Abstract
We report a probable case of Aspergillus basicranial infection diagnosed by pathogenic serological examination presenting atypical initial manifestations, and highlight the importance of serological examination to avoid treatment delay and disease management. An 84-year-old diabetic patient presented with right peripheral nerve palsy, intolerable otalgia, hearing loss, dysphagia, hoarseness, and bucking. The patient was diagnosed a probable Aspergillus skull base osteomyelitis with cranial neuritis and meningitis of central nervous system. Galactomannan test was used in combination with 1-3-β-D-glucan and magnetic resonance imaging to follow-up during the continuous treatment of voriconazole. To date, the patient has remained in clinical remission for over 39 months but the drug cannot be stopped safely.
Introduction
Fungal skull base osteomyelitis (SBO) is a rare and life-threatening opportunistic infection that occurs predominantly in immunosuppressed hosts.1,2 However, the diagnosis and therapeutic management are poorly defined. We present a probable case of Aspergillus basicranial infection in a senile diabetic patient. The purpose is to illustrate the importance of fungal serology examination in diagnosis and therapeutic management.
Case Report
An 84-year-old diabetic patient who presented with palsy of the right peripheral nerve, intolerable otalgia, hearing loss, dysphagia, hoarseness, and bucking was admitted. Three months prior to admission, fungal clumps were found in his right external auditory canal and treated with triamcinolone acetonide and econazole nitrate cream. Subsequently, he was diagnosed with otitis media and underwent multiple courses of antibiotic and steroid therapy.
Magnetic resonance imaging (MRI) and computed tomography (CT) revealed temporal bone osteomyelitis (Figures 1D and 2A1, B1, C1, and D1). The patient underwent a mastoidectomy and experienced relief from otalgia. Histological examination of intratemporal tissue showed hyperplastic fibrous tissue with partial hyalinization and calcification. No fungi were detected after methenamine silver staining (Figure 1F). High-throughput sequencing did not yield any reliable positive results. The 1-3-β-D-glucan (BDG) and galactomannan test (GM test) were 55.45 pg/ml and 1.85, respectively. During the same time, the patient developed a fever with a productive cough, Babani and Candida were isolated from a sputum analysis. Antibiotic treatment, combined with voriconazole, was administered and discontinued after 10 days.
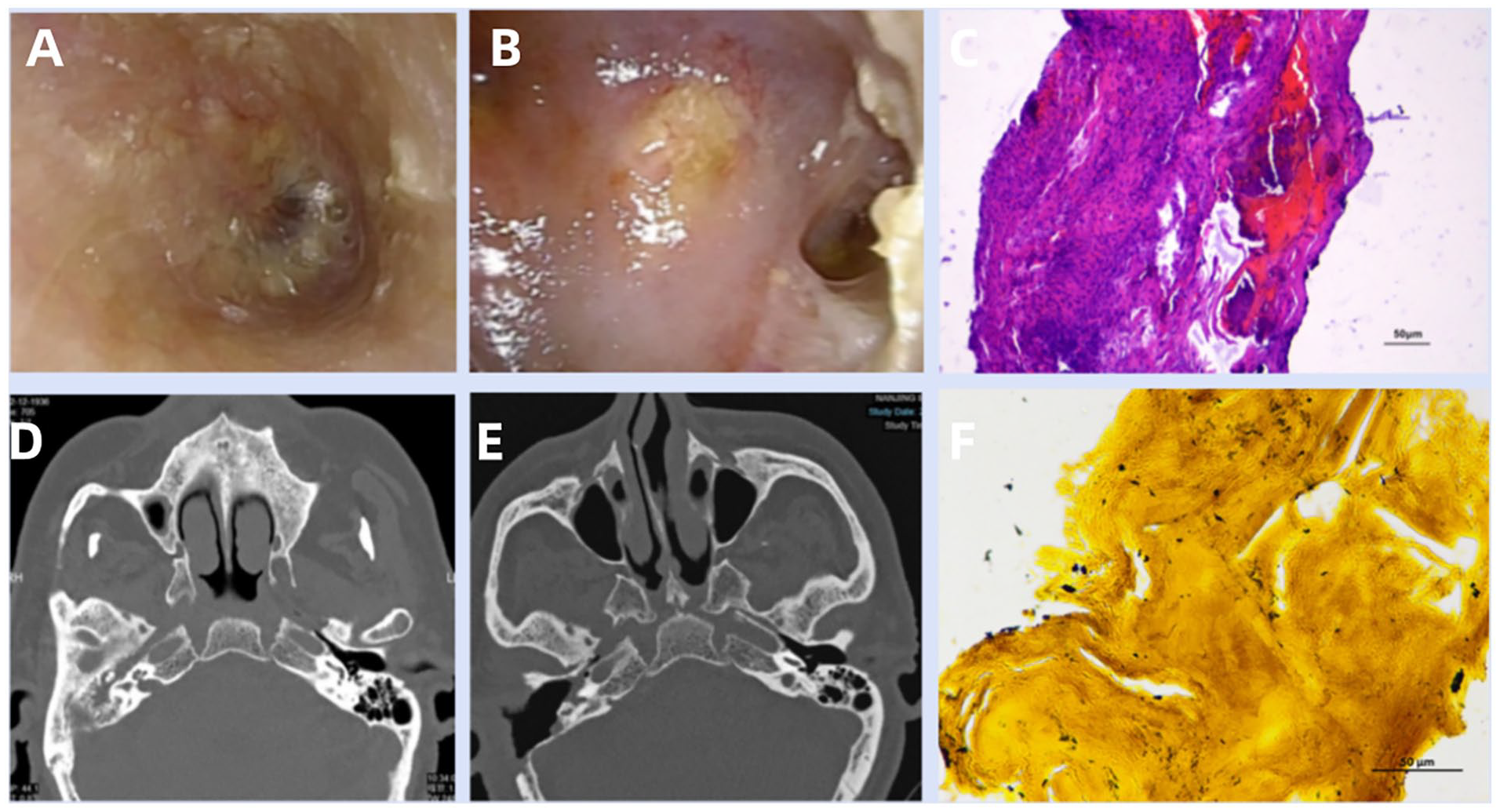
Results of endoscopic examination, CT examination, and pathological examination. (A) Otoscopy images show the external auditory canal after treatment with triamcinolone acetonide and econazole nitrate cream. (B) Images of the surgical cavity 5 weeks after mastoidectomy. (C) Histological examination in the mastoid mucosa shows hyperplastic fibrous tissue with partial hyalinization and calcification. (D) CT showed right mastoiditis with extensive bony erosions of the temporal bone, osteomyelitis, and involvement of the nasopharynx, petrous apex, inferior temporal fossa, and eustachian tube. (E) CT showed a postoperative surgical cavity 17 days after mastectomy. (F) No fungi were found after silver-methenamine staining. CT, computed tomography.
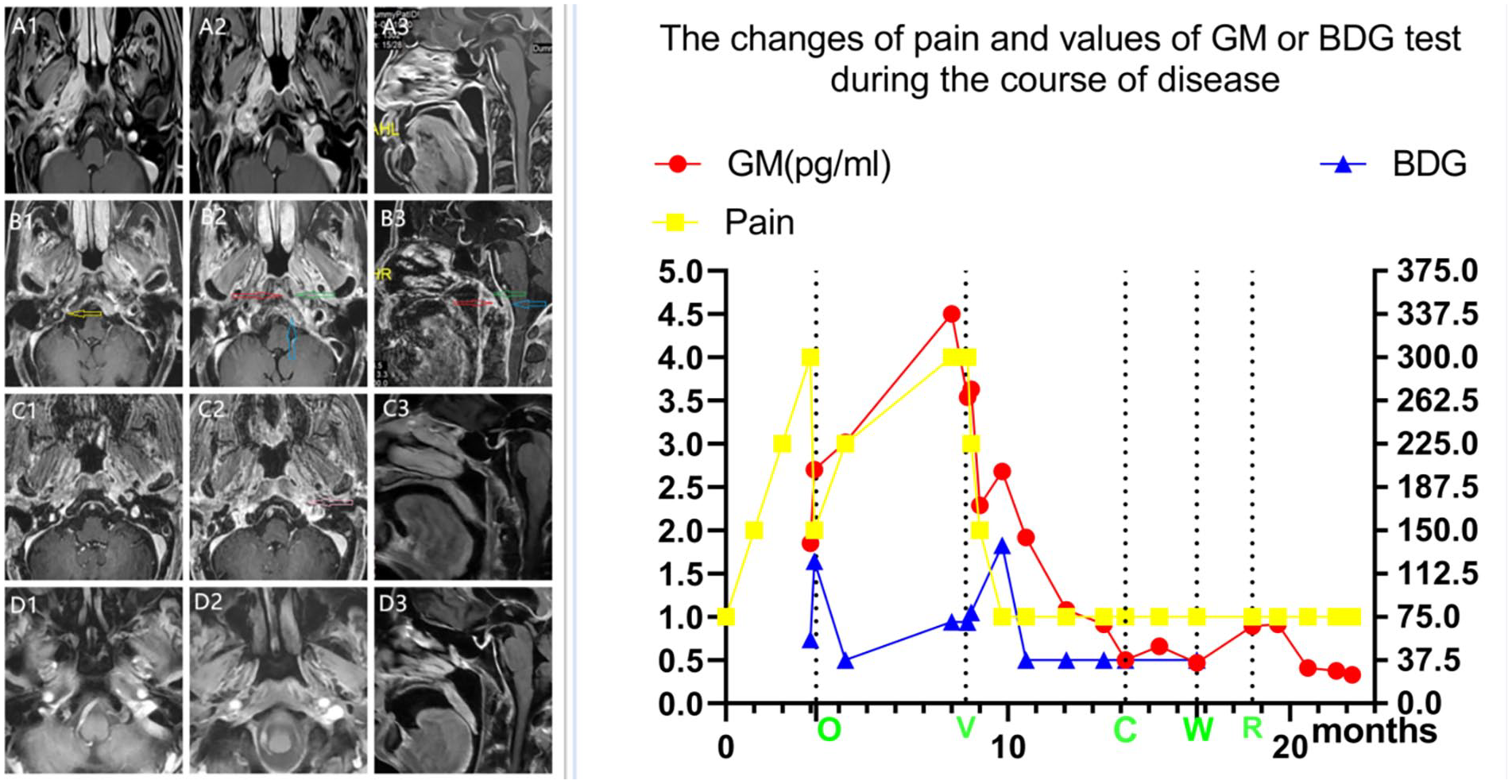
The following of MRI examination, symptoms, and pathogenic serological examination. In the left picture, axial and sagittal post-contrast T1-weighted enhanced magnetic resonance imaging around the eustachian tube that confirms the SBO extending from the right to the left along the cranial base bone, the prevertebral space (red arrow) with little abscess (green arrow), thickened dura (blue arrow), and parapharyngeal tissue. Meningeal enhancement is evidence of convincing intracranial extension. Images show diffuse infective lesions improved by mastoidectomy (yellow arrow) and voriconazole (pink arrow), but extended lesions cannot be prevented by surgery. (A) On admission. (B) Five months after right mastoidectomy, before voriconazole treatment. (C) Eleven months after right mastoideduractomy, 6 months after voriconazole treatment. (D) Seventeen months after right mastoidectomy,12 month after voriconazole treatment. MRI, magnetic resonance imaging; SBO, skull base osteomyelitis.
All symptoms improved, but the patient began to experience pain in the occiput and left otalgia over the following 4 months, eventually developing left hearing loss. An enhanced MRI showed temporal bone osteomyelitis progressing to SBO with small abscesses in the prevertebral space and enhanced thickened dura (Figure 2B2). The BDG assay and the GM test values increased to 70.7 pg/ml and 4.5, respectively. The patient was diagnosed with basicranial Aspergillus infection and treated with voriconazole. The blood drug concentration and the GM test were monitored monthly. The GM test value gradually decreased below 0.5 at 6 months, with clinical and radiological remission and no change in liver and renal function. The patient tried to change or stop voriconazole but had to resume the medication due to an increase in the GM test value (Figure 2). To date, the patient has remained in clinical remission for more than 39 months, but voriconazole cannot be stopped safely.
Discussion
Articles were found in PubMed using key words of “skull base osteomyelitis” and “fungal” from 1989 to the present in which 34 mastoiditis (28 articles) were identified.3-30 There were 24 male and 10 female patients, with a median age of 65 years (range 3-87 years).The most prevalent condition was diabetes mellitus for 58.8% (20/34), whereas 7 cases (20.6%) had no identified underlying condition.6,9,22,25,28,30 Twenty-two cases (64.7 %) were caused by fungal etiology predominately Aspergillus species,7,16,21,26,28 in which the reported isolated pathogens in 12 Aspergillus cases mixed infection with Candida, vancomycin-resistant Enterococcus, and methicillin-resistant Staphylococcus epidermidis,4,12,15,19 5 of which (14.7%) had a simple Candida strain infection,9,10,13,20,25 and 2 of which (5.9%) had a simple Scedosporium apiospermum infection.24,26 There were no pathogenic fungi were detected in 2 cases, 1 of them had positive results of serum BDG test and Aspergillus antigen test. 23 Sixteen facial palsy cases were found in 20 cranial nerve injury patients, and the meninges were invaded in 5 cases.12,16,23,26,27 Mastoidectomy were performed in 15 cases, and 7 cases accepted myringotomy with grommet insertion, 3 cases accepted extensive debridement of external auditory canal.15,17,19 Nearly one-half of all patients (15 cases) were treated with voriconazole, the treatment varied between 2 and 20 months.
Galactomannan is a polysaccharide component of Aspergillus cell walls and is released during tissue invasion by Aspergillus hyphae. BDG is present in the cell walls of almost all pathogenic fungi, such as Candida, Aspergillus, and several other genera (except Mucorales and Cryptococcus). These 2 serologic biomarkers have been introduced for the diagnosis of invasive aspergillosis (IA) over the past 4 decades. 31
The concentration of serum GM in vivo is determined not only by the rate of production and secretion by the growing fungus but also by the rate of uptake in the bloodstream, as well as the rate of elimination from the circulation. A meta-analysis from Mayo Clinic in 2018 shows that serum GM had a moderate mean sensitivity of 0.71 and a high specificity of 0.89. Increasing the cutoff index increased specificity and decreased sensitivity. A serum GM cutoff point of 1.0 appears to provide an optimal sensitivity of 0.79 and specificity of 0.88. 32 In another study, the sensitivity, in particular, was very heterogeneous at the cutoff values of the 0.5, 1.0, and 1.5 optical density index. Currently, due to the large number of false positives, a higher cutoff of 1.0 is proposed. 32 However, the GM test is difficult to consider to be a gold standard for IA diagnosis. The main sources of circulating BDG are invasive fungal disease, iatrogenic contamination, intestinal translocation, and intestinal contents: mycobiome and BDG translocation, liver function, bacterial infections, and manufacturing-associated contamination. A meta-analysis showed that the BDG assay in intensive care unit patients at risk for invasive candidiasis or candidemia had a mean sensitivity of 0.81 and mean specificity of 0.60. 32 The negative predictive value of BDG is very high and it could not identify Aspergillus infection. The limitation of BDG-based tests lies in their inability to identify pathogenic microbial species. So the BDG test is clearly less close to being a diagnostic gold standard of invasive fungal disease than the GM test.
In general, serological examination is not considered sufficient for a proven diagnosis.33-36 Serology is not applicable for criteria for proven invasive fungal disease except cryptococcal antigen,there was consensus that similar GM thresholds are appropriate for adults and children. Microscopic examination, culture, and polymerase chain reaction sequencing are required, according to the recent EORTC/MSG guidelines. 34 Invasive fungal diseases do not have specific symptoms, signs, or imaging findings. Some studies have evaluated the early diagnostic value of serum GM tests combined with CT for invasive pulmonary aspergillosis. The diagnostic and follow-up performance of the 2 biomarkers were further improved by using them as a combination of tests (parallel analysis improves sensitivity and negative predictive value) or continuous monitoring (serial analysis improves specificity and positive predictive value). In this report, pain appeared first and persisted throughout the progression of the disease; imaging studies provided localization but were not diagnostic. At the early stage of the disease, the BDG assay and GM test showed a slight increase, but the presence of Candida in the sputum examination made us consider them false positives. Fortunately, we persisted in monitoring the serological changes dynamically and made the decision to treat with voriconazole. Serological tests were sensitive and varied with the severity of the disease and remained valuable for observation even after the disappearance of symptoms and imaging changes.
Voriconazole is recommended as the first-line treatment for IA, particularly in central nervous system localizations. It has a good penetration across the blood-brain barrier and its central nervous system concentration exceeds inhibitory concentrations of Aspergillus spp.37-39 It is known to require longer periods of drug therapy for Aspergillus infections in the central nervous system. When the patient stopped taking the drug, the values of the GM test began to increase, raising concerns about a possible resurgence of aspergillosis. The current guidelines do not exhaustively describe these rare clinical cases, and the optimal duration of antifungal treatment and follow-up remains uncertain.
Only about half of all invasive fungal infections are diagnosed before death, the value of the GM test is not sensitive and specific enough to be considered a diagnostic gold standard, according to many sources. However, when used in combination with BDG and MR imaging and observed dynamically, it demonstrates significant value during the course of Aspergillus infections and therapeutic management.
Footnotes
Author Contributions
The data were collected by Zhengwen Zhu and Yating Wang. Xihong Cao analyzed all the data, and provided the treatments with Jie Huang. Pathological analysis was done by Lijuan Chen. Qingyu Zhang helped with imaging analysis.
Data Availability
Data cannot be shared openly to protect patient privacy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the project was supported by Nanjing Medical Science and Technology Development Fund Grant Number: YKK22250.
Ethical Approval
The treatment was approved by the Institute’s Human Research Committee of BenQ Medical Center.
Informed Consent
The patient gave his informed consent to participate and consent to publish.
