Abstract
Keywords
Introduction
Chronic rhinosinusitis (CRS) is a common global disorder, which is defined as chronic inflammation of the nose and paranasal cavity lasting for at least 12 weeks. 1 Its pathogenesis is multifactorial. Obstructive factors, allergies, microorganisms, and impaired ciliary function have been suggested as etiological factors. 2 In the past, CRS was often subdivided into two distinct clinical phenotypes based on the presence or absence of nasal polyps detected using nasal endoscopy: CRS with nasal polyps (CRSwNP) and CRS without nasal polyps (CRSsNP). 3 Recently, with the advancement of molecular biology, the importance of classifying CRS based on endotypes has been emphasized. 4
Gastrointestinal (GI) diseases are a significant health problem worldwide. Owing to their high prevalence and incidence, they have a major economic impact on society, including enormous costs and healthcare utilization. 5 With the improvement in genetics and immunology, we can fractionize more types of GI diseases based on different pathogenic mechanisms or immune pathways and deploy more therapeutic strategies.
The GI tract had been considered a possible component in the pathogenesis of CRS after Holmes et al. proposed a relationship between sinonasal disease and gastric hypersecretion in the 1950s. 6 Although related studies have advanced rapidly in the last few years, there has been no breakthrough because the mechanisms remain elusive. To date, only a few GI diseases have been strongly associated with CRS. Stewart et al. 7 supported a remarkable correlation between gastroesophageal reflux disease (GERD) and CRS in meta-analysis. Besides, a population-based case–control study identified the association between inflammatory bowel disease and CRS. 8 However, the results remain divergent in different randomized controlled trials.
The correlation between CRS and GI diseases remains ambiguous. Therefore, the primary aim of this population-based study was to determine the prevalence of GI diseases in patients with CRS, utilizing the National Health Insurance Research Database (NHIRD) in Taiwan. Several studies have supported the existence of distinct immune patterns between the Asian and Western populations in CRS patients. Through the population-based case–control study, we could compare the differences between various regions and provide further treatment strategies for subsequent studies in Asian CRS patients.9,10 The secondary aim was to assess whether different types of CRS influence the correlation with specific GI diseases. Understanding how different phenotypes or endotypes of CRS may relate to distinct GI disease patterns could provide valuable insights into the underlying mechanisms and potential shared pathways between these conditions.
Patients and Methods
Data Source
The Taiwanese government launched the National Health Insurance program in 1995. The dataset used in this study was the Longitudinal Generation Tracking Database (2005), which was derived from the NHIRD in Taiwan. The NHIRD contains demographic characteristics, records of admission and discharge, prescription drugs, and surgical procedures for approximately 99% of the population, comprising 23 million people residing in Taiwan. The LGTD is a sub-database of the NHIRD, which includes all the original claims data and registration files from 2000 to 2017 for 2 million individuals randomly sampled from the population of Taiwan. Disease diagnoses were coded using the International Classification of Diseases, 9th and 10th Revision, Clinical Modification. This study was approved by the Institutional Review Board of China Medical University Hospital Research Ethics Committee (CMUH109-REC2-031[CR-2]).
Study Population
In this study, we selected patients with newly diagnosed CRS between January 1, 2001 and December 31, 2017 as the case group, and the controls were defined as those without a history of CRS. The index date of patients with CRS was defined as the first prescription date of CRS and that of the control group was set as a random date between 2001 and 2017. The two cohorts were matched by sex, age (in 5-year intervals), and year of the index date. Propensity-score matching at a ratio of 1:4 was applied. To estimate the association between GI tract diseases and CRSwNP, we divided the case group into two groups: patients diagnosed with nasal polyps and patients without nasal polyps. The exclusion criteria were as follows: patients under 20 years of age, patients diagnosed with malignant neoplasm of the nasal cavity before the index date, diagnosed encounter for follow-up examination after completing treatment for conditions other than malignant neoplasm before the index date, diagnosed encounter for antineoplastic immunotherapy before the index date, and patients with missing sex and age data.
Exposure Assessment and Comorbidities
In this study, we assessed exposure to GI tract diseases. To consider GI tract diseases, we divided exposure into four groups based on different features: hepatitis C, hepatitis A, acute hepatitis B, and chronic hepatitis B. All patients who were prone to be immunocompromised were enrolled as group 1. Patients with GERD with esophagitis, GERD without esophagitis, achalasia cardia, peptic ulcer, gastrojejunal ulcer, and Helicobacter pylori, which were defined as functional GI diseases, were enrolled in group 2. Patients with autoimmune pancreatitis, Crohn’s disease, and ulcerative colitis defined as autoimmune GI diseases or classified as type 2 inflammation, were enrolled in group 3. Patients with diverticulitis, liver abscess, cholecystitis, and cholangitis, which are described as structural GI diseases but have weak associations with CRS based on limited research discussing their relationship, were enrolled in group 4.
We also adjusted for the potential confounding effects of other comorbidities, including diabetes mellitus, hypertension, pneumonia, and chronic obstructive pulmonary disease that occurred before the index date. In addition, related drugs, such as proton pump inhibitors (PPI) and histamine type-2 receptor antagonists (H2RA), were also considered.
Statistical Analysis
To examine the differences in the distribution of age, comorbidities, and medication between the two groups, the chi-square test and student’s t-test were used for categorical and continuous variables, respectively. The baseline categorical variables of patients in different groups were compared using a chi-square test, and the difference in mean age was estimated using student’s t-test. Logistic regression was used to assess the odds ratios (ORs) and 95% confidence intervals (CIs). There were two types of adjusted models: one was further adjusted for covariates, such as age, sex, monthly income, urbanization level, comorbidities, medications, and the rest of the exposure group. The other group was adjusted for age, sex, monthly income, urbanization level, comorbidities, medications, and other exposure diseases in the group. Statistical Analysis System (SAS) software version 9.4 (Windows version 9.4, SAS Institute Inc., Cary, NC, USA) was used for all the statistical analyses. Statistical significance was set at a P value less than .05.
Results
Between 2001 and 2017, 356,245 participants were enrolled in this study: 284,996 in the control group and 71,249 in the case group. Table 1 shows the baseline characteristics and demographics of the participants, including sex, age, monthly income level, urbanization level, comorbidities, and medication. The proportion of men was lower than that of women. Most of the patients were aged 40 to 64 years, and the mean age was 46.17 ± 15.70 years in the control cohort and 46.18 ± 15.66 years in the case group. Regardless of the group, participants with monthly incomes between 20,000 and 40,000 and moderate urbanization levels accounted for the majority. Furthermore, the distribution of comorbidities and medications between the two cohorts was significantly different. There were more comorbidities in patients with CRS than in patients without CRS, and the most common comorbidities in this study population were pneumonia (control: 22.05% vs case: 31.56%) and hypertension (control: 21.83% vs case: 25.31%). Regarding medications, there were more patients with CRS taking PPI (control: 18.07% vs case: 25.91%) and H2RA (control: 23.27% vs case: 29.47%) than controls.
Demographic Characteristics of Individuals With and Without Chronic Rhinosinusitis.
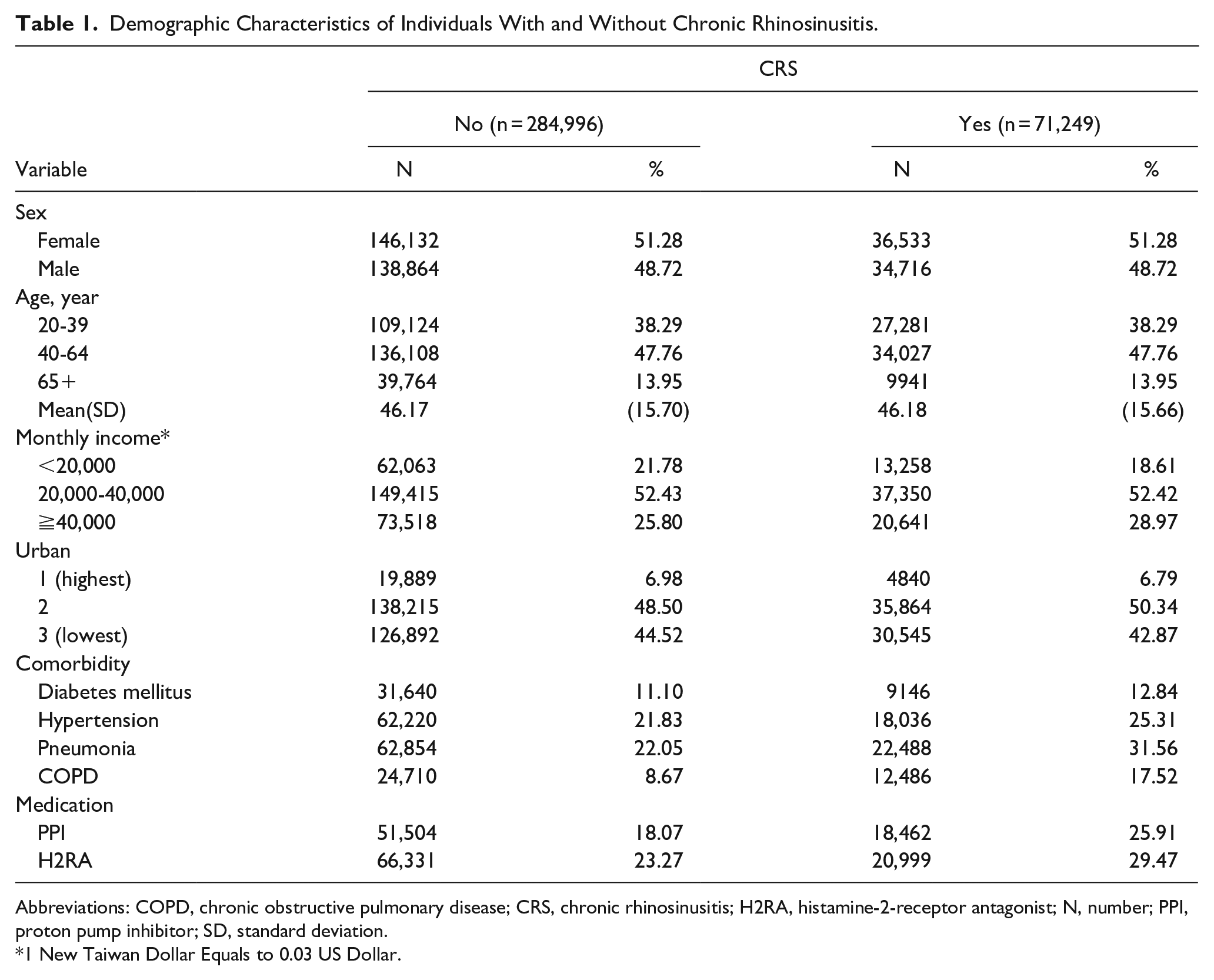
Abbreviations: COPD, chronic obstructive pulmonary disease; CRS, chronic rhinosinusitis; H2RA, histamine-2-receptor antagonist; N, number; PPI, proton pump inhibitor; SD, standard deviation.
1 New Taiwan Dollar Equals to 0.03 US Dollar.
Table 2 shows the association between GI tract diseases and CRS. After adjustment for age, sex, monthly income, urbanization level, comorbidities, medications, and the rest of the exposure group, those with exposure disease in group 1 had higher odds of developing CRS than those without exposure disease in group 1 (adjusted OR = 1.20; 95% CI = 1.15, 1.25). The association was also significant in groups 2 (adjusted OR = 1.55; 95% CI = 1.51, 1.58) and 3 (adjusted OR = 1.12; 95% CI = 1.07, 1.17). Conversely, compared to participants without a diagnosed disease in group 4, those diagnosed with diseases had a lower OR of CRS (adjusted OR = 0.87; 95% CI = 0.81, 0.94).
Univariate and Multivariate Logistic Regression for Four Groups of GI Tract Diseases Between the CRS Group and the Non-CRS Group.
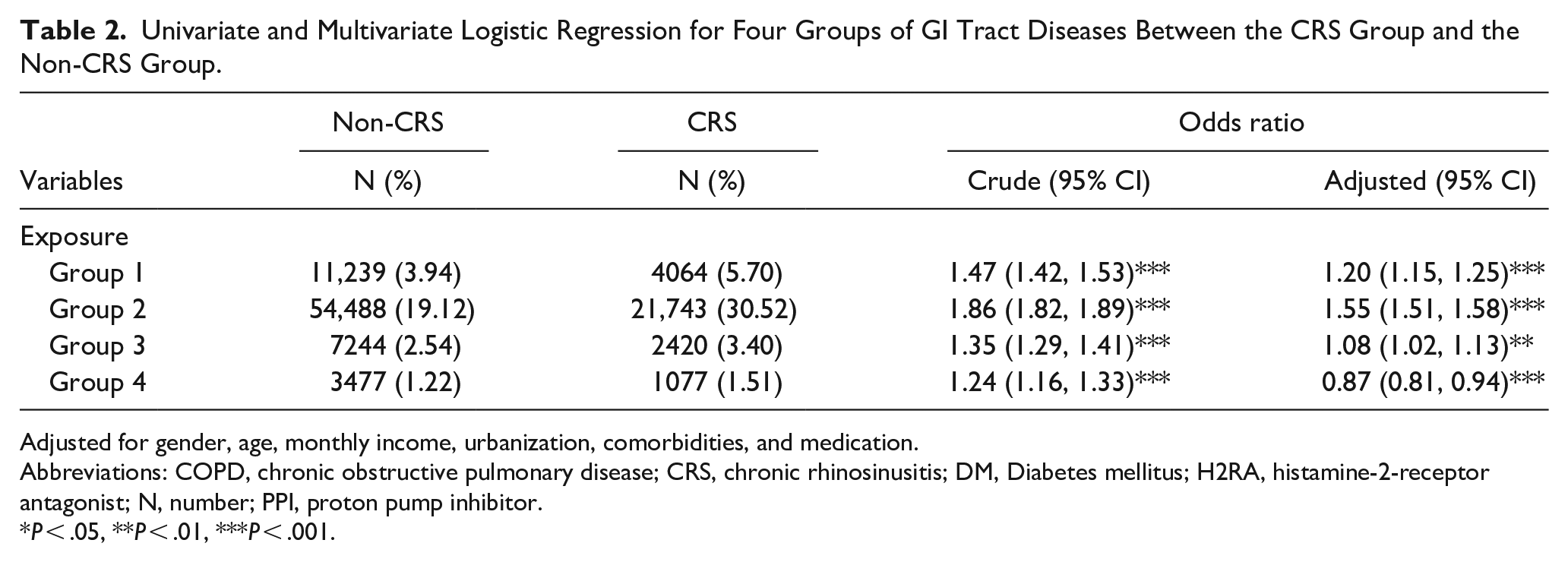
Adjusted for gender, age, monthly income, urbanization, comorbidities, and medication.
Abbreviations: COPD, chronic obstructive pulmonary disease; CRS, chronic rhinosinusitis; DM, Diabetes mellitus; H2RA, histamine-2-receptor antagonist; N, number; PPI, proton pump inhibitor.
P < .05, **P < .01, ***P < .001.
As shown in Table 3, after adjusting for covariates, comorbidities, medications, and other exposure diseases in group 1, patients with acute hepatitis B (adjusted OR = 1.30; 95% CI = 1.24, 1.37) and chronic hepatitis B (adjusted OR = 1.20; 95% CI = 1.12, 1.29) were more likely to develop CRS than those without them. In group 2, patients diagnosed with GERD with esophagitis (adjusted OR = 1.66; 95% CI = 1.60, 1.71), GERD without esophagitis (adjusted OR = 1.29; 95% CI = 1.21, 1.37), peptic ulcer (adjusted OR = 1.33; 95% CI = 1.30, 1.36), and gastrojejunal ulcer (adjusted OR = 1.25; 95% CI = 1.16, 1.36) had a higher risk of developing CRS than those without. By contrast, patients with H. pylori infection (adjusted OR = 0.82; 95% CI = 0.73, 0.91) had lower odds of developing CRS than those without it. In group 3, patients diagnosed with Crohn’s disease (adjusted OR = 1.19; 95% CI = 1.13, 1.26) and ulcerative colitis (adjusted OR = 1.31; 95% CI = 1.11, 1.55) were more likely to develop CRS than those without these diseases. By contrast, participants with autoimmune pancreatitis had significantly lower odds of developing CRS than those without autoimmune pancreatitis (adjusted OR = 0.89; 95% CI = 0.80, 1.00). There is no significant difference in odds ratio between CRS and non-CRS in group 4 GI tract diseases.
Univariate and Multivariate Logistic Regression for Premorbid GI Tract Diseases Between CRS Group and Non-CRS Group.
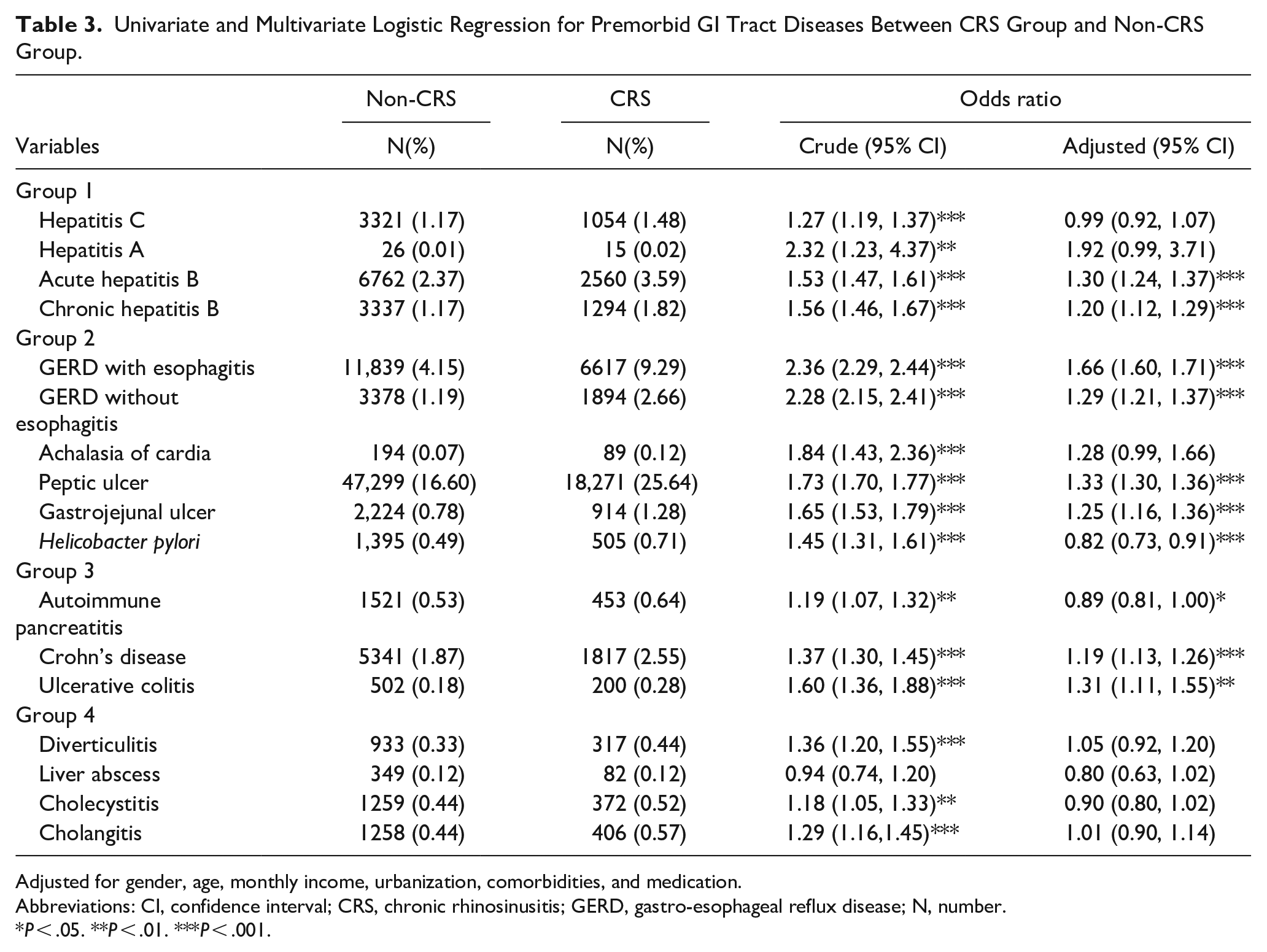
Adjusted for gender, age, monthly income, urbanization, comorbidities, and medication.
Abbreviations: CI, confidence interval; CRS, chronic rhinosinusitis; GERD, gastro-esophageal reflux disease; N, number.
P < .05. **P < .01. ***P < .001.
Table 4 shows the association between GI tract diseases and CRS with or without nasal polyps. Patients with GERD with esophagitis (adjusted OR = 0.80; 95% CI = 0.73, 0.87) and peptic ulcer (adjusted OR = 0.86; 95% CI = 0.82, 0.92) had significantly lower odds of CRSwNP than those without these diseases after adjusting for covariates, comorbidities, medications, and the rest of the diseases in the group. There is no significant difference in odds ratio between CRSwNP and CRSsNP in participants with other GI tract diseases.
Univariate and Multivariate Logistic Regression for Gastrointestinal Tract Diseases Between CRSwNP and CRSsNP.
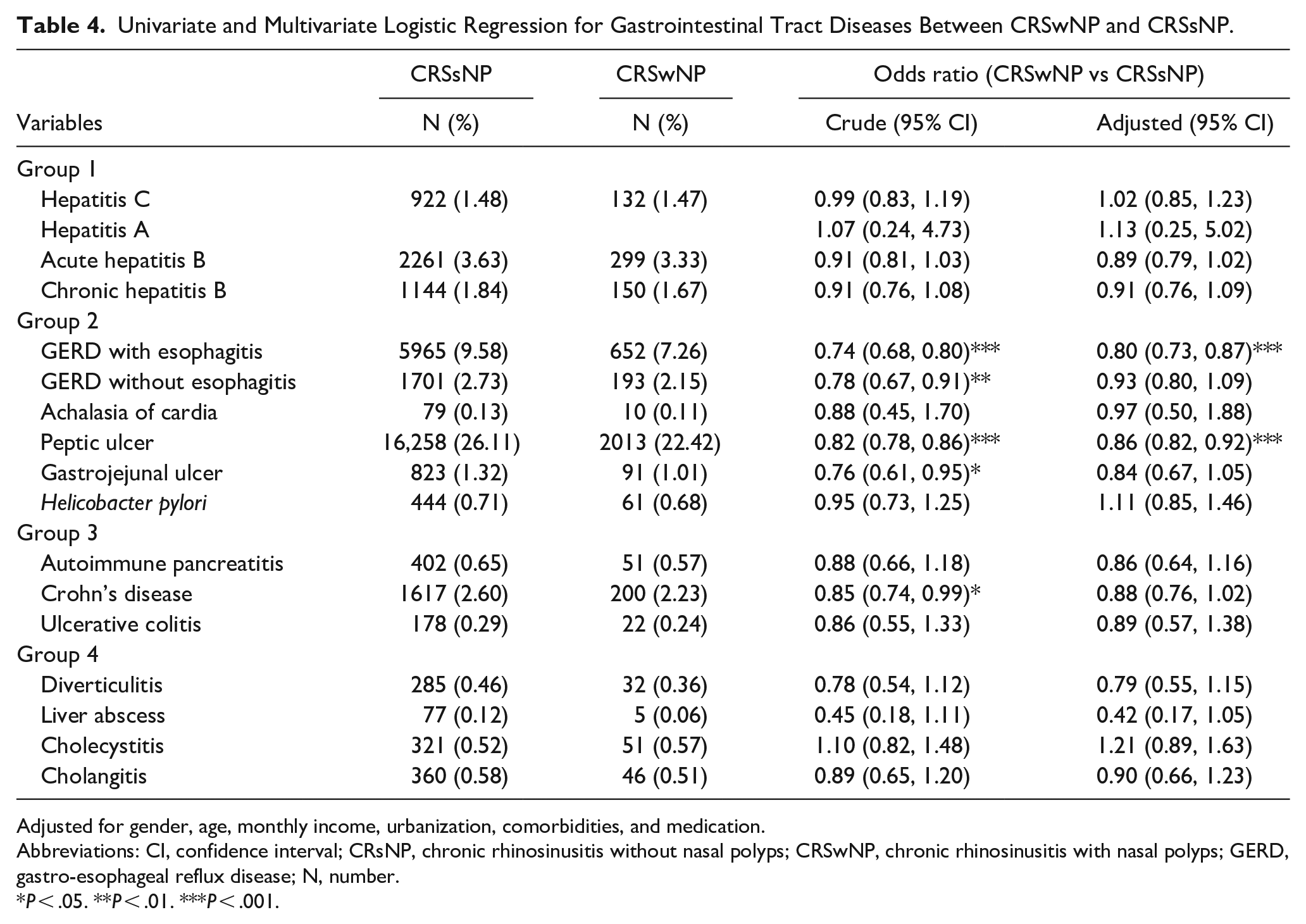
Adjusted for gender, age, monthly income, urbanization, comorbidities, and medication.
Abbreviations: CI, confidence interval; CRsNP, chronic rhinosinusitis without nasal polyps; CRSwNP, chronic rhinosinusitis with nasal polyps; GERD, gastro-esophageal reflux disease; N, number.
P < .05. **P < .01. ***P < .001.
Discussion
The strength of this study was that we used population-based data to select sufficient CRS cases (n = 71,249) and controls (n = 284,996) to investigate the occurrence of GI tract diseases. The LGTD was derived from the NHIRD, which is a large nationwide population database that covers more than 99% of the 23 million Taiwanese population. By utilizing the NHIRD, it allows researchers to study the actual clinical practices and outcomes. As a result, this study could serve as a guide for real-world evidence-based medicine for understanding of the prevalence, patterns, and correlation between CRS and GI tract diseases as diagnosed and treated by healthcare professionals.
In the present study, GI tract diseases were much more prevalent in patients with CRS than in those without CRS (group 1: 5.70% vs 3.94%; group 2: 30.52% vs 19.12%; group 3: 4.24% vs 2.94%; and group 4: 1.51% vs 1.22%). Compared with those without CRS, patients with CRS had a crude OR of 1.47, 1.86, 1.46, and 1.24, corresponding to groups 1, 2, 3, and 4, respectively. Except for group 4, the strength of the association between CRS and premorbid GI tract diseases remained after controlling for other confounding effects of comorbidities (adjusted OR: group 1: 1.20, 95% CI = 1.15, 1.25; group 2: 1.55, 95% CI = 1.51, 1.58; and group 3: 1.12, 95% CI = 1.07,1.17). Patients with CRS had a significantly higher association with acute and chronic hepatitis B in group 1, GERD with esophagitis, GERD without esophagitis, peptic ulcer, and gastrojejunal ulcer in group 2, and Crohn’s disease, and ulcerative colitis in group 3. When CRS was categorized into two phenotypes, CRSwNP and CRSsNP, we found that GERD with esophagitis (adjusted OR: 0.80, 95% CI = 0.73, 0.87) and peptic ulcer (adjusted OR: 0.86, 95% CI = 0.82, 0.92) in group 2, were more significantly associated with CRSsNP.
There is another study concluded that chronic and episodic upper airway disorders, lower airway disorders, GERD, epithelial tissue conditions, and psychiatric disorders have been associated with an eventual diagnosis of CRS. 11 A series of CRS cases and controls concluded that individuals who have both CRS and GERD are at a higher risk of having allergic conditions and asthma when compared to those with CRS alone and without GERD. One possible explanation for this connection is that having both GERD and atopic diseases may increase the risk of developing CRS. 12 A nationwide population-based study revealed that a personal history of inflammatory bowel disease (IBD) is associated with an increased risk of CRS. 13 In the present study, we observed that CRS had a more significant association with acute hepatitis B, chronic hepatitis B, GERD with esophagitis, GERD without esophagitis, peptic ulcer, gastrojejunal ulcer, Crohn’s disease, and ulcerative colitis. These findings are consistent with those of the aforementioned studies.
Paulo Borges Dinis et al. 14 carried out a controlled-cohort analysis and revealed that H. pylori colonization was not more common in patients with sinusitis than in controls, and it failed to support a pathogenic role for this organism in the sinuses. In the present study, we similarly found that patients with H. pylori infection had lower odds of CRS than those without it. However, a case–control analytical epidemiological survey showed a correlation between CRS and the presence of H. pylori in the sinonasal mucosa. 15
The mechanism by which premorbid GI tract diseases affect CRS development remains unclear. Genetic factors, environment (e.g., group 2, direct contact of refluxate), immunity, and inflammatory pathways (e.g., group 1 or 3, immunocompromised or autoimmune) may play important roles in the association between premorbid GI tract diseases and CRS. Interleukin (IL)-32, induced by IL-8, is a key modulator of the pathogenesis of various disease processes and enhances the severity of gastric inflammation and CRS. 16 Furthermore, Xu et al. 17 reported that IL-32 might be a key factor in the pathogenesis of chronic hepatitis B. Changes in the composition of innate lymphoid cells in inflamed gut tissues of patients with Crohn’s disease have been observed, suggesting a role for innate lymphoid cells in this disease. 18 A case report has suggested that there may be a higher prevalence of CRS in patients with autoimmune pancreatitis. It is not clear whether CRS developed as a nasal mucosal manifestation of systemic IgG4-related disease. Induction of remission by prednisolone for type 1 autoimmune pancreatitis led to a marked improvement in CRS. 19 However, in our present study, autoimmune pancreatitis may have a lower risk of developing CRS. Further research is needed to clarify the relationship between these two conditions. It seems that GI tract diseases in group 4, diverticulitis, liver abscess, cholecystitis, and cholangitis, have no significant association with CRS.
This study aimed to evaluate various GI tract diseases in patients with CRS and their association with different CRS phenotypes. According to The European Position Paper on Rhinosinusitis and Nasal Polyps 2020 (EPOS2020), CRS has been looked at in terms of primary and secondary CRS, and divided into localized and diffuse diseases based on anatomic distribution. In primary CRS, disease endotypes are classified as type 2 or non-type 2. For diffuse CRS, the clinical phenotypes are divided into predominantly eosinophilic chronic rhinosinusitis (eCRS) and non-eCRS based on the histologic quantification of eosinophilic numbers. Endotyping is useful for predicting the natural course of the disease and for determining pharmacotherapy and further surgical treatment. 20 Lin et al. 21 concluded that patients with GERD were at a greater risk of developing CRS, especially CRSsNP. The trefoil factor family peptides are overexpressed in CRSsNP and upregulated in peptic ulcer disease. 22 Our present findings showed that GERD with esophagitis and peptic ulcer in group 2 were more significantly associated with CRSsNP, consistent with previous studies.
This study had several limitations that should be considered. First, the study utilized data from LGTD, which included 2 million individuals from 2000 to 2017 and was derived from the NHIRD. The selected timeframe of 2000 to 2017 was chosen to maintain comprehensive and well-documented records for that specific period. We took measures to ensure that no variations in the demographic distribution were detected between LGTD and the original NHIRD. Second, the disease diagnoses were coded using the ICD format (for CRS, GI tract diseases, and comorbidities), which is strongly based on physician performance. However, periodic audits are performed to ensure that negligence and misdiagnosis are minimized. Third, we divided CRS into two subtypes, CRSwNP and CRSsNP, based on phenotypes; however, it cannot fully represent the endotypes. The relationship between phenotypes and endotypes in Asian patients is different from that in Western patients, and it still needs further investigation. Last, there is a variant of CRSwNP, central compartment atopic disease (CCAD), which represents a clinically distinct phenotype of CRSwNP associated with inhalant allergy and low prevalence of asthma. 23 However, it has not been coded in the ICD format; therefore, the association of CCAD with GI tract diseases still needs further investigation using different research methods.
In conclusion, a significant association between CRS and premorbid GI tract disease has been identified, particularly in acute/chronic hepatitis B, GERD with/without esophagitis, achalasia of cardia, peptic/gastrojejunal ulcer, Crohn’s disease, and ulcerative colitis. Remarkably, GERD with esophagitis and peptic ulcer were significantly associated with CRSsNP. In our study, it seems that GI tract diseases in group 4, structural GI diseases, have a weak correlation with CRS. Further studies are needed to better understand the underlying pathophysiological mechanisms and associations between certain GI tract diseases and CRS. Researchers can further investigate the mechanisms by referring to our classification method to determine the implications for diagnosis and treatment.
Supplemental Material
sj-docx-1-ear-10.1177_01455613231218143 – Supplemental material for Chronic Rhinosinusitis and Premorbid Gastrointestinal Tract Diseases: A Population-Based Case–Control Study
Supplemental material, sj-docx-1-ear-10.1177_01455613231218143 for Chronic Rhinosinusitis and Premorbid Gastrointestinal Tract Diseases: A Population-Based Case–Control Study by Sen-Sen Wu, Tzu-Hsun Hung, Pei-Shao Liao, Yung-An Tsou, Yu-Tung Hung, Chia-Der Lin, Chih-Jaan Tai, Te-Chun Shen and Liang-Chun Shih in Ear, Nose & Throat Journal
Footnotes
Data Availability Statement
Restrictions apply to the availability of these data. Data was obtained from NHIRD in Taiwan and are available from the authors with the permission of NHIRD in Taiwan.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported in part by the Taiwan Ministry of Health and Welfare Clinical Trial Center (MOHW111-TDU-B-212-134004), China Medical University Hospital (DMR-111-242). We are grateful to the Health Data Science Center, China Medical University Hospital for providing administrative, technical, and funding support. The funders had no role in the study design, data collection and analysis, the decision to publish, or the preparation of the manuscript. No additional external funding was received for this study.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of China Medical University & Hospital Research Ethics Center (protocol code CMUH111-REC3-139).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
