Abstract
Introduction
Rhinitis is a persistent and complicated inflammatory condition that affects the lining of the nose. 1 Allergic rhinitis (AR) is usually caused by environmental allergens, whereas non-AR may result from several non-allergic factors. 2 This condition is increasingly prevalent, affecting up to 30% of the global population, and can negatively impact an individual's well-being by affecting their quality of life, productivity, and sleep.3,4 Additionally, rhinitis places a substantial burden on healthcare systems worldwide, requiring more healthcare utilization and associated costs. 5 Although there are traditional treatments available, such as antihistamines, corticosteroids, and decongestants, their effectiveness is limited and may not address the complex nature of rhinitis, which highlights the need for innovative treatment options. 6
Effective and targeted therapies are crucial for managing the diverse symptoms and underlying causes of rhinitis. Advances in understanding the complex molecular mechanisms of this condition have led to the development of new treatments. Immune cells, cytokines, and other mediators play important roles in the development of rhinitis, which has led to the investigation of innovative therapeutic targets and approaches. 7 A variety of experimental therapies have emerged, including small-molecule inhibitors, monoclonal antibodies, and novel approaches to allergen immunotherapy.8,9 Additionally, stem cell therapies have shown promise for personalized treatment strategies. 10
This systematic review provides a detailed overview at both ongoing and completed clinical trials studying new treatments for rhinitis. The study critically reviews the stages of development, safety, tolerability, and effectiveness of these new treatments. The main goal is to give important insights into how these experimental treatments could change the way rhinitis is managed, encourage more research, and help in creating useful research questions.
By identifying areas where treatment needs are not being met and where more knowledge is needed, the review could help lead to better treatment options for rhinitis. This review includes information from completed trials and ongoing research to provide a complete picture of the progress being made in this field. This should contribute to improving treatments for rhinitis, ultimately improving the lives of patients with this condition.
Methods
Search Strategy
A comprehensive literature search was conducted to identify both ongoing and completed clinical trials investigating experimental therapies for rhinitis. The databases and engines searched included PubMed, Web of Science, Cochrane Library, ClinicalTrials.gov, the World Health Organization International Clinical Trials Registry Platform, and the European Union Clinical Trials Register. The search terms encompassed “rhinitis,” “experimental therapy,” “immunotherapy,” “probiotics,” and/or “stem cell therapy.” No restrictions were placed on the status of the trials, thereby including both ongoing/not yet completed trials and completed ones. The final search was conducted on June 2023. There were no language restrictions. The findings of the systematic review were reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement 2020 guidelines. 11
Included Experimental Therapies for Rhinitis
The therapies included for review in these trials were:
Immunotherapy: A treatment that exposes the immune system to small amounts of the allergen responsible for the allergic reaction, with the aim of decreasing the body’s sensitivity to it.
Probiotics: Live microorganisms which, when administered in adequate amounts, may confer health benefits. They are currently being investigated for their potential to modulate the immune response and reduce inflammation in the nasal passages.
Stem cell therapy: A treatment which uses stem cells to regenerate damaged tissue in the nasal passages with the aim of reducing inflammation and improving function.
Trial Selection
Two independent reviewers (ZS and AS) screened the search results for eligibility. Studies were included if they were clinical trials (both active and completed) involving immunotherapy, probiotics, or stem cell therapy as experimental treatments for rhinitis. Studies focusing on other types of experimental therapies or not specifically addressing rhinitis were excluded. Disagreements between reviewers were resolved through discussion and consensus. Inter-rater agreement was measured using the kappa statistic, calculated using SPSS v24.
Data Extraction and Synthesis
Two independent reviewers (ZS and AS) extracted data from the included studies. For completed clinical trials, the following data were collected: author, year, title, study type, intervention, population characteristics, key findings, and implications for care. For ongoing clinical trials, the data included: No. NCT number, title, status, conditions, interventions, outcome measures, age, phases, N, study design, and primary completion date. Any discrepancies between reviewers were resolved through discussion and consensus.
The data derived from the included studies were summarized using a narrative synthesis approach. The studies were categorized by intervention type (immunotherapy, probiotics, or stem cell therapy), and the results were presented separately for each group.
Protocol Registration and Role of Funding
The protocol was registered with the Open Science Framework: https://doi.org/10.17605/OSF.IO/PSDYJ. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Results
The PRISMA flowchart depicting the study selection process is depicted in Figure 1. The inter-rater kappa score was 0.913 suggesting excellent agreement.
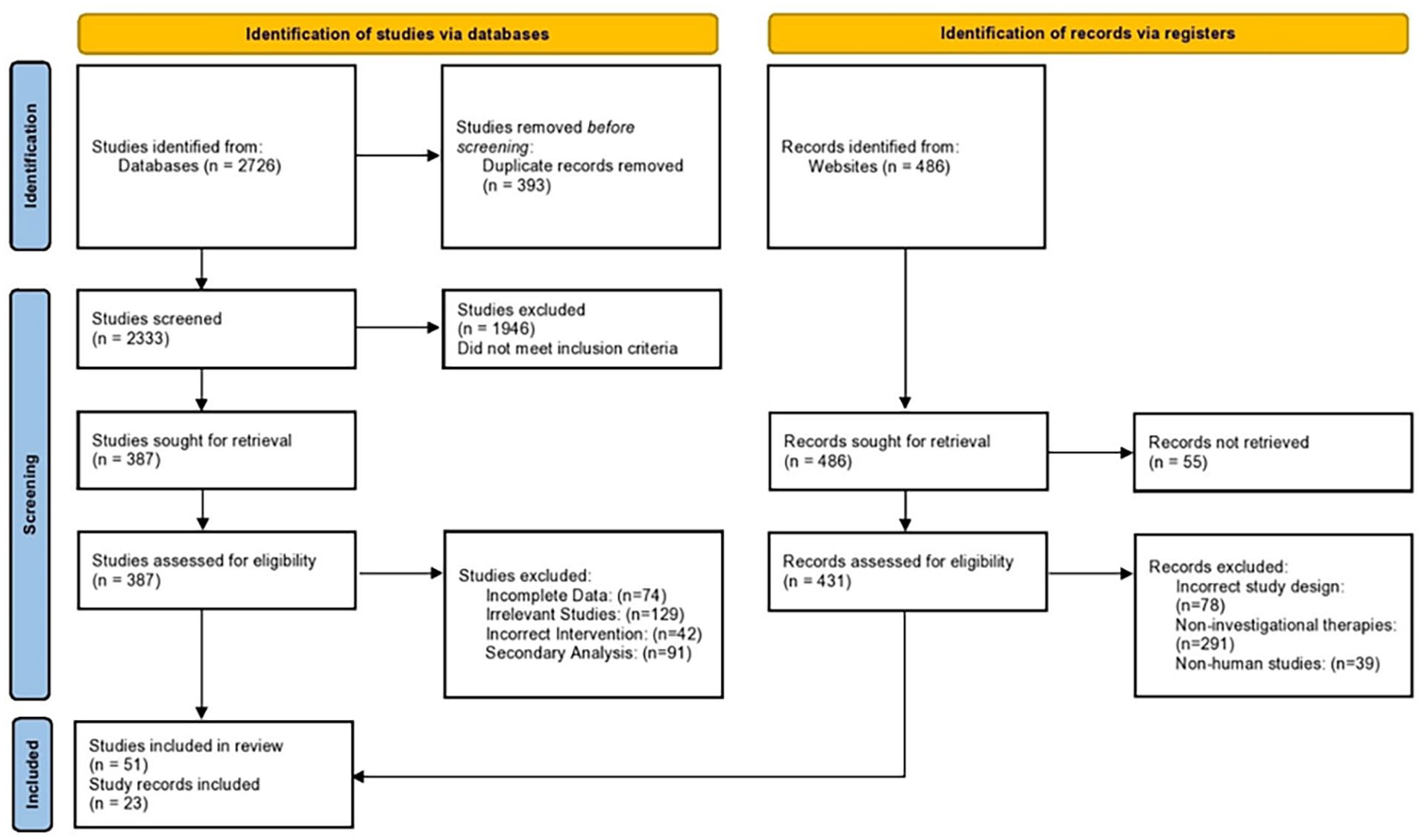
PRISMA flowchart depicting the study selection process. PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Completed Clinical Trials
Immunotherapy interventions for rhinitis
This section presents the therapeutic advancements of immunotherapeutic interventions in rhinitis across 44 studies conducted in the last 5 years, between 2018 and 2023.12–55 The studies are further synthesized and presented in Table 1.
Characteristics of the Completed Immunotherapy and Rhinitis Studies (N = 44).
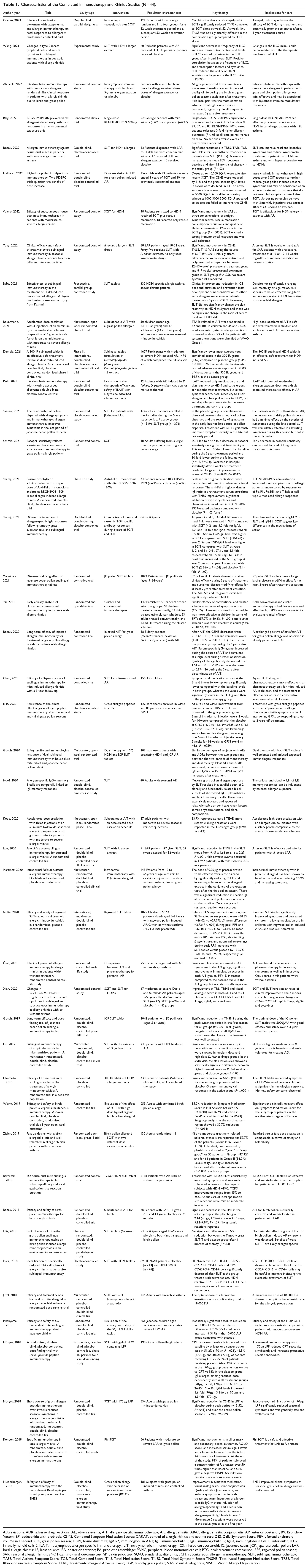
Abbreviations: ADR, adverse drug reactions; AE, adverse events; AIT, allergen-specific immunotherapy; AR, allergic rhinitis; AR/C, allergic rhinitis/conjunctivitis; AP, anterior posterior; BV, Broncho–Vaxom; BP, budesonide with probiotic; CSMS, Combined Symptom Medication Scores; CARAT, control of allergic rhinitis and asthma test; DSS, Daily Symptom Score; FEV1, forced expiratory volume in 1 second; GPS, grass pollen season; HDM, house dust mite; IgA1/2, immunoglobulin A1/2; IgE, immunoglobulin E; IgG, immunoglobulin G; IgG4, immunoglobulin G4; IL, interleukin; ILC2, innate lymphoid cells 2; ILAIT, intralymphatic allergen-specific immunotherapy; ILIT, intralymphatic immunotherapy; ICS, inhaled corticosteroid; JC, Japanese cedar; JCP, Japanese cedar pollen; LAR, local allergic rhinitis; LS, least squares; PA, posterior anterior; PA, probiotic assemblage; PBMC, peripheral blood mononuclear cell; PTC, peak treatment comparison; RPS, ragweed pollen season; SAR, seasonal allergic rhinitis; SNOT-22, sino-nasal outcome test; SPT, skin prick test; SQ-U, standard quality units; SCIT, subcutaneous allergen immunotherapy; SLIT, sublingual immunotherapy; TASS, Total Asthma Symptom Score; TCS, Total Combined Score; TMS, Total Medication Score; TNSS, Total Nasal Symptom Score; TNSMS, Total Nasal Symptom Medication Score; TRSS, Total Rhinoconjunctivitis Symptom Score; TEAE, Treatment-Emergent Adverse Event; TGP, timothy grass pollen; VAS, Visual Analog Scale; WAO, World Allergy Organization.
In 2023, Corren et al 12 conducted a double-blind parallel design trial to assess the combination of intravenous tezepelumab with subcutaneous allergen immunotherapy (SCIT) for cat allergy treatment. Their findings suggested that this combination therapy significantly reduced the Total Nasal Symptom Score (TNSS) during treatment. However, after a one-year observation period, no significant difference was observed compared to SCIT alone. This points toward the potential of tezepelumab to enhance the efficacy of SCIT during treatment and potentially promote tolerance after a one-year treatment course.
Another study by Wang et al 13 focused on an experimental approach involving sublingual immunotherapy (SLIT) with house dust mite (HDM) allergen extract. The study found significant decreases in the frequency of innate lymphoid cells 2 (ILC2), their transcription factors, and levels of ILC2-related cytokines. This was linked to the therapeutic mechanism of SLIT, suggesting a potential pathway for SLIT effectiveness in HDM allergy treatment.
In 2022, Ahlbeck et al 24 carried out a randomized control study focusing on intralymphatic immune therapy with birch and 5-grass allergen extracts. The study found that all treatment groups reported fewer symptoms, lower use of medication, and improved quality of life. This was accompanied by a decrease in IgE levels to birch and an increase in regulatory T-cell frequencies three years after treatment. The study, therefore, concluded that intralymphatic immunotherapy was safe, effective, and possibly associated with bystander immune modulatory responses.
In the same year, a randomized clinical trial conducted by de Blay et al 35 investigated the efficacy of single-dose REGN1908/1909 in cat-allergic patients with mild asthma. This study found that a single dose could effectively prevent reductions in forced expiratory volume in 1 second (FEV1) in cat-allergic patients with mild asthma.
A range of other studies conducted between 2021 and 2022 investigated various treatments such as SLIT for HDM allergies, dose escalation in intralymphatic immunotherapy for grass pollen-induced AR, SCIT for HDM allergies, Artemisia annua allergens SLIT, and several others. These studies consistently reported reductions in various symptom scores and improvements in quality of life, along with safety and tolerability of the treatments. This study synthesized various therapies for AR, including intravenous tezepelumab, SLIT, intralymphatic immune therapy, and subcutaneous allergen-specific immunotherapy (AIT). All these studies provide evidence toward the potential efficacy of different immunotherapies in managing allergies and AR and suggest possible therapeutic mechanisms.
Sakurai et al 16 showed that SLIT is effective for Japanese cedar (JC) pollen-induced AR, particularly in the late period of pollen dispersal. Yonekura et al 20 echoed this finding, indicating that JC pollen SLIT tablets have a long-lasting disease-modifying effect. Schmid et al 17 provided evidence that subcutaneous immunotherapy (SCIT) can significantly decrease basophil sensitivity, which can predict long-term treatment outcomes. Shamji et al 18 examined two different interventions, namely, anti-Fel d 1 monoclonal antibodies and SLIT versus SCIT for timothy grass pollen (TGP). Both interventions improved symptoms, albeit via different mechanisms. Chen et al 23 revealed that a 3-year SLIT in combination with pharmacotherapy showed greater efficacy than pharmacotherapy alone for mite-induced AR in children.
Alternative interventions included the use of anti-Fel d 1 monoclonal antibodies, 19 grass allergen peptides, 25 , and AIT. 22 These studies highlighted that treatments could be tailored to specific allergens and patient groups for enhanced efficacy. Comparative studies like those conducted by Shamji et al, 19 Ünal et al, 32 and Xian et al 33 showed that different forms of immunotherapy, such as SCIT and SLIT, can yield similar improvements in AR symptoms, but via distinct immunological pathways. Likewise, Ünal et al 32 highlighted that AIT was more effective than pharmacotherapy for perennial AR.
SLIT was widely employed in the studies reviewed, with various allergenic extracts used, including JC pollen, Dermatophagoides farinae extracts, and HDM allergen extracts. The study by Gotoh et al 34 found significant reductions in total nasal symptoms and medication scores among patients with JC pollinosis using JC pollen SLIT, especially in the 5000 JAU dosage group, over a 3-year period. Similarly, Liu et al 36 reported the benefits of SLIT with high- or medium dose D. farinae extracts in patients with HDMs-induced atopic dermatitis. Efficacy and safety of SLIT were further established by Okamoto et al 37 and Bernstein et al 40 in treating AR with HDM allergen extract tablets and 12 SQ-HDM SLIT-tablets respectively. However, Ellis et al 42 found no bystander effect of Timothy grass SLIT-T on birch pollen-induced AR symptoms, suggesting allergen-specificity of the treatment benefits. SCIT was also explored in these studies. Worm et al 38 found a significant effect on Symptom Medication Score with high-dose hypoallergenic birch pollen allergoid in a subgroup of patients in the north-eastern region of Europe. Zielen et al 39 evaluated two different dose escalation schedules of birch pollen allergoid SCIT and found comparable safety and tolerability. Bożek et al 41 also showed clinical effectiveness and well-tolerance of SCIT for birch pollen in patients with local AR.
Studies by Jutel et al 44 and Mösges et al47,48 evaluated SCIT with a D. pteronyssinus allergoid preparation and with gpASIT+™ containing Lolium perenne peptides (LPP), respectively, and found that the optimal dose of allergoid for a confirmatory trial is 18,000 TU, and a 3-week immunotherapy with 170 μg LPP significantly reduced CPT reactivity and increased protective specific antibodies. Finally, the studies by Rondón et al 49 and Niederberger et al 50 tested the Phleum pratense SCIT (Phl-SCIT) and a grass pollen allergy vaccine based on recombinant fusion proteins (BM32), respectively. Both found significant improvements in clinical outcomes and increased allergen tolerance.
Overall, these studies collectively highlight the potential of both subcutaneous and SLIT in effectively managing various allergic conditions, though the optimal dosage and allergen specificity appear crucial for the treatment benefits. The majority of the studies reported minimal adverse effects, suggesting the general safety and tolerability of these therapeutic strategies.
Probiotic interventions for rhinitis
The potential of probiotics as a treatment for AR has been explored in multiple studies. However, the findings have been mixed, demonstrating a wide range of effects on patients’ symptoms and quality of life. Table 2 presents key findings of 7 completed clinical trials administering probiotics to patients with rhinitis.56–62
Characteristics of the Completed Probiotics for Rhinitis Studies (N = 7).
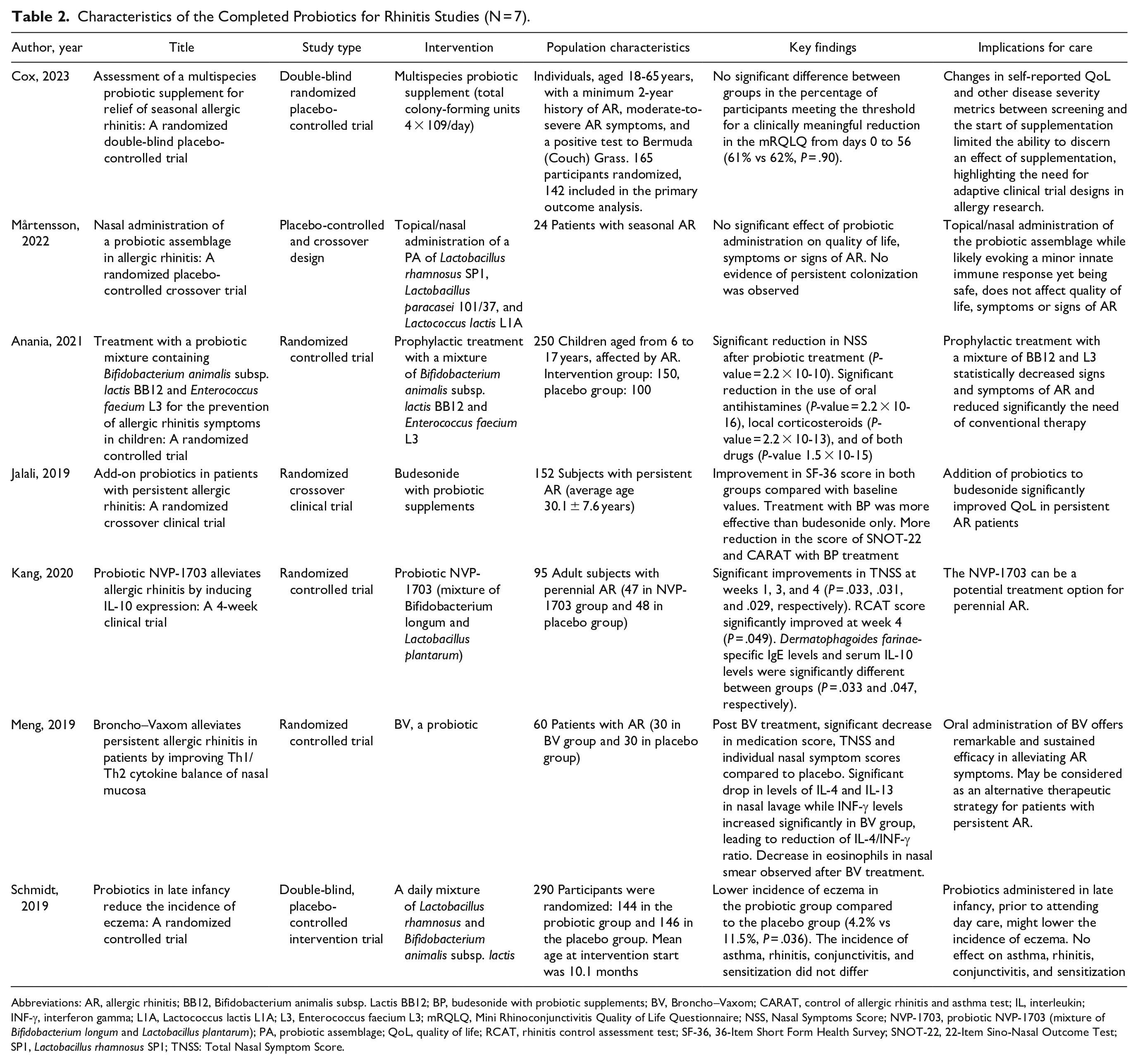
Abbreviations: AR, allergic rhinitis; BB12, Bifidobacterium animalis subsp. Lactis BB12; BP, budesonide with probiotic supplements; BV, Broncho–Vaxom; CARAT, control of allergic rhinitis and asthma test; IL, interleukin; INF-γ, interferon gamma; L1A, Lactococcus lactis L1A; L3, Enterococcus faecium L3; mRQLQ, Mini Rhinoconjunctivitis Quality of Life Questionnaire; NSS, Nasal Symptoms Score; NVP-1703, probiotic NVP-1703 (mixture of Bifidobacterium longum and Lactobacillus plantarum); PA, probiotic assemblage; QoL, quality of life; RCAT, rhinitis control assessment test; SF-36, 36-Item Short Form Health Survey; SNOT-22, 22-Item Sino-Nasal Outcome Test; SP1, Lactobacillus rhamnosus SP1; TNSS: Total Nasal Symptom Score.
In a randomized controlled trial by Anania et al, 58 250 children with AR were given a prophylactic treatment of a mixture of Bifidobacterium animalis subsp. Lactis BB12 and Enterococcus faecium L3. This resulted in a significant reduction in Nasal Symptoms Score, and a notable decrease in the use of oral antihistamines and local corticosteroids. This implies that such a probiotic mixture could be an effective way to lessen signs and symptoms of AR and reduce reliance on conventional therapy.
Similar positive results were reported by Kang et al 60 (2020) and Meng et al, 61 in studies involving probiotic mixtures of Bifidobacterium longum and Lactobacillus plantarum, and Broncho–Vaxom , respectively. Both studies reported significant improvements in nasal symptom scores and other measures of AR severity, suggesting potential benefits of these probiotics in AR management.
Jalali et al 59 also found that the addition of probiotics to budesonide, a corticosteroid used in AR treatment, improved quality of life in persistent AR patients. This finding demonstrates the potential synergistic effect of combining probiotics with existing AR treatments.
However, not all studies have reported beneficial effects of probiotics on AR. In a study by Mårtensson et al, 57 topical administration of a probiotic assemblage of different Lactobacillus and Lactococcus species did not affect quality of life, symptoms, or signs of AR in 24 patients with seasonal AR. A study by Cox et al 53 further reported mixed results. Using a multispecies probiotic supplement, this study found no significant difference in the improvement of quality of life between the probiotic and placebo groups in a population of 165 participants with AR. The authors suggested that changes in self-reported quality of life and other disease severity metrics between the start and end of supplementation may have obscured the effects of supplementation, stressing the need for adaptive clinical trial designs in allergy research. Finally, Schmidt et al 62 evaluated the preventive effects of a probiotic mixture on various allergic symptoms in infants. They found that the probiotics lowered the incidence of eczema, but did not affect the incidence of asthma, rhinitis, conjunctivitis, or sensitization.
Ongoing Clinical Trials
An index of all experimental therapies administered in these trials is given in Table 3.
Name, Mechanism of Action, and Proposed Application to Rhinitis of all the Interventions Included in This Study.
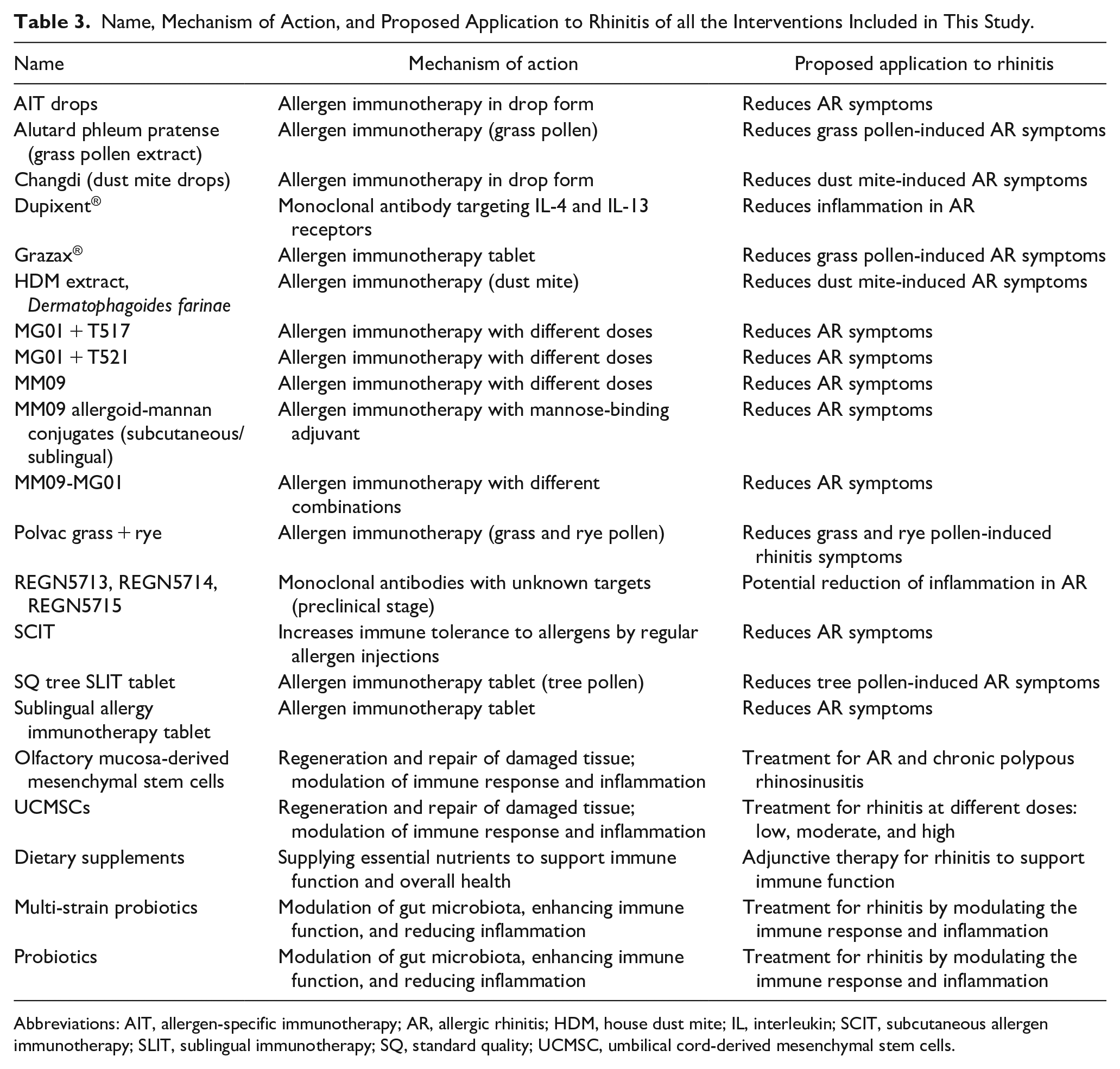
Abbreviations: AIT, allergen-specific immunotherapy; AR, allergic rhinitis; HDM, house dust mite; IL, interleukin; SCIT, subcutaneous allergen immunotherapy; SLIT, sublingual immunotherapy; SQ, standard quality; UCMSC, umbilical cord-derived mesenchymal stem cells.
Ongoing Immunotherapy Trials
A total of 18 trials were identified, pooling in 5745 participants. Two trials (11.1%) are classified as not applicable. One trial (5.6%) is in Phase 1, whereas 4 trials (22.2%) are in Phase 2, with one of these trials being a combined Phase 1 and Phase 2 trial. The majority of the trials, 10 in total (55.6%), are in Phase 3, and one of these trials is a combined Phase 2 and Phase 3 trial. Lastly, there is one trial (5.6%) in Phase 4. The completion dates for the 18 ongoing trials range from April 2023 to June 2026 (Table 4).
Characteristics of Ongoing Trials of Immunotherapy for Rhinitis.
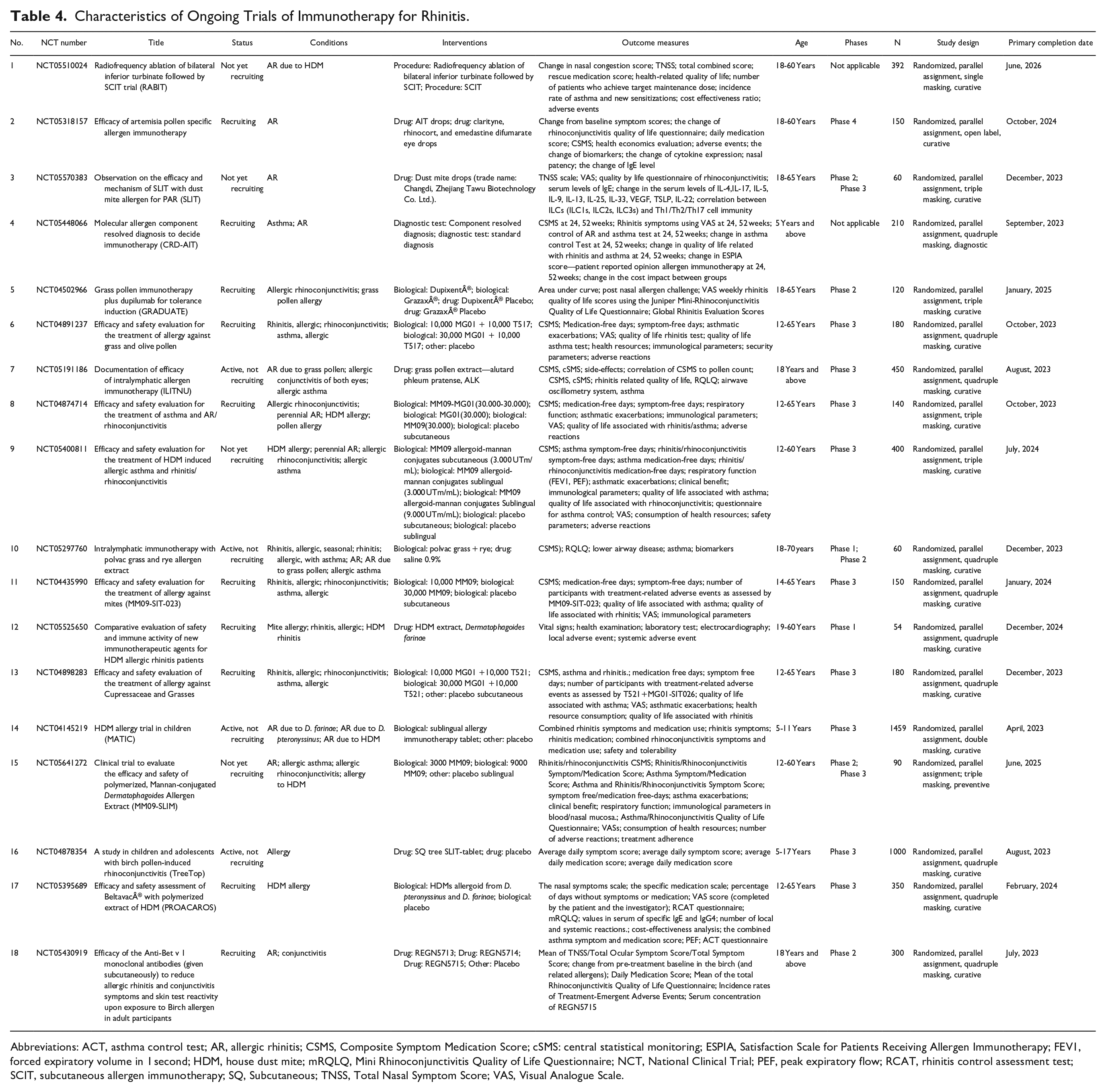
Abbreviations: ACT, asthma control test; AR, allergic rhinitis; CSMS, Composite Symptom Medication Score; cSMS: central statistical monitoring; ESPIA, Satisfaction Scale for Patients Receiving Allergen Immunotherapy; FEV1, forced expiratory volume in 1 second; HDM, house dust mite; mRQLQ, Mini Rhinoconjunctivitis Quality of Life Questionnaire; NCT, National Clinical Trial; PEF, peak expiratory flow; RCAT, rhinitis control assessment test; SCIT, subcutaneous allergen immunotherapy; SQ, Subcutaneous; TNSS, Total Nasal Symptom Score; VAS, Visual Analogue Scale.
Out of the 18 different treatments studied, 2 (11.1%) were allergen immunotherapy in drop form, 2 (11.1%) were allergen immunotherapy for grass pollen, 1 (5.6%) involved a monoclonal antibody targeting IL-4 and IL-13 receptors, and 3 (16.7%) were allergen immunotherapy tablets. Additionally, there was 1 (5.6%) allergen immunotherapy for dust mites, 3 (16.7%) allergen immunotherapies with different doses, 1 (5.6%) allergen immunotherapy with a mannose-binding adjuvant, and 1 (5.6%) allergen immunotherapy with different combinations. Furthermore, 1 (5.6%) study focused on allergen immunotherapy for both grass and rye pollen, 1 (5.6%) involved monoclonal antibodies with unknown targets in the preclinical stage, and 1 (5.6%) centered on SCIT, which increases immune tolerance to allergens through regular allergen injections. Finally, 1 (5.6%) study investigated an allergen immunotherapy tablet specifically for tree pollen.
The outcome measures for the 18 ongoing trials can be grouped under 6 themes as follows.
Symptom scores and quality of life: Change in nasal congestion score, TNSS; Combined Symptom and Medication Score (CSMS), Visual Analogue Scale (VAS), Rhinoconjunctivitis Quality of Life Questionnaire (RQLQ), control of AR and asthma test, asthma control test, Global Rhinitis Evaluation Scores, and ESPIA score—patient reported opinion allergen immunotherapy.
Medication and treatment-related outcomes: Rescue medication score, daily medication score, medication-free days, symptom-free days, number of patients who achieve target maintenance dose, treatment-related adverse events, asthmatic exacerbations, clinical benefit.
Immunological and biomarker changes: Change in IgE levels, change in cytokine expression (IL-4, IL-17, IL-5, IL-9, IL-13, IL-25, IL-33, VEGF, TSLP, IL-22), correlation between ILCs (ILC1s, ILC2s, ILC3s) and Th1/Th2/Th17 cell immunity, and immunological parameters.
Respiratory function and lower airway disease: Nasal patency, respiratory function (FEV1, PEF), lower airway disease, airwave oscillometry system, asthma.
Health economics and resource utilization: Cost-effectiveness ratio, health economics evaluation, change in the cost impact between groups, and consumption of health resources.
Safety and adverse events: Adverse events, the incidence rate of asthma and new sensitizations, local and systemic adverse events, number of adverse reactions, and safety parameters.
Probiotics
A total of three ongoing trials have been identified, which include 335 participants. Among these trials, two are classified as not applicable, and one is in Phase 4 of testing. The completion dates for these trials range from June to December 2023 (Table 5).
Characteristics of Ongoing Trials of Probiotics for Rhinitis.
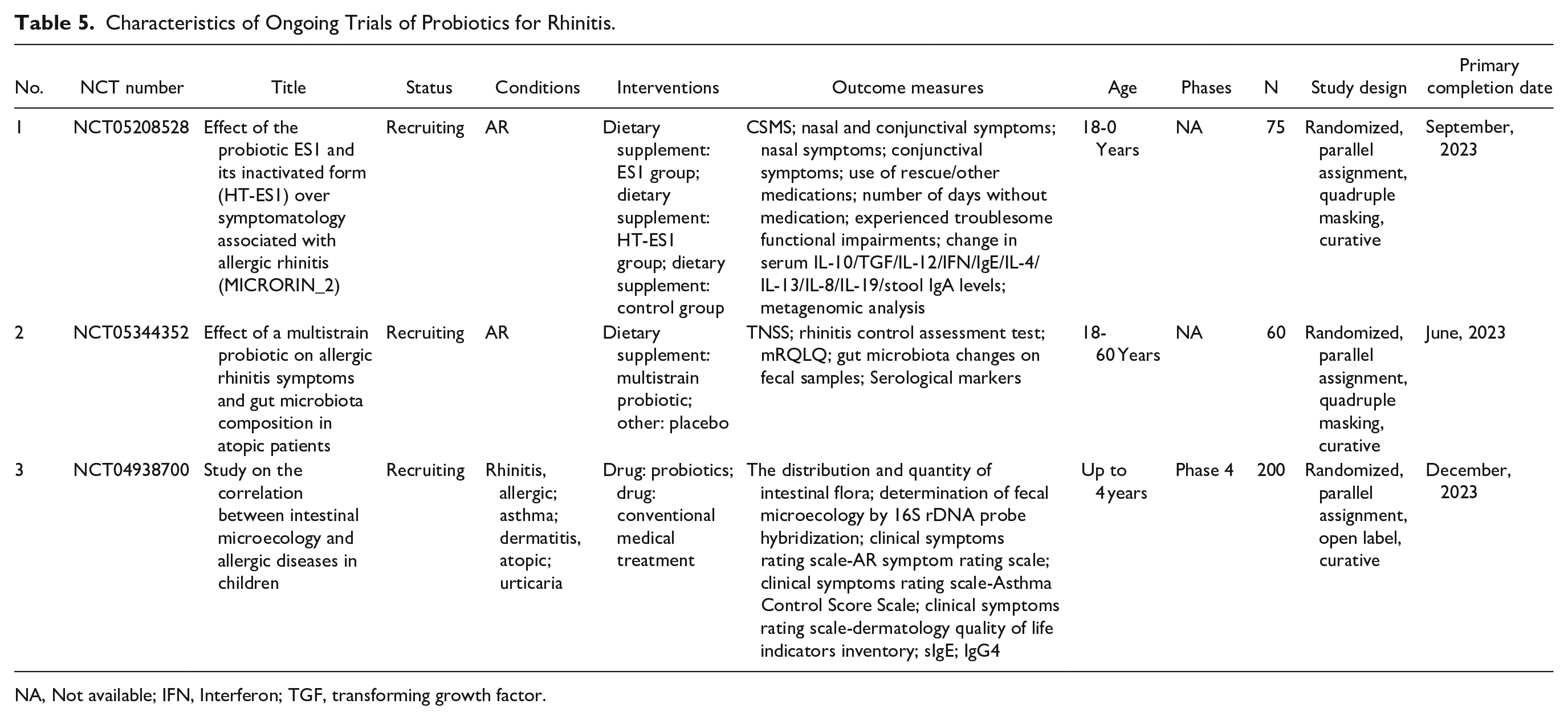
NA, Not available; IFN, Interferon; TGF, transforming growth factor.
The therapies administered in these trials are as follows: dietary supplements (ES1 group, heat treated version of ES1 [HT-ES1] group or control), multi-strain probiotics compared to placebo and probiotics versus conventional medical treatment.
The outcome measures for these trials include:
Symptom and medication assessments: CSMS, nasal and conjunctival symptoms, nasal symptoms, conjunctival symptoms, use of rescue/other medications, number of days without medication, experienced troublesome functional impairments, TNSS, rhinitis control assessment test.
Quality of life assessments: Mini Rhinoconjunctivitis Quality of Life Questionnaire, clinical symptoms rating scale-dermatology quality of life indicators inventory.
Immunological and serological markers: Change in serum IL-10/TGF/IL-12/IFN/IgE/IL-4/IL-13/IL-8/IL-19/stool IgA levels, serological markers, sIgE, IgG4.
Microbiota and metagenomic analysis: Gut microbiota changes on fecal samples, the distribution and quantity of intestinal flora, determination of fecal microecology by 16S rDNA probe hybridization, metagenomic analysis.
Disease-specific clinical assessments: Clinical symptoms rating scale-AR symptom rating scale, and clinical symptoms rating scale-asthma control score scale.
Stem Cell Therapy
A total of 2 trials are identified enrolling 78 participants. The trials are in Phases 1 to 2 of testing, with completion dates spanning December 2023 to December 2024 (Table 6). The therapies being administered in these trials include (1) biological treatment with olfactory mucosa-derived mesenchymal stem cells, alongside standard treatment for AR, and chronic polypous rhinosinusitis according to clinical protocols; and (2) biological treatment with umbilical cord-derived mesenchymal stem cells at three different doses: low dose, moderate dose, and high dose.
Characteristics of Ongoing Trials of Stem Cell Therapy for Rhinitis.
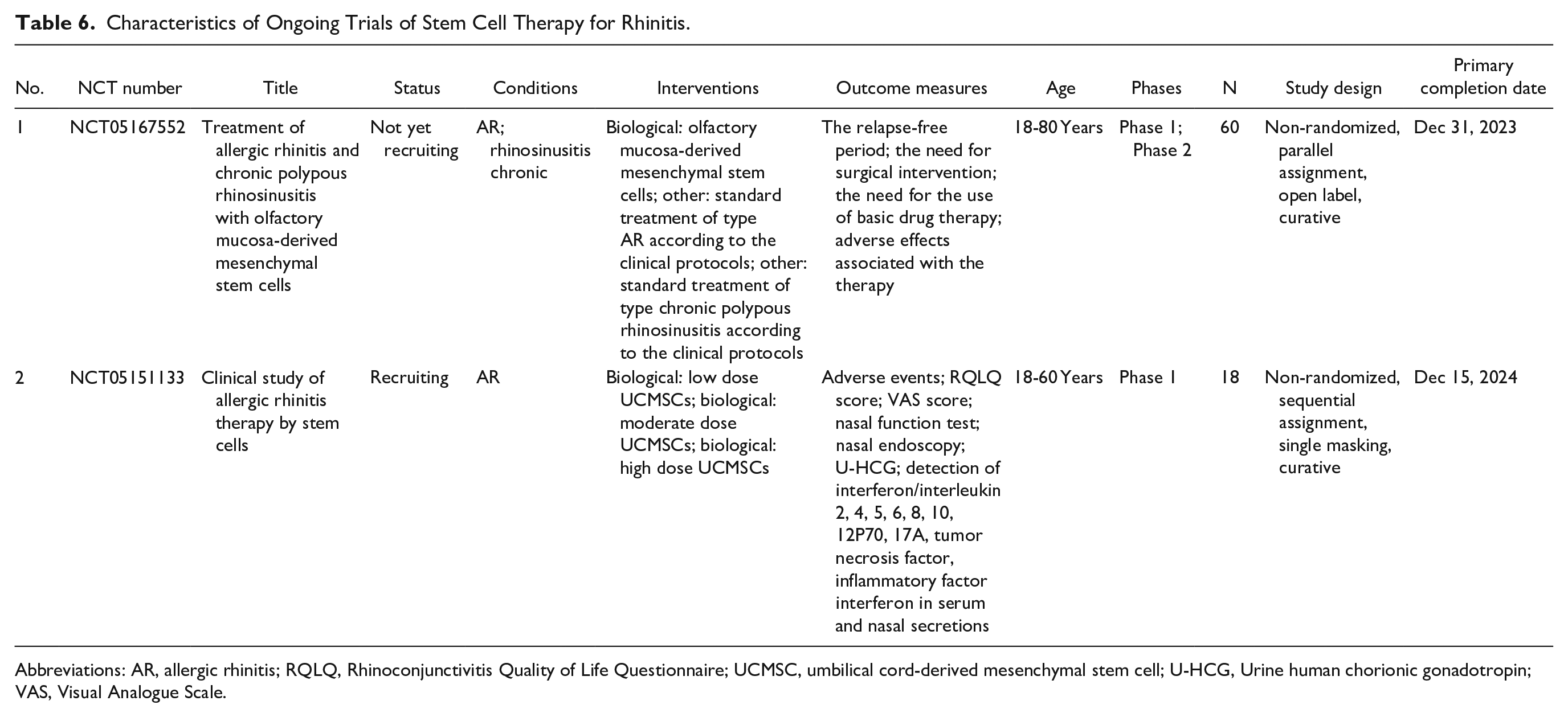
Abbreviations: AR, allergic rhinitis; RQLQ, Rhinoconjunctivitis Quality of Life Questionnaire; UCMSC, umbilical cord-derived mesenchymal stem cell; U-HCG, Urine human chorionic gonadotropin; VAS, Visual Analogue Scale.
The outcome measures for these trials encompass a variety of aspects, including the relapse-free period, the necessity for surgical intervention, the requirement for basic drug therapy, and any adverse effects. Additionally, the trials examine patient-centered outcomes such as the RQLQ score and the VAS score, which help to evaluate the therapy’s impact on patient’s daily lives. Furthermore, clinical assessments like the nasal function test, urine human chorionic gonadotropin, and the detection of various cytokines and inflammatory factors (interferon/interleukin 2, 4, 5, 6, 8, 10, 12P70, 17A, tumor necrosis factor) in serum and nasal secretions are used to determine the therapy’s effect on the immune response and inflammation.
Discussion
This systematic review has shown the wide scope of research being conducted on therapies for rhinitis, including 51 completed trials and 23 ongoing trials. Completed clinical trials have focused mainly on immunotherapy (44 out of 51 trials) and probiotics (7 out of 51 trials), contributing significantly to our understanding of the efficacy and safety of these interventions in rhinitis management; no completed studies were identified for stem cell therapy. Particularly, trials investigating probiotics demonstrate a shift in understanding of rhinitis treatment, with a focus on modulating the immune system and nasal microflora balance. Completed trials have investigated multiple facets of these therapies, such as symptom management, quality of life, immunological markers, and changes in nasal microbiota, to provide a holistic view of rhinitis treatment.
The 23 ongoing trials, on the other hand, are paving the way for the development of newer, more targeted therapies. Eighteen of these ongoing trials are investigating immunotherapies, 3 trials focus on probiotics, and 2 trials are researching stem cell therapies. Of these, a majority of immunotherapy trials have progressed to Phase 3, which indicates the potential imminent emergence of new treatment options. Meanwhile, stem cell therapy trials are currently in the early Phases 1 and 2, indicative of an exciting but exploratory phase in treatment possibilities for rhinitis.
The importance of personalized and targeted treatment strategies cannot be overstated. 63 This systematic review highlights a move toward individualized care plans for rhinitis patients, particularly with the advent of emerging therapies. 64 As clinicians become more familiar with these novel interventions, the management of rhinitis will likely evolve, necessitating modifications to existing treatment protocols to optimize patient outcomes.65,66
Moreover, this review has drawn attention to the potential for combination therapies and novel treatment modalities, suggesting a new frontier for the treatment of rhinitis. As these studies progress, we may see synergistic effects between therapies that enhance efficacy or even address aspects of the disease that were previously difficult to treat. With these advancements, the future of rhinitis treatment appears promising, and further research will undoubtedly continue to refine our understanding of these novel therapies and their roles in clinical practice.
Recent literature highlights the increasingly important role of immunotherapy, probiotics, and stem cell therapies in the management of rhinitis. For instance, Badorrek et al 67 reported promising results exploring a new form of immunotherapy, affirming the findings of our review that many ongoing trials are in the advanced stages of immunotherapy research. While our systematic review found most completed trials in immunotherapy further research is required particularly in identifying potential biomarkers for predicting the response to immunotherapy.
In terms of probiotics, the meta-analysis by Du et al 68 showed that while some trials demonstrated positive effects of probiotics in reducing rhinitis symptoms, others revealed no significant impact. This is consistent with the results of our review, where probiotic trials showed mixed results. It highlights the complexity of probiotic interventions, and the need for more research to understand the conditions under which probiotics might be most beneficial for patients with rhinitis.
Stem cell therapies represent an emerging field in rhinitis management. According to a study by Wang et al, 69 while preclinical trials show promise, few clinical trials have been completed. This contrasts with our findings, where two ongoing stem cell therapy trials were identified. It suggests that while this area is still in its infancy, the pace of research in stem cell therapies for rhinitis is increasing.
The current literature reflects the findings of our systematic review in many aspects. The rise of immunotherapy, the exploration of probiotics, and the emergence of stem cell therapies are all areas that require further research. However, the increasing number of completed and ongoing trials in these fields represents a significant step forward in rhinitis management, providing a greater understanding of the pathophysiology of the disease and paving the way for more effective and personalized treatments.
Conclusion
In summary, this systematic review provides an extensive and comprehensive overview of clinical trials in emerging therapeutic interventions for rhinitis. Our findings highlight the vast potential these novel approaches hold for augmenting patient outcomes and enriching the quality of life in individuals suffering from this pervasive condition. The spectrum of trials—spanning immunotherapy, probiotics, and burgeoning stem cell therapies—elucidates the dynamism of research in this field.
In light of the current landscape, there is a promising trend toward more personalized and precision-based treatment strategies, reflecting the complex nature of rhinitis and the need for tailor-made solutions. Continued innovation and rigorous scientific exploration in rhinitis therapy remain critical for propelling these emerging therapies from clinical trials to bedside, thus enhancing the armamentarium of options available to patients and clinicians. Ultimately, these developments harbor the promise of transforming the current paradigm of rhinitis management, leading to more efficient and targeted treatments that can mitigate the burdensome impact of this global condition.
Footnotes
Acknowledgements
The authors wish to acknowledge the following for their early contributions to the manuscript: Sidra Iqbal, Gohar Ur Rahman, and Mehak Khadim.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participant
All methods were carried out in accordance with relevant guidelines and regulations (declaration of Helsinki).
Data Availability Statements
All data utilized for the purpose of this study are available publicly and online.
