Abstract
Epstein-Barr virus (EBV) belongs to the group of human herpes virus and can cause clinical and subclinical infections. Although EBV-related disease presentations are similar, they can lead to oncogenic transformation with various clinical manifestations. A thorough workup with morphology, immunohistochemistry, and molecular studies is crucial for the diagnosis of EBV-positive polymorphic B-cell lymphoproliferative disorder, not otherwise specified (NOS), which is a new entity introduced by International Consensus Classification in 2022. We describe an interesting presentation of EBV-positive polymorphic B-cell lymphoproliferative disorder with laryngeal involvement to bring awareness to this entity and we would like to address the need for more accessible treatment options.
Introduction
The prevalence of dysphagia varies based on the characteristics of the patient population. Approximately 20% of the general population will experience dysphagia at some point in their life and 3% report dysphagia at least once a week. 1 Dysphonia is an indirect symptom of oropharyngeal dysphagia, which may lead to complications such as aspiration, chest infection, malnutrition, dehydration, and death. 2
Dysphagia with laryngeal involvement in the form of dysphonia has a broad differential diagnosis including infectious diseases such as tuberculosis, acute or chronic laryngitis, neoplasia, amyloidosis, and rheumatological diseases such as rheumatoid arthritis, granulomatosis with polyangiitis, relapsing polychondritis, and laryngeal sarcoidosis. 2 Furthermore, dysphonia may be the first symptom in neoplasms of the larynx, pharynx, lungs, thyroid gland, and lymphoma. 3
Epstein-Barr virus (EBV) associated B-cell lymphoproliferative diseases, which could be found in the gastrointestinal tract (GI), skin, or oropharyngeal mucosa, comprise 3%-15% of diffuse large B-cell lymphomas and affects individuals of all ages, both immunocompetent and immunocompromised. 4 Case reports indicate that this disorder can occur in immune-dysregulated settings, for example, after transplant surgeries or after engaging in immunosuppressant therapy. 5 The pathology diagnosis can be challenging since the manifestation of histology demonstrates a spectrum varying from follicular hyperplasia to polymorphic proliferation to diffuse large B-cell lymphoma. Management heavily relies on the clinical setting and pathology diagnosis since it could vary from wait-watch or simply withdrawal of immunosuppression medicine to aggressive chemotherapy. There are many reports and research conducted recently. In 2015, the workshop of the Society for Hematopathology and European Association of Hematopathology hosted the stage in review immune-dysregulation/immunodeficiency-associated B-cell proliferations of varied malignant potential associated with immunodeficiency; and a series of papers were published by the workshop organizers related to this subject.6-9 International Consensus Classification was published in 2022 and officially introduced a new entity within this group of disease, EBV-positive polymorphic B-cell lymphoproliferative disorder, NOS. 10
We report a case of dysphagia and dysphonia as the initial manifestation of subglottic EBV-positive polymorphic lymphoproliferative disorder, a rare presentation that represented a diagnostic challenge and required an interdisciplinary approach.
Case Description
A 36-year-old male presented with dysphonia, recurrent fever, and acute onset dysphagia. The patient was in his usual state of health until he developed a SARS-CoV-2 upper respiratory tract infection. After infection, he experienced persistent dysphonia, recorded fevers up to 102.8°F, voice fatigue, productive cough with clear sputum, and difficulty with increased vocal amplitude. Symptoms were not responsive to several courses of antibiotics and a trial of corticosteroids. Marked fatigue, generalized myalgias, diffuse arthralgias, and night sweats developed along with progressive dysphagia to solids with globus sensation. He experienced poor appetite and a 50 lb weight loss over 6 months. Serology study noted positive EBV titers for viral capsid antigen (VCA)-IgG antibody and Epstein-Barr virus nuclear antigen (EBNA) with negative VCA-IgM antibody. Antibody IgG is suggestive of reactivation of prior EBV infection.
Computed tomography (CT) neck demonstrated findings suggestive of a diffuse laryngeal mucosal-submucosal inflammatory process with no discrete mass lesion. CT chest angiogram described mild circumferential tracheal wall thickening. There was a mild narrowing of the upper trachea with mildly prominent subcentimeter upper mediastinal lymph nodes.
Magnetic resonance imaging (MRI) of the neck without and with intravenous contrast revealed diffuse edema of the larynx with enhancement including soft tissue thickening of the subglottis measuring up to 0.4 cm. Positron emission tomography (PET) demonstrated nonspecific uptake in the laryngeal supraglottic region. There was no PET evidence for nodal involvement in the disease.
Upper endoscopy and related pathology exam of the biopsies on the esophagus, stomach, and duodenum were unremarkable. Otolaryngology evaluation identified erythematous irregular mucosa of bilateral true and false vocal folds (Figure 1). Microsuspension direct laryngoscopy with laryngeal biopsies and cultures revealed benign squamous mucosa with reactive changes and dense lymphoid tissue suggesting an inflammatory process.
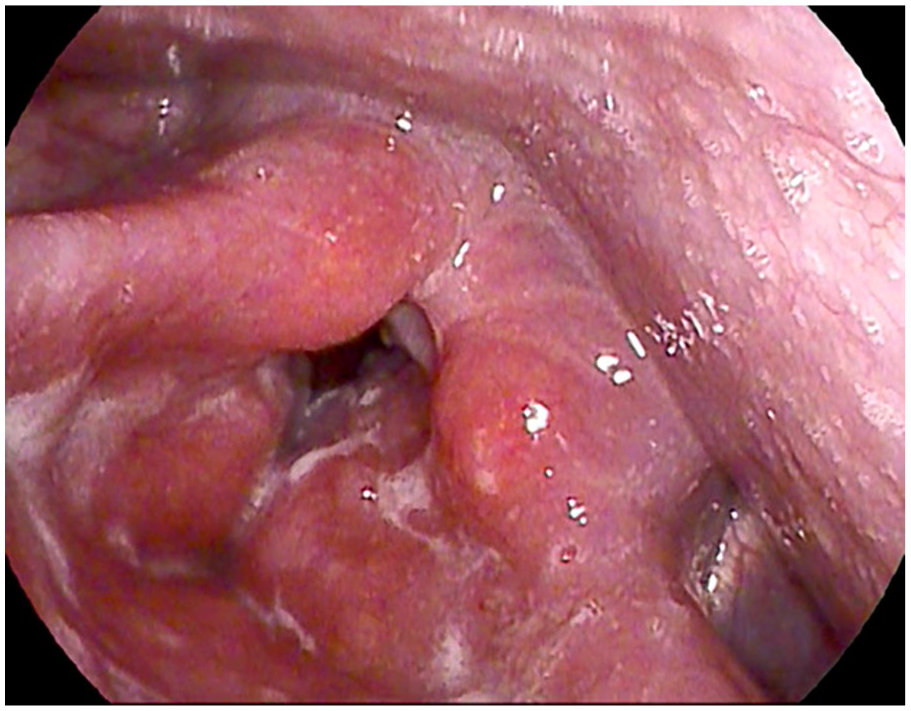
Image of patient’s vocal folds taken via Pentax scope at initial evaluation: severe inflammation of supraglottis, posterior glottis, false vocal cords, and arytenoids.
Subsequently, the patient underwent a repeat microsuspension direct laryngoscopy with examination under anesthesia and biopsies of bilateral vocal folds. Supraglottic biopsies revealed a dense and diffuse lymphoid proliferation with predominant small and occasionally medium to large cells; normal architectures such as reactive germinal centers were not present. Immunohistochemistry (IHC) studies with a large antibody panel were performed and demonstrated mixed B and T lymphocytes and polytypic plasma cells, and no evidence of IgG4-related disease was identified. EBER In situ hybridization stain highlighted EBV-encoded RNA in numerous small and medium cells, the distribution was in concordant with B lymphocytes. In addition, molecular study by polymerase chain reaction (PCR) was negative for either clonal immunoglobulin or T-cell receptor gene rearrangement. With the overall morphology, IHC, and molecular findings, the diagnosis of EBV-positive polymorphic lymphoproliferative disorder was rendered (Figure 2A and B).
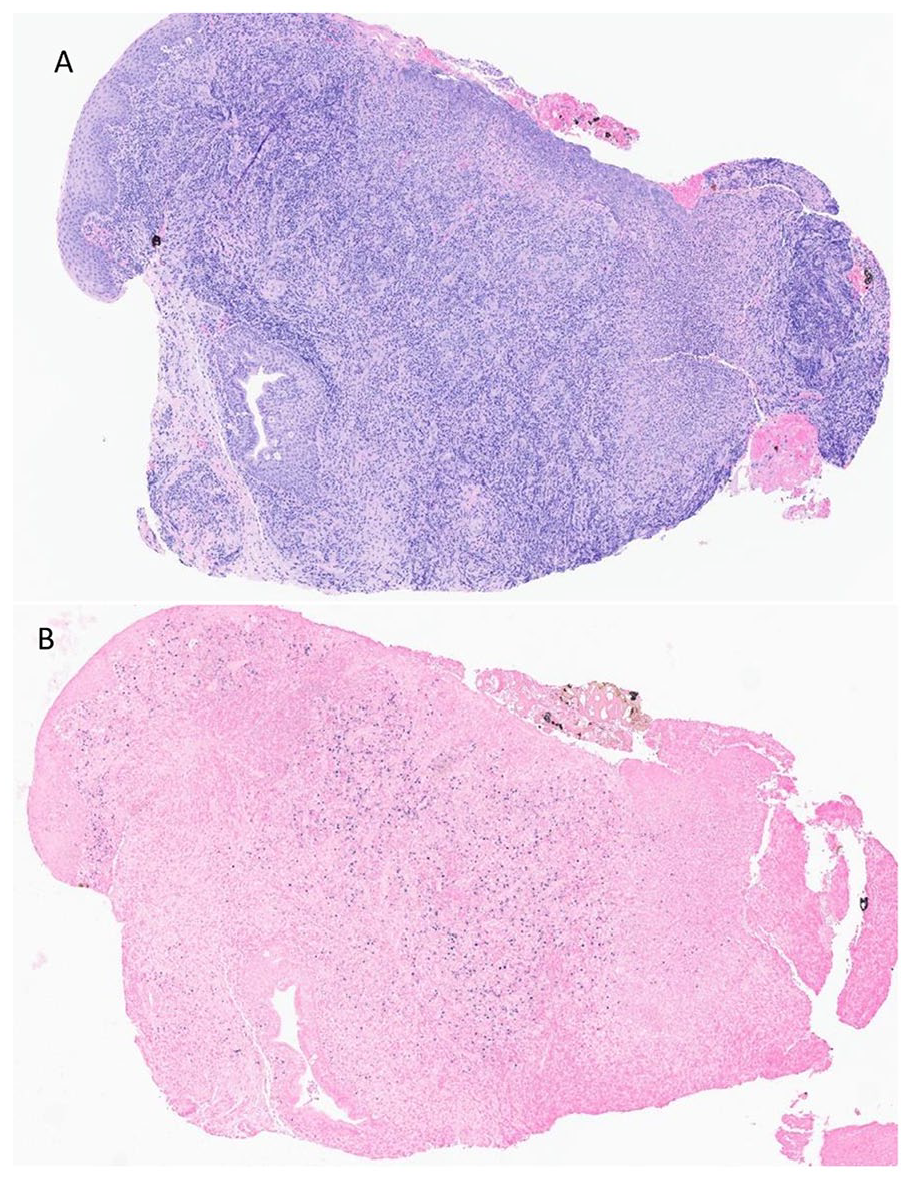
(A) Supraglottic biopsy showing a dense lymphoplasmacytic infiltrate with surface ulceration and associated acute inflammation (H&E stain). (B) In situ hybridization for EBV-encoded RNA, the presence of which is indicated by punctate dark blue staining of numerous nuclei within the infiltrate.
Follow-up serologic testing for EBV was positive for IgG but negative for IgM. EBV by PCR was positive in peripheral blood with over 700 copies. The patient was initiated on oral dexamethasone 4 mg twice a day and felt markedly improved with the resolution of fever and soreness in the upper throat. Hematology service was consulted, and the patient was initiated on rituximab IV 800 mg weekly for 4 doses. The patient noted resolution of all symptoms except hoarseness at 3 months of follow-up.
Discussion
Oropharyngeal dysphagia may be challenging to evaluate due to the chronology of symptom development. It can have far-reaching consequences ranging from poor nutritional status to the development of aspiration pneumonia and hypoxic respiratory failure. An extensive array of functional and structural abnormalities can lead to the development of oropharyngeal dysphagia. It has also been linked to viral infections including herpes simplex, cytomegalovirus, Kaposi sarcoma, and non-Hodgkin’s lymphoma. 11 Most of the infections are reactivation from prior exposure and immunocompromised hosts are at a greater risk. Ninety-five percent of the population is exposed to EBV during their lifetime due to the ease of transmissibility and although the majority of the infections are asymptomatic and fleeting, they have a strong association with oncogenic transformation and malignancy. 12
Notably, our patient was not documented to have a history of immunodeficiency or prior immunosuppressive treatments that would have put him at a greater risk for the development of EBV-positive B-cell lymphoproliferative disorder. Testing for HIV was performed multiple times with negative results. Woodward and Aybar described a case of rapidly progressive dysphagia secondary to acute EBV infection in a healthy male in 2012. 13 Oyama et al. 14 reported EBV-positive diffuse large B-cell lymphomas in elderly Japanese patients who were not immunodeficient implying aging may have a role in the pathogenesis. However, our patient was an otherwise healthy 36 years old with no clinical evidence of immunodeficiency or hematologic or non-hematologic malignancies, adding to the uniqueness of our case.
EBV-positive B-cell lymphoproliferative disorders at the head of a neck region could present in various diseases, ranging from benign (infectious mononucleosis) to malignant (i.e., Burkitt lymphoma or extranodal NK/T-cell lymphoma, nasal type), 15 as nodal and/or extranodal manifestation. These diseases could cause severe mucositis of the oropharynx, larynx, bronchial airway, and upper GI. Our patient does have a recent infection of the COVID-19 virus, which might play a role in immune dysregulation, which led to EBV reactivation. However, the loss of normal architecture and diffuse EBER ISH positivity in the B lymphocytes argues against a benign reactive change. In addition, our patient was not observed to have extensive ulceration in the mouth, respiratory tract, or GI, which does not fit in the diagnosis of the EBV-positive mucocutaneous ulcer, which is often associated with iatrogenic immunosuppression. 10
Thorough workup including morphology, IHCs, and molecular studies on the sufficient biopsy specimen, particularly the detection of EBV by EBER in situ hybridization are critical for an accurate diagnosis in EBV-related lymphoproliferative disorders; also, the newly introduced entities of EBV-related diseases in both ICC and WHO classification do require keen acknowledgment from both pathologists and clinicians. EBV-related disorders in immunocompetent people have high rates of spontaneous remission and regression with symptomatic management and supportive care. 11 In this case, the patient experienced multiple relapses without symptom resolution. Therefore, he was managed with corticosteroids and rituximab which has shown efficacious responses in the treatment of EBV-related lymphoproliferative disorders and B-cell lymphomas. 16 Additional treatment options include radiotherapy and hematopoietic stem cell transplant 17 could be considered in patients with more aggressive diseases such as EBV-positive diffuse large B-cell lymphoma.
Conclusion
We describe an atypical presentation of EBV-positive polymorphic B-cell lymphoproliferative disorder in an immunocompetent adult who developed a relapsing and progressive course of symptoms localized to subglottic oropharyngeal involvement with relative mucosal sparing. Future studies would be beneficial for earlier detection and enhanced characterization and classification of pathogenesis for EBV-associated lymphoproliferative disorders particularly with localized oropharyngeal involvement for the reduction in morbidity in such cases.
Footnotes
Acknowledgements
None.
Author Contributions
All the authors were involved in the care of the patient, editing, and approving the final manuscript.
Availability of Data and Materials
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval does not apply to this article.
Informed Consent
Written consent for publication was obtained from the patient.
Permission to Reproduce Material from Other Sources
Yes.
