Abstract
Keywords
Introduction
Phenol was described as a topical anesthetic for the tympanic membrane (TM) by Bonain in 1907. Concentrated phenol for myringotomy and tympanostomy tube (TT) placement in young children and adults was introduced by Storrs in 1956. 1 It is an effective analgesic agent which works by paralyzing nerve fibers in the TM, causing full-thickness analgesia. 2 Phenol is typically used as a topical anesthetic for office TT placement in adults, with pediatric TT placement traditionally being performed in the operating room under general anesthesia (GA).
The FDA has cleared single-use phenol kits with 89% phenol and 11% water with indications for application to the TM. 3 Many otolaryngologists also report using stock phenol preparation for TT placement in adults with a reusable and sterilized applicator, and short articles have been published on the best approach to the application of phenol for this use. Preparations of stock phenol used for this purpose range from 25% to 89%.4,5
However, there is concern among some practicing otolaryngologists that the use of phenol on the TM can lead to an increase of perforations or other complications, even with controlled use in cooperative individuals. As such, there remains concern that the use of phenol may not be suitable for awake and moving children, as movement could potentially result in an over application of phenol and cause potential damage to the TM. Additional concerns include discomfort and an adequately wide ear canal for application.6 -10
Further, there is a paucity of data on the use and safety of phenol on the TMs of awake children. The largest published article on the use of phenol 11 on pediatric TMs evaluated 46 children to determine whether phenol would reduce postoperative pain following TT placement under GA.
Recently, there have been publications demonstrating the transition of pediatric TT placement from the operating room under GA into the office to reduce costs, reduce risks, and improve convenience.12,13 The move to office placement of TT in children has been endorsed in a position statement by the American Academy of Otolaryngology—Head and Neck Surgery in 2019. 14 This shift in point of service of TT procedures for children has required alternative measures for local anesthetic of the TM. Previously reported agents to provide local anesthesia include EMLA cream and tetracaine. These agents can be difficult to work with, time-consuming to take effect, or give unreliable results.1,2,6,9,10
Lustig et al. 12 reported on an iontophoresis system for TM anesthetic in children using 2% lidocaine and 1:100,000 epinephrine. These results showed a 4.5% adverse event rate due to inadequate anesthesia. Further, iontophoresis requires additional time to anesthetize the TM and requires the surgeon to touch the TM with a dull instrument to determine if the anesthetic process was effective.
Given the recent shift of TT placement to the office and the concerns regarding use of phenol, we investigated and report on the use of phenol as a topical anesthetic on the TM for in-office TT placement in awake children.
Methods
Awake children were enrolled in a multisite, prospective, treatment-only study using phenol as a topical anesthetic and TT placement with the Hummingbird one-pass delivery system (manufactured by Preceptis Medical, Inc, Minneapolis, MN, USA). The study included 2 age groups of patients: 6 to 24 months old and 2 to 21 years old. The study took place in an office setting at 5 clinical sites: 2 academic institutions (Mayo Clinic—Rochester, MN, USA; Cedars-Sinai Medical Center, Los Angeles, CA, USA); 1 large multispecialty clinic (Park Nicollet Clinic, St. Louis Park, MN, USA); and 2 general otolaryngology private practice clinics (St. Cloud Ear Nose and Throat, St. Cloud, MN, USA; Prairie Sinus Ear Allergy, Bismarck, ND, USA). The study was approved by the Institutional Review Board (IRB) at the individual institution, except for St. Cloud Ear, Nose and Throat and Prairie Sinus Ear Allergy which used the Western Copernicus Group IRB.
Inclusion criteria were all pediatric patients greater than 6 months and under 21 years of age who were indicated to undergo TT insertion in the clinic. Investigators were guided by the criteria for TT placement defined by Rosenfeld et al., 15 including recurrent acute otitis media with middle ear effusion, chronic bilateral otitis media with effusion with hearing difficulty, and chronic otitis media with effusion with symptoms. Exclusion criteria included anatomy that precluded visualization and access to the TM, any condition that in the opinion of the Principal Investigator would place the patient at greater risk (eg, development delay), and patient had previously received tubes (patients 6-24 months only). A parent of each subject gave their informed consent before participation in the study. The study was conducted in accordance with the IRBs, and records of the informed consent for each subject is held by the corresponding author.
Children approximately 3 years of age and younger were swaddled and/or had a procedure immobilization board for body restraint, while also having their heads stabilized by an assistant in the office. The use of a procedure immobilization board and head restraint on children older than 3 years was optional, with the decision being made between the child, parent, and surgeon. At least 1 parent chose to be present during the majority of the procedures (68.0% documented). Parents were informed that they could stop the procedure at any time if they felt their child was undergoing undue pain or stress and that the procedure would be moved to the operating room under GA at a later date.
Phenol was either in kit form or using a stock bottle of 89% phenol applied with a metal phenol applicator with a forked end. Phenol was placed by the surgeon on the desired TM quadrant (typically in the anterior/inferior quadrant or posterior/inferior quadrant). The Hummingbird device was then used to deliver the TT in the location of the TM that underwent phenol application. An assessment of child recovery (defined as the child being calm and/or no inappropriate crying) was performed by the surgeon, and parents completed a procedure survey. Children were evaluated at the first post-procedure follow-up, typically between 3 and 10 weeks after TT placement, with an otologic exam and audiogram to evaluate for outcomes. Investigators also documented whether there was any evidence that the child was inappropriately reticent to have their ears examined upon follow-up compared to a standard of care ear tube procedure.
Additional follow-up visits beyond the first follow-up were obtained via a retrospective chart review that was also approved by the participating IRBs. Health records of study participants with at least 6 months follow-up were reviewed. The review focused on any reported complications, such as TM perforations, that did not self-resolve or otologic procedures such as tympanoplasty or myringoplasty.
Results
A cohort of 232 children aged 6 months to 15.9 years had in-office Hummingbird TT placement using phenol as a topical anesthetic. Four children were unable to complete an office insertion of TT placement due to excessive movement (n = 2), anatomical reasons (severe retraction n = 1), or severely scarred and thickened TM preventing adequate phenol application (n = 1). These 4 children were converted to TT placement in the operating room, and their data were excluded from this analysis.
Overall, successful in-office placement in patients using phenol occurred in 228 out of 232 patients (98%). Successful unilateral TT placement occurred in 21 children, for a total of 435 TMs successfully treated in the office with phenol. No procedures were halted or paused by the attending parent. Median bilateral procedure time was 4 minutes 43 seconds (4:43), with a range of 1:54 to 18:06. Demographics are noted in Table 1, with 79% of the patients in the 6 to 24 months age group.
Data Summary.
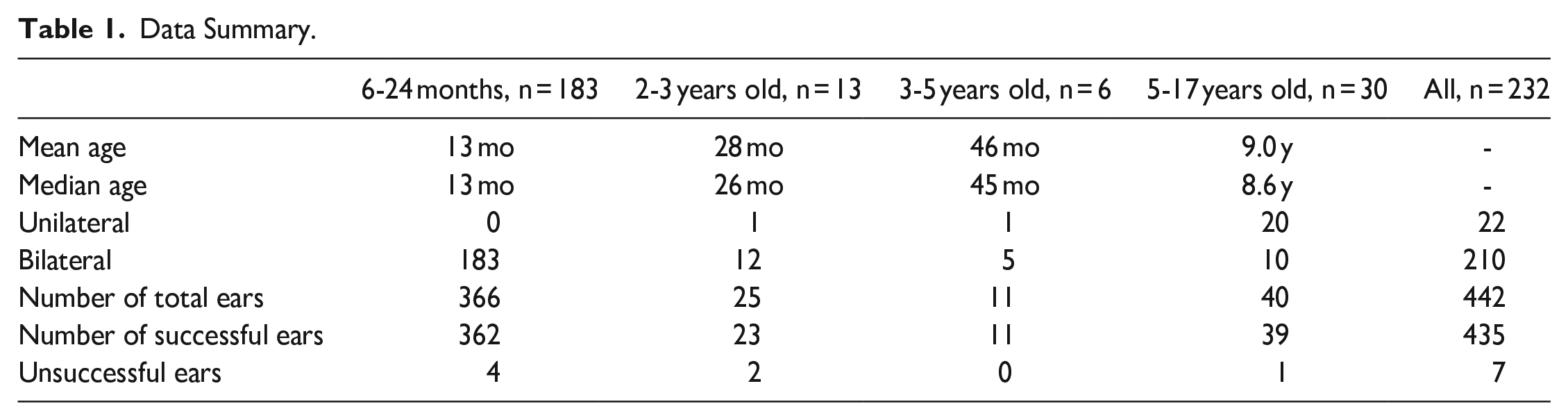
Per assessment of patient recovery by the surgeon and staff, 217 of the 228 children (95%) were judged to be recovered as soon as they were done with the procedure and back with their parents. The parents also completed a survey following the procedure (95.6% completed). Of this group, 96.8% strongly agreed or agree that they would recommend this in-office TT procedure to other parents, while the other 3.2% were neutral. The data that support the findings of this study are held by the corresponding author and available upon request.
For follow-up, 204 out of 228 children returned for the scheduled post-procedure visit defined by the protocol. Of the 204 children with a post-procedure visit, the investigators indicated reticence to having ears examined in 24 of the children (12%). There were no reports of TM complications in these 204 patients.
Additional follow-up information beyond the initial post-procedure visit was procured using a retrospective chart review. This review yielded 93 patients and 185 TMs with at least a 6-month or longer follow-up. The average follow-up in this group was 13.1 months (range 6.07-25.3 months). There were no reports of any TM complications, including perforations requiring tympanoplasty or myringoplasty, in these 93 patients.
Discussion
Phenol (Benzenol, also called carbolic acid or phenolic acid) is both an antiseptic and disinfectant, 16 as well as a mild acid with a pH between 5 and 6. Phenol is highly toxic on ingestion and causes a chemical burn on contact with the dermis. 1
Per its neurolysis properties, concentrated liquid phenol can be used topically as a local anesthetic for otology procedures, such as myringotomy and TT placement, as an alternative to GA or other local anesthetics. It has hemostatic and antiseptic properties that make it ideal for this use. 17 Use of concentrated phenol on the TM for grommet insertion was first described in 1956 by Storrs. He later claimed to have had no complications in 5000 cases.1,18
Following Storrs, there have been multiple publications on the use of phenol on the TM of adults (some with sample sizes as large as 5000) that concluded that the use of phenol as a local anesthetic is safe, efficacious, acceptable to patients, and has an advantage over other local anesthetic agents due to its immediate analgesic effect.1,2,4,5 In summary, phenol is inexpensive, easy to use, does not obscure visualization of the TM, and has rapid onset. The major disadvantages of phenol include brief discomfort on application and the possibility of significant permanent damage to tissue if misapplied.
Despite the above information detailing the safety and efficacy of phenol on the TM and the FDA clearing phenol kits for use on the TM, there are literature sources that have raised questions regarding the use of phenol on the adult TM. Hoffman et al. 9 in 2001 postulated that phenol was destructive and could result in a chemical otitis externa. Ng et al. 19 reported a single chronic perforation (successfully repaired at 2 months with myringoplasty) in a chart review of 205 patients (assumed adults) treated with electrocochleography and phenol on the TM. Santa Maria et al. 7 reported a case study in 2016 on a 21-year-old male who suffered serious iatrogenic injury from accidental application of copious amounts of phenol into the ear. Their recommendation was to use prepackaged single application kits.
There have been publications on the use of phenol on the TM of awake children, but we are not aware of any published studies on the complication rate associated with the use of phenol on the TM in awake children. Orobello et al. 11 reported on the use of phenol as an adjuvant anesthetic for TT insertion in children. However, this study was performed using GA where placement of phenol could be controlled on the non-moving child. This study did not discuss TM complications, and there was no follow-up evaluation.
Rosenfeld 20 reported on the use of phenol as a local anesthetic on the TM of children aged 2 years and older during TT insertion in the office using standard surgical instruments. There was no discussion of the tolerability or long-term effects of phenol on the child’s TM. Tunkel 21 reported that he has performed TT placement in the office for more than 30 years in carefully selected young children and teenagers using iontophoresis or phenol application but did not report on TM complications or long-term evaluation.
This analysis was designed to evaluate the success, safety, and efficacy of performing Hummingbird TT placement on awake pediatric patients in the office using phenol as a local anesthetic for the TM. The idea for performing TT placement in the office on children originated from the 2016 FDA Safety Alert on the potential long-term risk associated with the use of GA in children under 3 years of age 22 and the pioneering in-office tube pediatric insertion work of Rosenfeld et al. 23 Prior, in 2002 Hoffman 24 conducted a prospective review of 1000 children to determine the incidence of perioperative GA complications during bilateral TT placement in children and reported a major event rate of 1.9% and a minor adverse event rate of 8.8%.
The results showed that phenol was efficient, safe, and tolerable when placed on the TM of 228 children. In long-term retrospective follow-up of a subset of this group, there were no reports of TM injury, which compares favorably to the 3.0% perforation reported in UpToDate (a meta-analysis without age delineation) for TT placement under GA. 25 No other adverse effects related to the use of phenol (eg, acute otitis externa) were noted.
Additional benefits of phenol in the pediatric population are that it allowed for a short median total bilateral procedure time of 4 minutes 43 seconds for office placement of bilateral TT. A recent follow-up publication by Lustig, employing iontophoresis for in-office TT placement in children, stated that the time for iontophoresis anesthesia is 10 to 15 minutes, with a 33-minute average time for the total procedure. 6
Though Orobello et al.’s 11 study on phenol use in children did not find any effect on reducing postoperative discomfort in children receiving tubes under GA, the investigators of this study felt that phenol was clearly beneficial as noted by the 95% of children in this study who were deemed to be recovered as soon as the procedure was completed and in the presence of their parents. 13 The reason phenol was deemed effective in this study and not the study by Orobello et al. 11 may have to do with the complexity of causes that lead to a large percentage (57%) of postoperative emergence delirium in TT placement in children using GA. 26 To our knowledge, there have been no published reports of emergence delirium following in-office TT placement.
The primary limitation of this study is the lack of a direct comparison of phenol to other methods used for TM anesthesia. Additionally, per the potential for bias by the healthcare providers and caregivers, further research on this topic is recommended.
Conclusions
This is the first analysis to date on the long-term follow-up and complication rates of phenol on the TM of awake children for in-office TT placement. Phenol provides fast and efficient local anesthesia at a low cost but has been limited to adults per concerns that an uncooperative child could result in overapplication of phenol and/or cause long-term TM damage. This study provides data that demonstrate the safety, effectiveness, efficiency, and tolerability of phenol use on the TM of young children for in-office TT placement.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Franklin L. Rimell, MD is a part-time consultant for Preceptis Medical. Theodore Truitt, MD, has performed limited consulting work for Preceptis Medical. The remaining authors of this study (S.C., G.N., J.R.) have no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Preceptis Medical, Inc. was the sponsor and provided the funding for the prospective portion of the study. There was no funding provided for the retrospective portion of the study.
