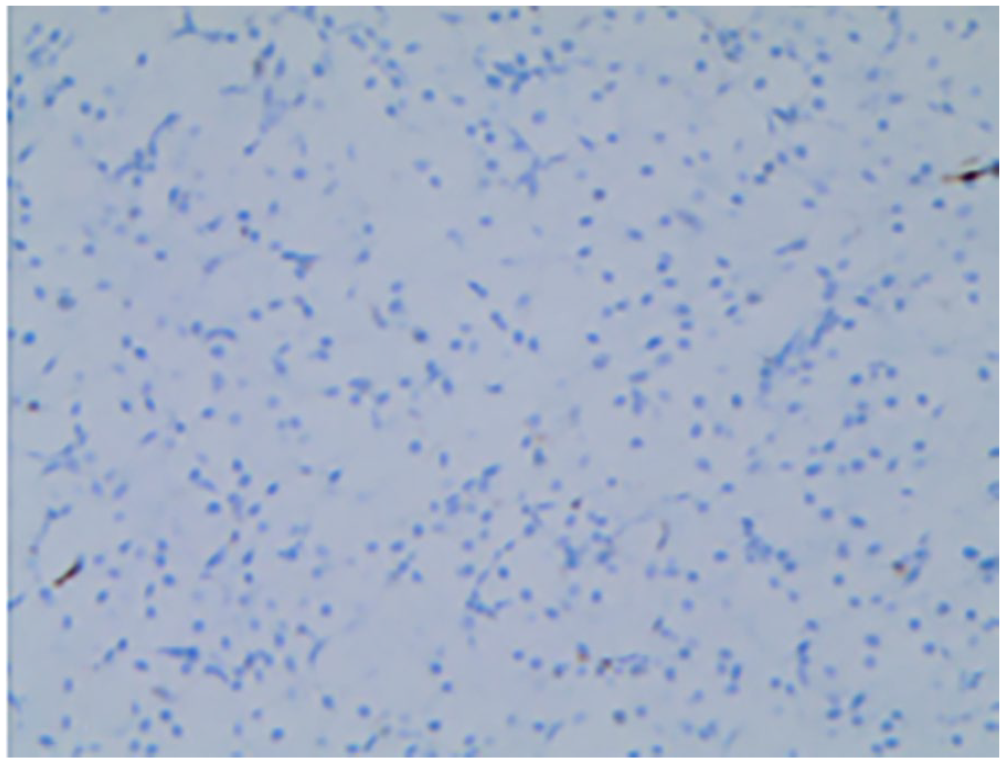
Section was negative for S-100 (immunohistochemical staining, ×10).
Discussion
CGCE, also known as congenital granular cell tumor (GCT), congenital epulis, congenital epulis of the newborn, congenital granular cell lesion, and gingival GCT of the newborn, represents a rare mucosal disease of the gingiva with an unknown etiology.5,6 It is predominantly located at the most apical part of the maxilla or mandible, with occurrences in the tongue being exceedingly rare.7,8 This case report focuses on a female newborn diagnosed with CGCE, presenting lesions in the gingivae of both the mandible and maxilla, as well as the tongue. The patient underwent surgical treatment, resulting in an ideal outcome over a 12 month follow-up period, thereby corroborating behaviors previously described in the literature.7,8
The pathogenesis of CGCE remains largely elusive, and the condition ceases growth at birth in neonates, often resolving spontaneously without intervention. 9 Potential histological origins include mesenchymal and epithelial cells, pericytes, smooth muscle, endothelial cells, myofibroblasts, neuromasts, and various fibroblastic cells. 10 Previous reports suggest that maternal, fetal, and neonatal endogenous glucocorticoid levels may influence the pathogenesis of CGCE. 11 Despite the absence of hormone receptor evidence in the tissues, CGCE is considered a reactive or degenerative lesion of mesenchymal origin.12,13 Occurring as an isolated anomaly, CGCE is not associated with other syndromes or anomalies. 14 CGCE is typically diagnosed postnatally; however, prenatal ultrasound and magnetic resonance imaging can facilitate early detection, with detailed sonographic examinations being straightforward as early as 26 weeks of gestation. 15 Such prenatal diagnostics aid in deciding the appropriate treatment approach. Large masses often necessitate Cesarean delivery to minimize the risk of trauma to the neonate if delivered vaginally. 16
Differentiating CGCE from GCT is crucial due to their similar histologic characteristics. 17 CGCE is more prevalent in newborns, with a higher incidence in females (female-to-male ratio of 8-10:1), 6 whereas GCT shows a predilection for adults, particularly females aged 30 to 60 years, with a female-to-male ratio of 2:1. Anatomically, CGCE is found in the gingiva, in contrast to GCT, which commonly occurs in the tongue, soft palate, and floor of the mouth. Histopathologically, both CGCE and GCT consist of large, polygonal cells with abundant eosinophilic cytoplasm and nuclei that are mostly rounded or oval. 6 However, GCT is characterized by cells arranged in layers, cords, or nests, with pseudoepitheliomatous hyperplasia of the epithelium and often accompanied by small peripheral nerves. 18
Immunohistochemical analysis demonstrates that GCT express the S-100 protein in the vast majority of cases, serving as the main method to differentiate CGCE from GCT. In the case described here, a female infant presented with a negative S100 protein and a thin squamous epithelium, with lesions occurring in 3 areas of the gingiva and 1 in the tongue. These characteristics fulfill the diagnostic criteria for CGCE.
Surgery represents the primary treatment option for CGCE, offering a relatively favorable postoperative prognosis, with no observed malignant transformation. 6 When the mass is too small, the condition is monitored without intervention, as it typically regresses spontaneously. 11 Surgical resection is recommended for patients experiencing feeding and breathing difficulties. It has been reported that, even when surgery does not result in complete resection, CGCE rarely recurs.19-21 Thus, diagnosing CGCE requires an integration of clinical, immunohistochemical, and histomorphometric findings. An accurate diagnosis and tailored treatment are essential for a favorable prognosis.
This case highlights the significance of accurate diagnosis and appropriate treatment for multiple CGCEs. Given the rarity of this condition, enhancing clinicians’ understanding of the disease and standardizing its treatment protocols can help mitigate undesired outcomes.
Footnotes
Data Availability
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
The studies involving human participants were reviewed and approved by the ethics committee of Qingdao Women and Children’s Hospital. The patients/participants provided their written informed consent to participate in this study.
