Abstract
Background:
Specific immunotherapy is an important immune-modifying treatment for patients with allergic rhinitis (AR). We compared the early efficacy and safety of cluster and conventional immunotherapies for patients with AR.
Methods:
One hundred forty-nine patients with persistent AR were enrolled in a randomized and open-label trial and were divided into the following 4 groups: 60 children treated conventionally, 33 children treated using the cluster schedule, 23 adults treated conventionally, and 33 adults treated using the cluster schedule. Patients in the cluster groups reached the maintenance dose within 6 weeks, while those receiving conventional therapy reached the maintenance dose within 14 weeks. Symptom scores and skin prick test scores (SPTs) were used to evaluate clinical efficacy and adverse reactions.
Results:
After buildup phase of treatment, symptom scores, and SPTs were significantly lower than those prior to treatment in each group (P < .05). No significant differences were found in the efficacy of nasal symptoms scores among four groups (P > .05).However, the efficacy of SPTs using conventional schedule was higher than cluster schedule in children groups (group A and B, 57.7 vs 30.2%, P = .001). Besides, the efficacy of SPTs in adults was higher than children when using the cluster treatment (group D and B, 53.0 vs 30.2%, P = .008). No severe adverse reaction occurred.
Conclusions:
Conventional and cluster immunotherapy schedules have similar efficacies, which do not vary with age; both schedules are safe and reliable. Also, SPT facilitate evaluation of clinical efficacy.
Introduction
Allergic rhinitis (AR) is mediated by immunoglobulin E (IgE) and a variety of immune cells and cytokines involved in inflammation of the nasal passages and is one of the most common otorhinolaryngological diseases. The prevalence of AR has in recent years increased significantly, concomitant with industrialization and increasing levels of pollution in the environment. 1 Although AR is not life-threatening, it has a marked effect on the quality of life of those afflicted and imposes an economic burden on society. 2,3 This condition, if poorly controlled, can also increase the risk of asthma. 4 Therefore, AR should be treated using effective modalities.
Therapies for AR include antihistamines, nasal steroids, and allergen-specific immunotherapy (SIT) as well as avoiding contact with allergens. 5 Specific immunotherapy is likely the only means of influencing the natural course of allergic diseases. 6 In addition to altering the natural course of AR, the clinically observed effects of SIT include preventing sensitization to new allergens and long-term efficacy after treatment cessation. Subcutaneous immunotherapy (SCIT) has many therapeutic benefits, but only a small proportion of AR patients select this treatment and comply with the regimen, 7 with inconvenience likely being the primary reason for treatment discontinuation. 8 Conventional immunotherapy includes 2 phases—a buildup phase and a dose-maintenance phase—with the buildup phase lasting nearly 4 months and the maintenance phase for at least 3 years. Thus, a new regimen that both shortens the therapeutic duration and does not increase morbidity due to side effects is needed. 9
Dermatophagoides pteronyssinus is one of the major allergens of AR and asthma. Up to 64.6% of patients with AR are positive for D pteronyssinus in cities in northern China, such as Beijing, 10 and up to 90.5% in southern cities, such as Wuhan. 11 Several clinical trials have demonstrated the effectiveness of SCIT for the treatment of AR, asthma, and allergic conjunctivitis. 12 -14 Conventional immunotherapy with the standard dust mite allergen vaccine has been effective in China. Here, we test the effectiveness of the so-called “cluster” immunotherapy, in which the treatment schedule is accelerated relative to that in conventional therapy. This has the advantage of a reduced number of hospital visits, which reduces the time and expense required. Several studies have compared the efficacy and safety of cluster schedules versus conventional approaches, 15 -17 but few reports on the efficacy and safety of the 2 SIT regimens in the initial stage have been published. In this randomized, open-label study, we compared the efficacy and safety of the build-up phase of the cluster schedule with that of the conventional schedule. We also compared the efficacy of the 2 schedules in children and adults allergic to D pteronyssinus and Dermatophagoides farinae.
Materials and Methods
This randomized, open-label study was approved by the ethics committee of the First Affiliated Hospital of Nanchang University.
Patients
In total, 149 patients were consecutively recruited from the AR clinic of the First Affiliated Hospital of Nanchang University. All patients underwent a skin prick test (SPT) using histamine (positive control) and standard extracts (ALK-Abelló, Hørsholm, Denmark) of the following allergens: D pteronyssinus, D farinae, German cockroach, ragweed, cat and dog dander, Alternaria tenuissima, Artemisia argyi, timothy grass, Taraxacum mongolicum, Platanus orientalis, Monilia albicans, and Gramineae. The mean of the largest wheal diameter and its perpendicular diameter were recorded as the result of the SPT. The skin test index (SI) was defined as the ratio of the mean diameter of a wheal produced in response to an allergen compared to that to histamine (positive control). 15 The following inclusion criteria were applied: a clear clinical history of AR, positive SPT (wheal diameter ≥3 mm and an SI ≥0.5) for D pteronyssinus and/or D farinae, and a positive specific IgE to D pteronyssinus and/or D farinae in serum of ≥0.7 kU/L (CAP Pharmacia, Uppsala, Sweden). None of the patients had a positive SPT response to German cockroach, ragweed, or any other inhalant allergen, with the exceptions of D pteronyssinus and D farina. Patients who had undergone allergen immunotherapy previously and had contraindications to immunotherapy 18 were excluded. The patients were divided into the following 4 groups: children (ie, patients <14 years of age) treated conventionally (group A), children treated with the cluster schedule (group B), adults (ie, patients ≥14 years of age) treated conventionally (group C), and adults treated with the cluster schedule (group D). All patients provided written informed consent to participate in this study. Specific immunotherapy was performed according to the World Health Organization recommendations 19 and the guidelines of the European Academy of Allergology and Clinical Immunology (EAACI). 18 Patients with asthma were diagnosed and classified as having mild asthma according to the Global Initiative for Asthma (http://www.ginasthma.org). Patients with atopic dermatitis were diagnosed as suggested by the British Association of Dermatologists. 20
Immunotherapy Schedules
Patients receiving SIT were pretreated with loratadine before each visit. Peak expiratory flow rates were recorded before and 30 minutes after injections and were required to attain 80% of the predicted value. The patients were observed for at least 30 minutes after each injection and adverse reactions were recorded. Patients were excluded from the study and the therapy was interrupted if severe adverse systemic reactions occurred.
Conventional immunotherapy
Specific immunotherapy was administered using a commercial standardized depot preparation of D pteronyssinus extract (Alutard SQ; ALK-Abelló). We used a standardized treatment schedule recommended by the Allergy Department of the University of Copenhagen. 21 The therapy began with 20 SQ-U, which was increased weekly according to the following scheme: 20, 40, 80, 200, 400, 800, 2000, 4000, 8000, 10 000, 20 000, 40 000, 60 000, 80 000, and 100 000 SQ-U, for a total of 15 injections over 14 weeks during the buildup phase (see Table 1). 15 The patients were given only 1 injection for each visit in the conventional schedule. The maintenance dose of 100 000 SQ-U was maintained during the maintenance phase, and the injection interval gradually extended to 6 weeks.
Conventional and Cluster Dosage Schedules.a
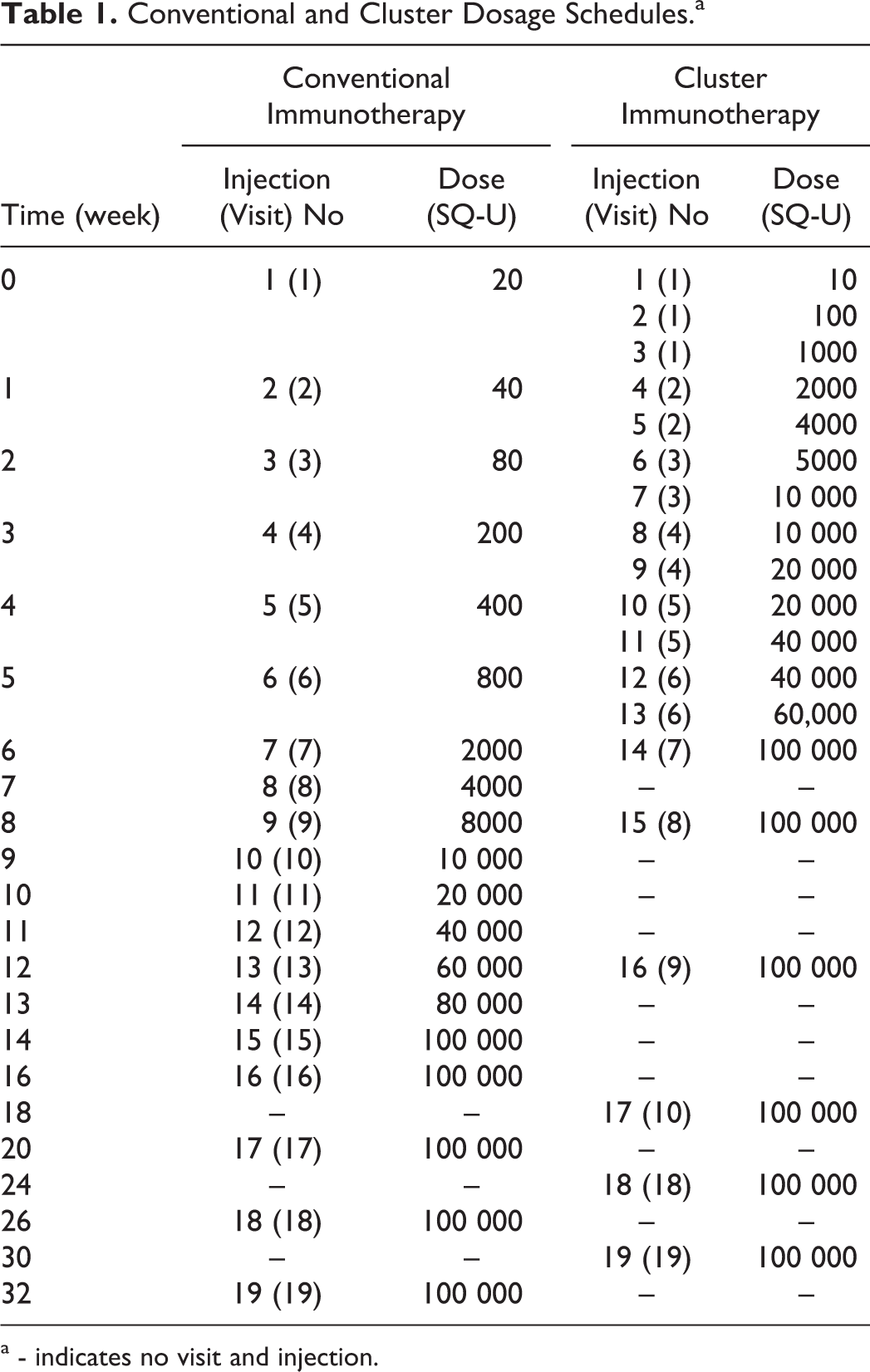
a - indicates no visit and injection.
Cluster immunotherapy
Patients received subcutaneous injections of Alutard SQ, with a first dose of 10 SQ-U. Three injections were given on the first visit and then 2 injections afterward, with an interval of 30 minutes between these injections to evaluate adverse reactions. The maximum dose during the buildup phase; that is, 100 000 SQ-U, was administered at week 6. This dose was continued during the subsequent maintenance phase, during which the interval between injections was gradually increased to 6 weeks (Table 1).
Evaluation of Clinical Efficacy
As mentioned above, patients were pretreated with loratadine before each visit. To evaluate clinical efficacy at the end of the buildup phase, we sought to exclude the influence of other drugs as much as possible. The patients were required to refrain from using a variety of systemic and intranasal topical agents for AR, including corticosteroids, antihistamines, antileukotrienes, and so on. During the study, patients took corticosteroids and other drugs only if adverse reactions occurred. When grade 1 (unspecific symptoms such as headache, discomfort or arthralgia, etc) or grade 2 (mild systemic reactions—mild rhinitis or asthma) adverse reactions occurred, the patients took antihistamines or β2-agonists. When grade 3 or 4 adverse reactions occurred, the patients took β2-agonists, systemic corticosteroids, or antihistamines. Nasal symptom and SPT scores were evaluated at least 2 weeks after taking the abovementioned drugs. Clinical efficacy was evaluated using a visual analog scale (VAS) at the end of the SIT buildup phase. 22 The improvement ratio—defined as (total score before treatment—after treatment)/total score before treatment × 100%—was used as the evaluation criterion; values ≥66% were considered high efficacy, those between 26% and 65% were considered moderate efficacy, and those ≤25% indicated no efficacy. 23 The efficacy %—defined as (number of high efficacy and moderate efficacy)/total number × 100%. Symptom scores and skin prick scores were recorded before and after the buildup phase.
Nasal symptom scores
Nasal symptom scores were recorded on VAS 24 ranging from 0 (very good) to 100 (very poor). This score included 5 specific symptoms (nasal congestion, nasal itching, sneezing, runny nose, and itchy eyes).
Skin prick test score
Before and after the buildup stage, each patient underwent an SPT (Soluprick SQ; ALK-Abelló S.A.). Histamine was used as the positive control and normal saline as the negative control. Skin pricks were conducted on the volar forearm, and the results were recorded after 15 minutes. The mean of the largest wheal diameter and its perpendicular diameter were recorded as the result of the SPT. The SI was defined as the ratio of the mean diameter of a wheal produced in response to an allergen compared to that to histamine (positive control). 15 The SI = 0 is (−), SI < 1/2 is (+), 1/2 < SI < 1 is (++), 1 < SI < 2 is (+++), and SI > 2 is (++++). And the levels corresponded to the difference of skin prick test before and after the buildup phase was, respectively, (−, 0 score), (+, 1 score), (++, 2 score), (+++, 3 score), and (++++, 4 score).
Adverse Reactions to SIT
Adverse reactions were categorized according to the time of appearance (immediate or delayed) and whether they were localized or systemic. Local reactions were quantified by measuring the wheal diameter. Immediate local reactions with a diameter less than 5 cm and delayed local reactions with a diameter less than 10 cm were considered to be clinically irrelevant. Systemic reactions were determined when any of urticaria, AR, asthma, angioedema, or anaphylactic shock was observed. Systemic reactions were graded following the standards of the EAACI. 18 Patients were required to report any symptoms immediately, which were addressed as soon as possible.
Statistical Analysis
Statistical analysis was carried out using SPSS for Windows statistical software (version 17.0; SPSS Inc, Chicago, Illinois). Mean (standard deviation) was used to express the symptom scores and SPTs of 4 groups before and after the buildup phase of treatment. Paired t test was used to analyze differences in symptom scores and SPTs in each group before and after the buildup phase of treatment. A χ2 test was used to determine differences in the efficacy of symptom scores and SPTs among 4 groups after the buildup phase. Differences were considered significant at values of P < .05.
Results
Patients
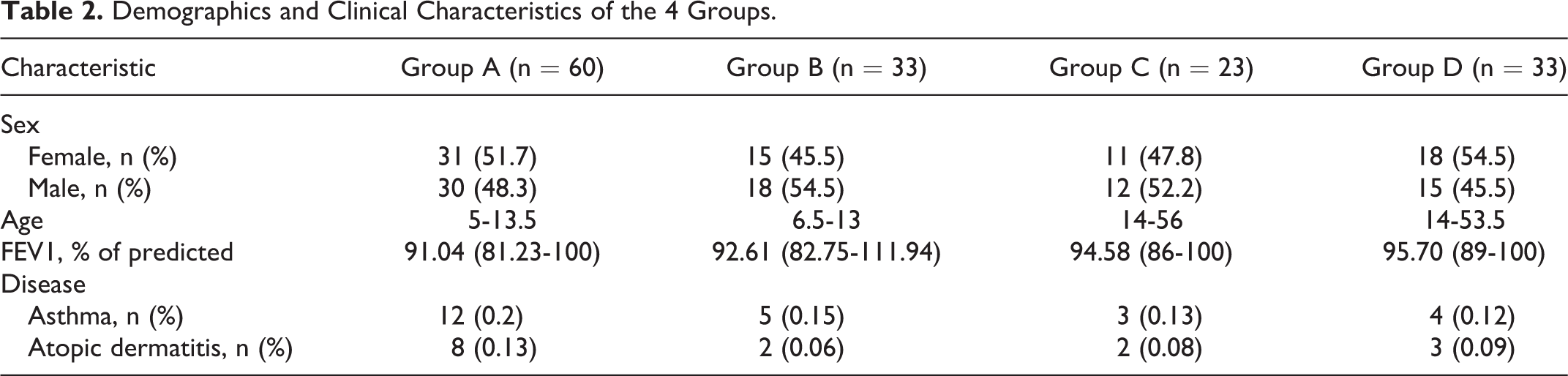
In total, 149 patients with AR were divided into 4 groups. None of the patients dropped out of the study. The sex, age, and clinical characteristics of the patients are summarized in Table 2. These characteristics did not differ significantly between patients in the cluster and conventional groups.
Demographics and Clinical Characteristics of the 4 Groups.
Clinical Efficacy
Symptom scores and SPTs before and after the buildup phase of treatment in each group
After the buildup phase of treatment, there were obvious reductions in symptom and SPTs in each group. In group A, the total symptom scores were 29.61 (6.47) and 10.65 (6.55) before and after the buildup phase of treatment, respectively. In group B, the total symptom scores were 23.45 (9.07) and 13.58 (5.63), respectively. In groups C and D, the total symptom scores before treatment were 23.37 (8.05) and 28.33 (9.47), compared to 11.33 (6.55) and 13.74 (9.87) after the buildup phase, respectively. And there were significantly lower in symptom scores and SPTs in each group at the end of the buildup phase (P < .05; Figure 1).
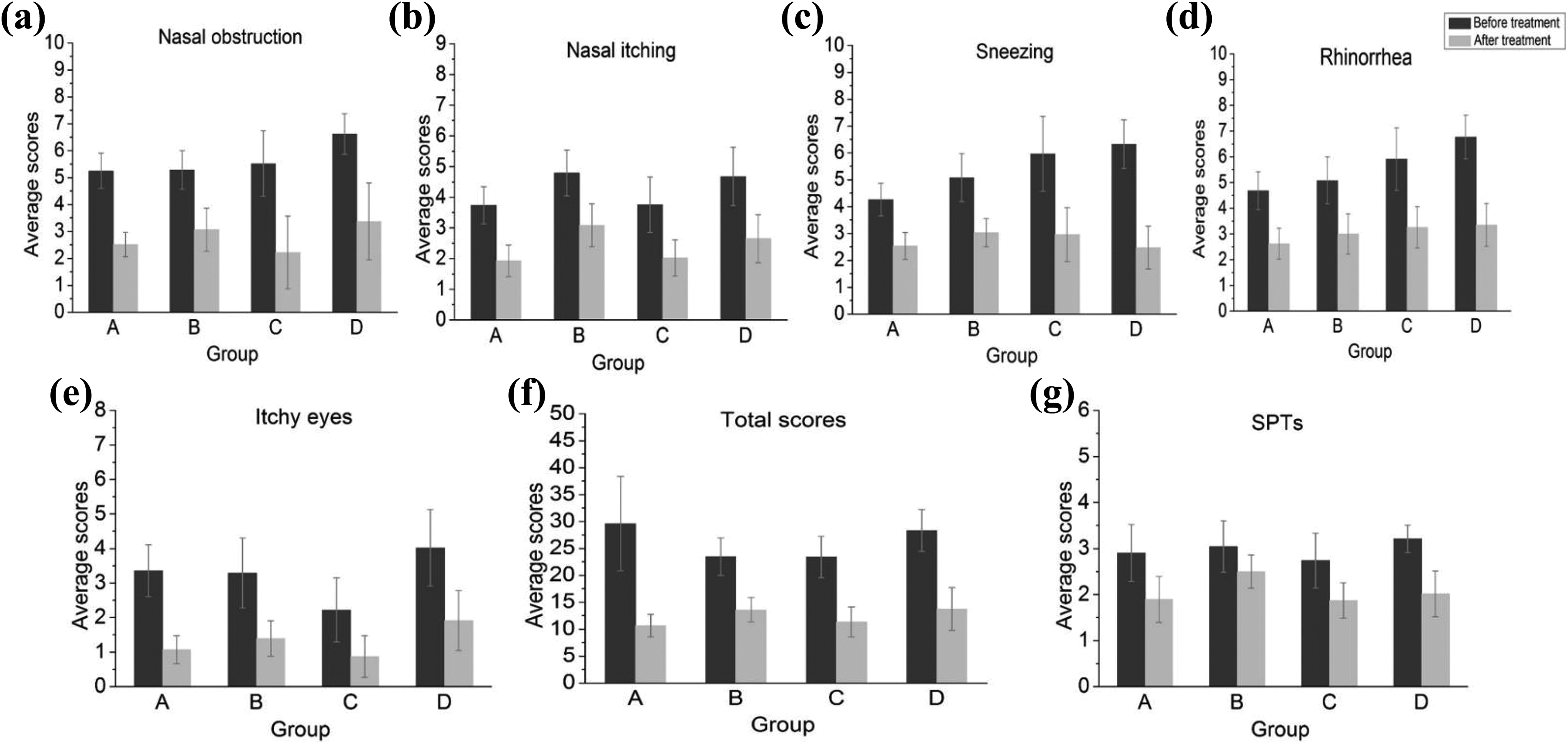
Symptom scores and SPTs before and after treatment (after the build-up stage of treatment) in each group. A, Children treated conventionally. B, Children treated with the cluster schedule. C, Adults treated conventionally. D, Adults treated with the cluster schedule. (a) Nasal obstruction. (b) Nasal itching. (c) Sneezing. (d) Running nose. (e) Itchy eyes. (f) Total scores. (g) SPTs (Skin prick test scores of Dermatophagoides pteronyssinus and Dermatophagoides farinae). (a-g) After the buildup stage of treatment, all the groups showed a significant reduction in symptom scores and SPTs. SPTs indicates skin prick test scores.
Efficacy of symptom scores and SPTs between conventional and cluster treatment in each age-group
In children groups (group A and B), no significant differences in the efficacy of nasal obstruction, nasal itching, sneezing, running nose, itchy eyes, and total symptom scores were observed between 2 schedules (P > .05, Figure 2A-F). However, there were differences in the efficacy of SPTs between group A and group B (57.7 vs 30.2%, P = .001, Figure 2G), suggesting that the efficiency of SPTs using conventional schedule is higher than cluster schedule in children. Whereas, in adult groups (group C and D), the efficacy of nasal symptom scores and SPTs did not differ significantly between 2 schedules (P > .05, Figure 2A-G).
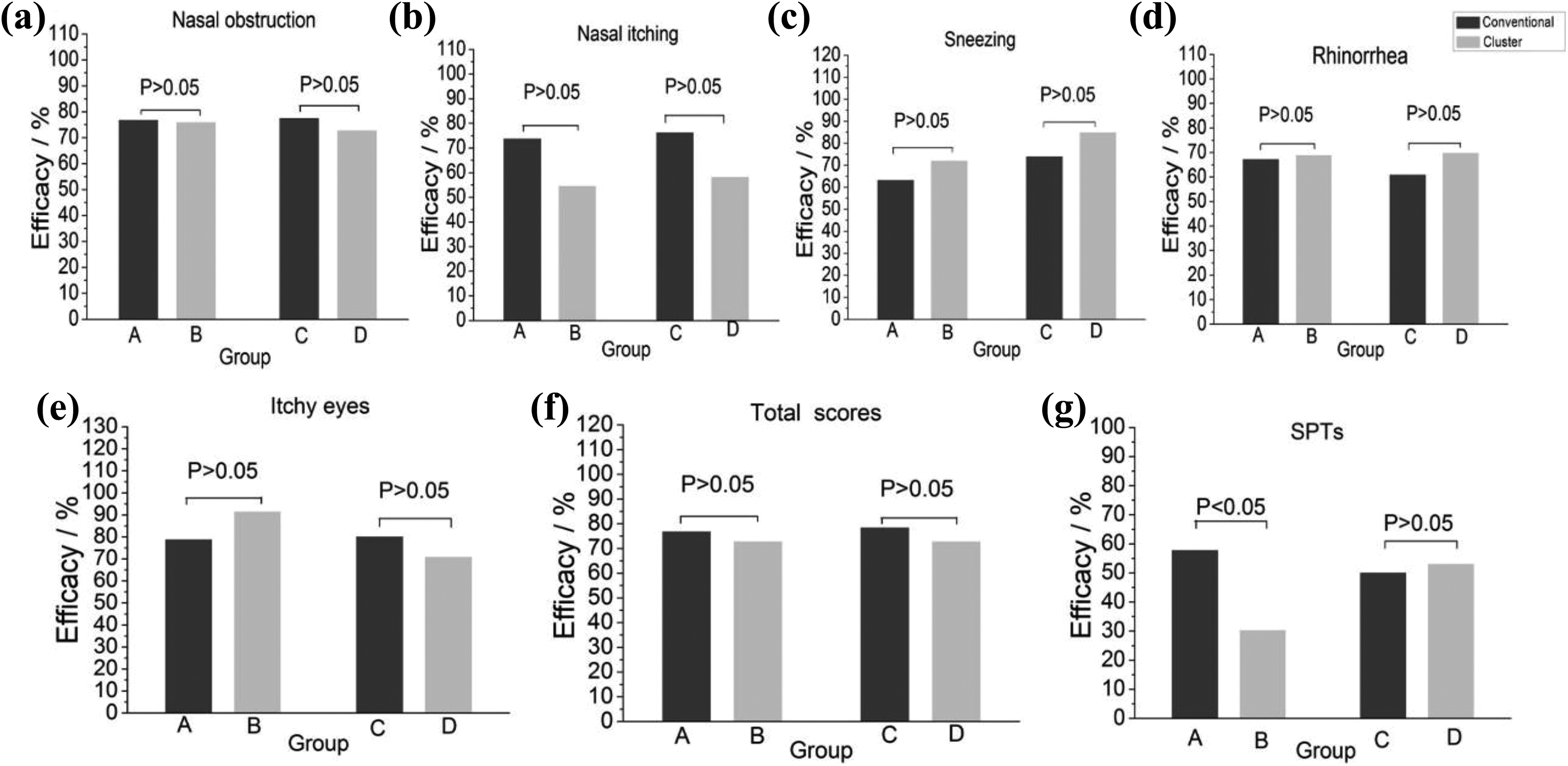
Efficacy of symptom scores and SPTs between conventional and cluster treatment in each age group. A, Children treated conventionally. B, Children treated with the cluster schedule. C, Adults treated conventionally. D, adults treated with the cluster schedule. (a-f) The efficacy of nasal obstruction (a), nasal itching (b), sneezing (c), running nose (d), itchy eyes (e), and total scores (f) between conventional and cluster treatment were similar in each age group. (g) SPTs (Dermatophagoides pteronyssinus and Dermatophagoides farinae), the efficacy of SPTs between conventional and cluster treatment were significantly different in children groups (group A and B). P < .05 indicates significant difference in each age-group. SPTs indicates skin prick test scores.
Efficacy of symptom scores and SPTs between children and adults in each treatment group
In the conventional treatment groups (group A and C), there were no significant differences in the efficacy of symptom scores and SPTs between children and adults (Figure 3A-G, P > .05). Again, in the cluster treatment groups (group B and D), no significant differences were found in the efficiency of nasal obstruction, nasal itching, sneezing, running nose, itchy eyes, and total symptom scores between children and adults (Figure 3A-G, P > .05). Nevertheless, there were differences in the efficacy of SPTs between children and adults (53.0 vs 30.2%, Figure 3G, P = .008), suggesting that the efficiency of SPTs in adults is higher than children when using the cluster treatment.
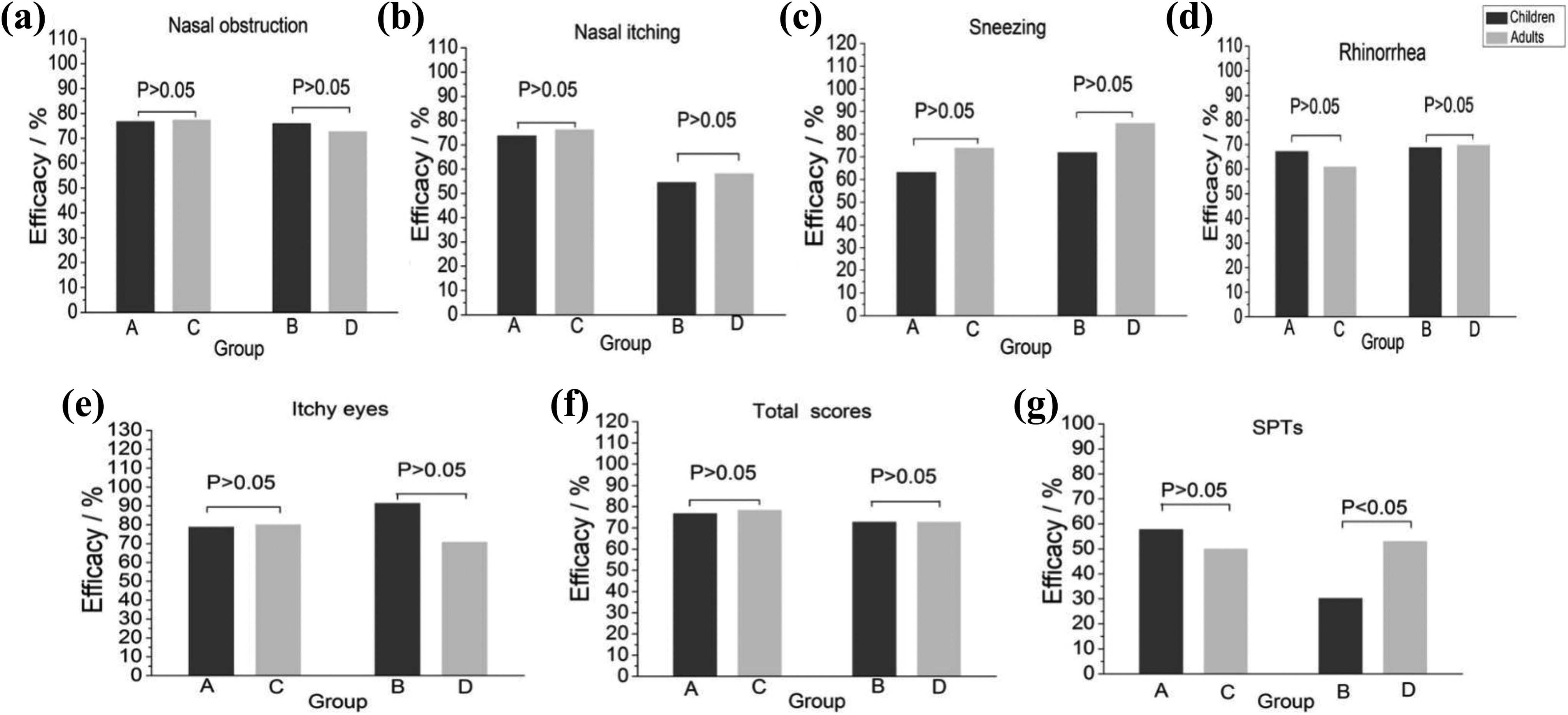
Efficacy of symptom scores and SPTs between children and adults in each treatment group. A, Children treated conventionally. C, Adults treated conventionally. B, Children treated with the cluster schedule. D, Adults treated with the cluster schedule. (a-f) The efficacy of nasal obstruction (a), nasal itching (b), sneezing (c), running nose (d), itchy eyes (e) and total scores (f) between children and adults were similar in each treatment group. (g) SPTs (Dermatophagoides pteronyssinus and Dermatophagoides farinae
Adverse Reactions
Of the 149 patients, 27 (18.1%) experienced adverse reactions (Table 3); 6 (4.03%) of the 149 patients, all from group C, experienced local reactions. In total, 21 (14.09%) of the patients displayed systemic adverse reactions, including 18 cases of grade 1 systemic adverse reactions, 2 cases of grade 2, 1 case of grade 3, and 0 cases of grade 4. The incidences of systemic reactions in the 4 groups were 16.67%, 12.12%, 21.74%, and 6.06%, respectively. The incidence of adverse reactions with cluster schedule was lower than with conventional schedule in children and adults groups, but no significant differences were found between them (group A 16.67% vs group B 12.12%, group C 21.74% vs group D 6.06%, P > .05). Meanwhile, there were no significant differences in the frequency of systemic reactions between children and adults when using both schedules (group A 16.67%vs group C 21.74%, group B 12.12% vs group D 6.06%, P > .05).
Adverse Reactions of the 4 Groups.
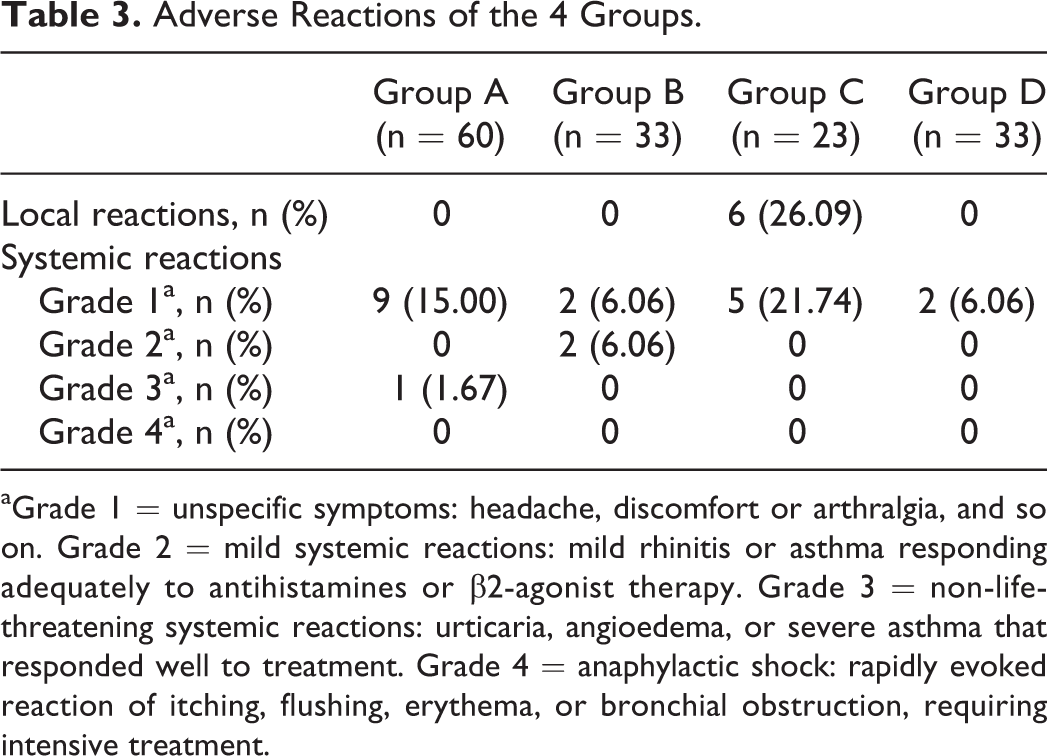
aGrade 1 = unspecific symptoms: headache, discomfort or arthralgia, and so on. Grade 2 = mild systemic reactions: mild rhinitis or asthma responding adequately to antihistamines or β2-agonist therapy. Grade 3 = non-life-threatening systemic reactions: urticaria, angioedema, or severe asthma that responded well to treatment. Grade 4 = anaphylactic shock: rapidly evoked reaction of itching, flushing, erythema, or bronchial obstruction, requiring intensive treatment.
Discussion
Allergic rhinitis causes disturbing chronic symptoms such as nasal blockage, sneezing, itchy nose, and rhinorrhea, which have negative effects on the psychological, physical, and social well-being of children, as well as on their school performance. 25 The main goal of treating AR is to improve the quality of life. Children with a relatively short course of the disease, and in the growth and development period, in whom the immune system is not fully developed, are the best subjects to accept SCIT. 26 However, although the beneficial effects of immunotherapy on AR are well-documented, 12,13 relatively few AR patients choose this treatment. This lack of interest in immunotherapy is mainly due to the inconvenience resulting from the long time required for patients to complete this treatment.
In the present study, we compared the efficacy of the buildup phase of conventional and cluster immunotherapies in both adults and children. When evaluating the effectiveness of immunotherapy, the common and optimal parameters representing efficacy are reductions in symptoms and/or drug intake. 27 In our study, we evaluated the effectiveness of immunotherapy through skin prick tests and symptom scores. The results indicated that, upon completion of the buildup phase of immunotherapy, whether using the conventional or accelerated cluster schedule, the symptom scores of the patients for the most part decreased significantly, which is consistent with the results of an earlier study. 17 In children, the total symptom efficacy were 76.7% and 72.7% in the conventional and cluster groups, respectively. In adults, these values were 78.3% and 72.7%, respectively. However, there was no difference in the efficacy between conventional SCIT and cluster SCIT, which indicates that cluster schedules may be considered an alternative but further investigation is required. In addition, our results are consistent with a study of Tabar, 17 which showed that the improvements in asthma symptoms of patients were similar in the 2 groups at the end of the buildup phase. Moreover, there was no difference in the efficacy of symptoms between adults and children using 2 schedules, indicating that age has nothing to do with efficacy. However, in the current study, we only summarized and analyzed the effects in patients who completed the buildup phase of therapy. It remains to be determined by further clinical observations and analyses whether the long-term efficacies of the cluster and conventional schedules are similar.
In addition, we found that, as the treatment progressed, the skin prick scores decreased, which coincided with an improvement in symptoms. Therefore, we believe that the skin prick test can prognosticate the clinical efficacy of treatment. Of course, since in the current study patients were only followed for the buildup phase of treatment, further observations are necessary to fully assess the relationship between skin prick test results and clinical efficacy. Surprisingly, in our research, we found that there were differences in the efficacy of SPTs between 2 schedules in children groups and the efficacy of SPTs was higher in conventional schedule than in cluster schedule (57.7% vs 30.2%). However, no differences were observed in the efficacy of SPTs between 2 schedules in adult groups. Besides, in the cluster treatment groups, there were differences in the efficacy of SPTs between children and adults, and the efficacy was higher in adults (53.0% vs 30.2%). These results are difficult to explain at present, but we will continue to observation it in the maintenance phase to see whether the results in the maintenance phase are consistent with that in the buildup phase.
The duration of cluster immunotherapy is markedly shorter than that of conventional SCIT. However, it may increase the risk of local and systemic adverse reactions. Adverse reactions mainly occur in the middle and late periods of the buildup stage and early in the maintenance phase, and the dose in this period change greatly, which is susceptible to adverse reactions, suggesting that we should focus on it. If a patient displays serious systemic adverse reactions during this process, the injected dose should be adjusted in a timely manner. In addition, although the majority of adverse reactions occur within 30 minutes of an injection, some adverse reactions occur after 24 hours. In view of this, patients should be observed after the injection in an observation room for at least half an hour and should be consistently followed up after administration of the therapy, to ensure timely treatment of any adverse reactions and thus to prevent the occurrence of complications and serious consequences. A wide range of incidences of systemic reactions have been reported in the literature, including <1% in patients receiving conventional immunotherapy and 0% to 79% in patients on the accelerated cluster schedule. 28 In our study of 149 patients, a total of 27 patients showed local or systemic adverse reactions. All 6 patients with local reactions were conventionally treated adults. Most cases involved grade 1 systemic adverse reactions in all groups. Two of the 33 children treated with the cluster schedule suffered grade 2 systemic adverse reactions, and only one case, a child treated conventionally, suffered a grade 3 systemic adverse reaction (Table 3). There were no grade 4 systemic adverse reactions. In an earlier study including 200 children, 66% of the systemic reactions were grade I and no grade III or IV ones occurred, 29 which is consistent with our study. An interesting result in our study show that the incidence of adverse reactions with the cluster schedule was lower than with the conventional schedule, but no significant differences were found between them. Some studies had found higher incidences of systemic reactions with a cluster schedule. 30 However, other studies did not show any significant differences in the frequency of systemic reactions when comparing conventional schedules with cluster schedules. 15,17 Though our study was not completely consistent with other studies, we indeed observed this result. Besides, our result was in accordance with Quiralte et al’ study, 16 which also found that the incidence of adverse reactions with cluster schedules is lower than with conventional schedules. On the one hand, we thought it was related to the sample size. The sample size was relatively small in our study. On the other hand, we inferred that the cluster schedule was carried out under well-controlled conditions, in hospital settings where the patients were evaluated carefully by qualified medical staff and were injected by experienced nurses, which might have contributed to good control and a better safety profile. Besides, in our study, no significant differences were found in the incidence of systemic reactions between children and adults with both schedules, suggesting that both schedules were safe for children and adults. Meanwhile, all grade 2 and grade 3 systemic adverse reactions occurred in children, suggesting that extra attention should be paid to the evaluation of children for side effects during immunotherapy. At the same time, there is one possible limitation in our research. This study mainly focused on a comparison of the efficacy between 2 immunotherapies. We didn’t compare the efficacy of immunotherapy according to the classification of the patients’ level of sensitivity. Therefore, in a future study, we will stratify them based on their level of reactivity, and conduct a more detailed analysis.
In conclusion, our results show that conventional immunotherapy and cluster immunotherapy are both effective and safe for children as well as for adults. Cluster immunotherapy, being on an accelerated schedule, reduces the visit times of the patients relative to those in conventional immunotherapy, in particular reducing the duration of the buildup stage of treatment, thereby improving the willingness of patients to be treated in this manner. In addition, while both schedules are safe, the incidence of adverse reactions was observed to be particularly low for patients following the cluster schedule. We also found that skin prick tests can provide significant guidance for assessing clinical outcomes.
Footnotes
Authors’ Note
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was supported by national natural science foundation of china (81860182) and Jiangxi nature science foundation(20181BAB205036, 20121BBG70028).
