Abstract
Introduction
Obstructive sleep apnea syndrome (OSAS) is a common condition in otorhinolaryngology that affects a large number of children. The clinical signs of obstructive sleep apnea and hypoventilation in children include snoring, sleep disorder, frequent decrease in blood oxygen saturation, and daytime sleepiness, all of which are detrimental to the health of children. There is no known age of onset for OSAS, and it is thought to affect anywhere from 1% to 5% of children.1,2 Without treatment, OSAS can lead to serious complications, most notably in the areas of neurocognition, behavior, and cardiovascular health, and increase the risk of stroke in young people.3,4 Upper airway obstruction is largely triggered by tonsil and adenoid hypertrophy. 5 Therefore, tonsillectomy and/or adenoidectomy are the primary treatments for OSAS in children. 6 However, the pathogenesis of OSAS in children is complex, resulting from a confluence of neuromuscular and structural abnormalities. 7 The pharynx, the nasal cavity, and the nasopharynx collectively make up the upper respiratory tract. Increased airway resistance can be caused by obstruction in any of these areas. Increased pharyngeal compliance is an independent risk factor for OSAS in children. 8 Previous research has shown that the normal nasal passage accounts for 50% to 75% of the total resistance of respiratory flow. 9 According to some research, patients with compromised nasal respiration have been found to have an increased risk of developing OSAS. 10 Cielo et al. found that there is a difference in the anterior nasal cavity narrowing between children with mild OSAS and moderate OSAS. 11 As a result, they hypothesize that nasal resistance (NR), in addition to structural narrowing from adenotonsillar hypertrophy, may be a cause of OSAS in children. Therefore, anterior NR is a crucial risk factor in the management of OSAS. Our previous research revealed an interesting phenomenon: in some children, the size of their tonsil adenoids did not change in response to drug treatment, despite a complete resolution of their symptoms. The results suggest that other factors are involved in the occurrence of OSAS in children. Therefore, in this study, we aimed to observe the nasal ventilation function (NVF) in children aged 6 to 12 years with OSAS and investigate the impact of NVF on the efficacy of drug treatment in such children.
Materials and Methods
Patients
Participants for the study were enrolled from the ear, nose, and throat (ENT) section of the Department of Otolaryngology, Children’s Hospital of Chongqing Medical University, from September 2019 to September 2021. The participants were children aged 6 to 12 years and were referred by pediatric otolaryngologists. Participants were suspected of having OSAS if they exhibited symptoms like restless sleep, excessive daytime sleepiness, snoring or inattention/hyperactivity, mouth breathing, and witnessed apneas. Inclusion criteria: (1) Children diagnosed with OSAS based on polysomnography (PSG) monitoring; (2) children with symptoms that persist for more than 12 months; (3) children who were willing to undergo treatment; and (4) children without any history of tonsillectomy and/or adenoidectomy. Exclusion criteria: (1) History of related diseases such as nasal disease (persistent sinusitis attacks or rhinitis), nasal anatomic anomalies (nasal septum flank curvature), cranial and facial deformity, immunodeficiency disease, cardiovascular disease, or who had recently undergone medical therapy involving corticosteroids (either oral, inhaled, or intranasal) or oral montelukast (OM), or had a histamine allergy; (2) Children who have had superior respiratory tract infections in the previous 2 weeks or who have used antibiotics within the last 4 weeks. Healthy participants who underwent physical examination in the Children’s Hospital during the same period were selected as the control group. The present study was approved by the Ethics Committee of the Affiliated Children’s Hospital of Chongqing Medical University, China. The caregivers of the children signed the informed consent and helped monitor and conduct follow-up for the children.
BMI Z-Score Calculation
The height and weight of all children were recorded before they underwent nighttime PSG. The Chongqing Center for Disease Control and Prevention’s online body mass index (BMI) Z-score calculator was used to calculate the Z-score. Children were classified as obese if they had a BMI Z-score greater than 1.65. 12
Overnight Sleep Study
A trained polysomnographic technologist used a PSG system (NOX A1, Iceland) to conduct an overnight PSG study in the sleep center. The following indices were measured: Apnea-hypopnea index (AHI), obstructive apnea-hypopnea index (OAHI), REM duration, and arterial oxygen saturation (SpO2). The number of apneas and hypopneas per hour of total sleep time were defined as the OAHI (TST). According to the Guidelines for the Diagnosis and Treatment of Obstructive Sleep Apnea in Chinese Children, OAHI > 1/h TST was used to diagnose OSAS in the children (2020). The children were classified based on the OAHI as follows: mild OSAS, OAHI = 1 to 5/h TST; moderate OSAS, OAHI = 5 to 10/h TST; severe OSAS, OAHI > 10/h TST. OAHI > 1/h TST was considered eligible for inclusion in this study. 13
Acoustic Rhinometry
The children underwent acoustic rhinometry (AR) after the PSG examination. The measurements were taken using an acoustic rhinometer (GM Instruments Ltd, A1, UK) in accordance with previously established protocols in our laboratory. The nasal minimal cross-sectional area (NMCA) was recorded in 3 different segments: NMCA1 (area corresponding to the nasal valve), NMCA2 (area corresponding to the anterior portion of the inferior nasal turbinate), and NMCA3 (area corresponding to the tail of the inferior nasal turbinate). The other parameters included: nasal cavity volume (NCV) from 0 to 5 cm and nasopharyngeal volume (NPV) from 6 to 8 cm (DCAN). 14 The mean values of NMCA2 and NMCA3 were used to evaluate the participants in our study.
The tests were performed and completed in a quiet room at a temperature ranging between 20°C and 25°C by the same experienced technician. The morning of the test, the technician guided the children on the test process, and it took about 30 minutes for the children to be acclimated to the test environment. The supine position was used to simulate nighttime sleeping positions to take the AR readings. The nares of the participants were in close proximity to the wand tip of the disposable pediatric plastic adaptor, with the nose of the wand pointing downward. Children were instructed to close their mouths and not move their tongue or to swallow during data acquisition (which was limited to 10 seconds to avoid interferences) and to breathe through their noses at the end of each exhale. The study was performed during a period of natural nasal respiration. 15
Rhinomanometry
The children underwent the NR test (ATMOS Diagnostic Cube, Germany) at the same time as the AR. 16 The children were tested while awake and lying in the supine position. The children were asked to breath normally with their mouth closed and the nasal probe parallel to the nasal ridge. This position kept the nasal alae tightly connected but did not alter the nasal shape. The inspiration and expiration rates of the participants were recorded every 12 breaths and were then averaged. NR was measured in both nostrils at a constant trans-nasal pressure of 150 Pa.
Questionnaires
The caregivers of the children completed the OSA-18 questionnaire, to assess the quality of life (QoL) of participants. 17 The questionnaire includes 18 items, divided into 5 categories (physical symptoms, daytime functioning, emotional symptoms, sleep disturbance, and caregiver concerns). To assess the frequency of specific symptoms, the 18 items were scored using a 7-point ordinal scale. The scale ran from 1 to 7, with 1 being “none of the time” and 7 being “all of the time.” Total, domain, and item scores were recorded. The aggregated score was generated by adding the scores for each of the 18 items, and the total was between 18 to 126 points. The effect of OSAS on QoL was evaluated using the overall QoL score. If the mean total QoL score was greater than 1.5, the change in QoL was considered significant.18,19
Medication
The children with OSAS received 12 weeks of treatment with intranasal corticosteroids (ICS) and OM. A single daily dose of 50 μg of mometasone furoate nasal spray was administered in each nostril. We administered 4 mg and 5 mg of montelukast once at night to children aged <6 and ≥6 years of age, respectively. The parents of the children were contacted every month to assess the compliance and potential side effects. Following the completion of the 12-week course, some of the children underwent a second PSG test, AR, and rhinomanometry.
Statistical Analysis
The results are presented as the mean ± standard deviation unless otherwise stated. The t-test was employed to compare the means of the 2 groups in case of normally distributed measurement data. We looked at age, gender, and BMI as explanatory variables. All of the data were analyzed using SPSS21.0 statistical software. The differences were statistically significant at a P value of .05.
Results
We enrolled 109 children who were diagnosed with OSAS from the ENT section of the Department of Otolaryngology, Children’s Hospital of Chongqing Medical University. Based on the PSG data, the children were grouped into 3 groups: mild OSAS, moderate OSAS, and severe OSAS. Furthermore, 116 healthy children (63 males and 53 females) were recruited from the physical examination center of the hospital as the control group. The PSG and demographic data of all the children in the study are shown in Table 1. There was no significant difference in the anthropometric and demographic data of the 3 OSAS groups and the control group, however the OSAS groups differed significantly from the control group with respect to the PSG values.
Demographic and Polysomnographic Characteristics of Subjects (n = 225).
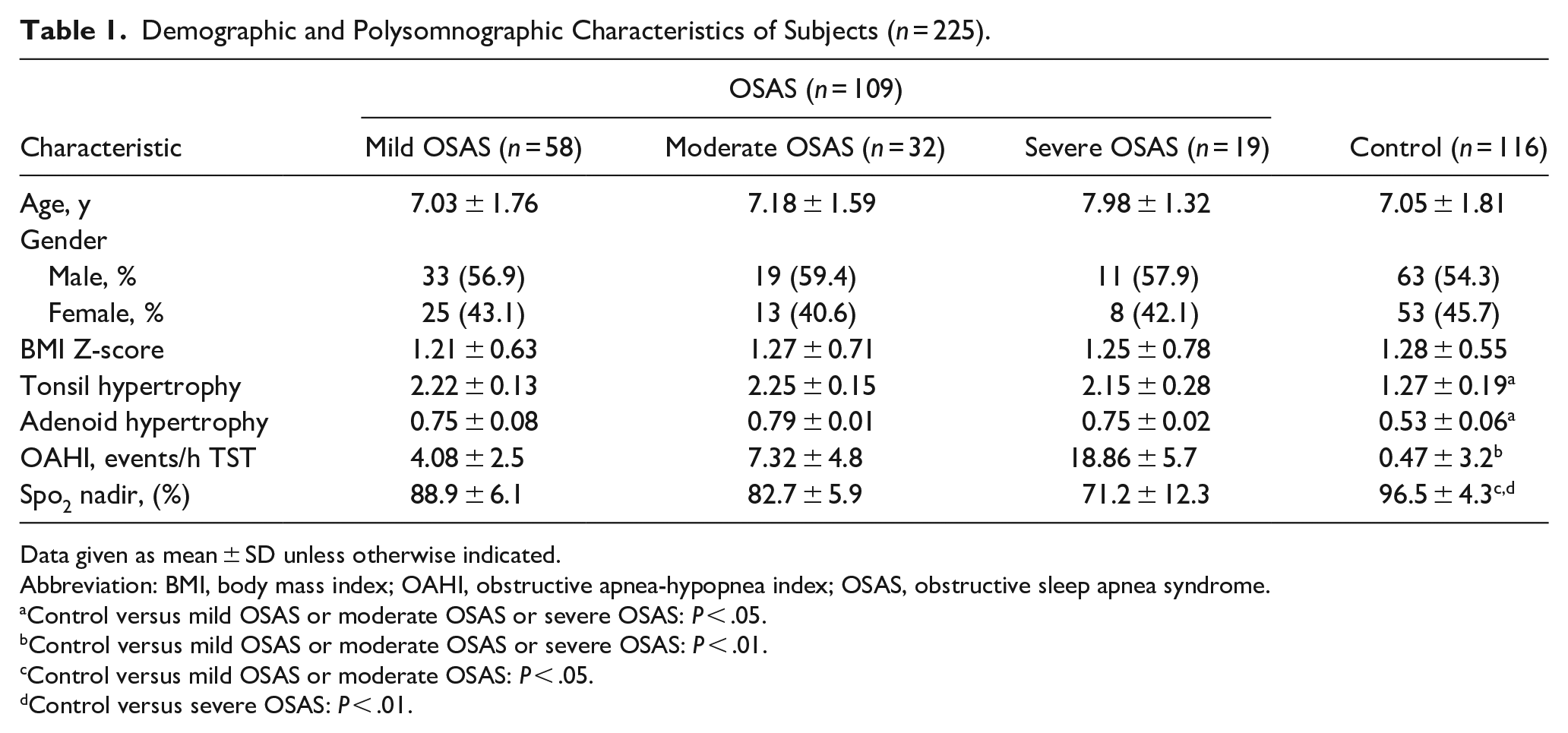
Data given as mean ± SD unless otherwise indicated.
Abbreviation: BMI, body mass index; OAHI, obstructive apnea-hypopnea index; OSAS, obstructive sleep apnea syndrome.
Control versus mild OSAS or moderate OSAS or severe OSAS: P < .05.
Control versus mild OSAS or moderate OSAS or severe OSAS: P < .01.
Control versus mild OSAS or moderate OSAS: P < .05.
Control versus severe OSAS: P < .01.
The parameters calculated from the NR and AR readings were analyzed and the outcomes are presented in Table 2. In comparison to the control group, the children in the OSAS groups had significantly higher NR (P < .05). In comparison to the control group, the OSAS groups had lower NMCA, NCV 0 to 5, and NPV values (P < .05 or P < .01). However, there was no significant difference in the DCAN value between the OSAS groups and the control group. There was no statistically significant difference in the AR and NR values in the 3 OSAS groups (P > .05).
Comparison of Nasal Ventilation Function in the OSAS Groups and the Control Groups (
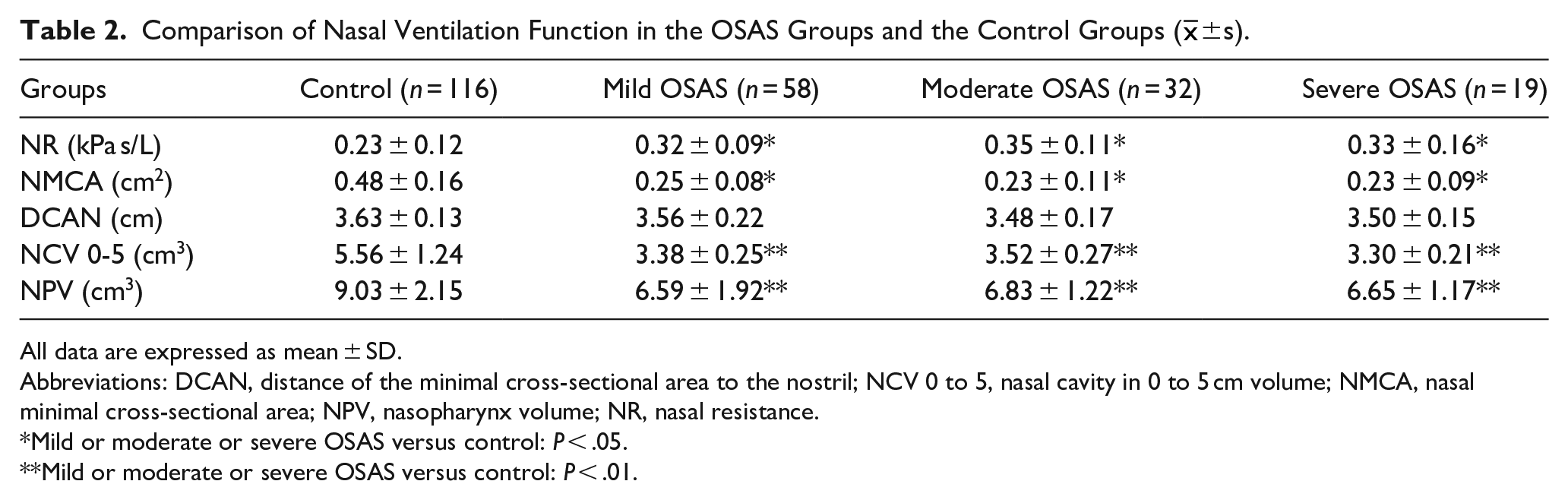
All data are expressed as mean ± SD.
Abbreviations: DCAN, distance of the minimal cross-sectional area to the nostril; NCV 0 to 5, nasal cavity in 0 to 5 cm volume; NMCA, nasal minimal cross-sectional area; NPV, nasopharynx volume; NR, nasal resistance.
Mild or moderate or severe OSAS versus control: P < .05.
Mild or moderate or severe OSAS versus control: P < .01.
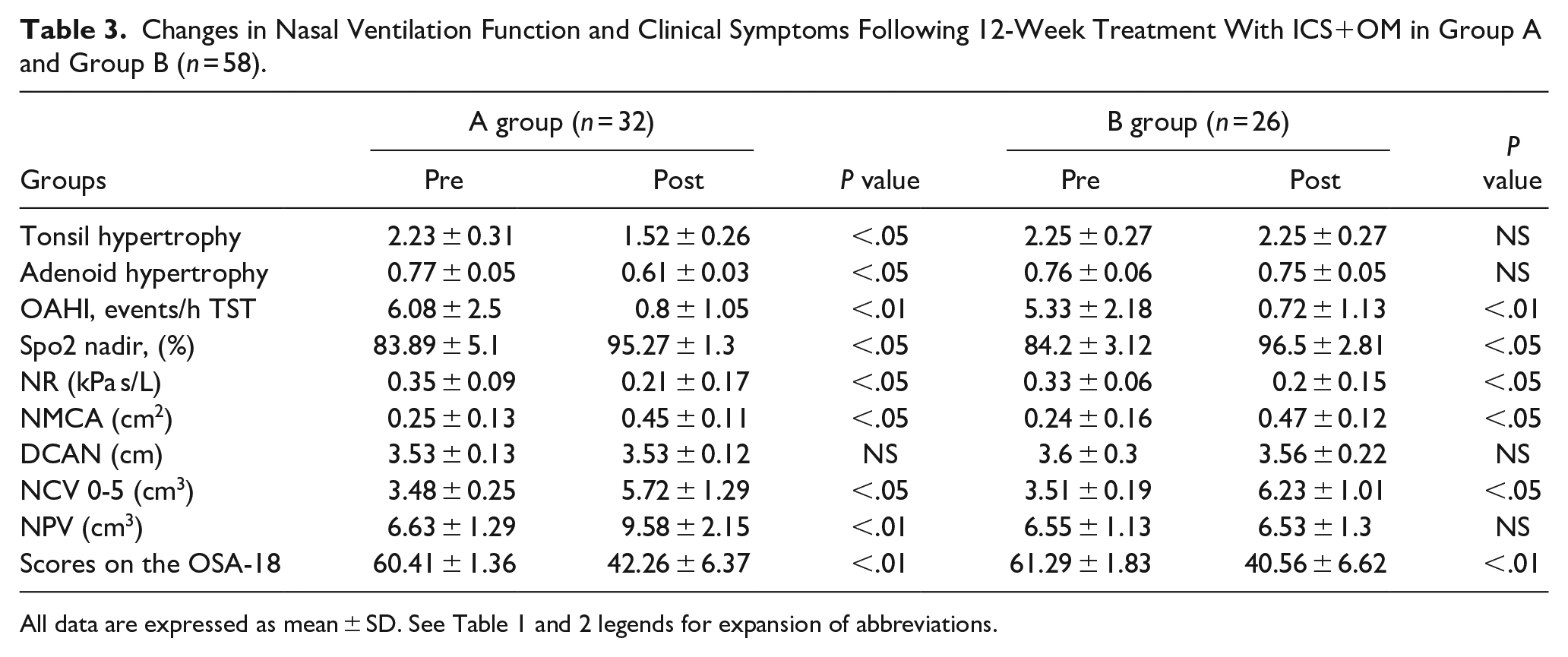
Treatment with ICS+OM was initiated for the 90 children with mild to moderate OSAS; 5 children (5.6%) dropped out or did not adhere to the treatment protocol after the first month, based on reports from their caregivers. Only 69 (81.2%) of the 85 children who completed the 12-week treatment course returned for their follow-up NPSG, AR, and NR evaluation. The 69 children were divided into 3 groups based on the treatment outcomes—group A: effectively cured, group B: successfully treated, and group C: treatment failure. There were 32 children in group A who were effectively cured, as evidenced by a significant reduction in tonsil adenoid size and improvements in AHI and SpO2 nadir. The 26 children in group B were also successfully treated. There were improvements in PSG values and clinical symptoms; however, there was no change in the size of tonsils and adenoids. The remaining 11 children in group C failed to respond to the treatment. The NVF of the children in group A and group B was further investigated. The mean measurements of NR and AR for the 2 groups are shown in Table 3. There were noticeable differences in NR, NMCA, and NCV values in both groups before initiation of ICS+OM treatment and after completion of the treatment, however, the differences between the initial DCAN values and those at the end of the third month in the groups were not statistically significant. As depicted in Table 3, there was a significant reduction in adenoid size and increased NPV value before and after treatment in group A. However, similar changes were not observed in group B.
Changes in Nasal Ventilation Function and Clinical Symptoms Following 12-Week Treatment With ICS+OM in Group A and Group B (n = 58).
Discussion
Here, we report on a 12-week study of NVF in children with OSAS, aged 6 to 12 years, and the changes in NVF brought on by treatment with ICS+OM. We enrolled 109 children with OSAS and 116 healthy children in this study. The NR and AR values of the OSAS groups were found to be significantly different from those of the control group. In addition, regardless of whether tonsil adenoid size improved, NVF in the OSAS groups showed significant changes after treatment.
Researchers have reached a consensus that OSAS in children has different causes than in adults. Obstruction of the upper airway due to enlarged tonsils and adenoids is undeniably a contributing factor. But as research has progressed, other factors have come to the fore, such as anti-inflammatory factors and NR. The nasal cavity is located in the middle of the upper airway and contributes significantly to airway resistance. Rhinomanometry and the acoustic rhinometer have seen increased use in clinical settings due to their convenience and lack of invasiveness. They are also extensively used in scientific research and clinical assessment of pediatric sleep apnea as they can be used to objectively assess nasal ventilation status. Researchers have been looking into the connection between nasal passage obstruction and OSAS for over a decade. Several studies have found a positive correlation between increasing levels of NR as measured by rhinomanometry and increasing levels of AHI in people with OSAS.20-22 The AR values in the OSAS group were also significantly higher compared to the control group, as discovered in our study. Importantly, all the AR measurements in our study were performed while the participants were in a supine position. When standing upright, children with OSAS had larger nasal airways than children without OSAS, according to earlier research, 14 which was the basis for measuring airway dimensions in this study. Furthermore, NR increased significantly in the OSAS group when the children were shifted from an upright to a supine position.23,24 Therefore, the supine position was used during the test to more accurately reflect the way people typically sleep. However, the role of NR in the development of OSAS is contentious. The connection between the 2 is poorly understood. Miljeteig et al. found a possible link between nasal factors and the development of OSAS in their study of 683 patients. 25 However, they found no discernible differences in AHI between patients with different levels of NR. Ersözlü et al. also found that although high NR is associated with OSA, there were no significant difference between rhinomanometric parameters and OSA severity in a larger sample size. 26 On the contrary, Rizzi et al. found a positive correlation between NR and the severity of OSAS in a pediatric study of children between the ages of 4 and 7 years. 27 Our results are in line with those of Miljeteig and Ersözlü. et al.25,26 who found that there was no strong link between NR and the severity of OSAS in children. Due to the small sample size, further research is required to confirm these results.
To further confirm the effect of NVF, the changes in the NR and AR values in children with OSAS after treatment with ICS+OM were continuously observed. Treatment options for children with OSAS currently include both surgical and nonsurgical methods. The effectiveness of tonsillectomy and adenoidectomy is well established in children. Although the benefits of ICS+OM therapy have gradually been well recognized in recent years,28,29 there are still many uncertainties. First, the treatment is only effective in children with mild to moderate disease and has no significant effect on children with severe disease. In addition, the response of children to the treatment in the mild to moderate category is not the same. Three different outcomes after ICS+OM treatment were observed in this study. The treatment was effective in some children, with significant reduction in tonsil adenoid size; however, some children did not respond to the treatment. Notably, there was also a group of children who responded to treatment without any change in the size of their tonsil adenoids or only a partial reduction in adenoid size. We observed the improvements in clinical symptoms and PSG values in these children. We hypothesized that when the nasopharyngeal and pharyngeal cavity volumes did not improve despite ICS+OM treatment, the resulting changes in NVF were responsible for the observed improvement in clinical symptoms and PSG findings. Studies on the role of NVF in OSAS have been reported earlier. Mitsuhiko et al. found that an increased NR could play an important pathogenetic role in hypoxemic apnea in obese patients with OSAS. 30 Lee et al. found that the NMCA value decreased in patients with OSAS, which resulted in increased nasal airflow and NR. 31 Nasal obstruction relief has been shown to improve arousal index and sleep structure in children with OSAS, according to some studies.32-34 Based on the results of our previous study and these studies, we further investigated NVF in children after treatment. The result indicated that there was a significant difference in NR, NMCA, and NCV values before initiation and after completion of ICS+OM treatment. Therefore, we suggest that it is critical to improve NVF in the treatment of children with OSAS. The result also indicates that NVF is involved in the pathogenesis of OSAS in children.
Limitations
Possible limitations of our study include its small sample size and the fact that NR was measured with the patients awake and sitting up in the supine position. Therefore, larger studies and double-blind trails are required to confirm this finding.
Conclusion
Improvement of NVF has been shown to significantly enhance clinical symptoms and PSG outcomes in children with OSAS. Therefore, the findings suggest that NVF plays a crucial pathogenetic role in OSAS-related pediatric apnea.
Footnotes
Acknowledgements
We are particularly grateful to all the people who have given us help on our article.
Availability of Data and Material
All data generated or analyzed during this study are included in this article. Further enquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Medical Project of Chongqing Municipal Science and Health Bureau of China (No. 2021MSXM258).
Ethics Approval and Consent to Participate
The study was conducted in accordance with the Declaration of Helsinki (as was revised in 2013). The study was approved by Ethics Committee of the Children’s Hospital of Chongqing Medical University [No.191(2020)]. Written informed consent was obtained from all participants.
