Abstract
Introduction
The incidence of salivary gland carcinoma (SGC) accounts for approximately 7% of all epithelial malignancies and approximately 2% of head and neck cancers.1,2 An epidemiological study involving approximately 1 million people found a significant 54% increase in the incidence of SGC over 30 years. 3 Patients with SGC often have a limited survival time after diagnosis, even in well-differentiated types, within 3 years. 4 The main treatment methods for SGC include surgery, radiotherapy, and chemotherapy. Due to the highly heterogeneous nature of SGC, treatment combinations are numerous and challenging, and the use of individualized approaches, while effective, is often costly. 5
Distant metastasis (DM) is one of the main reasons for the treatment failure of SGC. The locations of distant metastases include lung, bone, and liver. Among them, some studies reported that the lungs were the most common site of metastasis, with an incidence of over 50%.6,7 The rate of DM for different types of SGC is approximately 20% to 50%. 7 The higher the malignancy of the tumor, the higher the metastasis rate will be. According to the National Comprehensive Cancer Network guidelines, there is a lack of consensus on the treatment of DM in SGC. In addition, studies have indicated that distant metastases may occur early in SGC even after treatment.8,9 Currently, risk factors and survival rates associated with DM are rarely studied, and DM is relatively common in SGC. Among the histologic subtypes of SGC, DM in some subtypes is aggressive, progresses rapidly, and occurs frequently. Therefore, it is particularly important to study the risk factors of patients with distant metastases, which can help us to better manage salivary gland tumors and reduce the incidence of salivary gland tumors.
It is currently different to predict the occurrence and prognosis of distant metastases in salivary gland cancer patients, and clinicians are mostly using the e American Joint Committee on Cancer (AJCC) staging system. However, the AJCC staging system does not satisfy the growing demand for personalized medical care. The nomogram can be tailored to individual patient profiles, calculate the conversion between the probability of occurrence and prognosis through a function, and obtain a prognostic probability for each patient.10,11 The nomograms are currently widely utilized in the clinical prediction of various types of cancer, such as liver cancer, lung cancer, and breast cancer.12,13 Nomograms have found extensive application in cancer prognosis and recurrence prediction, mainly due to their ability to simplify intricate statistical prediction models into a single numerical estimate. This estimate corresponds to the probability of specific events, such as mortality or disease recurrence, based on individual patient profiles. 14 Equipped with a user-friendly graphical interface, they contribute to insights during clinical decision-making. 10 For numerous cancer types, nomograms surpass traditional Tumor-Node-Metastasis (TNM) staging systems, solidifying their status as a new standard.11,15 Currently, there is a scarcity of research in clinical studies on the use of nomograms to predict DM in salivary gland cancer. Therefore, this study analyzed the risk factors for distant metastases in patients with existing malignant salivary gland tumors and the prognosis of patients with salivary gland tumors with distant metastases based on the SEER database using the nomogram.
Methods
Data Collection
The data of patients in this study were downloaded from the SEER * Stat software (version 8.3.9.2, user name:21257-Nov2020). Before this study, we presented data usage consent to the SEER project and acquired admittance to the database. The data from the SEER database is open and anonymous, so this study did not need the requirement of the institutional research ethics committee approval, nor did it require additional informed consent. We included patients who were recently diagnosed in the SEER database from 2004 to 2015 in our study.
The inclusion criteria were as follows:
(1) Patients were diagnosed with malignant salivary tumors;
(2) Patients with complete demographic information, including age, sex, and race;
(3) Clinical pathological information was available, including primary tumor site, grade, histological type, TNM, and tumor size.
The exclusion criteria were:
(1) Patients with T0 or unknown T stage, N stage, and M stage status;
(2) Patients with autopsy or death certificates;
(3) Patients with follow-up time <6 months;
(4) Patients’ therapy type unknown.
A total of 7418 patients were included in our study, 307 of whom had DM.
Variable Selection
Age, sex, race, primary site, grade, histology type, T stage, N stage, tumor size, and treatments (including surgery, radiotherapy, and chemotherapy) were selected to identify the risk factors of DM in patients with SGC. Age was classified into 3 groups: ≤64, 65 to 80, and ≥80 years by using x-tile software. Race data were recorded as White, Black, or Other. Primary sites included parotid gland (PG), sublingual gland (SLG), submandibular gland (SMG), and other (minor salivary glands). The histologic grade was classified as grades I (Well-differentiated), II (Moderately differentiated), III (Poorly differentiated), and IV (Undifferentiated). Histological types including acinar cell carcinoma, adenocarcinoma (AC), pleomorphic adenoma, mucoepidermoid carcinoma (MEC), squamous cell carcinoma (SQC), and others (including 125 subdivisions of OCD-O-3 classification). Tumor size was categorized into 3 groups: ≤21 mm, 22~40 mm, and ≥41 mm.
Statistical Analysis
Analyzing data with R software (version 4.0.4), a P-value <0.05 (2 sides) was considered as statistical significance. All patients’ data were randomly divided into the training and validation sets on a 7:3 basis, while the chi-square test was used for categorical data. Patients in the training cohort were utilized to build the predicted nomogram, while patients in the validation cohort were utilized to approve the constructed nomogram.
To develop the diagnostic cohort, we utilized the univariate logistic analysis to identify DM-related risk factors. Then, multivariate logistic analysis was applied to determine independent risk factors of DM in SGC patients. Afterward, we used the rms package to build a nomogram and selected the best prediction model using likelihood ratio tests and Akaike information criterion (AIC) values. 16 Set in the analysis above, the ROC curve was finished, 17 and the calibration curves 18 and DCA curve 19 were used to evaluate the performance of the nomogram. Area Under Curve (AUC) value and DCA curve also exhibited that nomogram has better predictive power than TNM staging system.
As for the prognostic factors, the univariate Cox regression analysis was performed to determine overall survival (OS) rate-related factors for SGC patients with DM. Independent prognostic factors were further determined by referring to the significant variables at P < 0.05 in the previous step of the analysis, plus clinical experience to select factors into multivariate Cox analysis. Based on likelihood ratio tests and AIC value, the prognostic nomogram was constructed based on the best prediction model. In addition, we generated time-dependent ROC curves at 12, 24, and 36 months to compare the accuracy of the nomogram with the AJCC8 staging. Calibration curves and DCA curves of 12, 24, and 36 months were plotted to evaluate the nomogram. The Kaplan–Meier (K–M) survival curves with the log-rank test were used to compare the OS status between high-risk and low-risk SGC patients with DM.
Results
Baseline Characteristics of SGC Patients with DM
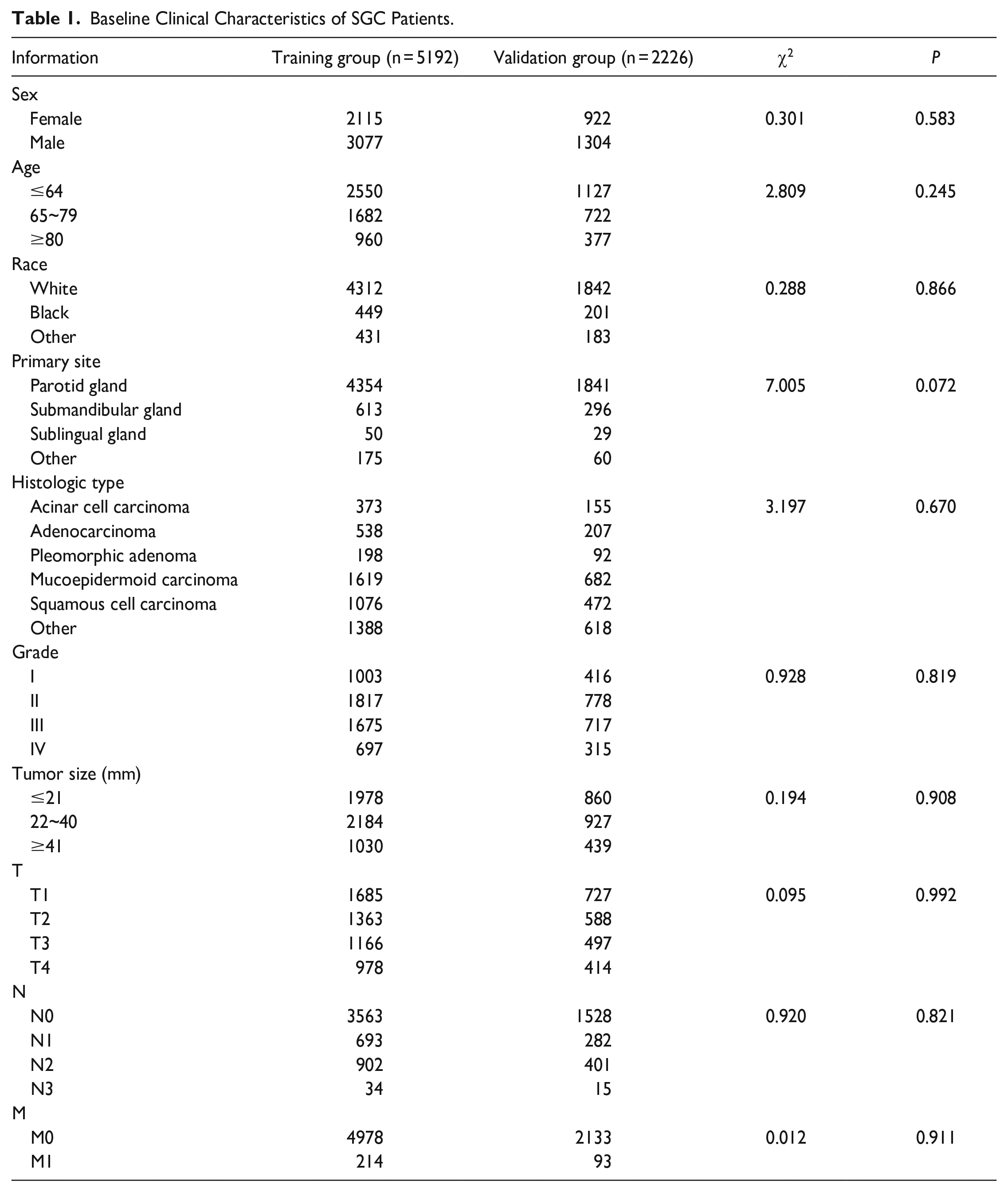
As shown in Table 1, a total of 7418 SGC patients with DM were enrolled in the study, including 5196 in the training group and 2226 in the validation group. The age of the enrolled groups ranged from a minimum of 0 years to a maximum of 85+ years, with approximately half of the patients being under 64 years old. The most common site was PG (83.6% in the training group and 82.7% in the validation group). Among the histologic types, MEC was the most common (31.2% in the training group and 20.7% in the validation group). The lowest number of T and N stages were T4 (18.8% in the training group and 18.6% in the validation group) and N3 (0.65% in the training group and 0.67% in the validation group). The majority of patients’ tumor sizes were smaller than 40 mm in size (80.2% in the training group and 80.3% in the validation group). The number of patients with histologic grade IV was the lowest in the grades stage (13.4% in the training group and 14.2% in the validation group).
Baseline Clinical Characteristics of SGC Patients.
Incidence and Risk Factors of DM in SGC Patients
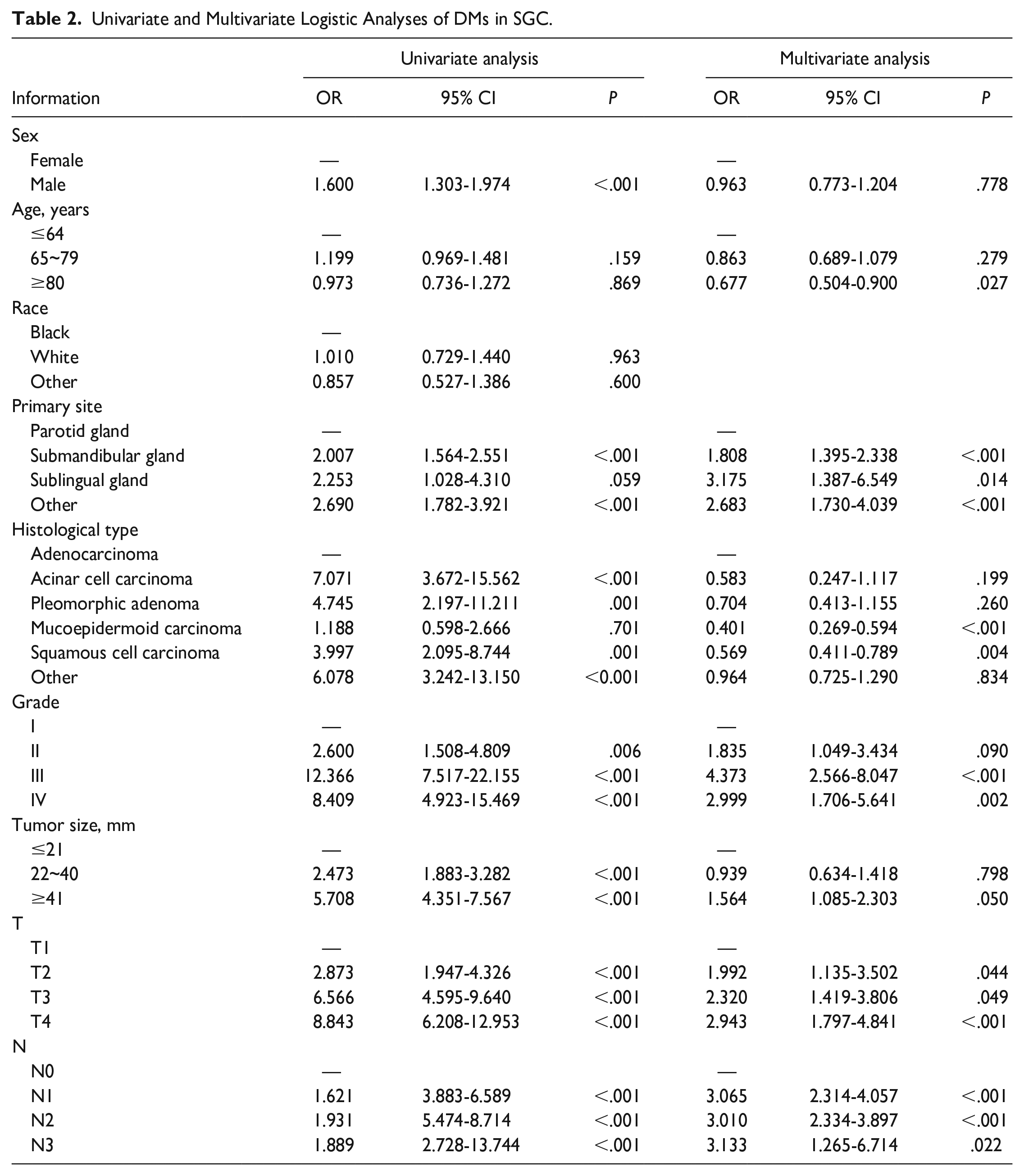
As seen in Table 2, 9 factors were analyzed by univariate analysis. Seven of these factors were prognostic factors related to DM, including sex, primary site, histologic type, T, N stage, grade, and tumor size. We included age in the multiple logistic regression analysis based on clinical experience. In addition, the result identified that non-parotid primary site, high histologic grade, T, and N stage are independent prognostic factors for DM in SGC patients.
Univariate and Multivariate Logistic Analyses of DMs in SGC.
Diagnostic Nomogram Development and Validation
A nomogram was established to predict the incidence of DM (Figure 1). AIC value was used to select the best-fitting model, we found that when including age, primary site, histologic type, T, N stage, histologic grade, and tumor size the model has the smallest AIC value (AIC = 2180). The model incorporating those 7 factors was significantly different from the model incorporating independent factors by likelihood ratio test, so the model incorporating those 7 factors was selected (χ2 = 59.681, P < .01).
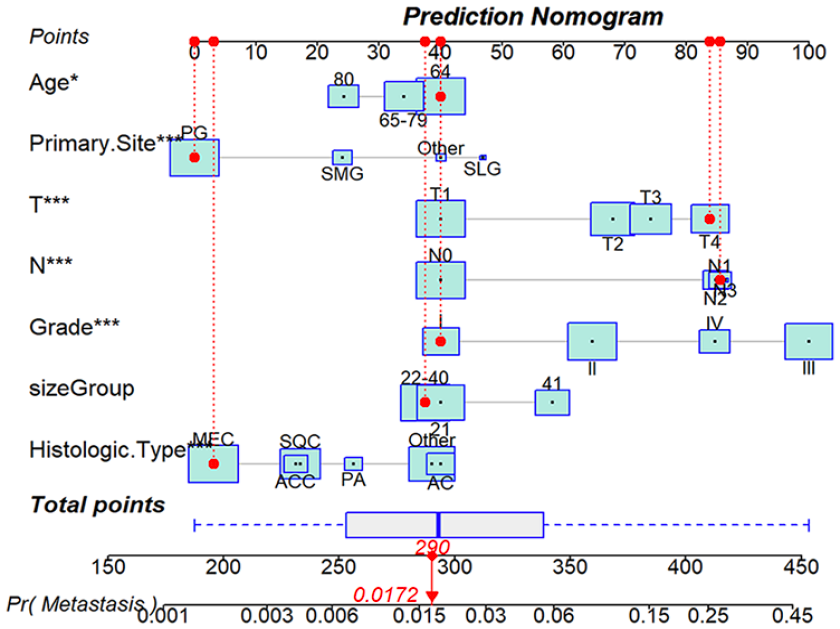
The diagnostic nomogram to estimate the risk of DM in SGC patients.
Then, ROC curves and AUC values were observed for the nomogram compared with those of AJCC staging in the training group (nomogram AUC = 0.821, AJCC staging AUC = 0.771) (Figure 2A) and validation group (nomogram AUC = 0.785, AJCC staging AUC = 0.748) (Figure 2B). In addition, the DCA curves (Figure 2C and D) further demonstrated that the diagnostic cohort nomogram of this study had better discrimination than the AJCC staging. The nomogram calibration curves were plotted to verify that there was good agreement between the predicted results and the actual observed results (Figure 3A and B). We also produced ROC curves for all independent predictors (Figure 3C and D), which showed that the nomogram has better discrimination than other factors in both the training and validation groups.
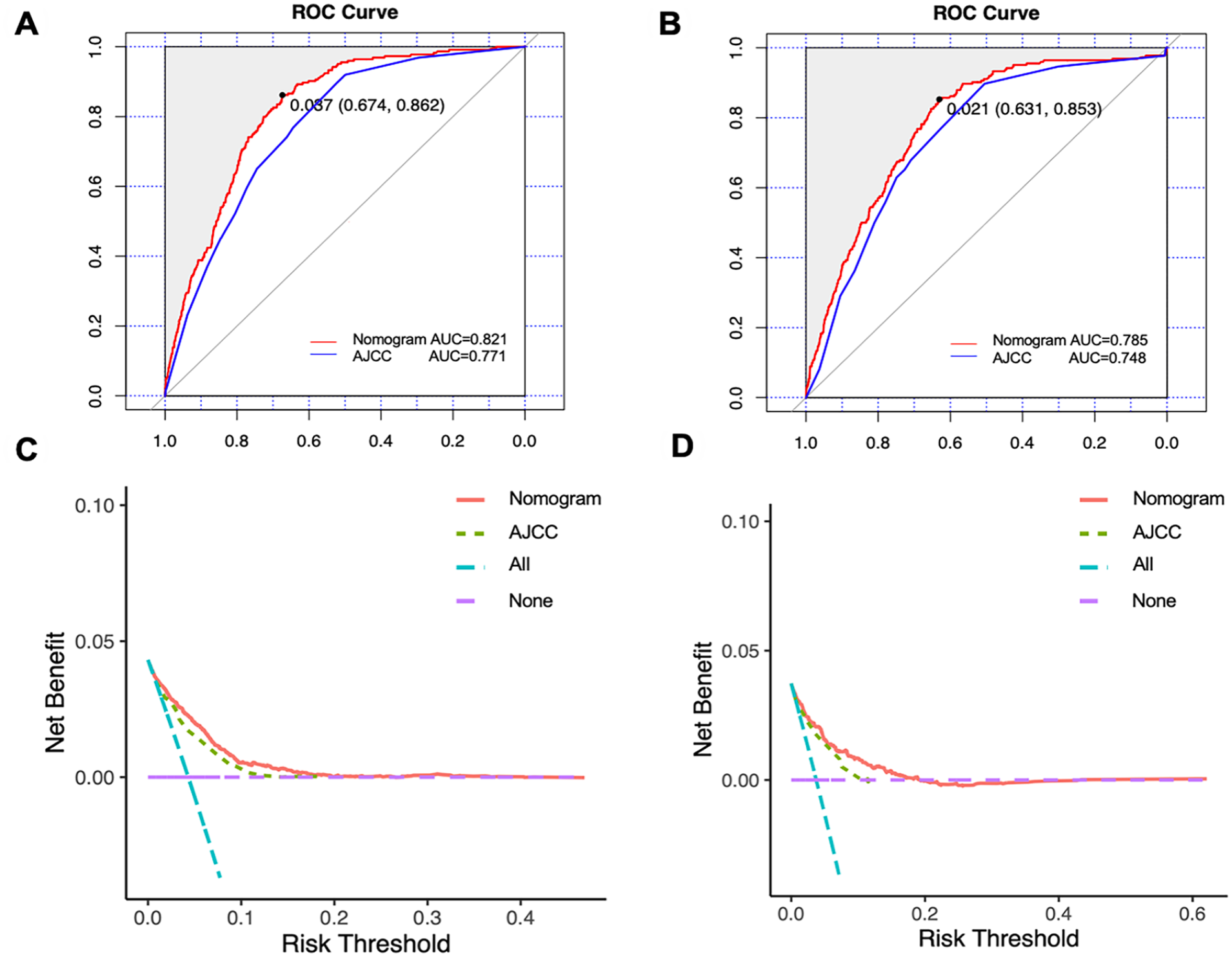
Comparison of predicting the incidence of distant metastases between AJCC TNM staging and the nomogram using ROC curves and DCA curves. The ROC curve of the training group (A). The ROC curve of the validation group (B). The DCA curve of the training group (C), and the validation group (D).
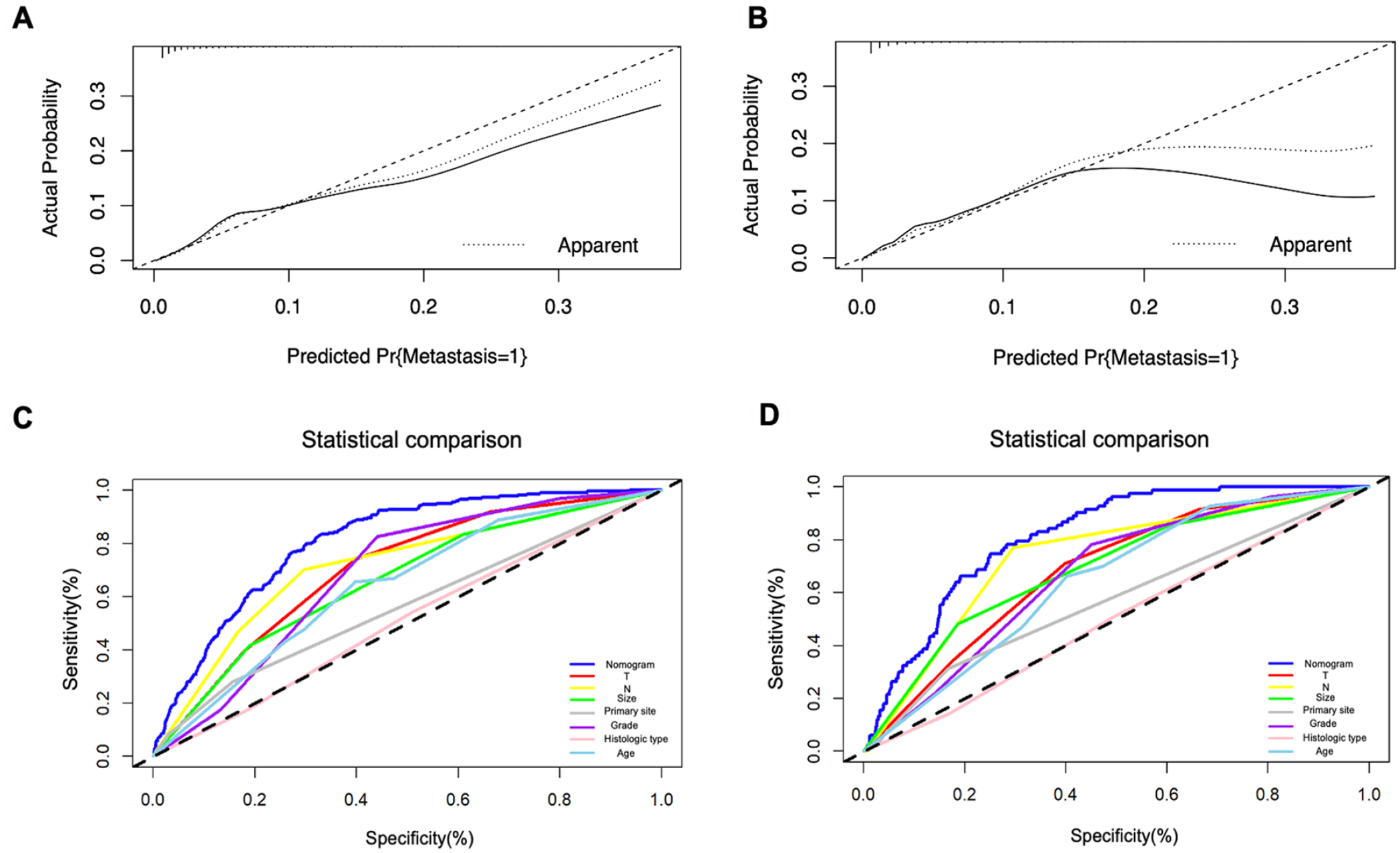
The nomogram calibration curves of the training group (A), and the validation group (B). The ROC curves of the nomogram and all independent factors, including nomogram, T, N stage, primary site, tumor size, grade, and histologic type in the training group (C) and the validation group (D).
Prognostic Factors for SGC Patients with DM
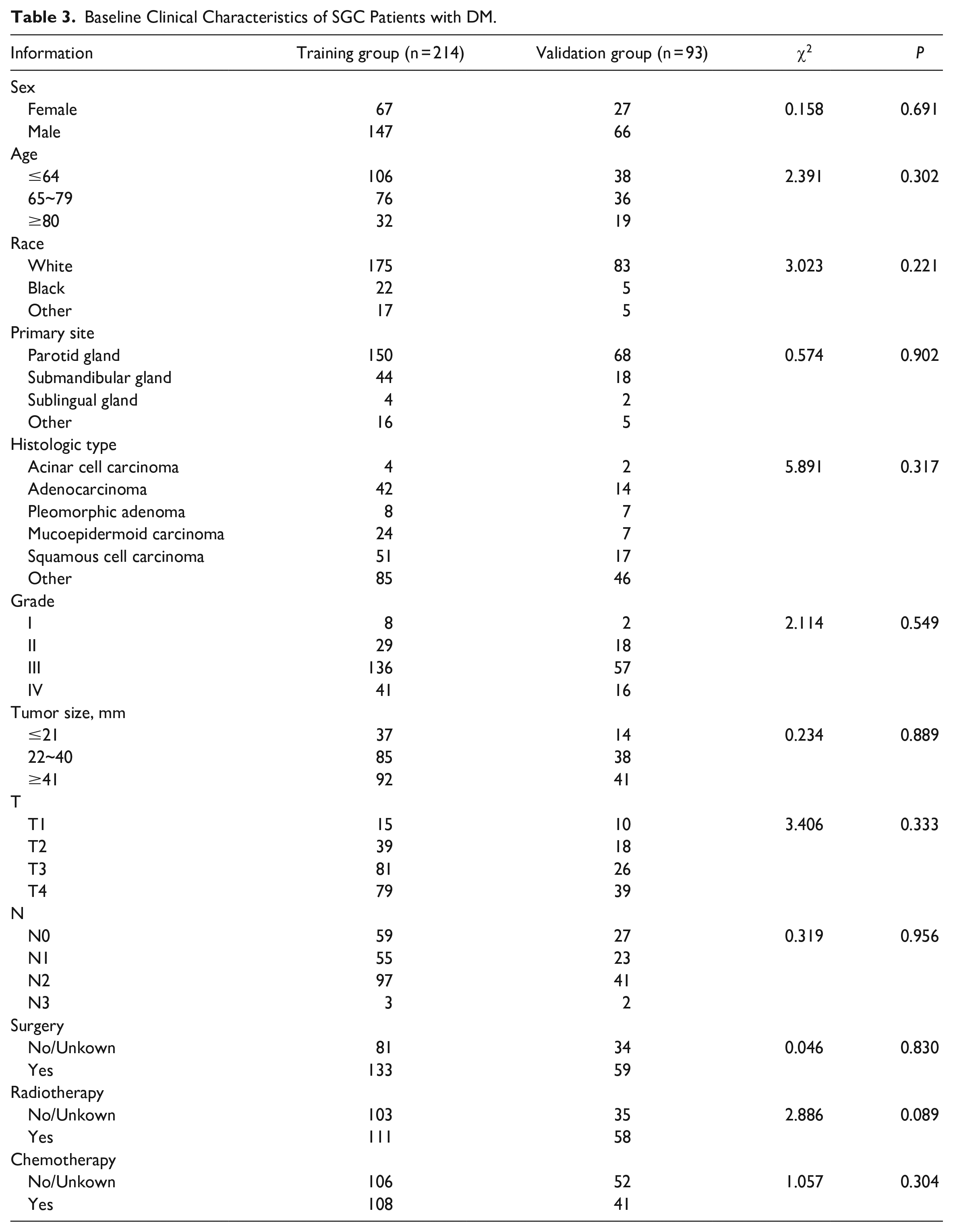
A total of 307 SGC patients with DM were included in the study. They were divided into a training group (214) and a validation group (93), and the chi-square test proved that this grouping was completely randomized. The majority of the primary sites were in the PG (70.1% in the training group and 73.1% in the validation group). In both groups, the number of Grade III patients was the highest (65.5% in the training group and 61.3% in the validation group). The majority of patients underwent surgery (70.1% in the training group and 73.1% in the validation group). About half of the SGC patients received chemotherapy (50% in the training group and 44% in the validation group), and radiotherapy (52% in the training group and 62% in the validation group). The specific demographic and clinical characteristics of SGC patients with DM are shown in Table 3.
Baseline Clinical Characteristics of SGC Patients with DM.
As shown in Table 4, twelve factors were analyzed by univariate Cox analysis. The outcome of multifactorial Cox regression analysis showed that age ≥80 years old, high histologic grade, and minor salivary glands primary site were independent prognostic factors for SGC patients with DM.
Univariate and Multivariate Cox Analyses in SGC Patients with DM.
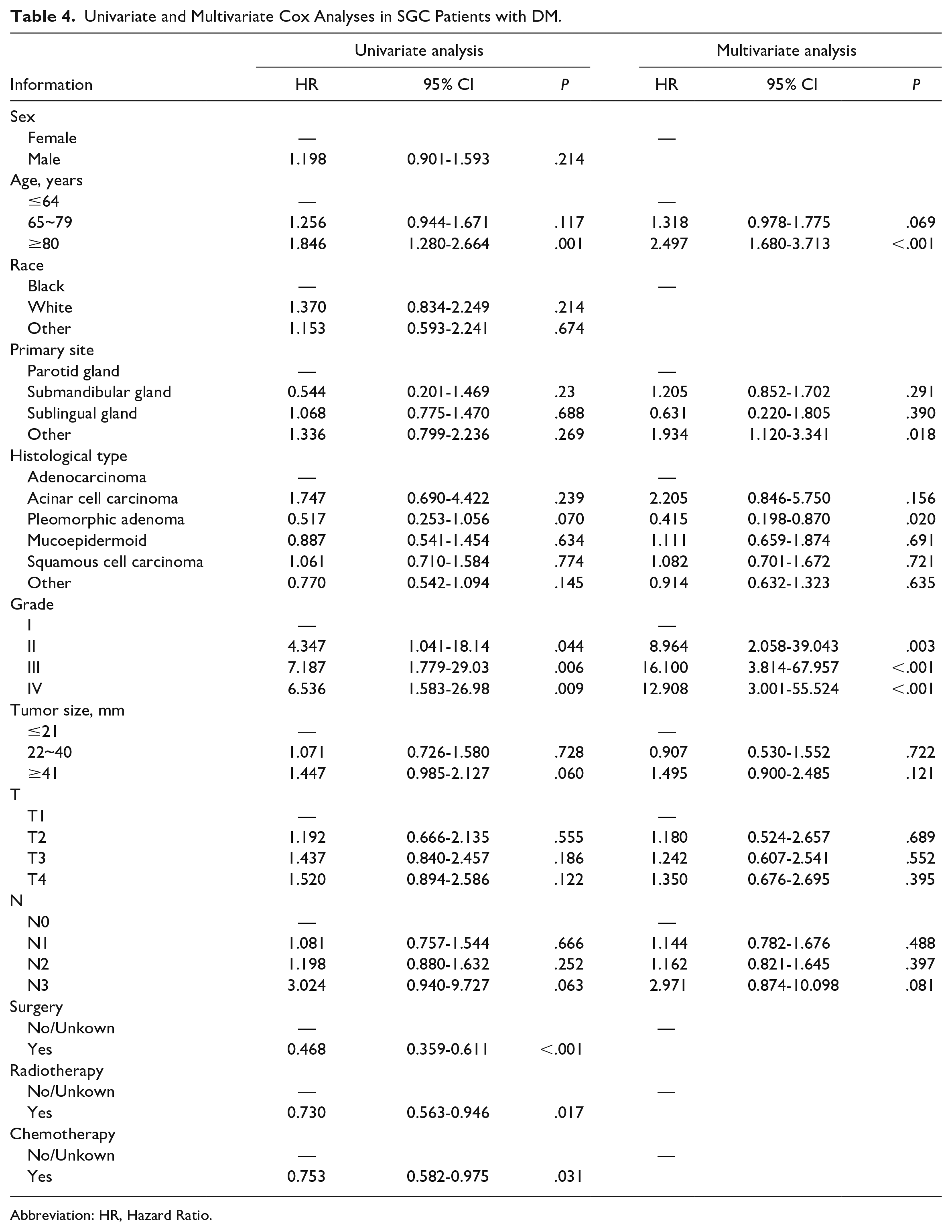
Abbreviation: HR, Hazard Ratio.
Prognostic Nomogram Development and Validation
Based on likelihood ratio test, the model incorporating age, histologic grade, T, N stage, primary site, histological type, and tumor size was significantly different from the model incorporating independent prognostic factors (χ2 = 27.134, P = 0.012), and we found that when including those 7 factors the model has the smallest AIC value (AIC = 2275). So 7 factors were selected into nomogram (Figure 4).
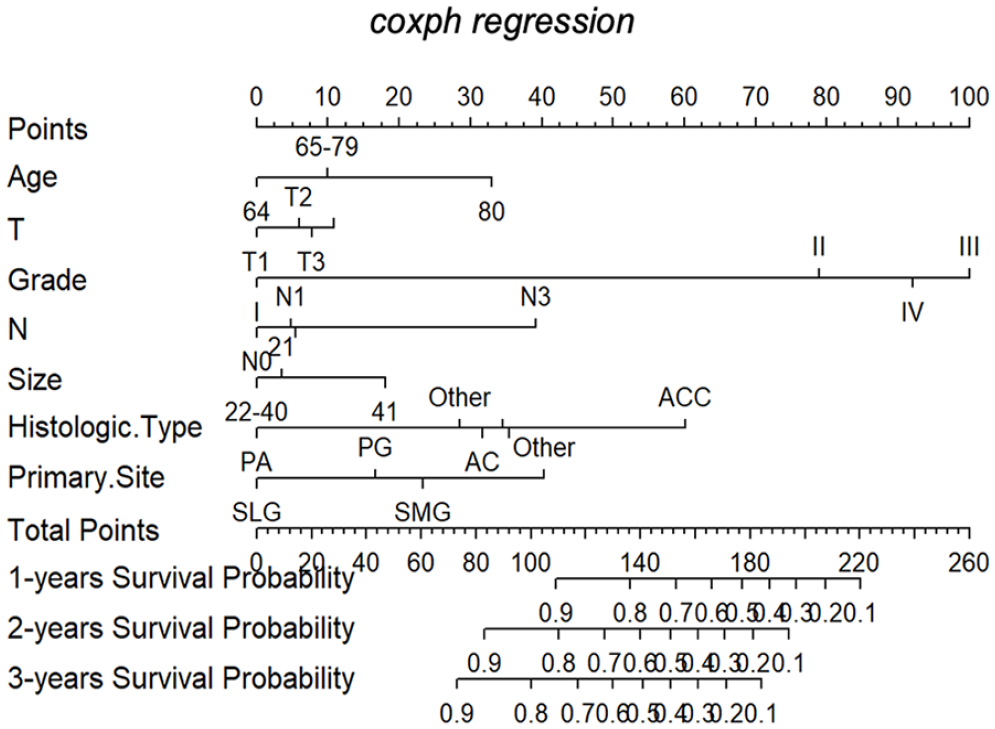
The prognostic nomogram to predict the survival of SGC patients with DM. The total Points line at the bottom of the nomogram is used to predict the survival rate of the patient for 12, 24, and 36 months.
ROC analysis results suggested better discrimination by the nomograms than by AJCC staging. Nomogram vs AJCC staging’s AUC value in the training group (12-months AUC: 0.724 vs 0.555, 24-months AUC: 0.708 vs 0.534, 35-months AUC: 0.676 vs 0.532) (Figure 5A-C) and validation cohort (12-months AUC: 0.749 vs 0.660, 24-months AUC: 0.711 vs 0.581, 36-months AUC: 0.776 vs 0.624) for OS (Figure 5D-F). the DCA curves determined that the nomogram had good performance in clinical practice at 12, 24, and 36 months in both training group (Figure 6A-C) and validation group (Figure 6D-F). In addition, the calibration curves of the nomogram for the probability of 12, 24, and 36 months OS exhibited a strong agreement between nomogram-predicted OS and the actual outcome in the training set (Figure 7A-C) and validation set (Figure 7D-F). Moreover, we further compared the discrimination between nomogram and each independent prognostic factor, and the results indicated that the accuracy of nomogram was better than all independent prognostic factors in the training group (Figure 8A) and validation group (Figure 8B). The results of the K–M survival analysis suggested that there existed different survival patterns among patients in the high- and low-risk group and the low-risk group had a favorable prognosis for OS (Figure 8C and D).
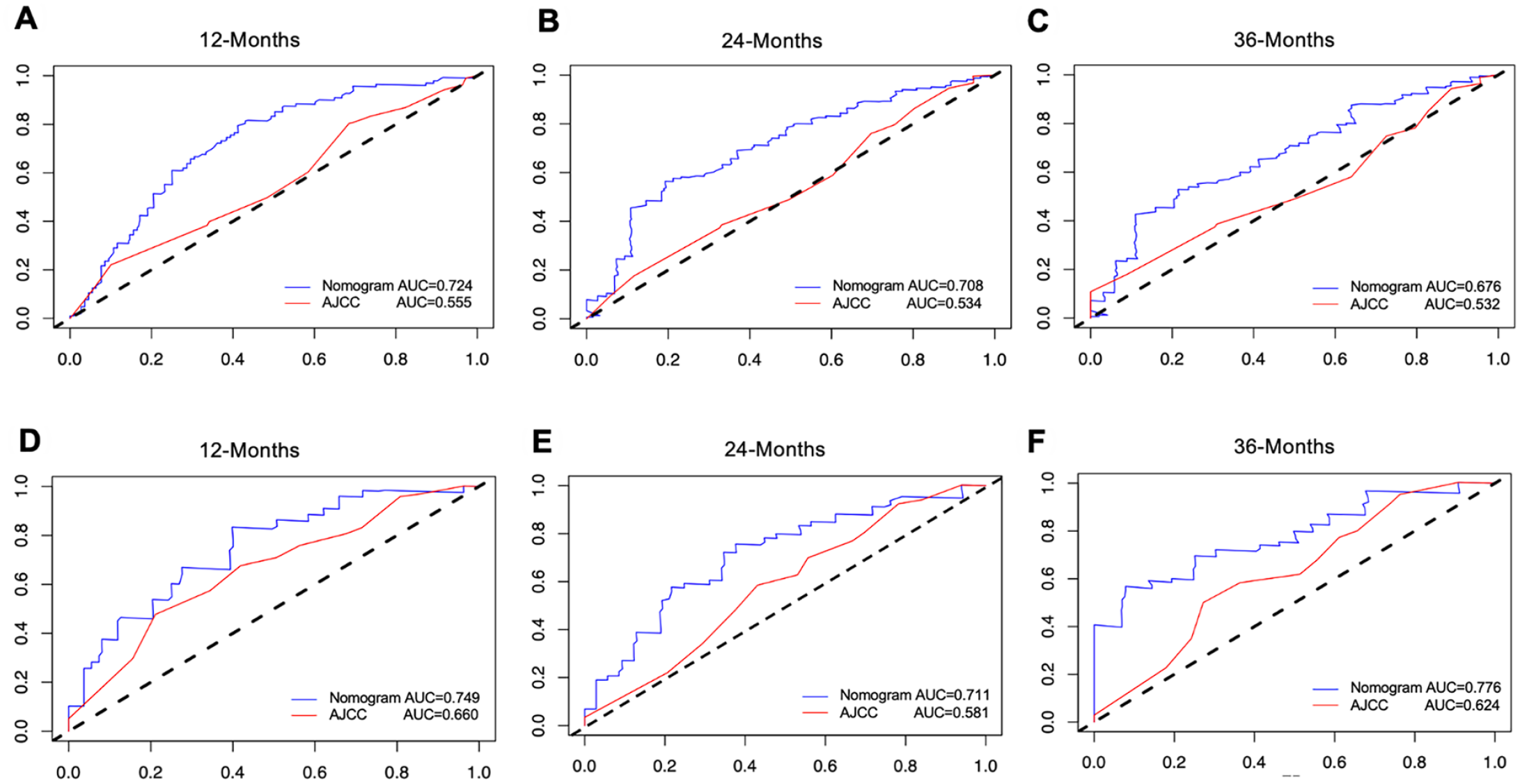
Comparison of survival prediction between AJCC TNM staging and the nomogram using time-dependent ROC curves for the 12-month (A), 24-month (B), and 36-month (C) in the training group. ROC curves for the 12-month (D), 24-month (E), and 36-month (F) in the validation group.
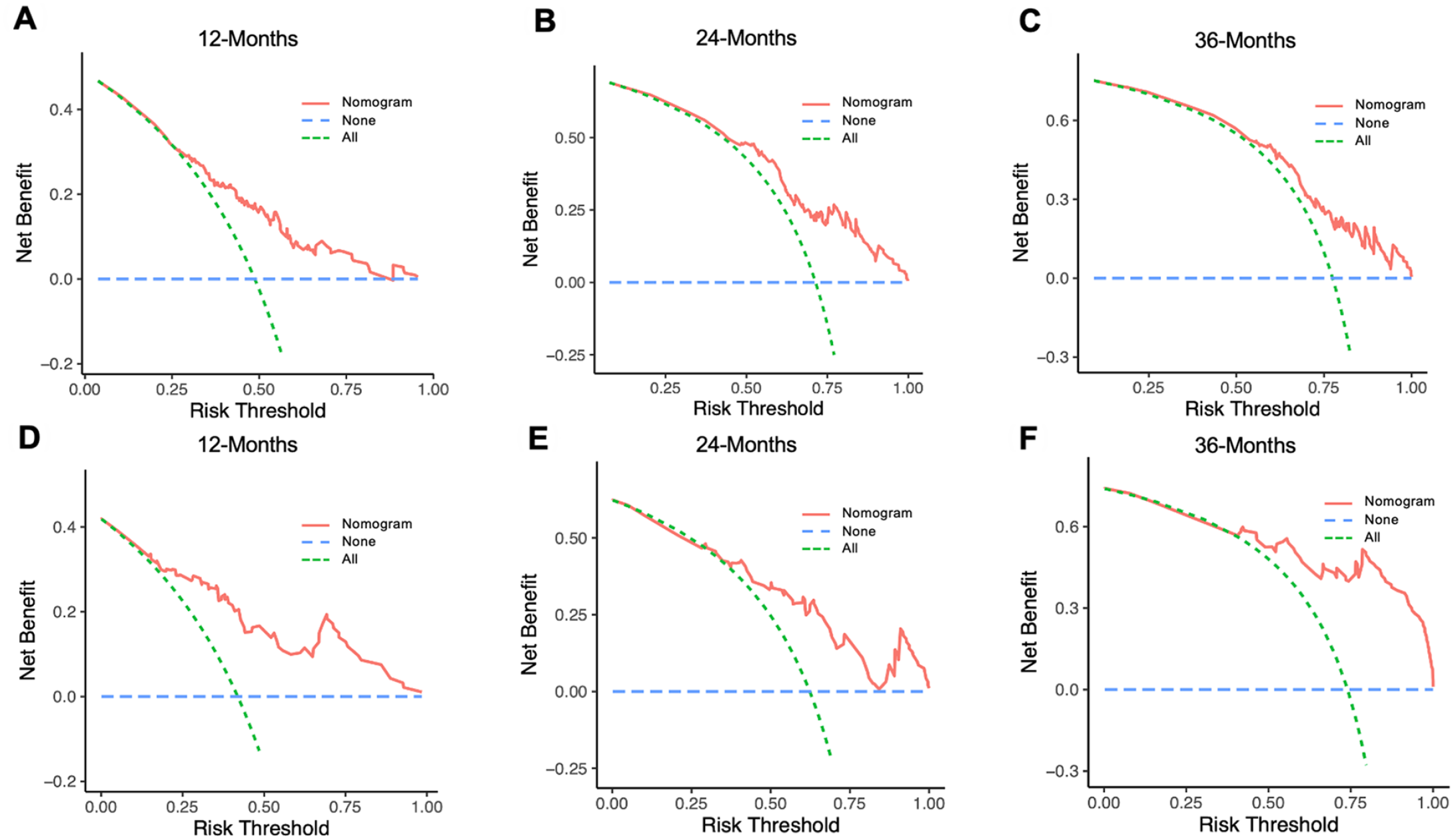
The DCA curve analysis of the nomogram for the 12-month (A), 24-month (B), and 36-month (C) in the training group. The DCA curve analysis of the nomogram for the 12-month (D), 24-month (E), and 36-month (F) in the validation group.
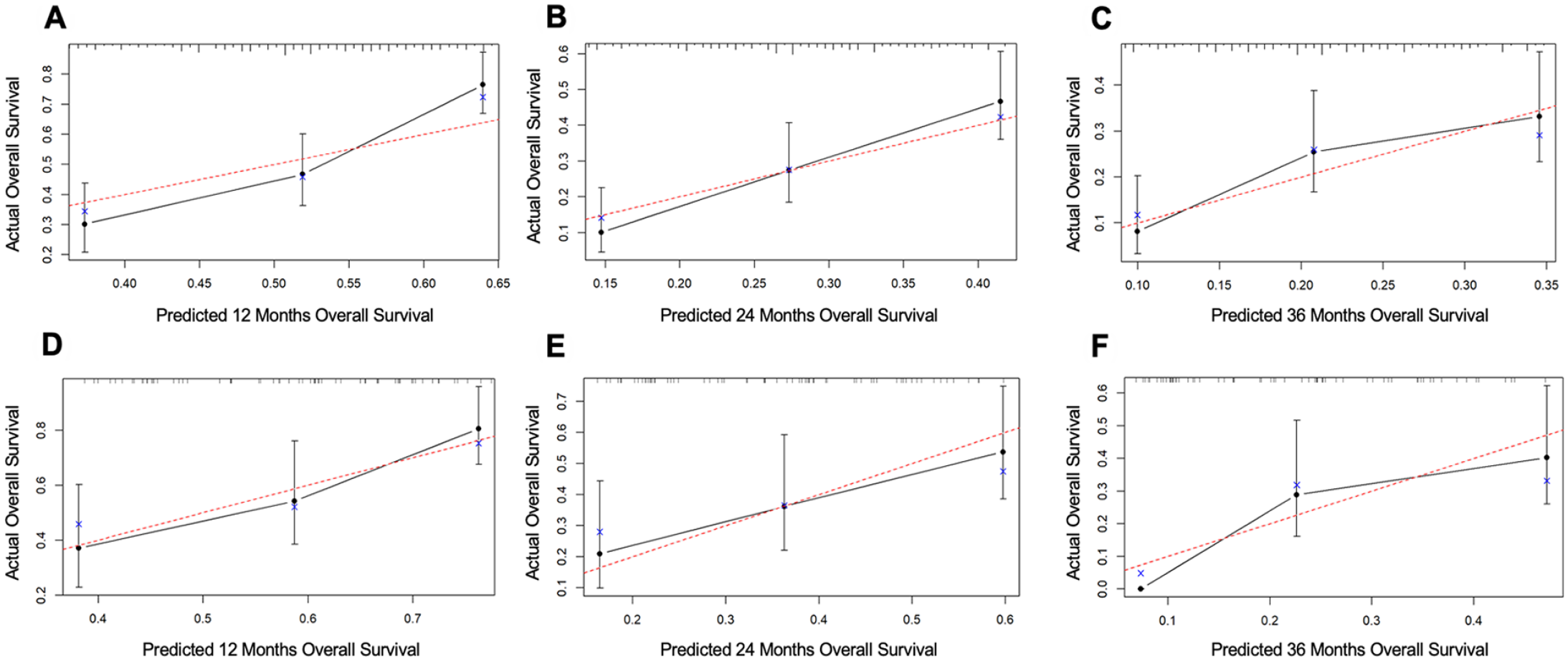
The nomogram calibration curves of the training group at 12-month (A), 24-month (B), and 36-month (C). The nomogram calibration curves of the validation group at 12-month (D), 24-month (E), and 36-month (F).
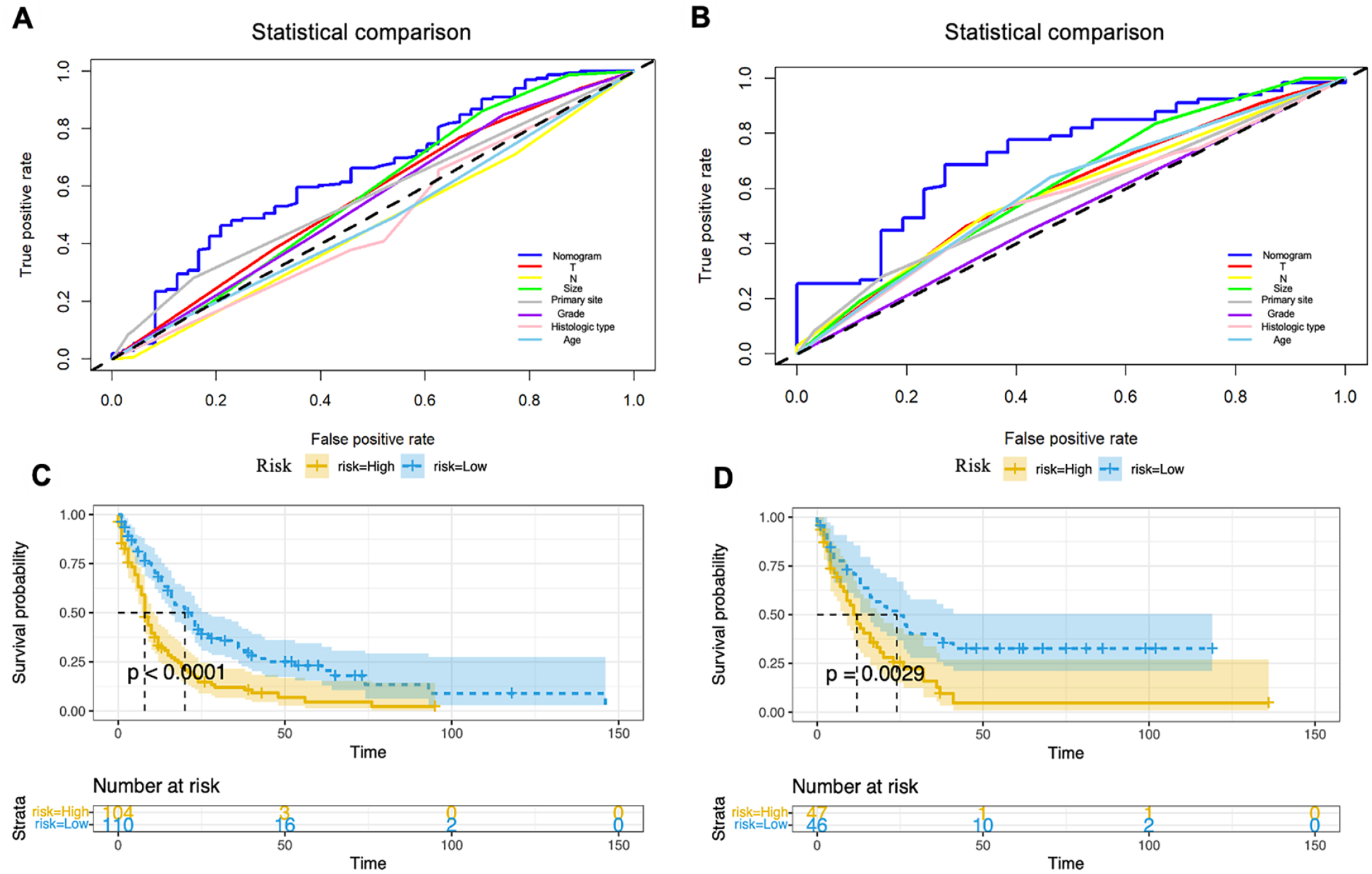
The Kaplan–Meier survival curves of the patients in the training set (A) and the validation set (B). The ROC curves of the nomogram and all independent factors, including nomogram, T, N stage, Primary site, tumor size, grade, and histologic type in the training group (C) and the validation group (D).
Discussion
There are several different types of malignant SGC. 20 While some are almost benign in their natural behavior, others are extremely aggressive. 5 When DM occurs, it can be devastating for the prognosis of SGC patients. 9 In the study of Soo Jung Nam et al., 8 DM as the independent risk factor of prognosis in SGC is associated with poor survival outcomes. They also reported that the median survival duration after DM development was only 15 months. Therefore, reliable risk and prognosis prediction tools are necessary for DM patients to enable individualized early diagnosis, early prevention, and prognosis assessment. Although the AJCC staging system is a widely recognized and commonly used prediction method by clinicians,21,22 its lack of important factors may reduce the accuracy of the prediction. The study about the prognosis of invasive micropapillary carcinoma of the breast reflected poorer predictive accuracy of AJCC staging than the established nomogram. 23 The nomogram has advantages over the traditional TNM staging system,23,24 allowing identification and stratification of patients participating in clinical trials and helping to provide more reliable information for clinical decisions. 25 This study constructed diagnostic and prognostic nomograms and compared them with the AJCC staging system, demonstrating that the nomograms were more accurate. Thus, the nomogram provides a more reliable reference for clinical assessment and treatment of distant metastases in terms of diagnostic and prognostic scores.
The use of nomograms in the development of predictive cancer models has increased over the past several years,10,14 but there are few studies of nomograms for SGC patients with DM diagnosis and prognosis. In our SEER database-based study, we screened 7418 eligible patients from real data over the course of 11 years. We identified 4 independent risk factors of DM in SGC patients by univariate and multifactorial regression analysis, including non-parotid primary site, high grade, T, and N stage. Age ≥ 80 years old, high grade, and minor salivary glands primary site were independent prognostic factors for SGC patients with DM. The majority of nomogram studies choose independent risk factors as selected variables to build nomograms,26-28 but we believe that using only independent risk factors to build predictive models is not accurate enough. For example, in our study, the univariate Cox regression analysis showed that age ≥80 years was a prognostic independent risk factor for distant metastases in SGC patients, but according to a global study in 2017, the average life expectancy was only 74 years. 29 Thus, the AIC was used to select variables for the nomogram. Seven variables (including age, primary site, histologic type, T, N stage, grade, and tumor size) were selected by the stepwise regression based on AIC minimum and incorporated into both diagnostic and prognostic nomogram. The development and validation of the nomogram illustrate it has a high degree of reliability.
For diagnostic cohort, compared with the AJCC8 staging system, our risk nomograms had higher time-dependent AUC values, demonstrating a superior discriminative power for predicting DM occurrence. Moreover, DCA curves also demonstrated that the nomogram was more accurate at predicting DM than the traditional tool. Furthermore, the actual survival and the nomogram-predicted survival agreed well according to the calibration plots, which indicated the reliability of the new models. The results of the ROC curves of the nomogram and all independent factors also supported the view that our prediction models were more effective and accurate than other factors. In the present study, high T stage, high N stage, high grade, and non-parotid tumor sites were significantly associated with the occurrence of DM. This is the same conclusion reached in some previous studies.7,8,30,31 Wang et al. 30 reported that histologic grade was the most significant risk factor influencing DM in patients with SGC. Although our study only considered histological type as a possible risk factor for DM occurrence, based on the complexity of the pathological staging of SGC, we believe that the impact of SGC patients’ histological type on DM occurrence deserves more study.
The prognostic nomogram established in this study showed higher AUC values compared to the AJCC8 staging criteria at 12, 24, and 36 months, respectively, demonstrating excellent discriminatory ability to predict the onset of DM and OS. In addition, the reliability of the new model was demonstrated based on the DCA curves and calibration curves. The ROC curves compared to the independent factors show that the nomogram also exhibits a better level of prediction. Patients were classified into high-risk or low-risk groups based on their total nomogram scores. In the high-risk group, survival rates were statistically significantly lower than in the low-risk group. Therefore, nomogram-based risk stratifications can provide clinicians with useful information to differentiate between high-risk patients and those who should receive a more active treatment plan. Our research shows that age ≥ 80 years old, high grade, and minor salivary glands primary site were significantly associated with the prognosis of DM. Especially the HR values of grades II, III, and IV were significantly higher than the others, indicating that the grade stage is an important factor in the progression of SGC patients. There are fewer studies focused on the prognosis of SGC patients with DM, and we did not search for similar results. Currently, several researchers have studied the factors influencing the prognosis of SGC. According to this study, 32 surgery-radiation-chemotherapy, intraparenchymal extension, and tumor size >40 mm were prognostic factors. In a 30-year-long study, 33 multivariate analysis confirmed the presence of lymphovascular invasion and shorter disease-free intervals are independent risk factors.
Our study listed factors that can be easily obtained from clinical and pathological data by using the nomogram. Through calibration and validation, our nomograms have high accuracy and reliability and better accuracy than the AJCC staging system. Thus, the diagnostic and prognostic nomogram that we developed can be combined with other factors in the clinic to provide a reliable reference for physicians. However, our study still has some shortcomings. Firstly, because of the low incidence of DM in SGC patients, no relevant data on DM was found in other open databases. This resulted in the lack of an external validation group for further validation of the results in this study. Secondly, the histologic type of SGC is very complex. Despite analyzing common tissue typing, the study did not go into any further detail about some rare tissue typing, which may have overshadowed the effects of certain rare tissue typing. Finally, as most of the patients involved in this study were white, further studies are required to determine if the findings of this study can be applied to other ethnic groups.
Conclusions
Total 7418 SGC patients and 307 SGC patients with DM were screened through the SEER database. In contrast to the traditional AJCC staging system, Risk and prognostic nomograms demonstrated excellent discrimination and clinical efficacy. In validation, the nomogram-based prediction tools demonstrated good accuracy, which can show that risk and diagnostic nomograms can be used as visual graphical tools to quantitatively assess the risk and prognosis of SGC patients with DM and to help physicians make clinical treatment decisions.
Footnotes
Author’s Contributions
Jiayu Shi and Yunjian Fan proposed the ideas. Jiayu Shi and Yunjian Fan collected and investigated data. Jiayu Shi, Yunjian Fan, Jiazhen Long, and Shuqi Zhang performed the formal analysis and validation. Jiayu Shi, Zhen Zhang, and Wenyue Chen drafted the original manuscript. Shuguang Liu and Jin Tang reviewed and edited the manuscript. Shuguang Liu also supervised and administrated this project. All authors read and approved the final manuscript.
Availability of Data and Materials
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors would like to thank SEER * Stat software for giving statistical assistance, and the funding: Guangdong Provincial Key R&D Program of the stomatological hospital, Southern Medical University (Grant No. 2018B090906001).
Ethics Approval and Consent to Participate
The present study used previously collected anonymized and de-identified data from the SEER database. Therefore, no additional informed consent was required.
Consent for Publication
All authors agree to publish.
