Abstract
Introduction
Among tumors of the parotid gland, 15% to 25% are reported to be malignant. 1 Pain/tenderness, one of the symptoms of parotid carcinoma, is experienced by about half of the patients and is thus considered to be an important diagnostic sign. 2 In addition, facial nerve paralysis has been reported to occur in 14% to 30% of patients with parotid carcinoma,3,4 but it is extremely rare in benign tumors. Therefore, a finding of facial nerve paralysis can help differentiate between benign and malignant parotid tumors. Moreover, facial nerve paralysis is a known poor prognostic factor in patients with parotid carcinoma.5-7
The well-known causes of facial nerve paralysis include Bell’s palsy and Ramsay Hunt syndrome. Idiopathic Bell’s palsy accounts for roughly 80% of cases of peripheral facial nerve paralysis, and about 70% of these cases are said to completely resolve.8-10 On the other hand, only about 5% of cases of facial nerve paralysis are due to tumors, including parotid carcinoma. 11 For this reason, there is a risk that general practitioners and general ENT doctors may overlook parotid carcinoma when treating patients with facial nerve paralysis. Occasionally, parotid carcinoma is diagnosed for the first time by diagnostic imaging, such as magnetic resonance imaging (MRI), after steroid therapy is administered for facial nerve paralysis.
At our department, we have treated 209 cases of parotid carcinoma over the past 23 years by using uniform diagnostic and treatment strategies. The present study aimed to examine differences in the prognosis of parotid carcinoma according to the presence and absence of facial nerve paralysis and to capture the features of patients with facial nerve paralysis. In addition, we examined whether earlier diagnosis of parotid carcinoma would have been possible if doctors had been able to recognize the relevant characteristics of undiagnosed patients at the onset of their facial nerve paralysis.
Patients and Methods
Patients
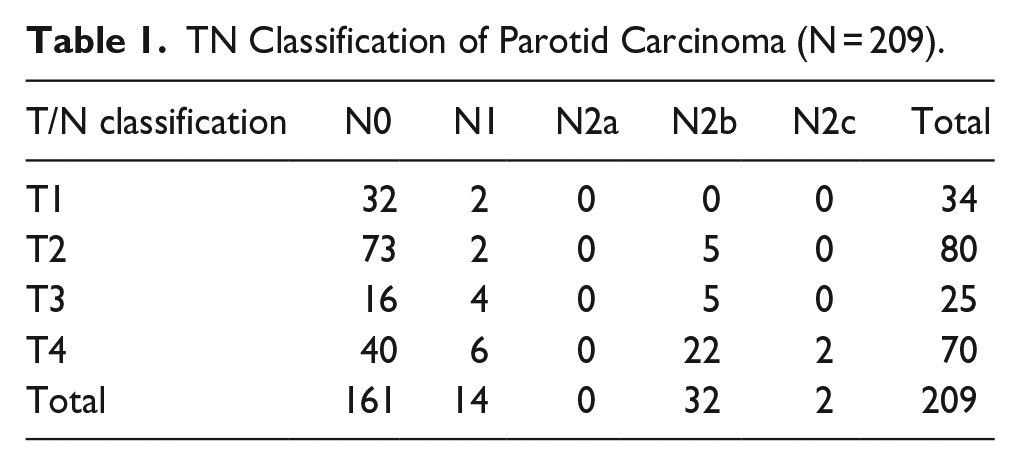
A total of 209 patients with parotid carcinoma who received initial treatment for the carcinoma at the Department of Otorhinolaryngology—Head and Surgery, Osaka Medical and Pharmaceutical University during a 23 year period from September 1999 to August 2022 were included in the study. Table 1 shows the TN classification of the 209 carcinomas. The main histopathological types were mucoepidermoid carcinoma (56 patients), adenoid cystic carcinoma (28 patients), carcinoma ex pleomorphic adenoma (25 patients), and salivary duct carcinoma (24 patients). The histological grade was low/intermediate in a larger number of cases (Table 2). The study was approved by the ethics committee of Osaka Medical and Pharmaceutical University (Approval No. 2621).
TN Classification of Parotid Carcinoma (N = 209).
Histopathological Types of Parotid Carcinoma (N = 209).
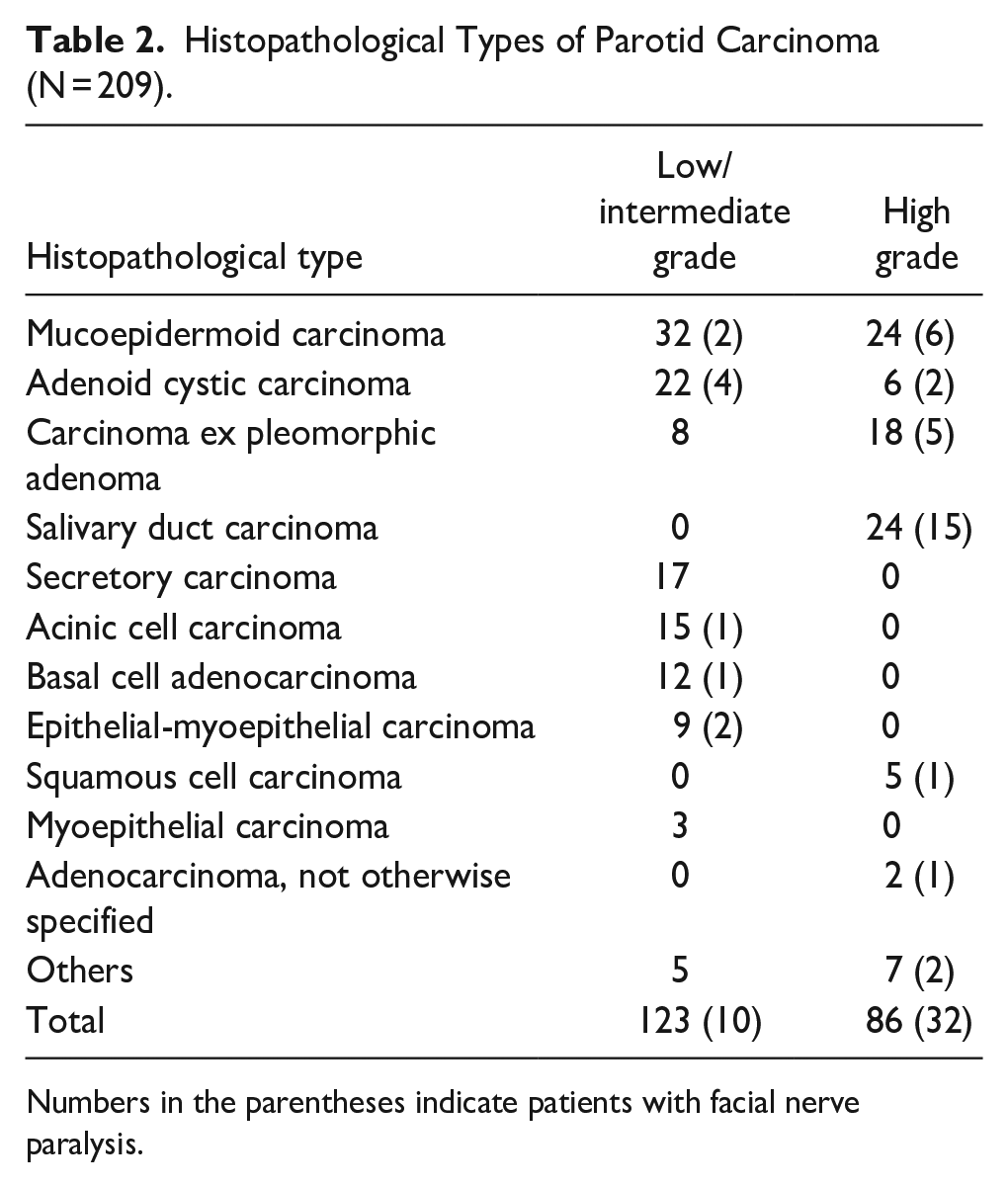
Numbers in the parentheses indicate patients with facial nerve paralysis.
Presence and Absence of Facial Nerve Paralysis at the Time of the Initial Visit
At the initial visit to our department, 42 patients had facial nerve paralysis and 167 patients did not (Figure 1). We studied the diagnosis, treatment, and prognosis for these patients retrospectively. This design is a cross-sectional study. Table 2 shows the number of patients with facial nerve paralysis by histopathological type and grade. Patient characteristics, signs/symptoms, stage classification, tumor size and localization, histological grade, treatment, and 5 year disease-specific and disease-free survival were compared between the 2 groups. With regard to tumor localization, tumors were classified according to whether the center of the tumor was located in the superficial, deep lobe, or lower pole.
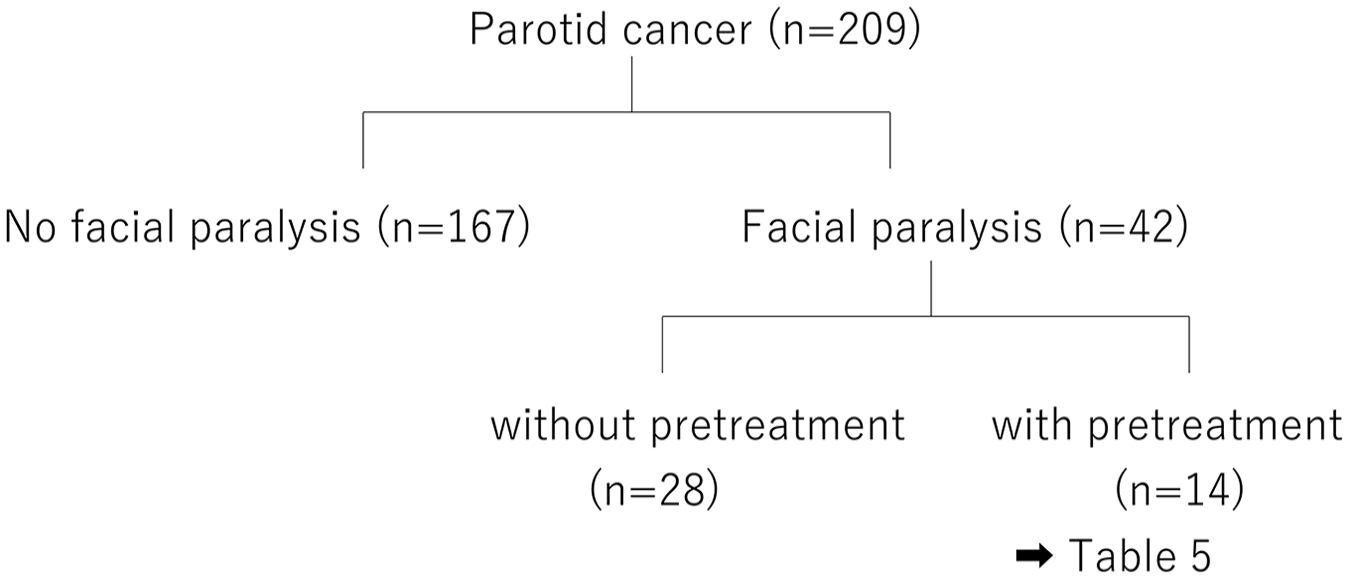
Flow chart of facial nerve paralysis for parotid cancer.
Presence and Absence of Pretreatment for Facial Nerve Paralysis
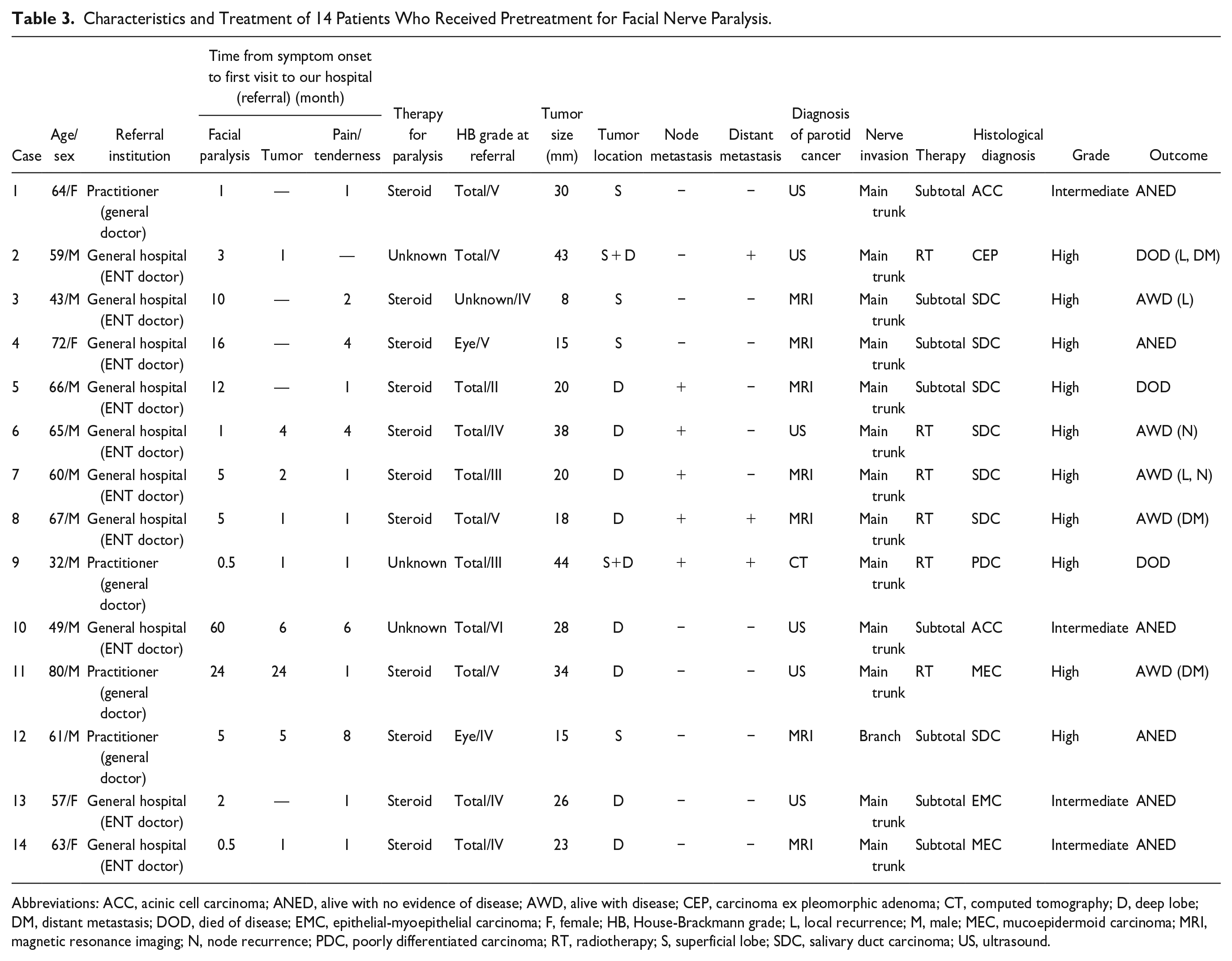
Among the 42 patients with parotid carcinoma who presented with facial nerve paralysis at their initial visit to our department, 14 were initially diagnosed with and treated for Bell’s palsy (pretreatment group; Figure 1); in these patients, parotid carcinoma was not diagnosed at the initial visit to another facility. The referral sources for the 14 patients are shown in Table 3. Ten patients were inquired from the general hospital and 4 cases were from practitioners. The remaining 28 patients were diagnosed with parotid carcinoma at their initial visit to our department and did not receive pretreatment for facial nerve paralysis (no pretreatment group; Figure 1). All patients were treated for parotid carcinoma by using uniform diagnostic and treatment strategies in our department.
Characteristics and Treatment of 14 Patients Who Received Pretreatment for Facial Nerve Paralysis.
Abbreviations: ACC, acinic cell carcinoma; ANED, alive with no evidence of disease; AWD, alive with disease; CEP, carcinoma ex pleomorphic adenoma; CT, computed tomography; D, deep lobe; DM, distant metastasis; DOD, died of disease; EMC, epithelial-myoepithelial carcinoma; F, female; HB, House-Brackmann grade; L, local recurrence; M, male; MEC, mucoepidermoid carcinoma; MRI, magnetic resonance imaging; N, node recurrence; PDC, poorly differentiated carcinoma; RT, radiotherapy; S, superficial lobe; SDC, salivary duct carcinoma; US, ultrasound.
Patients Who Underwent Pretreatment for Facial Nerve Paralysis (Pretreatment Group)
In the 14 patients who were initially treated for Bell’s palsy (pretreatment group), we examined patient characteristics, signs/symptoms, degree of paralysis, tumor localization and size, treatment, histopathological type, and outcome.
Statistical Analysis
Disease-specific and disease-free 5 year survival rates according to the presence and absence of facial nerve paralysis and the presence and absence of pretreatment for facial nerve paralysis were analyzed by the Kaplan-Meier method, and prognostic factors were evaluated by univariate and multivariate analysis.
The univariate analysis according to the presence or absence of facial nerve paralysis included the following factors: age, sex, the time from symptom onset to first visit, pain/tenderness, adhesion, T classification, N classification, stage, tumor localization, tumor size, histological grade, operation, and postoperative radiotherapy. The chi-squared test was used to analyze sex, pain/tenderness, N classification, tumor localization, histological grade, and postoperative radiotherapy. Fisher’s exact test was used to analyze adhesion, T classification, stage, and operation, and the Mann-Whitney U test was used to analyze age, the time from symptom onset to the first visit, and tumor size.
The univariate analysis according to the presence or absence of pretreatment for facial nerve paralysis included patient characteristics, symptoms, stage classification, tumor size, localization, histological grade, and treatment. The chi-squared test was used to analyze N classification. The Fisher exact test was used to analyze sex, pain/tenderness, adhesion, T classification, stage, tumor localization, histological grade, operation, and postoperative radiotherapy, and the Mann-Whitney U test was used to analyze age, the time from symptom onset to the first visit, and tumor size. Multivariate analysis was performed for items for which a significant difference was observed in the univariate analysis, that is, pain/tenderness, adhesion, N classification, tumor localization, and histological grade.
A P value of less than .05 was considered statistically significant.
Results
Presence and Absence of Facial Nerve Paralysis at the Initial Visit to Our Department
A significantly higher proportion of patients with facial nerve paralysis had pain/tenderness and adhesion with surrounding tissues (both P < .001). In all these patients, the tumors were classified as T4a and stage IV. In addition, deep lobe tumors and high-grade tumors were observed significantly more frequently in the group with than in the group without facial nerve paralysis (both P < .001). We found no significant relationship between histological grade and tumor localization. In terms of treatment, extended resection and postoperative radiotherapy were performed significantly more frequently in the group of patients with facial nerve paralysis (both P < .001). Factors that were significantly more common in the group with facial nerve paralysis were examined by multivariate analysis. The odds ratio was highest for adhesion, followed by localization in the deep lobe and high-grade malignancy (Table 4).
Univariate and Multivariate Analyses of Factors Influencing Facial Nerve Paralysis.
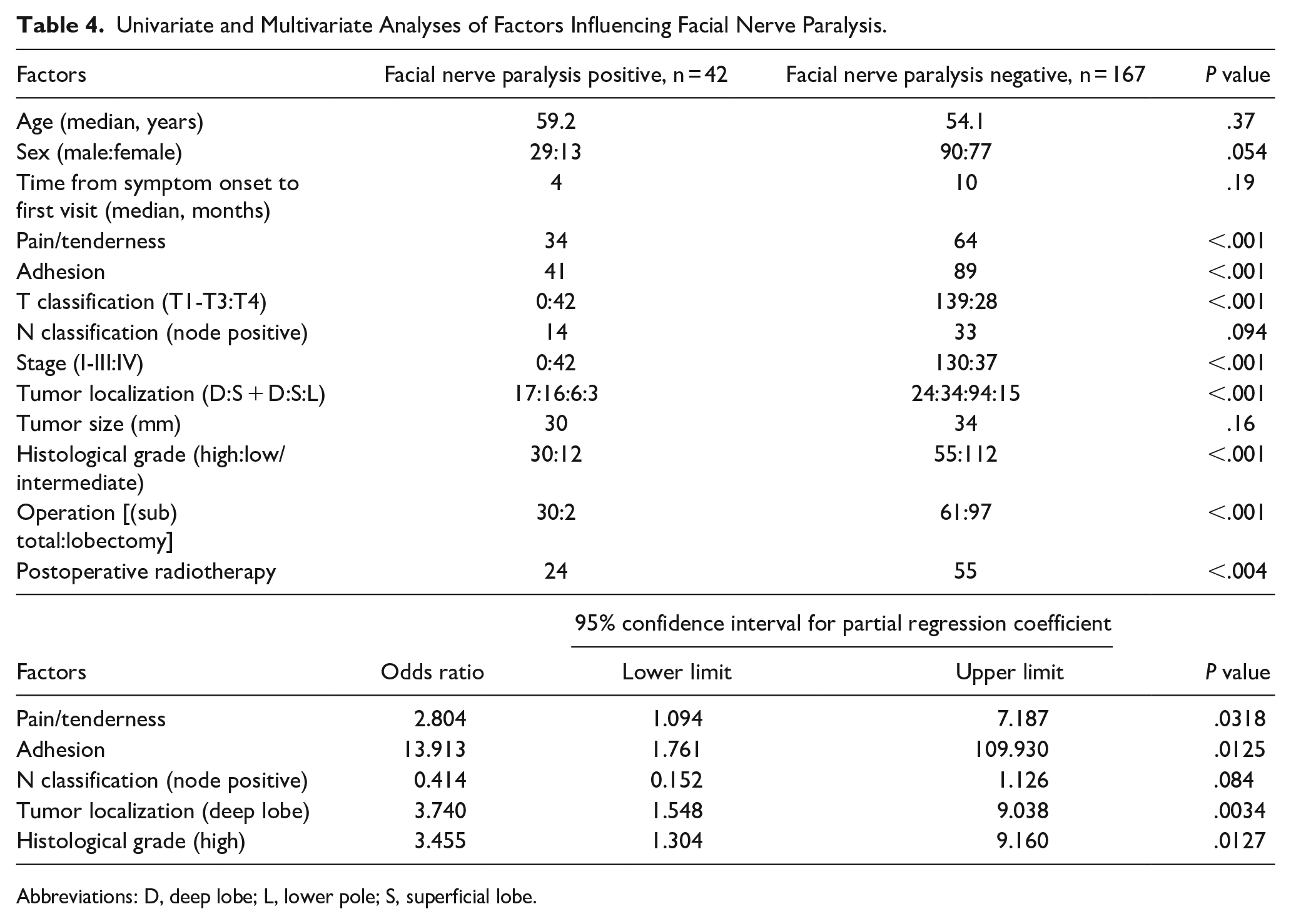
Abbreviations: D, deep lobe; L, lower pole; S, superficial lobe.
Disease-specific 5 year survival rates were significantly lower in the group with than in the group without facial nerve paralysis (48.0% vs 88.7%, respectively; P < .01), as were disease-free 5 year survival rates (34.5% and 77.4%, respectively; P < .01; Figures 2 and 3).
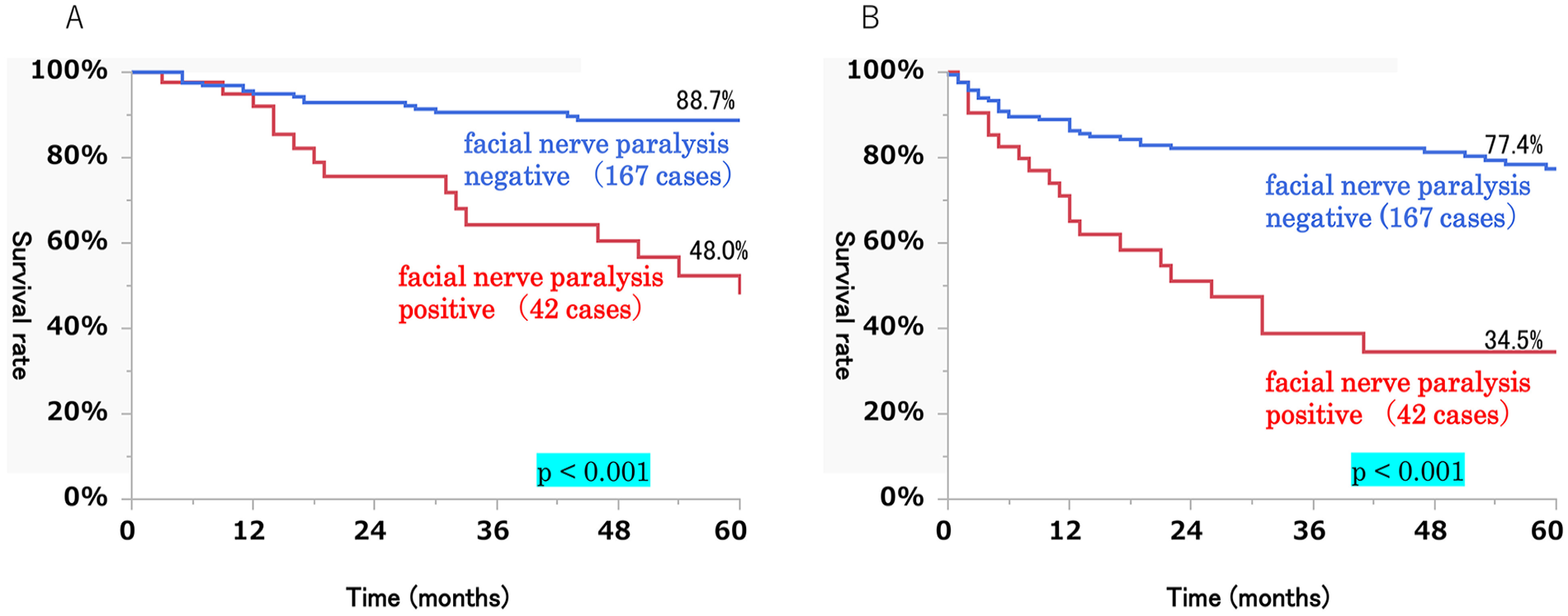
Disease-specific 5 year survival rate (A) and disease-free 5 year survival rate (B) of 209 patients with or without facial nerve paralysis at the initial visit.
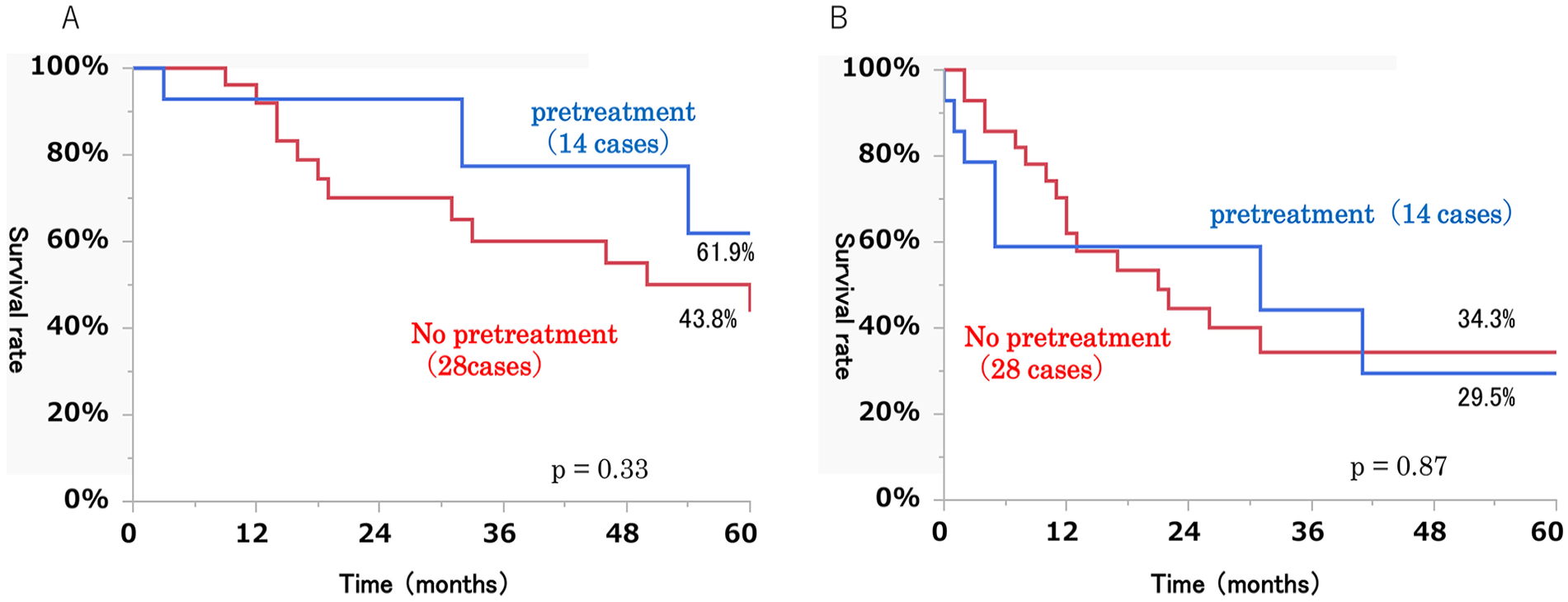
Disease-specific 5 year survival rate (A) and disease-free 5 year survival rate (B) of 42 patients with or without pretreatment for facial nerve paralysis.
Presence and Absence of Pretreatment for Facial Nerve Paralysis
No significant differences were found in any of the factors compared between the pretreatment and no pretreatment groups (Table 5).
Univariate Analysis of Factors Influencing Pretreatment Facial Nerve Paralysis.
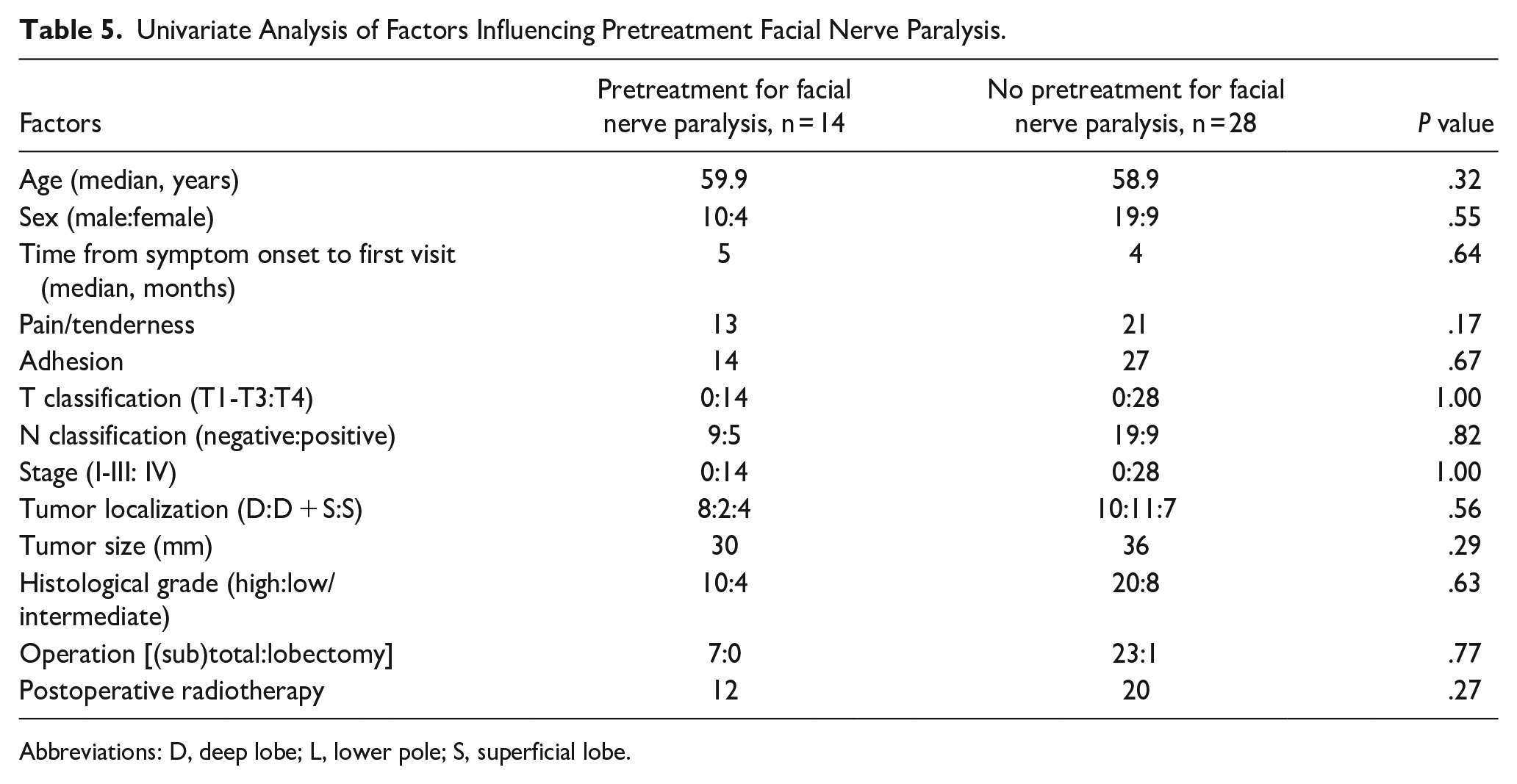
Abbreviations: D, deep lobe; L, lower pole; S, superficial lobe.
Disease-specific 5 year survival rates were not significantly different between the pretreatment group (61.9%) and the no pretreatment group (43.8%; P = .33). The same was true for disease-free 5 year survival rates (29.5% vs 34.3%, respectively; P = .87; Figure 2).
Patients Who Underwent Pretreatment for Facial Nerve Paralysis (Pretreatment Group)
Table 3 shows the details of the 14 patients in the pretreatment group. The time from the first observation of facial nerve paralysis to the initial visit to our department ranged from 0.5 to 60 months (median, 5 months). At the time of their first visit to our department, 9 of these patients had noticed palpable tumors and 13 complained of pain/tenderness. At the time of or before the onset of facial nerve paralysis, 4 patients noticed palpable tumors and 5 patients complained of pain/tenderness. Treatment for facial nerve paralysis was performed shortly after the appearance of paralysis in all patients, and steroids were administered to all except for 3 patients with unknown status. Among the 8 patients who underwent surgery, 7 showed facial nerve invasion at the main trunk and 1 showed facial nerve invasion at the branches. Invasion to the main trunk of the facial nerve was suspected in all 6 patients who underwent radiotherapy, based on imaging results. With regard to tumor localization, the presence of deep lobe tumors was noted in 10 patients, and of these patients, 8 had tumors only located in the deep lobe. In terms of histological grade, 10 patients were high-grade malignancy, and of these patients, 7 had salivary duct carcinoma. Cervical lymph node metastases were noted in 5 patients, and distant metastases were observed in 3 patients.
Discussion
Bell’s palsy accounts for roughly 80% of cases of peripheral facial nerve paralysis, and among these cases, 70% are assumed to achieve recovery.8-10 On the other hand, facial nerve paralysis due to tumors is rare and represents only about 5% of all cases. 11 Therefore, there is a risk of parotid carcinoma being overlooked by general practitioners and general ENT doctors. In general clinical practice, it is not common to perform MRI or computed tomography in patients suspected of having Bell’s palsy.
According to Boahene et al, 12 facial nerve paralysis due to occult malignancy should be suspected when the following 4 important findings are observed: prolonged paralysis with no sign of recovery, a complaint of pain/tenderness, the presence of other cranial nerve symptoms, and a history of localized skin cancer. In parotid carcinoma, pain/tenderness is an important sign, and its frequency is proportional to the degree of malignancy. One study reported a frequency of 67% for high-grade malignancy and 44% for low/intermediate-grade malignancy. 2 Parotid carcinoma accompanied by facial nerve paralysis is highly malignant in most cases. In the present study, 81% of the whole group of patients and 93% (13/14) of the pretreatment group complained of pain/tenderness; these findings suggest that pain/tenderness is an important sign of parotid carcinoma. However, careful diagnosis is necessary because Ramsay Hunt syndrome, another cause of facial nerve paralysis, is also often accompanied by pain/tenderness. 13
Chung et al 14 reported that in 9 of 240 patients, facial nerve paralysis was initially diagnosed as Bell’s palsy and the real cause (ie, tumors) was revealed later on. Among those patients, 3 had parotid carcinoma and 2 of these 3 had adenoid cystic carcinoma. Quesnel et al 15 reported 4 cases of facial nerve paralysis in which parotid tumors were not identified in diagnostic imaging at the time of the initial visit. As for histopathological type, 2 patients had adenocarcinoma, 1 patient had squamous cell carcinoma, and 1 patient had adenoid cystic carcinoma. In adenoid cystic carcinoma, nerve invasion is often observed, and although the degree of malignancy is not high in general, this histological type requires particular attention from the perspective of facial nerve paralysis. 16
Among the 42 patients with facial nerve paralysis, 79% had tumors located in the deep lobe, whereas only 35% of the 167 patients without facial nerve paralysis did. On the other hand, the group without paralysis included significantly more patients with superficial lobe tumors than the group with paralysis (56% vs 14%, respectively). Because the deep lobe has a smaller volume than the superficial lobe, 17 a tumor located in the deep lobe is more likely to invade the facial nerve. One could hypothesize that superficial lobe tumors are more likely to be detected early because the tumors are more easily palpable; however, this hypothesis does not appear to be true because we did not observe any significant differences in the duration of symptoms or tumor size between the 2 groups. The proportion of patients with high malignancy grade tumors was higher in the group with than in the group without paralysis (71% vs 33%, respectively). Park et al 6 also reported that 87% of parotid carcinomas with facial nerve paralysis were highly malignant. In highly malignant cases, the frequency of facial nerve paralysis may be higher because tumors are more likely to infiltrate surrounding tissues. In the present study, we found no significant relationship between tumor localization and histological grade, thus suggesting that high-grade malignancy is an independent factor in patients with facial nerve paralysis. In the multivariate analysis, adhesion with surrounding tissues was found to be the factor most strongly associated with facial nerve paralysis, but this is presumed to be because highly malignant tumors tend to infiltrate surrounding tissues more readily, and it is difficult to diagnose the mobility of deep lobe tumors by palpation.
On the other hand, the comparison between the pretreatment and no pretreatment groups revealed no significant differences in any of the factors nor in survival. In patients with pain/tenderness, tumors located in the superficial lobe, and/or large tumor sizes, these observations are likely to lead to a diagnosis of a tumor, but no significant differences in these factors were seen between the 2 groups. However, among the 14 patients in the pretreatment group, 4 patients noticed palpable tumors and 5 had pain/tenderness at the time when facial nerve paralysis was first noted or even beforehand. Therefore, performing a more careful examination might have enabled an earlier diagnosis of parotid carcinoma. In cases with prolonged paralysis and a complaint of pain/tenderness, a detailed examination, such as an ultrasound, should be performed. Since the tumor is often located in the deep lobe of the parotid gland, an MRI should also be performed in case where the tumor is not detected by ultrasound. Given that the duration of symptoms to the first visit to our hospital did not differ significantly between the 2 groups, it is understandable that there was no significant difference in survival rates between the 2 groups. However, an earlier diagnosis of parotid carcinoma in the pretreatment group might have improved the prognosis.
One limitation of the present study is that the initial examination was not performed at the same facility in all cases. In Japan, patients are free to visit and be examined by any medical facility, whether it is a small clinic or a large hospital. For this reason, patients in the pretreatment group received treatment for facial nerve paralysis at various facilities with varying levels of expertise. In addition, patients in the no pretreatment group were referred to our hospital by various medical institutions. Most cases were diagnosed or misdiagnosed in other hospitals concerning the cause of the facial nerve paralysis, while all cases, not only the “pretreatment” but also the “no pretreatment” groups, were treated in our hospital. Therefore, diagnosis and treatment policy, and treatment results were interpreted as a single-institute study. In that regard, this was not a single-institute study.
Conclusions
Patients with parotid carcinoma who present with facial nerve paralysis at their initial visit have a significantly poorer prognosis than those who do not present with facial nerve paralysis. In the group with facial nerve paralysis, deep lobe tumors, and high-grade malignancy are seen significantly more frequently. About 30% of the patients who presented with facial nerve paralysis at the time of their first visit to our department had not been diagnosed with parotid carcinoma at their first visit to another facility after the onset of paralysis, where they received treatment for Bell’s palsy. In some patients, findings may have been present at the time of the onset of paralysis that indicated the possible presence of parotid carcinoma. There were no significant differences in the prognosis between the “pretreatment” and “no pretreatment” groups. A lot of factors may affect their prognosis. However, an earlier diagnosis for facial nerve paralysis caused by parotid cancer may improve the prognosis. Therefore, we would like to emphasize the necessity of avoiding the overlooking of parotid cancer. The important points to avoid misdiagnosis are the cases with prolonged paralysis with no sign of recovery, and a complaint of pain/tenderness. In particular, deep lobe tumors of parotid cancer require special attention. By conducting a detailed examination in all patients who present with facial nerve paralysis, the number of missed cases of parotid carcinoma may be reduced, thereby shortening the time to diagnosis and possibly improving the prognosis.
Footnotes
Author Contributions
Ryo Kawata designed and supervised the study. Eisuke Suzuki and Ryo Kawata wrote the manuscript. Yusuke Ayani, Masaaki Higashino, Tetsuya Terada, and Shin-Ichi Haginomori acquired all clinical data. All authors performed data analysis and interpretation, and approved the manuscript.
Data Availability
Data are available on request due to privacy/ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by our Institutional Review Board.
