Abstract
Introduction
Globally, about 177,422 individuals are newly diagnosed with laryngeal carcinoma, which represents 1.5% of all cancers, and over 94,000 patients die from this disease each year. 1 From a pathological standpoint, squamous cell carcinoma (SCC) accounts for 95% of all laryngeal malignancies. Although the cure rates for early-stage laryngeal SCCs treated with radiotherapy or surgery alone are high, the advanced stage may lead to a substantial decline in 5-year overall survival (OS) rates: 74.4% for localized cases, 37.8% for regional cases, and 7.2% for metastatic cases. 2 The available treatment options for laryngeal squamous cell carcinoma (LSCC) remain limited, especially for patients who have not responded to or are unable to tolerate surgery and/or chemo-radiotherapy. In recent years, cancer immunotherapy targeting immune checkpoint inhibitors, for example, PD-1/PDL-1 inhibitors, has emerged as a novel and promising therapeutic strategy for head and neck SCCs.
IL-17 represents the hallmark cytokine of a subset of CD4+ T helper cells, known as T-helper 17 (Th17) cells, which are characterized by the presence of RORγt and activated through exposure to IL-23. The IL-23/IL-17 axis participates in the occurrence of autoimmune diseases, including psoriasis and arthritis. 3 Growing evidence suggests that the inflammatory mediators and cytokines are important components of the tumor microenvironment 4 and can directly influence the pathophysiological characteristics of human tumors. IL-17 may contribute to tumor development by stimulating chronic tissue inflammation. There is evidence that the IL-17 level in peripheral blood or cancer tissues was elevated in different human cancers, including breast cancer, esophageal cancer, cervical cancer, and laryngeal cancer.5-7 The development of LSCC is well known and associated with chronic inflammation caused by irritants such as long-term smoking, acid reflux, etc8,9. According to our previous study, patients with LSCC showed an increase in serum IL-17 concentrations. 10 IL-17 may participate in the regulation of tumor immunity by affecting cancer angiogenesis and modulating tumor-infiltrating lymphocytes. 3 However, the effect of endogenous IL-17 on tumor immunity is currently a subject of controversy, as suggested by recent studies conducted across a variety of tumor types. 11
We hypothesize that IL-17 expression can be a prognostic marker for LSCC. In our study, the expressions of vascular endothelial growth factor (VEGF), CD31 which is a surface marker of neovascular endothelial cells, and CD8 which is a surface marker of cytotoxic T lymphocytes, were detected in both LSCC and adjacent normal (AN) tissues by immunohistochemical staining (IHC), to explore the relationships between IL-17 expression and angiogenesis and cytotoxic T-lymphocyte infiltration.
Patients and Methods
Patients and Specimens
Between May 2010 and January 2013, 67 male LSCC patients who underwent curative resection at the Second Affiliated Hospital, Zhejiang University School of Medicine (Hangzhou, Zhejiang Province, PR China) were recruited as the study population. The clinical data were reviewed retrospectively. The exclusion criteria were as follows: the participants with a concomitant autoimmune disorder, had a malignant tumor in another organ, received any antitumor treatment such as chemotherapy and radiotherapy, or had an incomplete medical record. All patients underwent curative resection through partial or total laryngectomy with or without cervical lymph node dissections according to the NCCN Clinical Practice Guidelines in Oncology. Three female patients during the period were not included in this study because they did not undergo radical surgery. The LSCC and AN tissues were collected from all the participants. All tissue samples were embedded in paraffin to perform IHC and hematoxylin and eosin staining. Clinicopathologic variables, including age, the site of the tumor, TNM stage, differentiation, and outcome, are presented in Table 1. The staging of tumors was determined by the AJCC TNM classification system (Edition 8).
Correlations Between IL-17 Expression in LSCC Tissues and Clinical Variables.
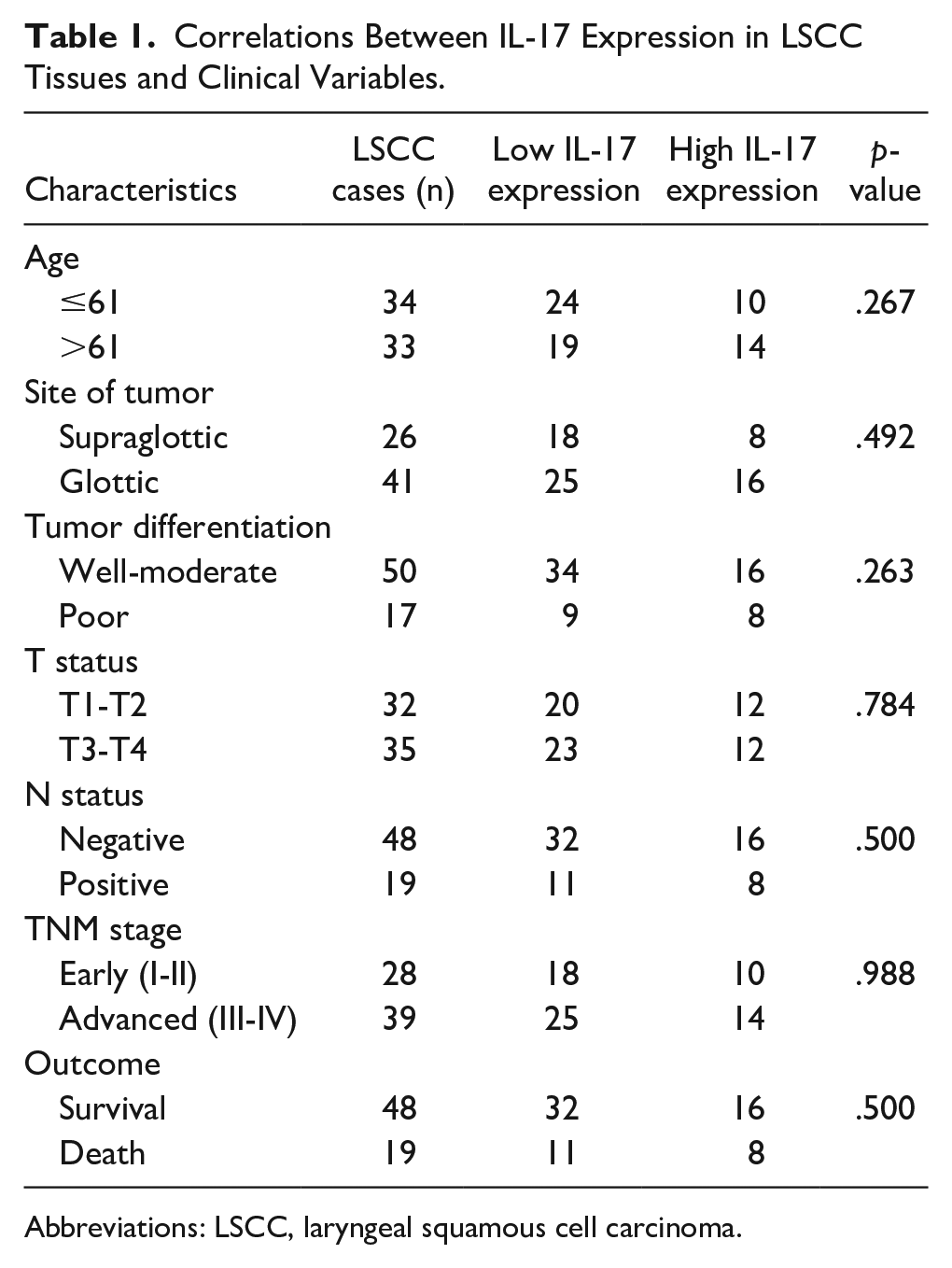
Abbreviations: LSCC, laryngeal squamous cell carcinoma.
OS period was calculated as the time interval between the operation and the decease of the patient. Data for living patients were censored at the date of the last follow-up, including outpatient follow-ups and telephone follow-ups.
This research was approved by the Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (2022-0061), and informed consent was obtained from each participant.
Immunohistochemistry
For IHC studies, the tissue section was deparaffinized, and subsequently rehydrated with graded ethanol. Microwave antigen retrieval was conducted in citrate buffer (pH 6.0). H2O2 (0.3%) was utilized for blocking endogenous peroxidase activities. Subsequently, primary antibodies including anti-human IL-17, IL-23, CD8, CD31, or VEGF were incubated overnight at 4°C. An Envision two-step system with diaminobenzidine (Dako A/S, Glostrup, Denmark) was used to perform the immunostaining. The stained slices were visualized by 3,3-diaminobenzidine tetrahydrochloride and then counterstained with hematoxylin.
Two pathologists who were blind to the clinical data assessed and scored the IHC grades for tissue sections. The number of positive-stained cells (PSCs) was counted in five high-power fields (400×), and the average number of PSCs was measured. The IHC scores of CD8 and VEGF were defined by the percentages of PSCs: 0% of PSCs was scored as 0, ≤25% as 1, 25%-50% as 2, 50%-75% as 3, and ≥75% as 4. Based on the low expression of IL-17 and IL-23 in our previous work, the IHC scores of IL-17 and IL-23 were defined as follows: 0% of PSCs was scored as 0, ≤10% as 1, 10%-25% as 2, 25%-50% as 3, and ≥50% as 4. If the IHC scores were different from the two pathologists, the final score was the average of two scores.
For microvessel quantitation, 12 a single, calculable microvessel was defined as any CD31-positive staining endothelial cells or endothelial cell cluster that were separated from adjacent microvessels, regardless of the presence of a vessel lumen. Microvessels with thick muscular walls or lumen diameters greater than 8 red blood cells were not counted. The slides were examined at 100× magnification to locate the hotspot area with the highest vascular density within the tumor. The microvessel density (MVD) was determined as the average microvessel number in the five most vascular regions under 200× magnification.
Statistical Analysis
Data were cut off by the median value for all immunohistochemical variables. The Pearson chi2 test was employed to determine the relationships among immunohistochemical variables and clinicopathological features. Correlations between variables with different properties were assessed by Spearman correlation analysis. Cumulative survival time was measured using the Kaplan-Meier approach and then compared using the log-rank test. The univariable and multivariable Cox proportional-hazards models were applied to identify the prognostic factors for OS. The variables with P < .20 in the univariable Cox proportional-hazards model were further analyzed by multivariable analysis using a forward stepwise selection model. P < .05 was deemed significant. All statistical tests were conducted with SPSS software v19.0 (SPSS Inc, an IBM Company, USA).
Results
Patient Characteristics
The clinical features of 67 patients are displayed in Table 1. All patients were male, with a median age of 61 years (range, 44-76 years). In all, 26 patients (38.8%) were with supraglottic LSCC, 41 (61.2%) were with glottic LSCC, and none were with subglottic LSCC. None of these patients had distant metastases before initial treatment, and 19 (28.4%) had cervical lymph node metastasis (CLNM). In the initial diagnosis, 28 (41.8%) and 39 (58.2%) patients had early (I/II) and late (III/IV) stages.
IL-17 PSCs are Elevated in LSCC Tumors
To detect the expression of IL-17 in both LSCC and AN tissues, we conducted IHC for IL-17 on paraffin-embedded tissues. IL-17 PSCs were observed in both LSCC and AN tissues, mainly in the tumor stroma (Figure 1). The nonparametric test revealed that the immunochemistry score of IL-17 was remarkably higher in LSCC tissue than those in AN tissue (P < .001). The immunochemistry scores of IL-23, CD8, VEGF, and MVD in LSCC tissue were also markedly higher than in AN tissue (P < .001).
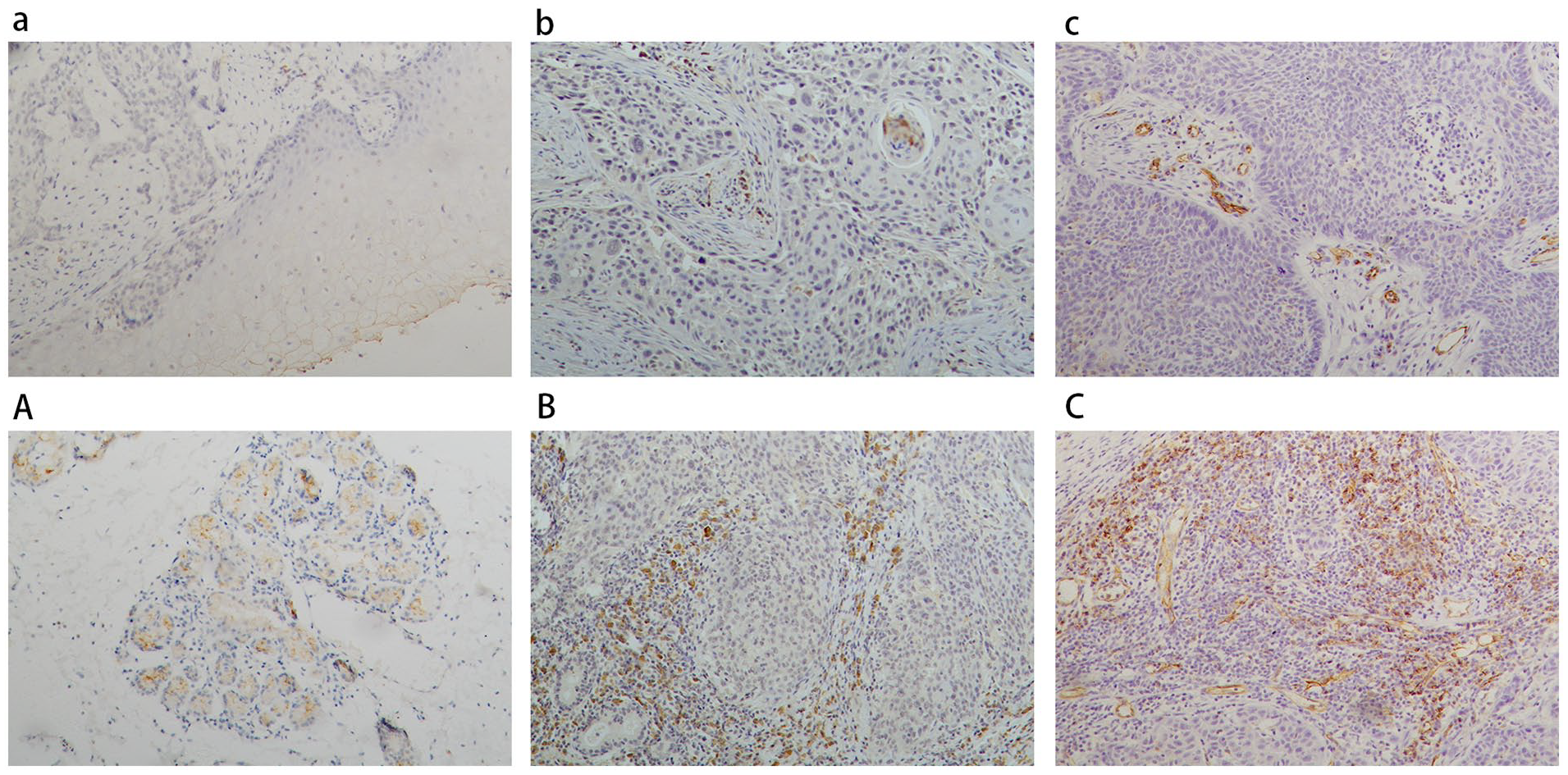
Representative pictures showing immunohistochemical staining of IL-17, IL-23, and CD31 in LSCC tissues (×100). Low (a–c) and high (A–C) expression of IL-17, IL-23, and CD31. LSCC, laryngeal squamous cell carcinoma.
Next, the correlations among IL-17, IL-23, CD8, CD31, and VEGF were evaluated (Figure 2). IL-17 expression was positively correlated with IL-23 expression in both LSCC (ρ = .293, P = .016) and AN (ρ = .260, P = .034) tissues. However, IL-17 expression was not correlated with CD8, VEGF, and MVD in LSCC and AN tissues. VEGF expression was positively correlated with MVD (ρ = .244, P = .046) in LSCC tissues.
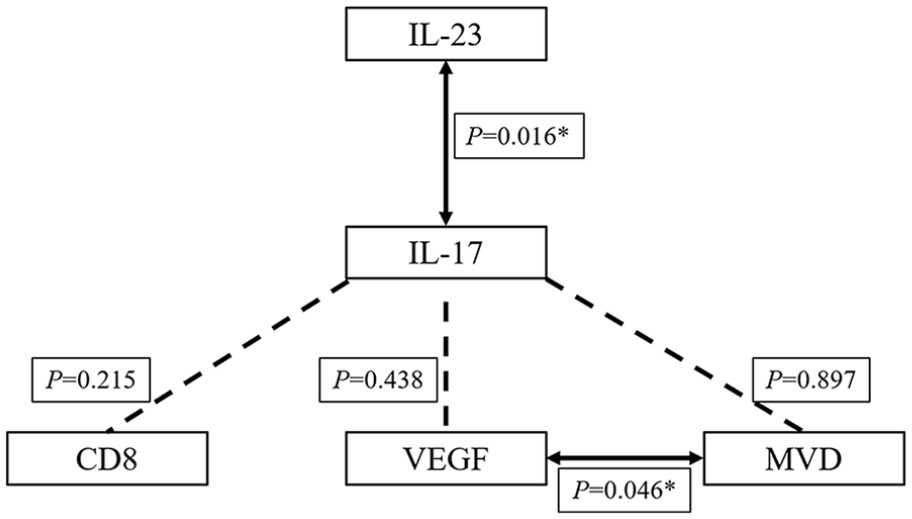
The relationships between expression of IL-17 and IL-23, VEGF, CD8, MVD in LSCC tissues using Spearman correlation analysis. The solid arrows represent significant correlations. Dashed lines represent no significant correlation. LSCC, laryngeal squamous cell carcinoma; MVD, microvessel density; VEGF, vascular endothelial growth factor.
Association Between IL-17 Expression and Clinical Features
The relationships between clinical characteristics and IL-17 expression are presented in Table 1. However, IL-17 expression did not correlate with age, site of tumor, TNM stage, T status, N status, and tumor differentiation (all P > .05).
Prognostic Factors for Survival Analysis
The median follow-up period was 84 months (range, 2-113 months). The OS rate at 5 years was 75.0% for the entire cohort. The Kaplan-Meier analysis indicated that patients with CLNM (P < .001), advanced TNM stage (P = .025), poor-differentiated grade (P = .005), higher MVD in LSCC tissues (P = .009), and higher IL-17 expression in AN tissues (P = .039) had poorer OS (Figure 3). However, LSCC patients with IL-17 upregulation did not show significantly different OS compared to those with IL-17 downregulation.
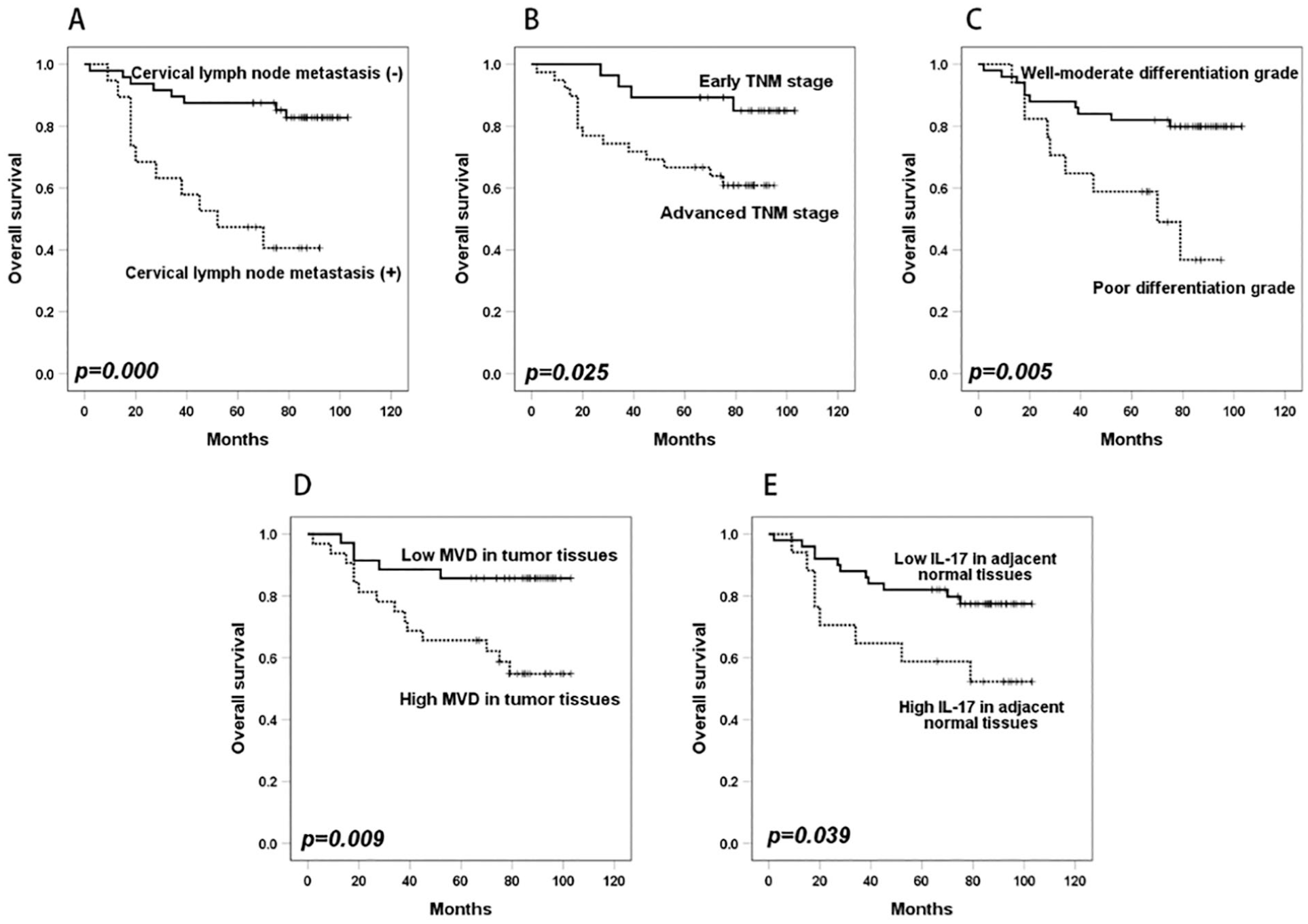
Kaplan-Meier survival curves for overall survival according to the status of CLNM (A), TNM stage (B), differentiation grade (C), microvessel density in LSCC tissues (D), and IL-17 expression in AN tissues (E). CLNM, cervical lymph node metastasis; LSCC, laryngeal squamous cell carcinoma.
Site (P = .141), tumor differentiated grade (P = .009), N status (P = .001), TNM stage (P = .035), IL-17 expression in AN tissues (P = .048), CD8 expression in AN tissues (P = .196), and MVD in tumor tissues (P = .015) in the univariable Cox proportional-hazards model were included in multivariable analysis (Table 2). It was found that CLNM (HR = 7.246; 95% CI: 2.598-20.208; P < .001), high MVD in tumor tissues (HR = 4.233; 95% CI: 1.482-12.092; P = .007), and low CD8 expression in AN tissues (HR = 3.996; 95% CI: 1.442-11.074; P = .008) were significantly associated with worse OS in LSCC, while IL-17 expression did not reveal prognostic significance (Table 2).
Univariable and Multivariable Analyses of the Prognostic Variables in LSCC.
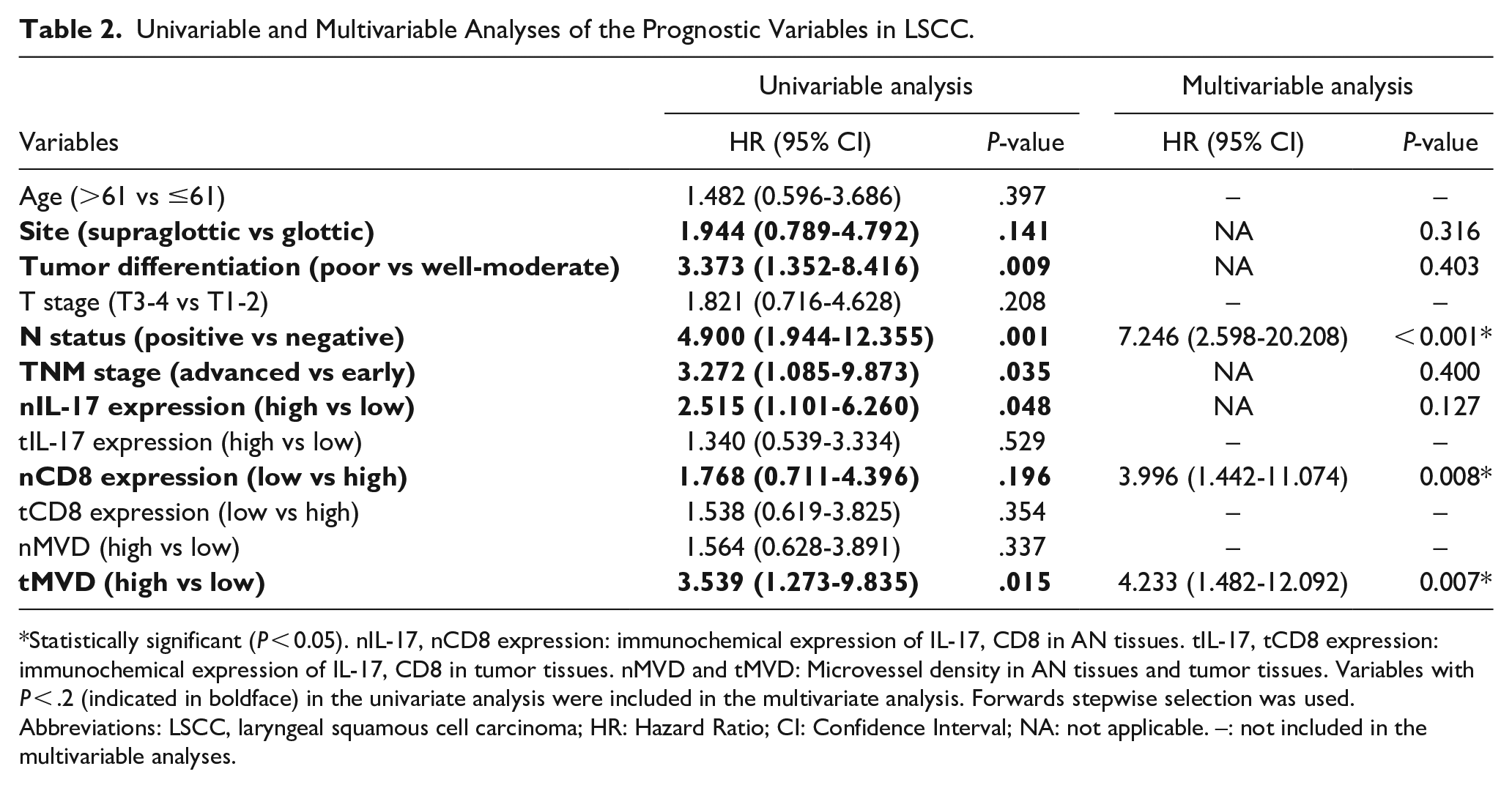
Statistically significant (P < 0.05). nIL-17, nCD8 expression: immunochemical expression of IL-17, CD8 in AN tissues. tIL-17, tCD8 expression: immunochemical expression of IL-17, CD8 in tumor tissues. nMVD and tMVD: Microvessel density in AN tissues and tumor tissues. Variables with P < .2 (indicated in boldface) in the univariate analysis were included in the multivariate analysis. Forwards stepwise selection was used.
Abbreviations: LSCC, laryngeal squamous cell carcinoma; HR: Hazard Ratio; CI: Confidence Interval; NA: not applicable. –: not included in the multivariable analyses.
Discussion
It is well known that chronic inflammation can promote immune tolerance, tumor progression, and metastasis. Previous reports have proven a significant association between laryngeal cancer and chronic inflammation, particularly due to smoking and acid reflux. 9 The role of IL-17, as a pro-inflammatory factor, remains to be studied in LSCC. This retrospective research indicated that IL-17 was highly expressed in AN tissue, rather than in LSCC tissue, which could indicate worse survival in LSCC, as well as CLNM, advanced TNM stage, poor-differentiated grade, and higher MVD in tumor tissues. However, multivariate analysis verified that IL-17 expression was not an independent prognostic factor for LSCC.
Despite that inflammation is tightly correlated to cancer oncogenesis and progression, the effect of the pro-inflammatory cytokine IL-17 on malignancies remains a controversial issue. This retrospective study confirmed elevated expression of IL-17 in LSCC tissues. A meta-analysis 11 reviewed a series of literature on the relationships between IL-17 and OS in different types of human malignancies and showed that upregulation of IL-17-producing cells in tumor tissues using immunohistochemistry was associated with worse OS in 18 out of 27 studies. Two previous studies in laryngeal cancer also reported that IL-17 upregulation in LSCC tissue was related to poor prognosis.13,14 Interestingly, no correlation was observed between IL-17 expression and LSCC prognosis in this study. Instead, we observed that high IL-17 expression in AN tissues was associated with worse OS. Apart from the tumor tissues, IL-17 was overexpressed at the tumor-stroma interface. 15 Zhang and co-workers 16 also found that IL-17 expression in tumor tissues and margins of oral SCC was remarkably increased compared to the normal tissues by IHC and that IL-17 expression in the adjacent stroma was positively correlated with tumor budding located in the tumor invasion front. Tumor budding, defined as the occurrence of sporadic or small clusters of cancer cells at the invasive margin of the tumor, has been confirmed as a prognostic biomarker for different types of tumors, predictive of tumor progression and unfavorable survival. 17 This may be one of the mechanisms underlying the correlations between IL-17 expression and worse prognosis in LSCC.
The effect of IL-17 on tumorigenesis remains ascertained. For example, it has been suggested that IL-17 exerts a pro-tumor effect by increasing tumor angiogenesis 18 and inhibiting infiltration of CD8+ T lymphocytes,19,20 or an antitumor effect by increasing IFN-γ-producing NK and T cells. 21 Angiogenesis is an important hallmark of malignancy and is closely associated with prognosis. Previous reports have demonstrated that IL-17 upregulation is markedly related to high MVD in different types of malignancies, including non-small-cell lung carcinoma, hepatocellular carcinoma, colorectal carcinoma, and ovarian cancer. Our study also confirmed that MVD was an independent prognostic factor for OS of LSCC; however, IL-17 expression did not show a significant correlation with VEGF expression and MVD in both LSCC and AN tissues. Since angiogenesis is mediated by dynamic changes between pro- and anti-angiogenesis factors, IL-17 may be not the major pro-angiogenic factor in the complex and dynamic tumor microenvironment of LSCC.
CD8+ T lymphocytes are important antitumor effector cells that induce tumor-specific cytolysis. There is an association between CD8+ T lymphocyte infiltration and prolonged patient survival time in many human cancers. 20 Previous in vitro experiments and tumor-bearing mouse models have confirmed that IL-17 can inhibit the infiltration of CD8+ T lymphocytes and exhibit a suppressive effect on CD8+ T-cell responses.19,20 In our study, low CD8 expression in AN tissues was associated with poorer OS in LSCC by Cox regression analysis, which is consistent with the antitumor effect of CD8+ T-lymphocyte infiltration. CD8 expression in tumor tissues did not show a correlation with OS. This may be attributed to the fact that CD8+ T lymphocytes in the AN tissues reflect the frontline counter-attack of antitumor immunity against tumors, while CD8+ T lymphocytes in the tumor microenvironment show functional exhaustion due to chronic exposure to tumor antigens. However, IL-17 expression was not significantly associated with CD8 expression in our study.
While not initially mandatory, IL-23 plays a crucial role in the terminal differentiation and maintenance of Th17 cells. Our findings also proved the significant correlation between IL-17 and IL-23 expression. Previous research has demonstrated that IL-23 is responsible for tumor immunosurveillance, and IL-23R overexpression in tumor cells is associated with an unfavorable prognosis. 22 Herein, IL-23 expression was not associated with the clinical features and OS of LSCC (data not shown).
Nevertheless, there were a few limitations in this research. A small sample size of this study might compromise the results of Cox proportional-hazards regression models. Thus, more efforts should be focused on the specific cell subpopulation expressing IL-17, and the actual role of IL-17 in the development of LSCC.
In summary, we confirmed that IL-17 expression was upregulated in both tumor tissue and AN of LSCC. The univariable Cox regression showed that IL-17 upregulation in AN tissue was related to worse prognosis, while the multivariable Cox regression indicated that it may not be an independent prognostic factor. In addition, CLNM, higher MVD in LSCC tissues, and CD8 expression in AN tissues were independent prognostic factors for patients with LSCC.
Footnotes
Data Availability Statement
The datasets generated and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Zhejiang Provincial Natural Science Foundation of China under Grant No. LQ20H130001 and No. LY23H130002.
Ethical Approval
The study was approved by the Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine (2022-0061).
