Abstract
Introduction
Coronavirus disease 2019 (COVID-19), which was a pandemic that started in 2020 and lasted until 2022, is known to cause acute respiratory failure and a following death in the acute form. Also it is associated with symptoms during the course of the disease and even afterward in the form of prolonged symptoms, such as anosmia, which is the loss or reduced sense of smell, and ageusia, which is the complete loss or reduced sense of taste. Two main pathophysiological pathways are involved in patients with COVID-19 1 (Figure 1). It has been reported that the leading factor that causes these symptoms is a small change in the gray matter of the brain.2,3 However, both anosmia and ageusia are related to the partial demyelination of related nerves with the involvement of the central nervous system (CNS) and peripheral nervous system (PNS). 4 Anosmia and ageusia are not only limited to the course of the disease but also occur very rarely after the COVID-19 vaccination. Similarly, there have been reports of anosmia and ageusia after influenza or human papillomavirus vaccination in both pediatrics and adults. 5 The main pathophysiological causes that lead to this dilemma are poorly understood but are considered to be similar to the causes that occur during the course of the disease. 6 The mentioned symptoms are very rare post-COVID-19 vaccination adverse events. To the best of our knowledge, our study is the very first study that has gathered reports of post-COVID-19 vaccination anosmia and ageusia cases in a systematic literature review.
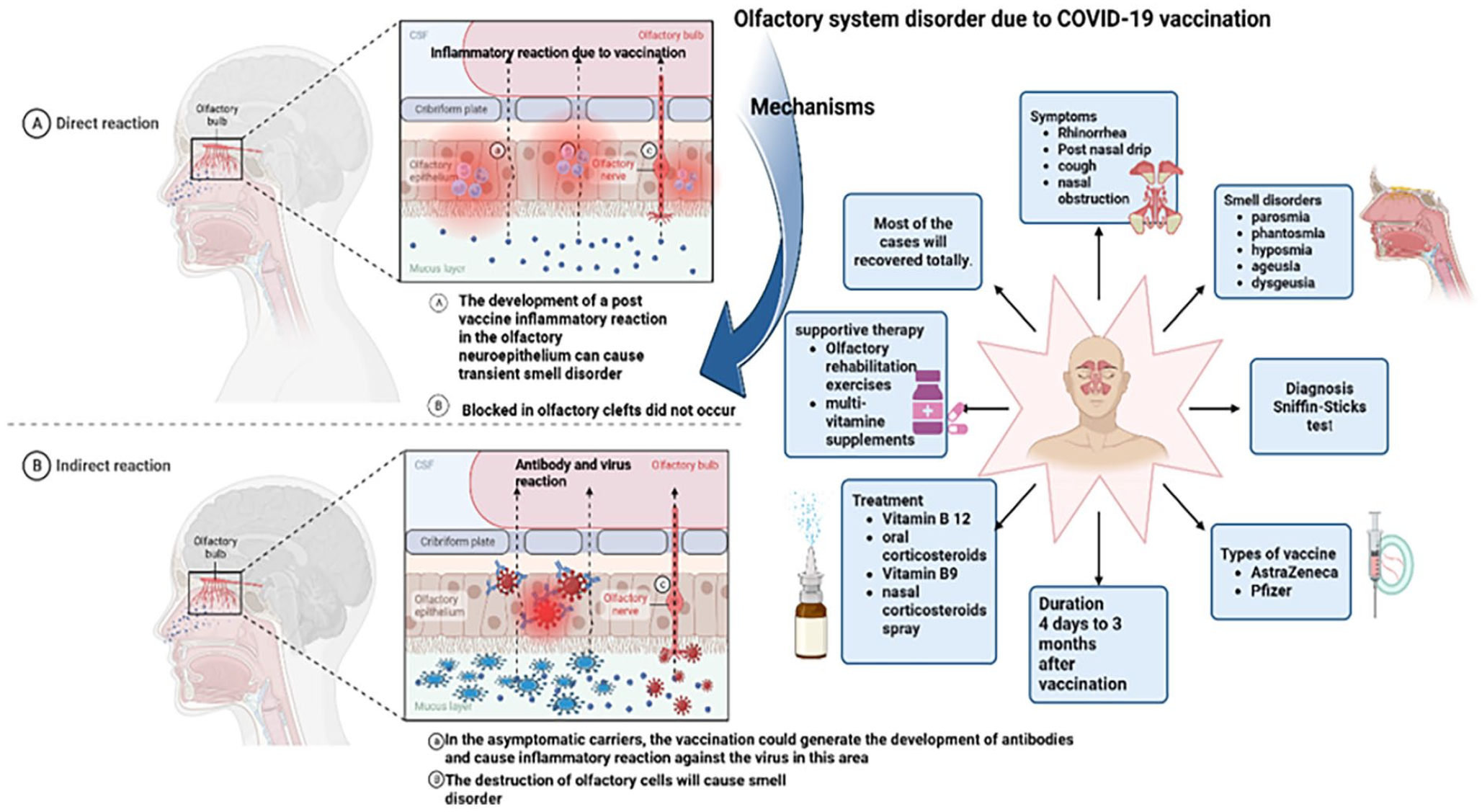
An overview of the mechanisms of olfactory system disorder following COVID-19 vaccination. COVID-19, coronavirus disease 2019.
Methods
This systematic review was performed to comprehensively review the studies that evaluated and reported anosmia or ageusia following COVID-19 vaccination. All the methods used for conducting this study were consisted in the Cochrane book and Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA).
Search Strategy
A systematic search was conducted in international bibliometric databases, including Web of Science, Scopus, Embase, and PubMed, from their inception to February 27, 2023. No limitation was imposed on the original language of the studies. We categorized the keywords into 2 groups, including the COVID-19 vaccine group and anosmia or ageusia group. In the COVID-19 vaccine group, we used any possible keyword, such as covid 19 vaccine, sars-cov-2 vaccine, human coronavirus 2019 vaccine, or 2019-ncov vaccine. In the anosmia or ageusia group, we used all possible keywords, including anosmia, loss of smell, ageusia, or gustatory loss. The keywords were combined with “AND” between the groups and with “OR” in each group.
Eligibility Criteria
We included any published study that evaluated the anosmia or ageusia after receiving the COVID-19 vaccine, including case reports, case series, letter to editor articles with reported cases regarding our topic, or observational studies with at least 1 eligible patient consistent with our criteria. We excluded the studies that reported anosmia or ageusia due to COVID-19 infection and non-COVID-19 vaccines.
Data Extraction and Quality Assessment
The screening of the identified studies was performed by 2 reviewers, independently based on their abstracts and titles, to exclude irrelevant studies. The full texts of the initially screened articles were then reviewed for confirmation of the inclusion criteria and the data extraction. Any discrepancy between the reviewers was resolved by a third reviewer. The data extraction was conducted using an Excel-based sheet by 2 reviewers, with the following items: (a) names of the first authors; (b) year of publication; (c) type of the study; (d) country of origin; (e) sample size; (f) age and gender of the patients; (g) vaccine type and dose; (h) the major disorder; (i) history of COVID-19 infection; (j) any symptom of the patients; (k) the main treatment for the disorder; and (l) the main outcomes. The methodological quality assessment of the included studies was performed using the Joanna Briggs Institute (JBI) critical appraisal tool by 2 independent reviewers. Any disagreement was resolved by a third reviewer.
Results
Overview of the Studies
The systematic search for the present study yielded 1069 articles. After removing duplicate articles, 661 remained for initial screening of their titles and abstracts. A total of 91 articles were selected and enrolled for evaluation of their full texts and 5 studies were included in this systematic review4,6-9 (Figure 2). The included studies were conducted in 5 different countries, including France (n = 1), 6 Germany (n = 1), 4 the United States (n = 1), 9 Italy (n = 1), 8 and Greece (n = 1). 7 All the included studies were published in 2021 and 2022. A total of 11 patients, including 8 females and 3 males with a mean age of 42.90 ± 14.96 years (range: 25-76), were evaluated. The characteristics of the articles included are presented in Table 1.
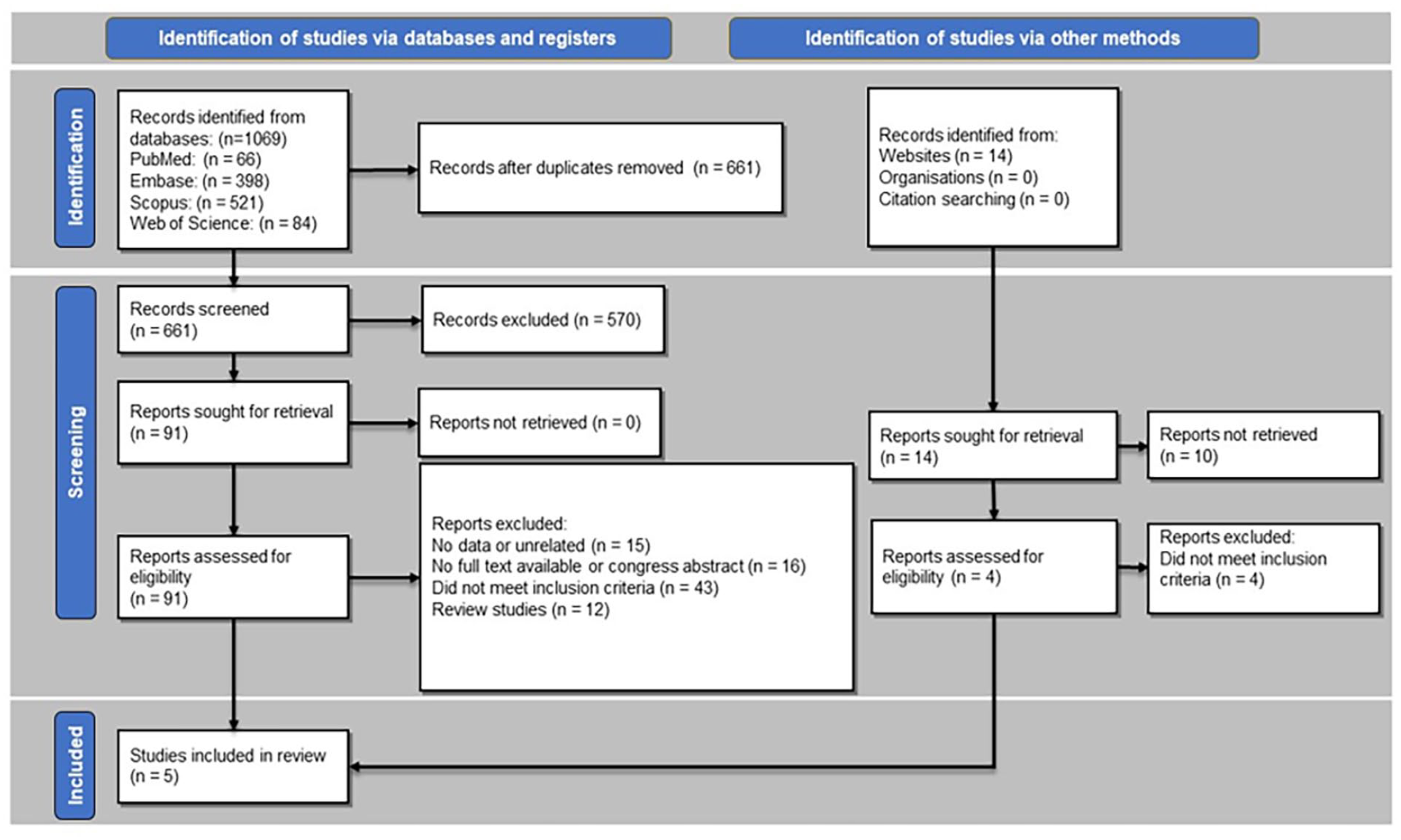
PRISMA flowchart of the literature search and selection of the articles. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Characteristics of Included Studies.
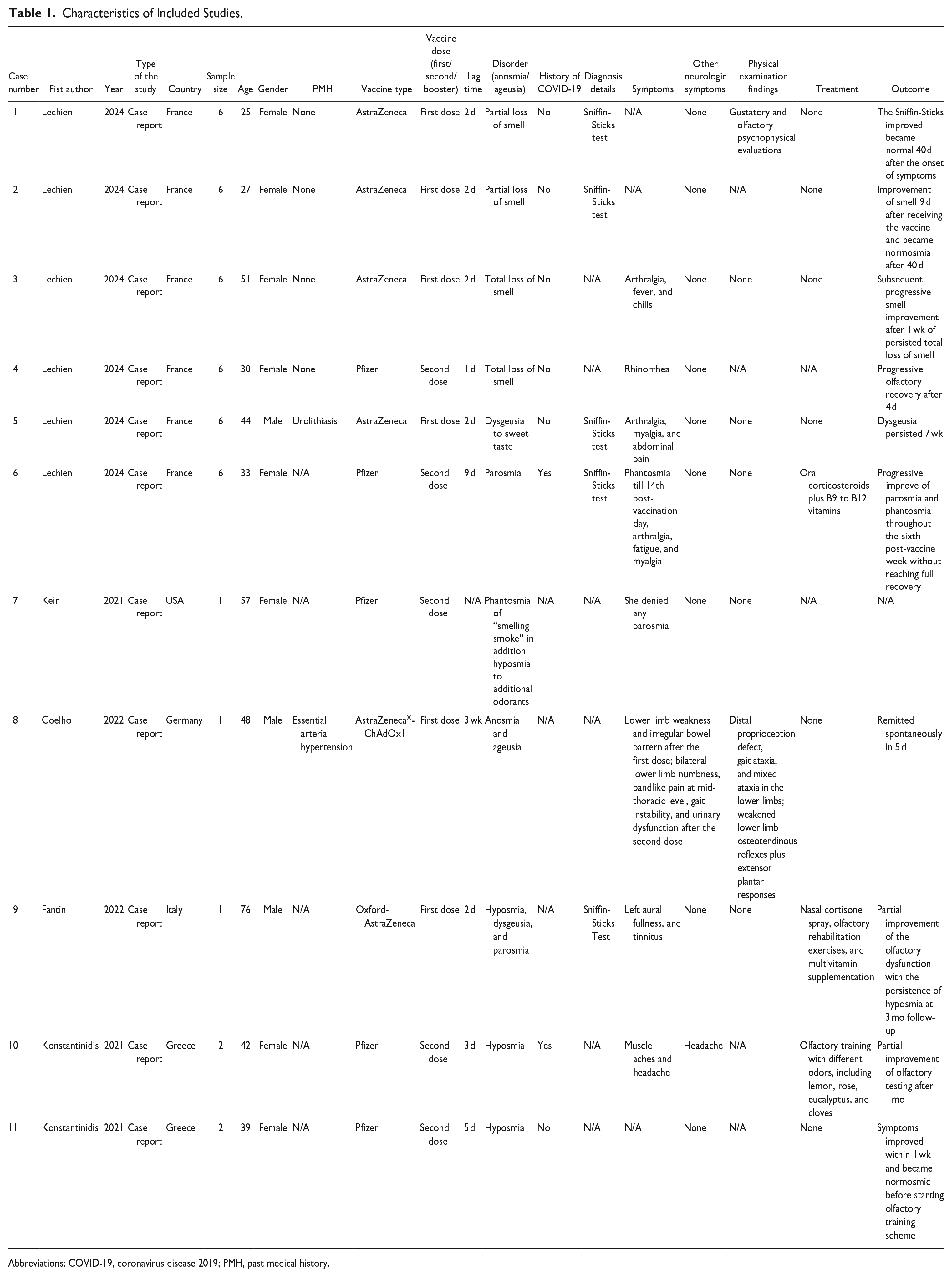
Abbreviations: COVID-19, coronavirus disease 2019; PMH, past medical history.
Vaccine Type
Of the 11 patients, 5 patients had received the Pfizer COVID-19 vaccine and 6 patients received the Oxford-AstraZeneca COVID-19 vaccine, of which 6 patients developed symptoms after the first dose of vaccination and 5 patients were symptomatic after the second vaccine dose.
Symptoms
Most of the patients developed symptoms within 1 week after the vaccination. However, 2 patients were symptomatic after 9 days and 3 weeks, respectively (Table 1).
The disorders of the patients included partial or total anosmia, parosmia, phantosmia, hyposmia, ageusia, and dysgeusia. Also, the patients had some symptoms in addition to smell or taste disorders, including arthralgia, fever, chills, rhinorrhea, myalgia, abdominal pain, fatigue, muscle weakness, altered bowel pattern, aural fullness, tinnitus, and headache.
Diagnosis and Treatment
The Sniffin-Sticks test was the diagnostic test in 2 studies,6,8 which was used for the diagnosis of anosmia or ageusia. No abnormal findings in the physical examinations of the patients were reported except in the study by Coelho et al, 4 which reported distal proprioception defect, weakened lower extremities reflexes, gait ataxia, and mixed ataxia in the lower extremities.
Most of the evaluated patients did not receive any treatment as for their disorders. However, 1 patient in the study by Lechien et al 6 received oral corticosteroids, vitamin B12, and vitamin B9. They reported that the parosmia and phantosmia of the patient improved after 6 weeks, although a full recovery was not achieved. Fantin and colleagues 8 reported a patient with hyposmia, dysgeusia, and parosmia after receiving the first dose of COVID-19 vaccine. A nasal cortisone spray, olfactory rehabilitation exercises, and multivitamin supplementation were prescribed for the patient. The olfactory dysfunction of the patient was partially improved after 3 months; however, the hyposmia was still present. In the study by Konstantinidis et al, 7 olfactory training was suggested regarding the hyposmia of the patient, which showed partial improvement after 1 month.
Discussion
Main Findings
This systematic review showed that anosmia and ageusia could appear as complications of COVID-19 vaccination. We have studied 5 articles, including 11 patients, of whom all received COVID-19 vaccines (including Pfizer or Oxford-AstraZeneca). After a while, they developed symptoms like partial or total anosmia, ageusia, parosmia, phantosmia, hyposmia, and dysgeusia. In 3 studies, patients have not shown any recovery despite receiving treatments (eg, corticosteroids, vitamin B12, and olfactory rehabilitation exercises).
Hypothesis
The development of losing smell and taste after the COVID-19 vaccination is an uncommon reaction. We discuss different hypotheses proposed to justify these complications. Most of them are based on structural changes. Central and peripheral demyelination is an infrequent neurological condition involving both CNS and PNS with demyelinating lesions. One trigger for this situation is infections and vaccination. Also, The SARS-CoV-2 spike protein can connect to the cell surface and activate this pathway. We can explain both anosmia and ageusia with the partial demyelination of related nerves. 4 The reduction in the olfactory bulb volume in MRI can explain another hypothesis. This has been reported in postinfection olfactory loss, but we must remember that the correct comparison cannot be done because of the lack of access to the previous MRI. 8 The third hypothesis is based on imaging findings, too. Imaging COVID-19 patients with anosmia/ageusia and comparing them to the radiographic findings of patients with olfactory problems after vaccination show similar results. Imaging COVID-19 patients indicates microvascular disruption, microhemorrhages, and olfactory CNS system damage. This hypothesis states that vaccines may have similar effects to the attachment of SARS-CoV-2 to related receptors (olfactory epithelial and perivascular ACE-2 receptors), causing the same inflammatory reaction. Both finally lead to olfactory edema and clumping of olfactory filia. Another hypothesis suggests that protein induced by mRNA vaccine (spike protein) can itself damage olfactory epithelium or can cause immune responses. The fifth theory for this reaction could be interpreted by the inflammation caused by the vaccine in the olfactory neuroepithelium and its blockage. Besides these structural hypotheses, we can assume that the presence of the virus and the person being a carrier at the time of vaccination are the reasons for this occurrence, and the vaccination was a trigger to activate the virus and its symptoms.
We have stated 6 relevant hypotheses in this matter, but our assumptions are only partially acceptable, and all need future studies. However, many reasons have yet to be proposed to explain ageusia, and most information is about anosmia.
Strengths and Limitations
Our article is the first systematic review in the field of COVID-19 vaccination and its relation to anosmia and ageusia. We operated this review using 5 data sources; we followed standard methods and used proper tools to reduce the risk of bias. Nevertheless, we need to admit that there are limitations. We did not hold on to a specific definition of anosmia or dysgeusia, and we tried to collect as much evidence as possible; against this decision, the search did not give us many results. Our selected reports represent confirming information, but they studied a limited number of patients. Also, there are limited tools to diagnose anosmia and ageusia, which could be a topic for future studies.
Conclusion
Anosmia and ageusia have been previously considered as viral infection symptoms; however, in the present study, we revealed that these symptoms can also be the consequences of COVID-19 vaccination. As a result, in examining the benefits and harms of the COVID-19 vaccine, its benefits are far more significant, and the risk of the discussed side effects is still infrequent. Also, more studies are needed to investigate the pathology and mechanisms of the aforementioned complications.
Footnotes
Data Availability
All relevant data are within the article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
