Abstract
Keywords
Introduction
Turicella otitidis and Staphylococcus auricularis are part of the normal flora of the external auditory canal. 1 Isolation of T. otitidis and S. auricularis from otorrhea in patients presenting with active infection is considered normal and not a treatment target. 1 Treatment is focused on common pathogens: Streptococcus pneumoniae, Haemophilus influenzae, Moraxella catarrhalis, and Pseudomonas aeruginosa.
Prior reports demonstrate the possible pathogenicity of T. otitidis and S. auricularis, while the degree remains controversial.1-3 These cases focused on pediatric patients. One study isolated T. otitidis via spontaneous drainage or direct tympanocentesis from 7 pediatric patients presenting with otitis media. 2 Another case report described a 3-year-old female with a posterior auricular abscess; T. otitidis was isolated in pure culture in the setting of obvious infection. 3 The ability of T. otitidis to cause inflammation in a location other than the external auditory canal points toward possible pathogenicity and its ability as an organism to cause infection.
While T. otitidis is generally considered normal aural flora in the external auditory canal and found in 12% of healthy external auditory canals in young adults (ages 19-22 years), 4 its prevalence in middle ear fluid is lower at 6.3% based on a cohort of pediatric patients in Madrid. 1 A total of 71% of those with T. otitidis in middle ear fluid had a history of prior perforation. 1 Comparatively, S. auricularis is found in 20% of healthy external auditory canals in young adults (ages 19-22 years), making it a common organism. 4 However, the pathogenicity of S. auricularis has been described such as its capacity to form biofilms on implanted medical devices and ability to adhere to and destroy epithelial cells. 5 There have been little to no reports describing S. auricularis in middle ear fluid or its role in otitis media.
Despite a few prior case reports describing the potential pathogenicity of the organisms that were considered normal aural flora, no study has synthesized the clinical presentation, treatment outcomes, and sensitivity results of the organisms. This case series presents 13 patients with otorrhea whose fluid culture isolated T. otitidis and S. auricularis with their clinical history, examination findings, and outcomes.
Patients and Methods
A retrospective chart review was performed over a 5 year period (2017-2022) at a tertiary surgery center in Minnesota to identify patients who had active otorrhea on otologic microscopy examination, and the culture from the external auditory canal isolated T. otitidis or S. auricularis. Eligible patients were aged 18 years and older. Patient selection is outlined in Figure 1. This study was approved by the University of Minnesota Institutional Review Board (STUDY00016223).
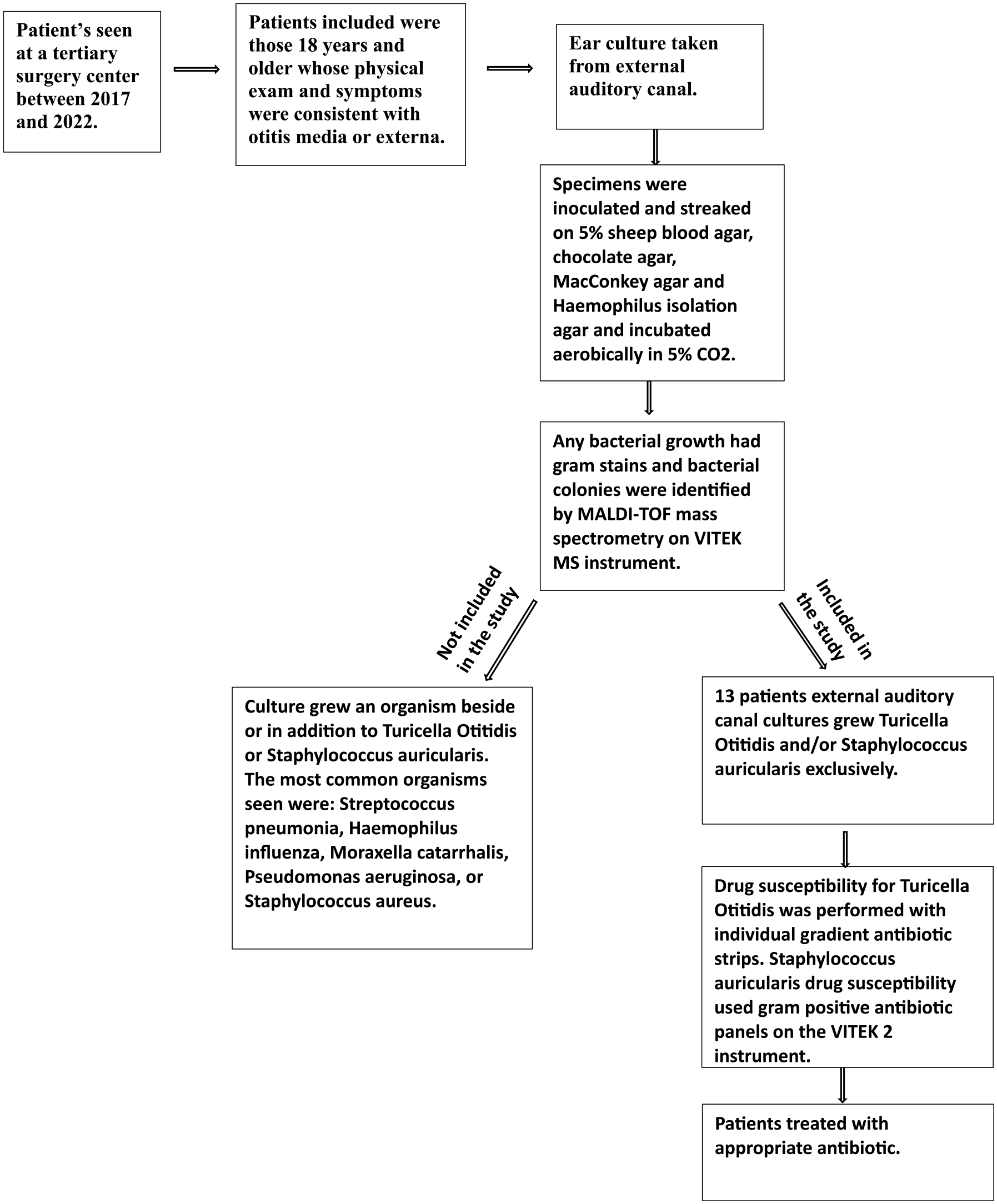
Algorithm for patient selection.
Patient demographics (age, sex, and race) and detailed clinical characteristics (history, otologic microscopy examination, prior history of ear surgeries, presence of tympanic membrane perforation, interventions given, and outcomes) were collected from chart review.
The isolation technique for T. otitidis and S. auricularis after external auditory canal collection was as follows. The specimens were inoculated and streaked on 5% sheep blood agar, chocolate agar, MacConkey agar, and Haemophilus isolation agar, and incubated aerobically in 5% CO2. Growth was observed at daily intervals. Any bacterial growth had gram stains performed, and bacterial colonies were identified by MALDI-TOP (matrix-assisted laser desorption/ionization time of flight) mass spectrometry on a VITEK MS instrument (bioMerieux). Any bacteria that was written as “normal flora” was then speciated to ensure all flora isolated were specifically named.
Antibiotic sensitivity results were available among a subset of patients per discretion of the microbiology laboratory at the time of culture. The drug susceptibility testing for T. otitidis used individual gradient antibiotic strips, the ETEST (bioMerieux). S. auricularis used the gram-positive antibiotic panels on the VITEK 2 instrument (bioMerieux). Antibiotics were then chosen based on these results.
Descriptive statistics and logistic regression analysis was performed to explore the factors associated with resolution of otorrhea.
Results
Patient characteristics are summarized in Table 1. The mean age was 38 years (SD: 17.8, range 18-79 years), and 62% were female. Majority of patients had a history of prior otologic surgery [including pressure-equalizing (PE) insertion; (n = 12, 92%)] and/or tympanic membrane perforation (n = 8, 61.5%). Only 4 of the patients had a major otologic surgery (30.8%).
Summary of Case Report Patient Characteristics (N = 13).
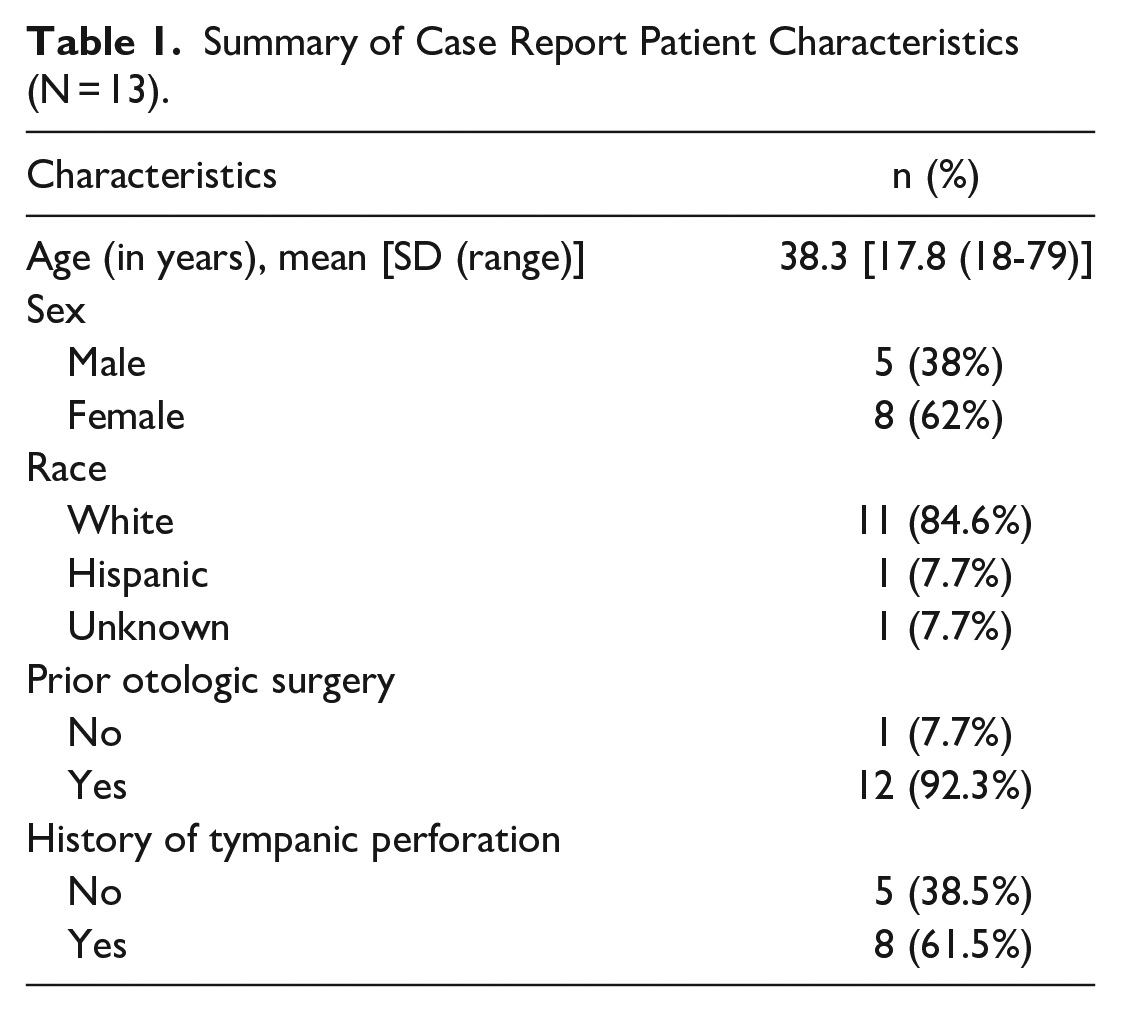
Table 2 summarizes the patient history, the otologic microscope findings, interventions, and outcomes. There was evidence of active infection on examination for all patients, while the otorrhea culture only grew T. otitidis among 10 patients and S. auricularis among 3 patients. Common pathogens accountable for otitis media or externa like Streptococcus pneumonia, Haemophilus influenzae, Moraxella catarrhalis, Pseudomonas aeruginosa, or Staphylococcus aureus were not cultured. In our clinical practice, P. aeruginosa has been the most common bacteria in patients with chronic ear disease.
Summary of Patient Clinical History, Physical Examination Findings, Treatment, and Outcomes.
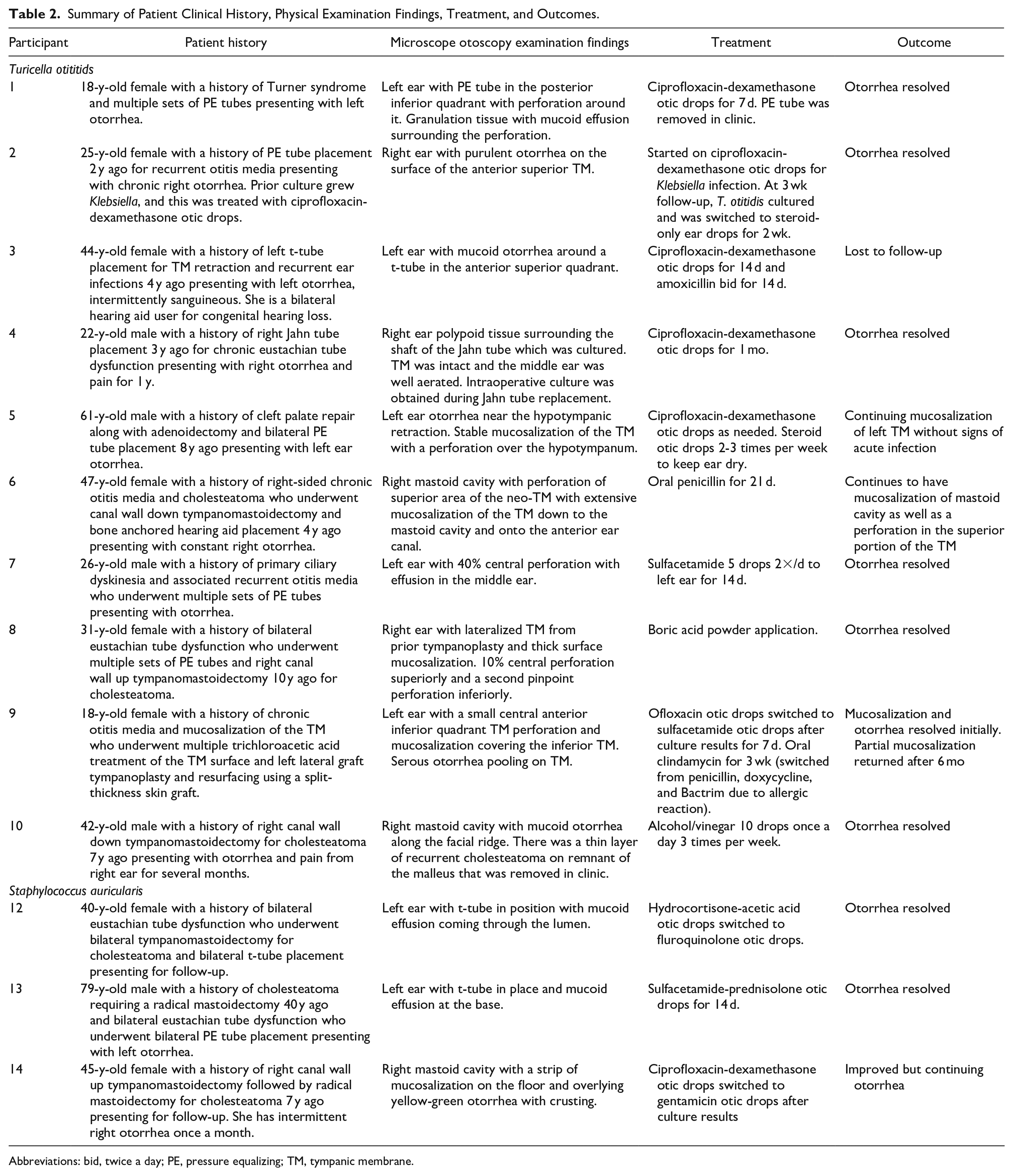
Abbreviations: bid, twice a day; PE, pressure equalizing; TM, tympanic membrane.
Ten out of 13 (77%) patients took antibiotic otic drops. Among the 10 patients whose culture isolated T. otitidis, 6 patients started fluoroquinolone-based otic drops, and 1 patient began sulfacetamide otic drops. Two patients completed a course of oral antibiotics: amoxicillin or penicillin. Among 3 patients whose ear canal culture isolated S. auricularis, 2 patients were started on fluoroquinolone-based otic drops and 1 on sulfacetamide otic drops. The majority of the drainage resolved after treatment. However, in 4 patients (31%), drainage and mucosalization of the tympanic membrane returned after treatment. Logistic regression analysis was performed to explore the factors associated with resolution of otorrhea. There was no significant association of otorrhea resolution with age, sex, history of perforation, or surgery. Interpretation of the results were limited due to low sample size.
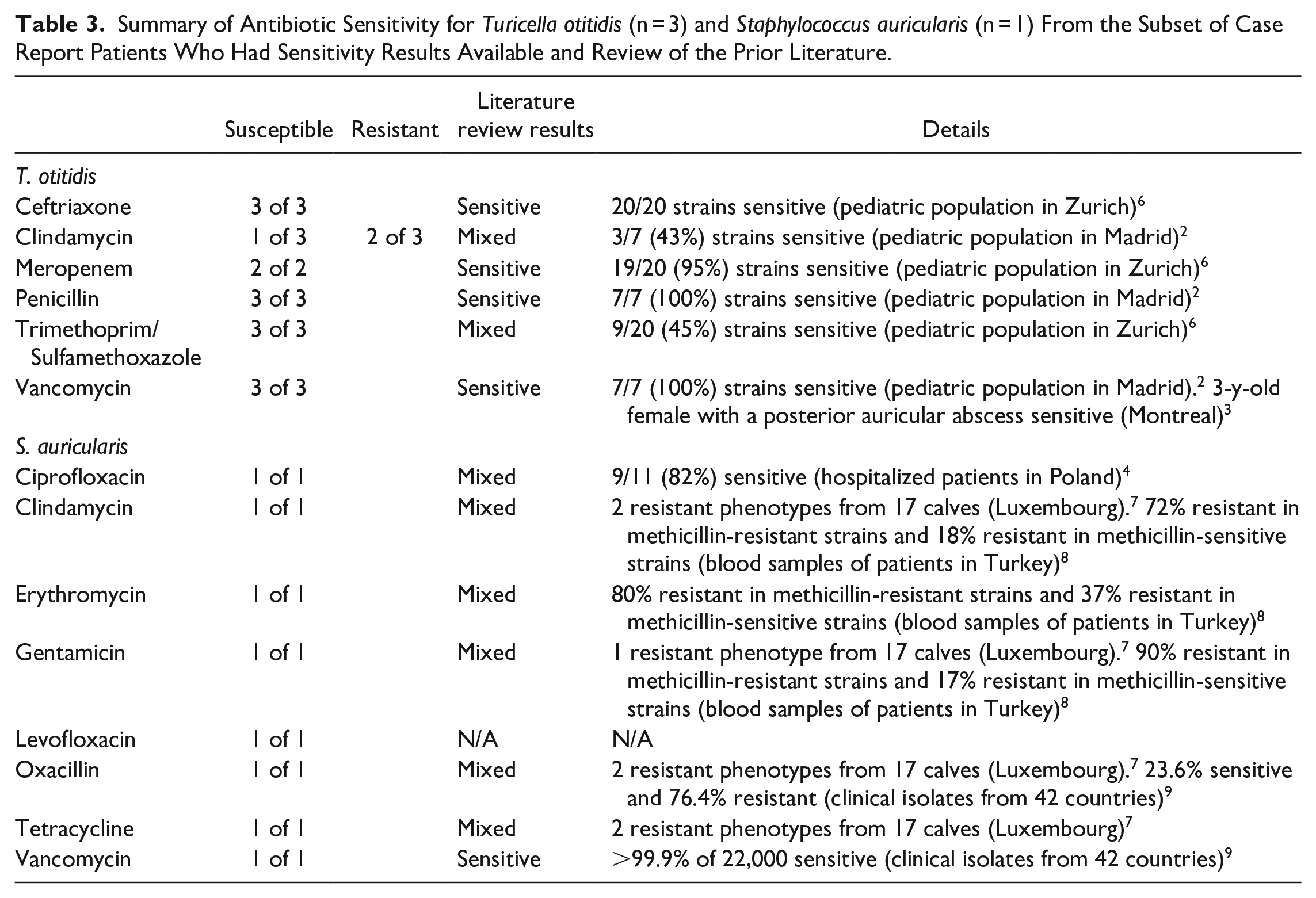
Table 3 summarizes the antibiotic sensitivity results against T. otitidis and S. auricularis among a subset of patients. Of the 10 patients whose otorrhea isolated T. otitidis, 3 patients had sensitivity results available. All 3 strains were sensitive to ceftriaxone, penicillin, trimethoprim/sulfamethoxazole, and vancomycin. Two strains were resistant to clindamycin. One patient had sensitivity results available for S. auricularis. This strain of S. auricularis was sensitive to ciprofloxacin, clindamycin, erythromycin, gentamicin, levofloxacin, oxacillin, tetracycline, and vancomycin, and was not resistant to any tested antibiotics. Additional antibiotic sensitivity results for both pathogens based on prior literature are summarized in Table 3.
Summary of Antibiotic Sensitivity for Turicella otitidis (n = 3) and Staphylococcus auricularis (n = 1) From the Subset of Case Report Patients Who Had Sensitivity Results Available and Review of the Prior Literature.
Discussion
We report a series of patients who had evidence of active ear infection on the examination while the culture of the otorrhea isolated only T. otitidis or S. auricularis. Prior case reports suggest T. otitidis and S. auricularis as pathogens of complicated otitis media including mastoiditis, 1 subperiosteal abscess, 3 and otorrhea with maxilla-labio-palatine cleft surgery. 3 Our findings add to the prior reports and suggest T. otitidis and S. auricularis as potential pathogens of the ear despite the standard of care considering them normal aural pathogens that do not merit treatment.
The previous notion that T. otitidis was not pathogenic comes from findings of a low rate of T. otitidis cultured in the middle ear effusion compared to the external auditory canal of children with otitis media with effusion. T. otitidis was found simultaneously in both the external auditory canal and middle ear effusion in only 10% of patients. 10 This led to the conclusion that T. otitidis grows exclusively in the external auditory canal and should not be a treatment target. 10
In this cohort, all patients presenting with otorrhea with isolation of T. otitidis and S. auricularis had a prior history of otologic surgery and/or tympanic membrane perforation. The etiology for each patient’s tympanic membrane perforation is not completely understood, it does suggest that a violation of the ear canal skin from previous surgery or tympanic membrane perforation may change the microbiome in the canal and middle ear, causing normal aural flora to become pathogenic. The most common surgeries among this case series were PE tubes and tympanomastoidectomy.
A perforated tympanic membrane also provides a route of transmission for T. otitidis and S. auricularis. Prior studies found that a history of tympanic membrane perforation increases the incidence of T. otitidis in the middle ear fluid. 1 T. otitidis does not colonize the nasopharynx or ascend from the eustachian tube to the middle ear as other common pathogens of otitis media. 1 Less than 1% of nasopharyngeal swabs showed T. otitidis, 11 suggesting that tympanic membrane perforation may allow T. otitidis to colonize the middle ear space, leading to an active infection.
Studies have focused on the possible pathogenicity of T. otitidis in otitis media, leaving the role of S. auricularis in question. The similarities of S. auricularis to T. otitidis in habitat and clinical presentation strongly suggest that prior otologic surgeries or tympanic membrane perforation could leave the middle ear susceptible to S. auricularis infection. Past research found S. auricularis to have pathogenic potential through adherence to epithelial cells, biofilm formation, and destruction of human cells. 5 This was determined from bacterial strains isolated from hospitalized patients and biofilm formation was found using crystal violet. 5 One patient in the cohort had an osseointegrated middle ear ventilating tube in bilateral ears which may have created an environment for S. auricularis to form biofilm and become pathogenic. S. auricularis has been isolated in 2.5% of cases with otitis media. However, when present in middle ear fluid, it was present in a larger quantity which suggested its ability to replicate in the middle ear, contributing to its infectious potential. 12
The sensitivity results for T. otitidis from our cohort were similar to prior studies that reported sensitivity to tetracycline and amoxicillin and resistance against clindamycin. Clinical samples over 5 years in Zurich reported resistance against sulfamethoxazole. 1 Varying sensitivity of different strains of T. otitidis should be considered when deciding on a course of antibiotic otic drops or oral antibiotics. The sensitivity of S. auricularis is less documented. It is sensitive to tigecycline with rifampin when growing on implanted medical devices. 5 We did not find any antibiotic resistance for S. auricularis in our patients.
There are several limitations in this study. First, the small sample size limits our ability to determine the pathogenic potential of T. otitidis and S. auricularis (n = 13). Still, our report represents the largest series of adult patients presenting with active infection and isolation of T. otitidis and S. auricularis. Second, our report is a retrospective chart review. We are unable to establish the causality of the active infection from the pathogens isolated from culture. There may have been other pathogenic organisms that were not isolated from the culture due to sampling errors of the swab or these pathogens may have been isolated from contamination. The lack of longitudinal culture results at multiple time points from this study also makes it difficult to establish causality. Third, sensitivity results were available among a limited subset of patients as sensitivities are not typically obtained for these organisms in our laboratory and required a separate call to the microbiology laboratory to run sensitivity studies. Further study including a larger number of patients and strains of T. otitidis and S. auricularis may demonstrate other antibiotic resistance.
Our findings add to prior literature suggesting T. otitidis and S. auricularis are potential pathogens of otologic infections. Treatment against these organisms warrants consideration when isolated, especially among patients with a history of prior otologic surgery or tympanic membrane perforation. Further research is needed to examine the degree of pathogenic potential of T. otitidis and S. auricularis and their role in acute and chronic otitis media.
Footnotes
Acknowledgements
The authors thank the technical staff of the Infectious Diseases Diagnostic Laboratory at the University of Minnesota Medical Center, MHealth Fairview, and the senior Medical Director, Patricia Ferrieri, MD.
Data Availability
Data supporting this case series are not publicly available due to ethical and legal reasons as it contains sensitive patient information that patients, in this case series, have not released to the public.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
