Abstract
Tularemia is a rare disease but shows an approximately 10-fold increase in reported cases over the last 15 years in Germany. Clinical symptoms of acute tularemia infection are various, which often delays diagnosis. This case report gives an overview of the clinical manifestations of acute tularemia and shows the importance of interdisciplinary work to shorten the time from the onset of symptoms to effective treatment in infection with Francisella tularensis. Since some cases of tularemia are life-threatening, early diagnosis is vital. This case report serves as a reminder that rare diseases need to be considered in cervical lymphadenopathy.
Introduction
Tularemia is a zoonotic disease caused by the gram-negative, facultative intracellular coccobacilli Francisella tularensis. 1 The bacteria is found in over 250 different animal species but the main reservoir and important source for human infection are wild animals, such as small rodents.2,3 Although F. tularensis is not spore-forming, it has a high ability to survive in humid and cool environments over many months. 4 Therefore, arthropods, for example, ticks and mosquitoes, are also possible vectors for human infection, either by transmission of contaminated environmental water sources or through direct transmission from the animal reservoir.5-7 Three major ways of human pathogen transmission are known: direct transmission from the animal reservoir (ingestion of meat, handling, and bites of infected animals), ingestion of contaminated environment (eg, water sources), or through arthropods (eg, tick bites, mosquito bites).
Four subspecies of the bacteria are known. These occur with variable prevalence mainly in the northern hemisphere of the world: Subsp. tularensis (primarily northern America), subsp. holarctica (primarily Europe), subsp. mediaasiatica (primarily central Asia), and subsp. novicida (primarily N. America). 1 The pathogenicity of the four subspecies varies. While cases of tularemia with subsp. novicida are rare and mostly seen in patients with immunodeficiency, subsp. tularensis and holarctica induce the most documented outbreaks. 8
The first description of tularemia as the cause for a case of conjunctivitis with regional lymphadenopathy was published in 1914. 9 Since then, the global incidence of tularemia has declined, probably because of the urban sprawl and decreasing contact with wild animals. 10
Figure 1 shows the number of cases since 1949 in Germany. During the last 6 decades, the incidence decreased to <0.1/100.000 citizens/year reporting only 6 to 34 cases per year. 11 The real incidence could be higher due to undiagnosed cases and difficulties of laboratory verification of the pathogen. 1
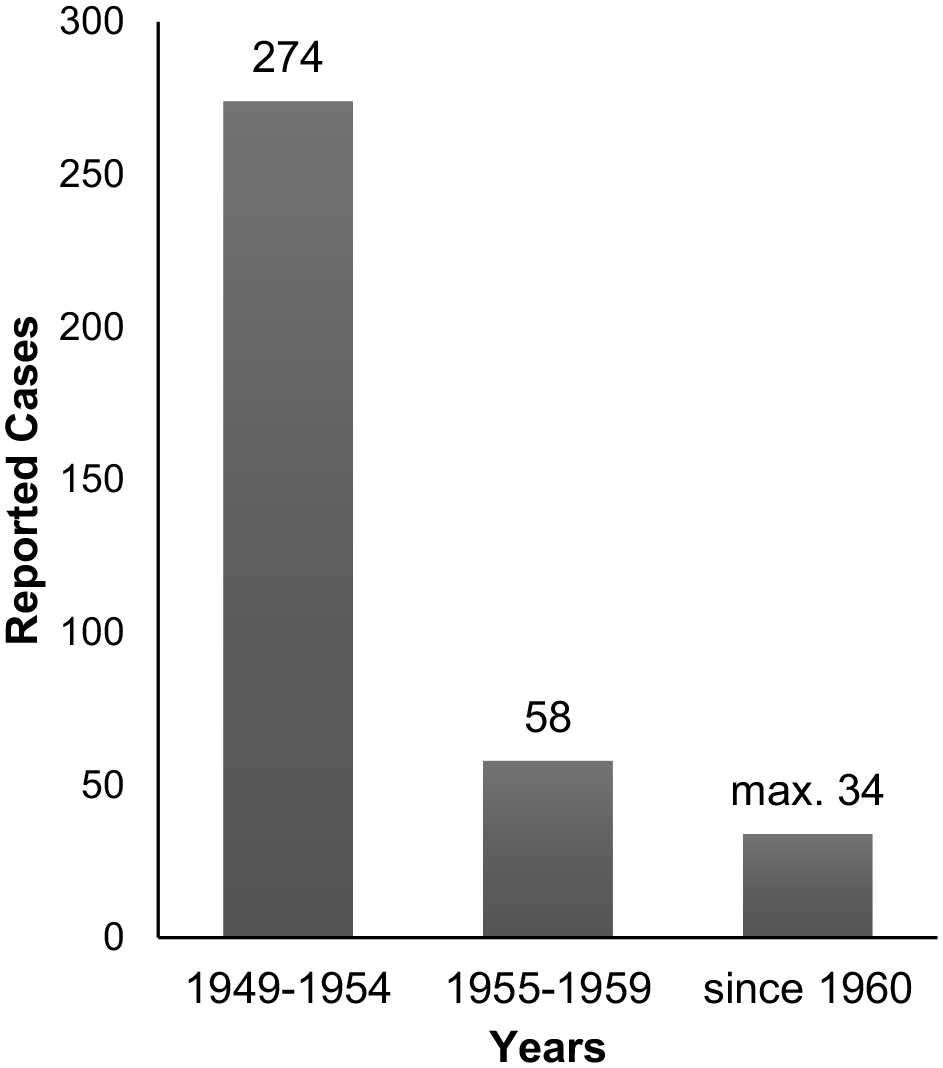
Number of tularemia cases in Germany.
Clinical presentation of tularemia depends on the route of entry and subtype of the pathogen. After an incubation period of 3 to 5 days, flu-like symptoms with fever and headache occur. Six clinical forms of tularemia can commonly be differentiated. Typical clinical signs of ulceroglandular and glandular tularemia are either skin ulcer (gate of entry) and enlarged lymph nodes, or sole lymphadenopathy which is the predominant clinical presentation of tularemia. 5 In Germany, glandular and ulceroglandular tularemia accounted for 50% of the reported cases from 2002 to 2016. 12 Patients with oropharyngeal tularemia, due to ingestion of contaminated food or water, present with lymphadenitis in combination with tonsillitis, pharyngitis, or stomatitis and represent a more infrequent variation of tularemia with only 7% of the reported cases in Germany from 2002 to 2016.5,12
Inhalation of contaminated aerosols can cause respiratory tularemia, which in difference to the F. tularensis subtype potentially results in a life-threatening pneumonia. 5 In Germany, 11% of the 257 cases of tularemia from 2002 to 2016 suffered from pneumonic tularemia. 12 Tularemia rarely occurs with clinical symptoms of conjunctivitis and lymphadenitis. Such oculoglandular tularemia, with potential ocular transmission of the pathogen, is reported in only 1% of the registered cases in Germany from 2002 to 2016. 12 If the synopsis of symptoms does not fit the inclusion criteria of one of the tularemia forms, and the patient presents with high fever, headaches, myalgia, neurologic symptoms as a severe systemic disease without any visible lymphadenopathy or ulcer, it is specified as typhoidal tularemia. 10
In Germany, 15% of the recorded tularemia cases from 2002 to 2016 had met the inclusion criteria “only fever.” 12 The diagnosis of tularemia is often delayed due to the rarity of the disease and the specialized reagents, which are required by laboratories to detect the pathogen. Various methods can be used to diagnose a tularemia infection, including serology, culture, antigen detection, and molecular detection. 13 Serological methods, for example, enzyme-linked immunosorbent assay and microagglutination assay, are the most common established tests for diagnosis of tularemia. 14
Prompt diagnosis and early antibiotic regime are essential for successful treatment of tularemia. 13 Beta-lactams and macrolides are often ineffective as a result of antibiotic resistance. 15 For ambulant treatment of tularemia, oral ciprofloxacin or doxycycline is recommended. In severe cases of tularemia, a parenteral dosage of gentamicin or streptomycin is reasonable. 13
Case Report
The 27-year-old male patient was initially referred by his ear, nose, and throat specialist to our department of otorhinolaryngology at the university hospital in Mannheim, Germany with unclear swelling of his face and jaw on the right side over the last 4 weeks. The patient reported a conjunctivitis of his right eye which had directly preceded the facial swelling. The conjunctivitis was successfully treated by topical therapy with eye ointment. The patient could not remember the type of eye ointment he received but it probably contained ciprofloxacin as an antibiotic agent. He negated any form of fever, weight loss, or night sweats and had no long-term medication, pre-existing conditions, or known allergies. He reported a visit to the Czech Republic but no further journeys. He remembered no animal contact, no tick bites, or other wounds.
The clinical examination showed a young adult with an asthenic physique, in good health condition. The oral examination showed no pathologies, no signs of infection, no palpable sialolith, and normal saliva flow. Laryngoscopy showed normal pharynx and larynx anatomy without any pathological swelling or mucosa transformation. In the manual palpation of the head and neck, a relocatable, soft tumor in the right preauricular region in close proximity to the parotid gland with a diameter of approximately 3 cm and another one in the right submandibular region with a diameter of approximately 2 cm were palpable.
Sonography of the right cervical region and parotid gland showed an inhomogeneous tumor with a diameter of approximately 2 × 3 cm, with diffuse margins in the area of the caudal pole of the parotid gland, and another one with the same characteristics with a diameter of approximately 3 × 3 cm in the region of the submandibular gland, with multiple surrounding enlarged cervical lymph nodes with an otherwise normal configuration.
To address a potential infectious genesis of the lymphadenopathy, a general and serological blood test was performed. We tested for the following pathogens: Bartonella henselae, Borrelia burgdorferi, Toxoplasma gondii, cytomegalovirus, Epstein–Barr virus, and herpes simplex. To precisely assess tumor size and location, we referred the patient to the radiology department for cervical magnetic resonance imaging. The pressure-sensitive tumor indicated an acute infection; therefore, the patient received an oral antibiotic treatment with amoxicillin/clavulanic acid for 1 week. In the first follow-up visit, clinical symptoms were unchanged. Blood test results showed no pathologies in the hemogram or leukogram, electrolytes in the normal range, and no signs of acute infection. Besides an Epstein–Barr virus infection in the past, no infection was detected. The radiologic report described multiple, partially necrotic tumors in and around the parotid gland and pathologically enlarged cervical lymph nodes in Levels I to V on the right side. Similar but much smaller lesions and cervical lymph nodes were found on the left side, as shown in Figure 2.
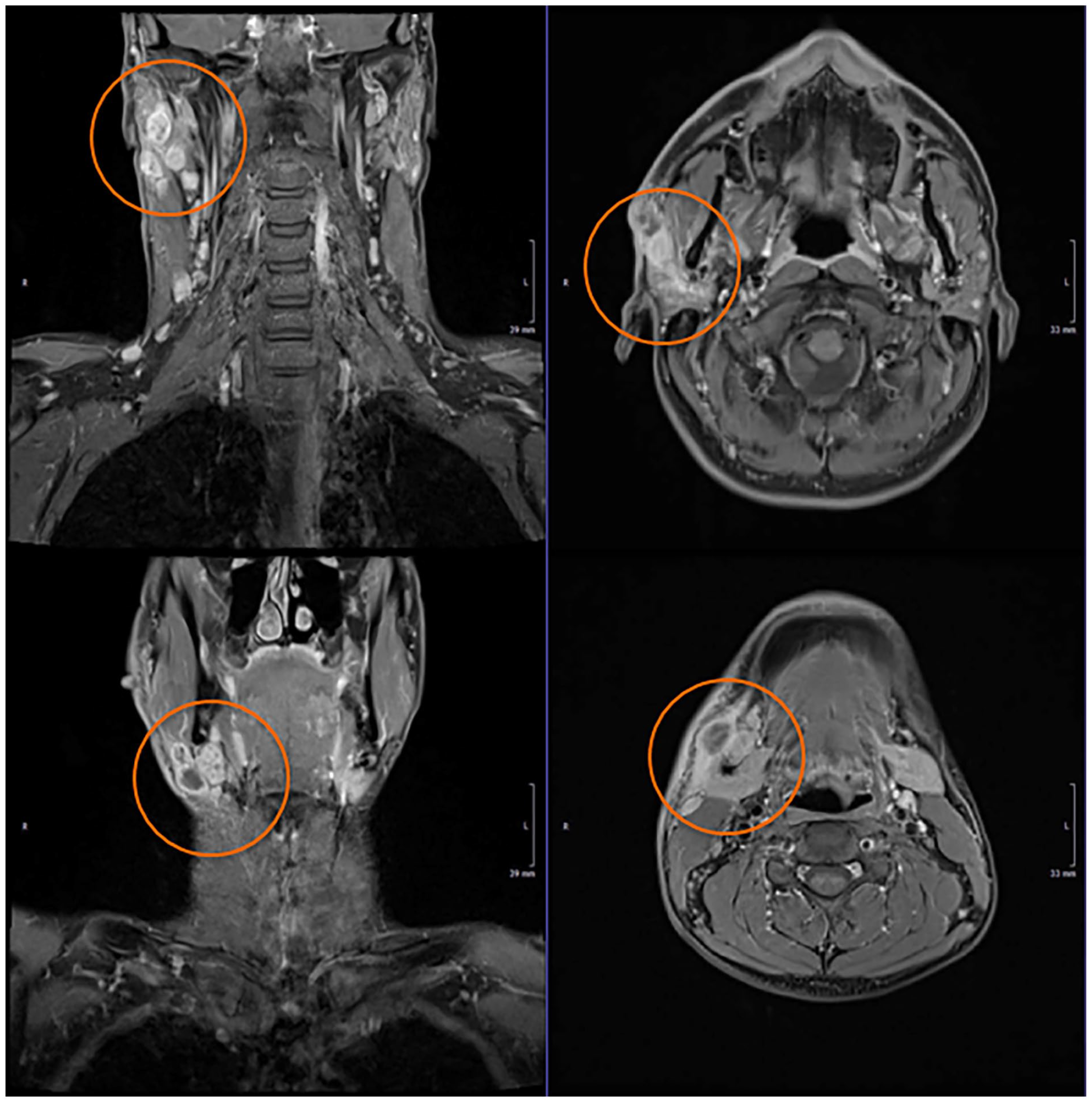
T1 magnetic resonance imaging coronary and sagittal imaging shows multiple, partially necrotic tumors in and around the parotid gland and pathologically enlarged cervical lymph nodes in each cervical level right > left.
To address potential malignant differential diagnosis, for example, carcinoma, lymphoma, a biopsy in local anesthesia in Level Ib was indicated. Under ultrasound surveillance, six core-needle biopsies of the undulating swelling in the submandibular region on the right site were performed. The tumor in the region of the caudal pole of the parotid gland showed the same ultrasound configuration as the tumor in the submandibular region. To avoid potential harm to the facial nerve, the core-needle biopsies were only performed in the submandibular region, not in the parotid region.
The biopsies were sent to the department of pathology, and an additional sample of the putrid-serous secretion in the region of the core-needle biopsies was sent for microbiological testing. The already prescribed antibiotic treatment with amoxicillin/clavulanic acid was prolonged for another week. The microbiological testing of the collected secretion showed no bacterial growth within 96-hour incubation time. The initial histopathological report of the tissue showed insinuated granulomatous growth but no signs of malignant carcinoma, as shown in Figure 3. Further immunohistochemistry was performed. There was no indication for a granulomatous forming lymphoma, such as Morbus Hodgkin, no evidence for light chain restriction of kappa or lambda, and no signs of immunoglobulin G4 (IgG4)-associated illness. A sarcoidosis seemed rather unlikely, further molecular pathological testing was performed. There was no proof of typical or atypical mycobacteria. The pathological processing of the punch biopsy excluded carcinoma and lymphoma from the potential list of differential diagnoses but a pathogen was not identified.
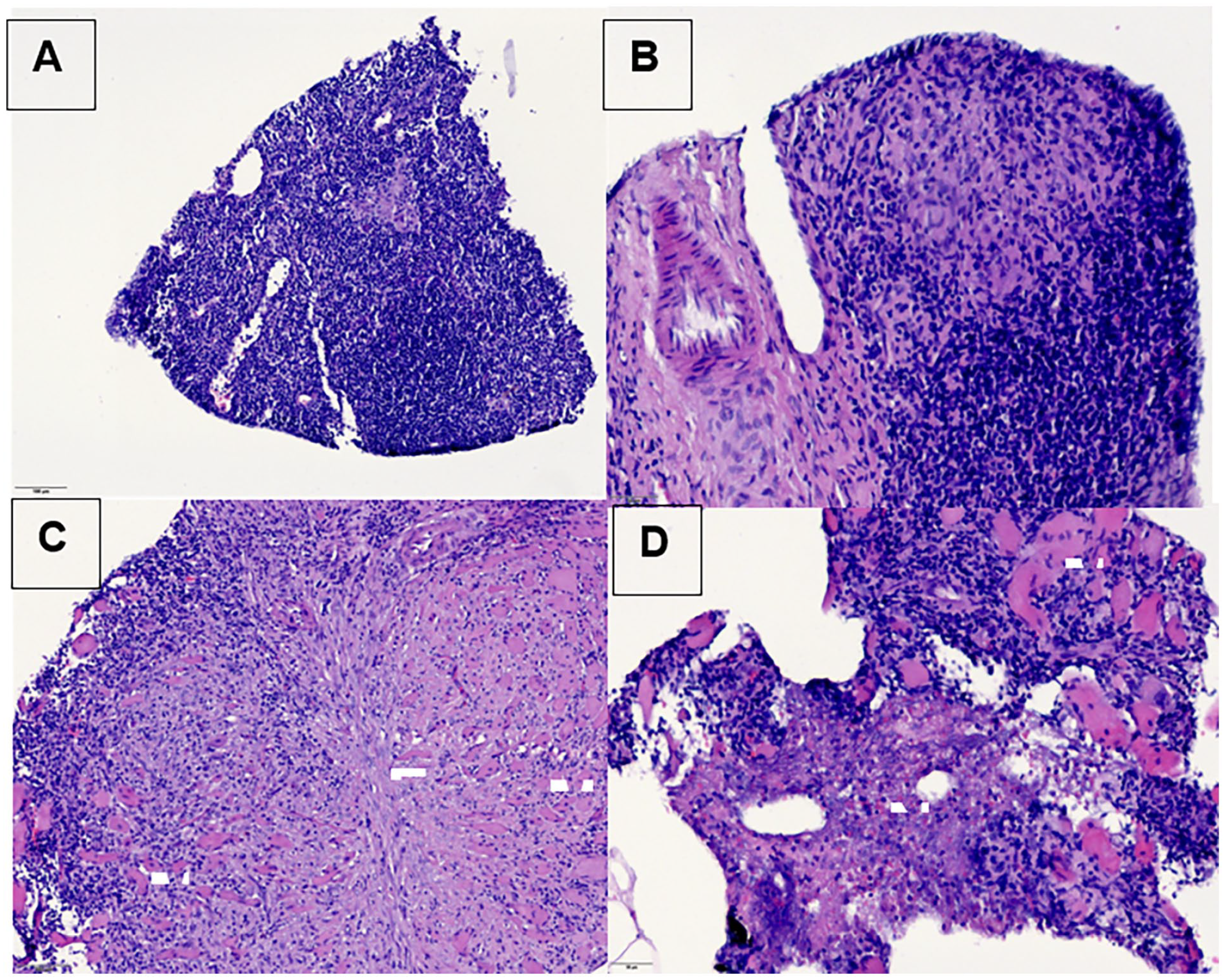
Biopsy Level Ib right side: Biopsy with skin, skin adnexa, subcutaneous fat, skeletal muscles, and partial lymph nodes (A). Unspecific, variable pattern of inflammation with lymphadenitis forming non-caseating granulomas, without certain definable giant cells (B). As a result of lymph node capsule exceeding inflammation with soft tissue effect and destruction of skeletal muscles scarred transformation occurs (C). In small parts, necrosis is detectable (D). A histomorphologic proof of the pathogen could not be assigned.
Either an infectious or rheumatological cause for the medical condition came into question, therefore, the Institute of Infectiology and Rheumatology was consulted. The infectiologist performed a selective anamnesis, including potential infection sources in social anamnesis, sexual anamnesis, travel history, and vaccination status. The physical examination reported the same palpable tumor in the region of the parotic gland as previously mentioned. The remaining examination showed no pathologies. The abdominal sonography showed regular diagnostic findings. A liver elastography indicated no signs of liver fibrosis or liver cirrhosis. Lymph node sonography showed cervical lymphadenopathy on both sides and axillar lymphadenopathy with no signs of enlarged lymph nodes in the right axillar, clavicula, inguinal, or retroperitoneal region.
The repeated blood testing showed regular results for white blood cells and the inflammation parameter C-reaktive Protein (CRP) as well as the blood sedimentation rate. To complete the serological testing, brucellosis, mumps, and F. tularensis were assessed and the aforetested pathogens checked again (B. henselae, B. burgdorferi, T. gondii, cytomegalovirus, Epstein–Barr virus, and herpes simplex). The physical examination performed during the rheumatologic consultation revealed no new findings. No reported vasculitis or collagenosis-associated discomfort was found. There was no family history of rheumatologic diseases. Blood testing showed negative Antineutrophil Cytoplasmic Antibodies (ANCA), Antinuclear Antibody (ANA), and complement factors. Inflammation parameters showed regular parameters as before. No decreased IgG, IgA, or IgM was seen. Autoantibodies showed negative results besides threefold increased Anti-Jo-1 antibodies were verified but had no clinical correlation to a potential anti-synthetase syndrome associated with polymyositis.
The completed serological testing requested from the Department of Infectiology brought evidence for acute infection with F. tularensis, showing a >300.00 U/ml IgG, >400.00 U/ml IgM and F, and tularensis-antibody titer of 1:320.
The patient received antibiotic treatment with doxycycline 100 mg for 21 days. The follow-up appointment showed declining cervical lymphadenopathy and constantly enlarged axillary lymph nodes. A serological gradient monitoring for the F. tularensis antibodies was recommended 3 months after initial testing but was rejected by the patient because of private travel plans. The patient reported a complete remission of the clinical presentation after 3 months.
Discussion
The uncommon clinical presentation of this case with the lack of any initial flu-like symptoms, and the rarity of the disease caused the prolonged time period until the final diagnosis. This issue is commonly known and often reported in literature. For example, the reported mean delay from onset of symptoms to diagnosis was 47.5 days in a tularemia outbreak with 142 cases in Spain. 16 The subspecies of tularemia in our case is unknown because only serological testing was performed. The pathogen was not identified in the harvested tissue or secretion and therefore polymerase chain reaction testing of the subspecies was not possible. The most likely pathogen in this case is subsp. holarctica, the most common subspecies in our region. The route of entry cannot be evaluated with absolute certainty. The patient described a conjunctivitis that had occurred right before the facial swelling. The patient remembered a mosquito in his right eye before the onset of the ocular symptoms. Therefore, the route of entry could be ocular and would define the subtype as oculoglandular tularemia. However, this theory of the entry route cannot be proven and as the conjunctivitis fully recovered only with topical therapy it could also be a confirmation bias. Considering only lymphadenopathy as the clinical symptom of the acute tularemia infection, the subtype glandular tularemia would be appropriate. The histopathological report, describing granulomatous growth, likely from the unspecific type of infection is often associated with tularemia. Core-needle biopsy or fine-needle aspiration is frequently performed in reported cases of tularemia. Mostly, they are not applied for evidence of the tularemia pathogen but executed as part of the routine workup for lymphadenopathy. 17
Footnotes
Acknowledgements
We thank our colleagues from the Institute of Pathology and Infectiology who provided insight and expertise that greatly assisted the research of this case report.
Authors’ Note
Presented at the 95th ENT Congress (DGHNO) in Leipzig on 20th of May 2023
Data Availability Statement
The data that support the findings of this study are available on request from the corresponding author, J. Kornmann.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
